Abstract
Abstract
Background
The typical pattern of thyroid function tests (TFTs) associated with hypopituitarism consists of subnormal free T4 (fT4) or total T4 and normal or marginally elevated thyroid-stimulating hormone (TSH). A previous study calculated an incidence of hypopituitarism of 3.2 cases/100,000/year by following up abnormal TFTs. The aim of this study was to verify the incidence of unsuspected hypopituitarism diagnosed by reflective testing on such samples in a Scottish population.
Methods
Prospective audit of TFT results over 15 months. Individuals with suitable results (fT4 < 9 pmol/L, TSH < 10 mU/L) were identified by the laboratory information system at the Biochemistry Department, Glasgow Royal Infirmary, serving a population of 200,000. fT4 (repeat analysis following assay recalibration), total T3, testosterone (males), luteinizing hormone, follicle-stimulating hormone, prolactin and cortisol were analysed on appropriate samples.
Results
Three hundred and eleven suitable results from 266 adult individuals were identified from a total of 73,650 TFT results, leading to the diagnosis of 10 new cases (age range 28–90 years) of hypopituitarism (approximately 4 cases/100,000/year). Nine patients now attend endocrine clinics. Pituitary imaging was abnormal in five cases (2 large pituitary tumours, one macroadenoma, 2 empty sellae), normal in three cases and two patients were not scanned.
Conclusions
A significant number of cases of unsuspected hypopituitarism can be diagnosed by reflective testing on appropriate samples. fT4 should be an integral part of frontline TFTs as TSH alone cannot assist in the identification of possible hypopituitarism. Laboratories are in an excellent position to assist in early identification of hypopituitarism, which may lead to improved outcomes in such patients.
Introduction
Hypopituitarism is a condition in which secretion of pituitary hormones is inappropriately reduced. It is a rare condition, the incidence of which has been estimated at 4.2 cases/100,000 adult population per year in a prospective study in Spain. 1 Using a combination of an abnormal baseline thyroid function test (TFT) plus additional hormonal tests and referrals to local endocrine clinics, Wardle et al. calculated the incidence of hypopituitarism to be 5.5 cases/100,000/year. 2 Of these, 3.2 cases/100,000 population per year were initially identified due to abnormal baseline TFTs suggesting that TFTs are useful in diagnosing this condition. The authors concluded that hypopituitarism may be more common than usually reported and warned against the use of thyroid-stimulating hormone (TSH) alone, rather than TSH together with total T4 (tT4) or free T4 (fT4), as first-line TFT.
TFTs are the most commonly requested endocrine investigations, used to investigate a wide variety of symptoms and signs. Just as in hypothyroidism, symptoms of hypopituitarism may be vague with low clinical suspicion of the condition. Classically, in central hypothyroidism, TFT results consist of subnormal fT4 or tT4, normal or marginally elevated TSH and normal T3 concentrations. 3 The literature contains many examples of case reports where the diagnosis of previously unsuspected hypopituitarism has followed recognition of such TFT results. 4–6 While endocrinologists will be well aware of this result pattern and likely to elicit clinical evidence of hypopituitarism, other clinicians who request the vast majority of TFTs are more likely to miss such cases. Suggestive TFT results may therefore present an opportunity for diagnosis and it is here that biochemistry laboratories may make a contribution.
T3 is a useful additional test, which may assist differentiation between non-thyroidal illness, usually with a low T3, and central hypothyroidism. One prospective study, however, found that in 37 individuals with central hypothyroidism, 19% presented with low total T3 (tT3). 7 One explanation for the slight increase in TSH sometimes seen in hypopituitarism is an increase in immunoreactive TSH with a decrease in biological activity. 8–10
The aim of our prospective study was to assess the number of cases of unsuspected hypopituitarism that would be diagnosed in a Scottish population by reflective testing on appropriate samples and, thereby, to verify previously reported data. 1,2
Methods
The study was carried out at Glasgow Royal Infirmary which serves an outpatient population of approximately 200,000. In the Biochemistry Department Laboratory, samples were identified from TFT results using the local laboratory information system, Telepath, over a 15-month period from April 2005 to July 2006. To identify possible cases of central hypothyroidism, the following TFT result parameters were selected and Telepath was programmed to identify suitable samples:
TSH < 10 mU/L (reference interval 0.3–5.0 mU/L); FT4 < 9 pmol/L (reference interval 9–25 pmol/L). Non-thyroidal illness: any admission to intensive care unit (ITU), moderately raised inflammatory markers (CRP > 50 mg/L), clinical information consistent with sepsis or systemic illness,
13
chronic renal failure on dialysis.
14
Thyroid-related disease: individuals on antithyroid medication, thyroxine or T3 replacement, and previous thyroidectomy
The TSH cut-off value of 10 mU/L selected was based on two studies showing a range of TSH concentrations in patients with central hypothyroidism up to 10 mU/L (immunoradiometric assay)
11
and 11.1 mU/L (immunofluorometric assay),
12
respectively, and an attempt to exclude primary hypothyroidism. It is routine practice at our laboratory to analyse both TSH and fT4 on samples for all TFT requests. Previously confirmed cases of hypopituitarism, samples from children and repeat samples on the same individuals were excluded from TFT results meeting these criteria, as were insufficient samples, lost samples and those already referred to reference laboratories for additional tests. For repeat requests on the same individual, only the first TFT sample received was investigated further. Also, individuals who had normal TFT results within the previous 3 months, and no reference to a clear clinical change, were excluded. Current and previous clinical information and other biochemistry results were used to identify and exclude cases of probable non-thyroidal illness and specific thyroid conditions as follows:
In order to exclude significant analytical error with the fT4 assay, the remaining samples had repeat analysis of fT4 carried out on all identified samples directly after assay recalibration. This decision was based on our experience of poor internal quality control (IQC) precision at low fT4 concentrations (see below). Samples with normal repeat fT4 were not analysed any further and the laboratory report suggested that TFTs be rechecked after three months. Confirmed subnormal fT4 (<9 pmol/L) prompted the addition of further hormonal investigations, namely gonadotrophins, cortisol, prolactin, tT3 and testosterone (males) on the same sample. Clearly subnormal gonadotrophins in postmenopausal females, subnormal testosterone concentrations in men with inappropriately low gonadotrophins and clearly subnormal morning cortisol were considered indicative of further pituitary dysfunction. This allowed identification of cases of hypopituitarism (≥2 pituitary-target organ axis hormone deficiencies). These cases were discussed with the relevant clinician (general practitioner or hospital specialist) to obtain clinical information regarding past medical history and to recommend further investigation.
Assays were carried out by standard automated immunoassay on a Bayer Centaur analyser (Bayer Plc, Newbury, UK) from April 2005 to September 2005. From September 2005 to July 2006, assays were carried out on an Abbott Architect analyser (Abbott Diagnostics, Maidenhead, UK) apart from cortisol analysis, which was carried out on a DPC Immulite analyser (DPC, Gwynedd, Wales, UK). The change in analysers occurred following local procurement negotiations for laboratory analytical platforms. Comparison studies between the Bayer Centaur and Abbott Architect confirmed that no changes to the TSH or fT4 reference intervals were required. Apart from poorer fT4 precision at low concentrations on the Bayer Centaur, the performance characteristics for the relevant assays on the three analysers were comparable, as detailed below.
Bayer Centaur analyser
All methods were chemiluminescent microparticle immunoassays using magnetic particle separation and acridinium ester for light generation. IQC coefficients of variation (CV) for the assays: fT4 CV, 13.5% at 7.6 pmol/L and 9.6% at 24.5 pmol/L; tT3 CV, 8.2% at 1.3 nmol/L; TSH CV, 7.9% at 0.38 mU/L and 4.0% at 5.20 mU/L; cortisol CV, 7.9% at 75 nmol/L and 9.3% at 555 nmol/L; follicle-stimulating hormone (FSH) CV, <4% at all concentrations; luteinizing hormone (LH) CV, <4% at all concentrations; prolactin CV, 3.2% at 302 mU/L; testosterone, 8.2% at 3.2 nmol/L and 5.7% at 20.0 nmol/L.
Abbott Architect analyser
All methods were chemiluminescent microparticle immunoassays using magnetic particle separation and acridinium ester for light generation. IQC CVs for the assays: fT4 CV, 5.8% at 9.0 pmol/L and 5.4% at 31.0 pmol/L; tT3 CV, 5.7% at 1.3 nmol/L; TSH CV, 4.6% at 0.34 mU/L and 4.3% at 4.8 mU/L; FSH CV, <6% at all concentrations; LH CV, <8% at all concentrations; prolactin CV, 5.0% at 394 mU/L; testosterone CV, 7.8% at 2.4 nmol/L and 4.6% at 17.4 nmol/L.
DPC Immulite analyser
The cortisol method is a solid phase, competitive chemiluminescent enzyme immunoassay. IQC CV was 5.2% at 235 nmol/L.
Results
Over 15 months, 73,650 TFT requests were received at Glasgow Royal Infirmary. This includes requests from primary care, hospital wards and outpatient clinics including specialist thyroid and ENT clinics. Three hundred and eleven samples were identified meeting the Telepath search criteria. Figure 1 summarizes subsequent sample handling.
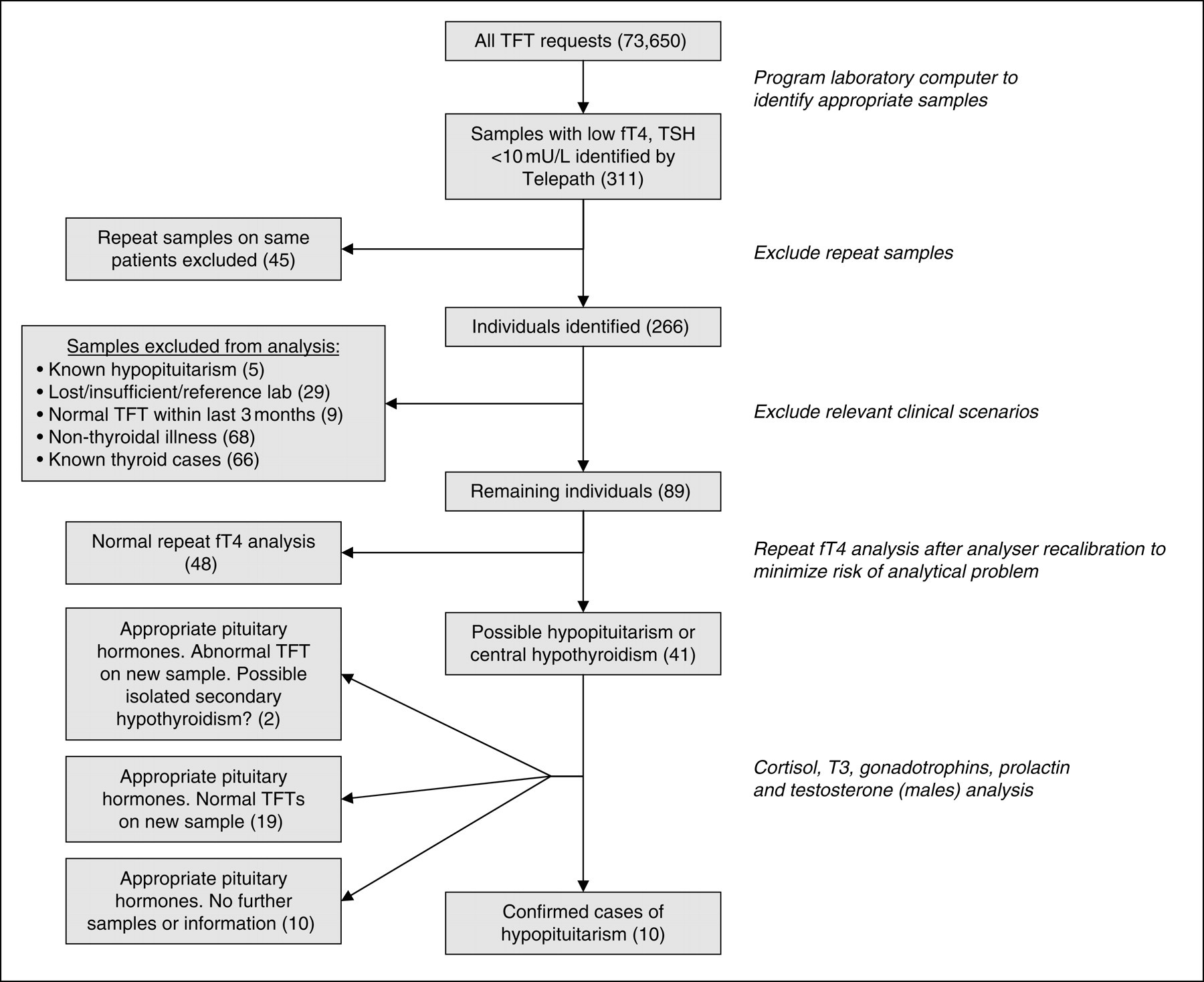
Identification of 10 cases of hypopituitarism from suggestive thyroid function test (TFT) results. fT4, free T4; TSH, thyroid-stimulating hormone
Of the 266 individuals identified, 68 (25.6%) were classified as having non-thyroidal illness, 66 (24.8%) had specific thyroid-related treatment, and samples from 29 (10.9%) individuals were either insufficient for further analysis, had already been forwarded to reference laboratories for initially requested analyses or were no longer traceable. Furthermore, nine (3.4%) individuals had normal TFTs taken within the preceding three months and five (1.9%) were already attending endocrine specialist clinics with relevant diagnoses including hypopituitarism. These samples were excluded from further analysis.
Repeat fT4 analysis was carried out on the same samples taken from the remaining 89 individuals, identified as potentially having hypopituitarism, shortly after assay recalibration. Post-calibration fT4s were, on average, 1.47 pmol/L higher than initial results (95% CI 1.15–1.79 pmol/L; P < 0.0001), increasing from 7.69 to 9.16 pmol/L and leading to normalization of results (fT4 ≥ 9 pmol/L) in 48 cases. These samples were also excluded from further analysis and follow-up samples requested.
The remaining 41 samples with confirmed subnormal fT4 were analysed for tT3, prolactin, gonadotrophins, testosterone (males) and random cortisol, leading to the diagnosis of hypopituitarism in 10 individuals (median age 71.5 years, range 28–90 years; 5 men and 5 women). Immunoassay and imaging results of these 10 individuals are available in Table 1.
Immunoassay and imaging results for 10 individuals diagnosed with hypopituitarism
fT4, free T4; F, female; M, male; TSH, thyroid-stimulating hormone; FSH, follicle-stimulating hormone; LH, luteinizing hormone; MRI, magnetic resonance imaging; CT, computed tomography
*Random samples (taken between 09:00 and 14:30)
†Not applicable
‡Data unavailable, insufficient serum
All 10 individuals were resident in the geographical area covered by Glasgow Royal Infirmary (a population of approximately 200,000). Four individuals were attending outpatient clinics and six had samples taken at their general practitioner (GP) surgeries. None were inpatients and none were acutely unwell or clinically infected at the time of blood sampling. Hypopituitarism was not clinically suspected in any of the cases, as verified by discussion with the relevant clinicians, and there was no suggestion of hypopituitarism in any of the clinical information provided on the biochemistry request forms. This represents an incidence of hypopituitarism of four cases/100,000 adults/year. Nine of the 10 are now attending local specialist endocrine clinics for follow up and one has declined further investigation.
Thus far, 29 of the 48 individuals whose samples produced a normal fT4 on repeat analysis, have had a follow-up sample sent for TFTs as recommended on the results issued. In all 29 cases, fT4 has been within the reference interval (≥9 pmol/L) and therefore, no samples required further analysis according to the Telepath search criteria. There is no further information available regarding the other 19 of these 48 patients. Of the 31 individuals with low-repeat fT4 results (on the initial sample), but appropriate FSH, LH, prolactin, testosterone (in men) and cortisol concentrations, 19 have had further samples sent which have yielded euthyroid results. In these cases, the explanation for the initial abnormal TFTs is not clear. Follow-up samples have not been received for a further 10 of these 31 individuals, and results on follow-up samples for the remaining two patients have again shown low fT4 results suggesting possible isolated central hypothyroidism although no thyrotrophin-releasing hormone (TRH) tests have been performed.
Discussion
Reflective testing led to the diagnosis of non-traumatic hypopituitarism in 10 unsuspected cases, representing an incidence of four cases/100,000 adults/year. Only one of the 10 cases would have been identified if our laboratory offered only TSH as frontline TFT, rather than fT4 and TSH. These findings support the recently published Association for Clinical Biochemistry and British Thyroid Association Guidelines 3 and conclusion of the study by Wardle et al. 2 that a first-line TSH strategy will miss cases of hypopituitarism. That study, carried out in an area with a population of 471,000, concluded that the incidence of hypopituitarism may be somewhat higher than previously quoted. The authors diagnosed 15 cases of hypopituitarism based on TFT results only and during the same year a further 11 patients were diagnosed with hypopituitarism following referral on clinical grounds; this equated to an estimated incidence of hypopituitarism of 3.2 cases/100,000/year based on samples with abnormal TFTs alone and 5.5 cases/100,000/year in total. When compared with other published data on the incidence of hypopituitarism (4.2 cases/100,000 adults/year), 1 our study and that of Wardle et al. 2 suggests that the majority of cases may be identified by recognition of abnormal TFTs with subsequent reflective testing.
Reflective testing implies that the addition of further tests to identify possible hypopituitarism is discretionary, based on the clinical judgement of the reporting biochemist after taking into account the relevant clinical and biochemical information. 15 This contrasts with reflex testing, where additional tests are added automatically. This would have led to testing on a further 222 samples in our study.
The finding that fT4 results were significantly higher when repeated after assay recalibration is consistent with experience that competitive immunoassay performance is worse at low analyte concentrations. The poor precision of the Bayer Centaur fT4 assay (IQC CV 13.5% at 7.6 pmol/L) in particular is evidence of this. We therefore took the pragmatic approach of repeating fT4 analysis in the best available conditions (i.e. following instrument recalibration) before considering additional investigations. Requests for follow-up samples were made when results on the initial TFT analysis and analysis following assay calibration were discrepant. Of the 48 individuals with normal repeat fT4 following instrument recalibration, 29 have had further samples sent for TFT analysis and the biochemistry has been euthyroid in all cases. This supports the decision to only further investigate samples when subnormal fT4 has been confirmed after assay recalibration.
Strengths of our study include its prospective design, description of an easily followed protocol and evidence of improved diagnosis of a condition that is clinically important and often unsuspected. It also highlights an important pattern of TFT results which clinicians should be aware of. Weaknesses of the study include the lack of inclusion of 29 relevant samples and lack of data to compare diagnostic rate with inclusion of low-normal fT4 samples. Furthermore, continued audit will be required to confirm whether the calculated incidence of hypopituitarism diagnosed based on TFTs is indeed accurate or whether it is more closely related to the prevalence of hypopituitarism. We also do not have follow-up data for a number of individuals where suggested resampling after three months has not occurred. While we recognize that acute illness may have a significant impact on both gonadotrophins in postmenopausal women 16 and TFTs, 17,18 these conditions were excluded as far as possible and there was no evidence of acute illness in any of the 10 individuals identified. It is important to exclude acute illness as it is clear that the diagnosis of hypopituitarism based on reflective testing is often based on low gonadotrophin concentrations in postmenopausal women. Samples of the 10 individuals diagnosed with hypopituitarism were taken at different times of the day and the addition of random cortisol assays did not prove useful in case identification.
Accurate measurement at low fT4 concentrations is required to limit the number of samples identified for further investigation. An assay with high random analytical error (i.e. precision) or significant negative systematic error (i.e. bias) will increase the number of samples identified at the lower reference interval limit (fT4 < 9 pmol/L) leading to a higher number of false-positives. We found a significant change in assay performance over time following fT4 assay recalibration. This indicated that search criteria were less likely to identify false-positives shortly after assay recalibration. The fT4 value chosen for search criteria is debatable. All 10 individuals identified in our study and 12 out of 17 from the study of Wardle et al. 2 were outpatients with low fT4s and TSH concentrations comfortably within the reference range. This is in contrast to acutely presenting cases of pituitary apoplexy, where low TSH and low-normal fT4 may occur. 19 Loosening of the search criteria to include such cases (by including low-normal fT4) will lead to a significant increase in the number of samples identified, increasing the false-positive rate and weakening the positive predictive value of further testing.
The number of tests needed to obtain a diagnosis (NND), as described by Paterson, 20 is 31.5 in this study, comparable with the NND of 18.8 for diagnosing hereditary haemochromatosis in that study. This represents an acceptably low level of additional testing in the context of a serious diagnosis. The cost for all the additional analyses was approximately £1050 (£105 for each new case of hypopituitarism).
Addition of further analyses by the reporting biochemist without the express consent of the patient, based on TFT results and clinical information, raises ethical questions about reflective testing. For example, the diagnosis of a previously unsuspected pituitary lesion or tumour, particularly in a very elderly patient, may be unwelcome. However, a recent study found that the vast majority of patients favour reflective testing by laboratory professionals, above simply highlighting an abnormality and recommending further tests, if it is likely to be in their best interest. 21
In summary therefore, review of suggestive TFT results with appropriate reflective testing appears to constitute a reasonable opportunistic screening test for hypopituitarism. While the published thyroid guidelines 3 do point out the likely pattern of TFTs in central hypothyroidism and suggest that referral to an endocrinologist for further pituitary function testing (prolactin, FSH, LH, ACTH/cortisol) may be required to make a diagnosis of hypopituitarism, they do not suggest laboratory initiated reflective testing. We recommend that biochemistry laboratories should have a formal standard operating procedure in place for further investigations of abnormal TFT results. Further prospective studies are required to evaluate any change in patient outcome and cost-effectiveness of earlier diagnosis of hypopituitarism identified from abnormal TFT results.
Footnotes
Acknowledgements
Many thanks to Prof Naveed Sattar and Karen Smith for their helpful comments on the manuscript. We are also grateful to Celia Carroll, Biomedical Scientist at Glasgow Royal Infirmary, for sample analyses and to Kevin Deans, Specialist Registrar, for assisting with sample collection.
