Abstract
Abstract
Background
D-lactate is essentially a product of bacterial metabolism, and its assessment in plasma has been mainly used to diagnose D-lactic acidosis in patients with short bowel syndrome. In the last few years, there has been growing interest in the use of subclinical elevations of D-lactate concentrations as a diagnostic tool in a variety of clinical conditions such as ischaemia, trauma or infection.
Methods
An endpoint enzymatic spectrophotometric assay to measure plasma D-lactate with a sample blank correction was validated on our routine clinical chemistry analyser (Olympus AU640). An ultrafiltration procedure was used in samples with a high L-lactate dehydrogenase (L-LDH) activity in order to avoid underestimation of the D-lactate concentration, when a sample blank was processed.
Results
The intra- and inter-assay imprecision were <5.1% and <13.3%, respectively and the mean recovery for the D-lactate assay was 95% (range 88–103%). Samples with L-LDH activity greater than 1500 IU/L required the use of ultrafiltration devices. Plasma D-lactate concentration in our ‘non-diseased’ paediatric population showed a non-Gaussian distribution – 95th percentile equal to 19 μmol/L – and no difference based on gender or age was observed.
Conclusion
We have established an accurate, sensitive and precise routine assay for D-lactate measurement in plasma. The assay was used to formulate paediatric reference ranges and will be used to assist clinicians to evaluate ‘D-lactate toxicity’ in patients with a variety of conditions such as short bowel syndrome, small bowel transplantation and as an early marker of intestinal ischaemia.
Introduction
Clinical significance
Patients with short bowel syndrome may present with high plasma concentrations of D-lactate (4000–10,000 μmol/L) 1 due to bacterial fermentation of unabsorbed carbohydrates in the colon and absorption of this metabolite from the intestinal lumen. The clinical presentation of the ‘D-lactic acidosis syndrome’ is characterized by episodes of encephalopathy and metabolic acidosis. Recognition is important to avoid further unnecessary investigations and to prompt treatment with restricted carbohydrate intake and metronidazole. 2 Various pathogenic bacteria such as Bacteroides fragilis, Escherichia coli, Klebsiella pneumonia and Staphylococcus aureus can produce D-lactate and its measurement in pleural, ascitic, synovial and cerebrospinal fluid might be useful for the early diagnosis of bacterial infection. 3 Several articles have suggested that subclinical elevations of plasma D-lactate concentration may be of value as a clinical diagnostic tool in a variety of disorders: (1) D-lactate might contribute to the development of osteomalacia in patients with malabsorption receiving long-term parenteral nutrition; 4 (2) Measurement of plasma D-lactate concentration has a lower false-negative rate than C-reactive protein or leukocyte count when used as a marker in the diagnosis of appendicitis; 5 (3) Serum D-lactate concentration is increased in critically sick patients with septic shock; 6 (4) Plasma D-lactate is a reliable marker of colonic ischaemia and a very sensitive marker for gut failure; 7 (5) Plasma and urine D-lactate might be used as a biochemical marker to support the diagnosis of neonates suspected of necrotizing enterocolitis (NEC); 8 (6) Plasma D-lactate and urine excretion of D-lactate are markedly increased in triosephosphate isomerase deficiency and can be used to support its diagnosis. 9
Production of D-lactate, metabolism and excretion
D-lactate is usually present in human blood at very low concentrations as a product of methylglyoxal metabolism, which is produced in small amounts from fat, protein and carbohydrate metabolism. D-lactate is also produced by bacteria of the gastrointestinal tract and is absorbed in the small intestine and colon. D-lactate is thought to be metabolized in the liver to pyruvate by the enzyme D-α-hydroxy acid dehydrogenase at one-fifth the rate at which L-lactate dehydrogenase (L-LDH) metabolizes L-lactate. 10 About 10% of D-lactate is excreted in the urine and it has been reported that L-lactate interferes in its renal reabsorption. 11 Thus, metabolism and excretion of D-lactate and L-lactate are closely related.
Assays to measure D-lactate
Several methods have been reported to measure D-lactate in blood, plasma, urine or biological fluids. 12–15 The most widely accepted method is based on conversion of D-lactate to pyruvate in the presence of nicotinamide-adenine dinucleotide (NAD+) and D-lactate dehydrogenase (D-LDH) and monitored by a spectrophotometric technique. The main problem with these assays is the non-specific transformation of NAD+ to NADH that results in an overestimation of D-lactate measurement. Some solutions have been proposed: (1) Pre-incubation of the sample with all the reagents except D-LDH. This approach is difficult to perform in a routine clinical chemistry analyser where the timing of adding sample and reagents is fixed by the instrument; (2) Sample deproteinization with percloric acid to make the sample protein-free, removing the oxido-reductases present in the sample. This approach requires a further step of alkalinization that results in a sample dilution which makes it difficult to measure D-lactate at physiological concentrations; (3) Performance of a sample blank and subtraction from the total absorbance. This approach is probably the most convenient with plasma samples, but increasing concentrations of L-lactate and L-LDH in the sample makes this subtraction inappropriate resulting in an underestimation of the D-lactate concentration; (4) Sample deproteinization using an ultrafiltration procedure. This approach removes the main interferences in the sample and avoids sample dilution.
Material and methods
Reagents
A D-lactate kit from Megazyme International (Ireland Ltd, Ireland) was used to set up the method on our routine clinical chemistry analyser. A standard preparation of D-lactate was purchased from Sigma Chemical Co (St Louis, MO, USA) and was used to spike the serum/plasma pools used in further experiments. Centrifugal filter devices (Microcon YM-10, nominal molecular weight limit [NMWL] = 10,000) from Millipore Corporation (MA, USA) were used to make samples protein-free.
Sample requirements
One hundred microlitres of serum or plasma from blood collected into lithium heparin or fluoride/oxalate was required for the analysis. When the ultrafiltration procedure was necessary, the minimum volume of serum or plasma required to perform the analysis was 300 μL.
D-lactate assay
The quantification of serum/plasma or ultrafiltrate D-lactate requires two coupled reactions (Figure 1). In the first reaction, D-lactate is oxidized to pyruvate by NAD+ in the presence of the enzyme D-LDH. A second reaction is needed to shift the equilibrium to the formation of pyruvate and NADH. This is achieved by the conversion of pyruvate to D-alanine and 2-oxoglutarate, catalysed by the enzyme D-alanine-amino-transferase (D-ALT) in the presence of an excess of D-glutamate. The amount of NADH formed during the reaction is stoichiometric to the amount of D-lactate in the sample, and it is measured by the increase in absorbance at 340 nm. A second wavelength (700 nm) was used to correct small variations of the lamp intensity during the monitoring of the chemical reaction. The method was automated on our routine chemistry analyser using a two-reagent format with two channels linked by the analyser software. The first channel was used to measure the plasma D-lactate concentration in the sample plus the non-specific transformation of NAD+ to NADH, and the second channel was used to perform a sample blank that accounts for the non-specific transformation of NAD+ to NADH. The ratio of final volume to sample volume was 13.5:1 and the final concentrations of the reagents were as follows: NAD+ (4.19 mmol/L), glycylglycine buffer (104 mmol/L, pH 10.0), D-glutamate (104 mmol/L), D-ALT (11.3 KU/L). D-LDH (EC 1.1.1.28, from Escherichia coli) suspension (1250 KU/L). Chemical reaction sequence for the measurement of D-lactate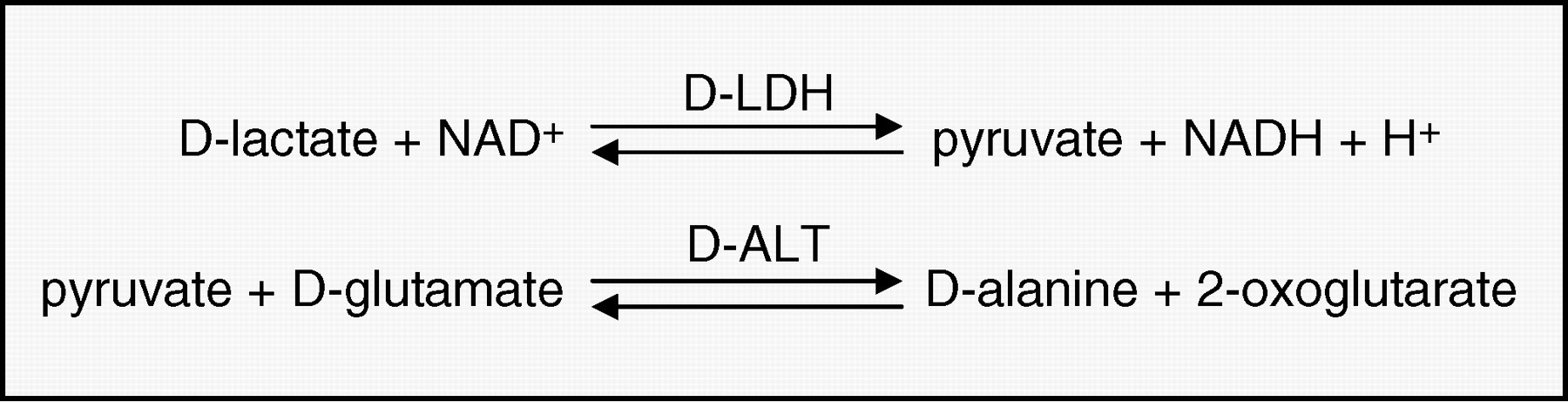
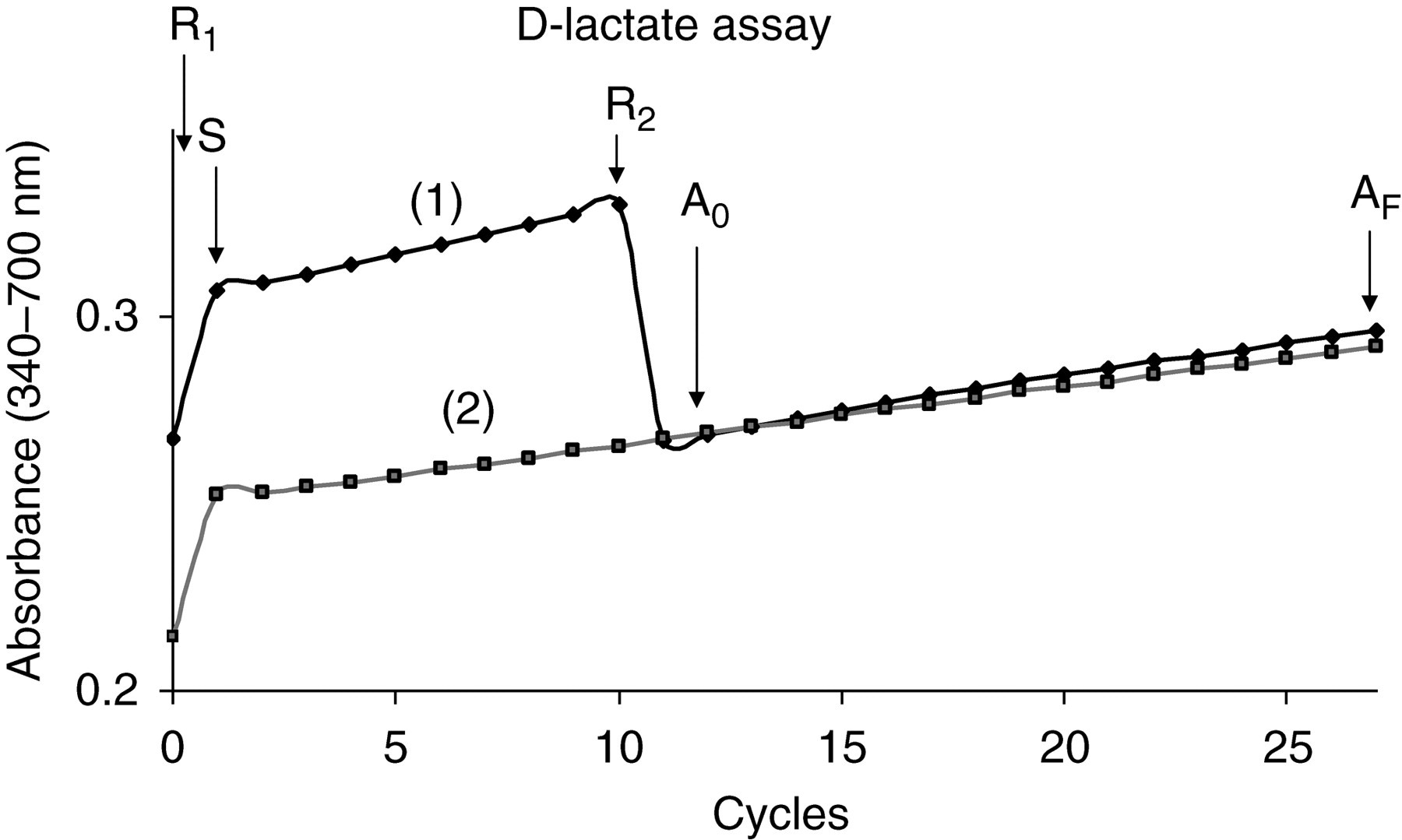
Monitor reaction of the D-lactate assay. The reagent 1 (R1) was added at t = 0, the sample (S) 20 s later and the reaction started after adding the reagent 2 (R2; D-LDH). The initial absorbance (A0) and final absorbance (AF) were measured respectively 20 s and 5 min after the reagent 2 was added. The sample blank was performed using the same analytical conditions but without adding the enzyme D-LDH (R2). The reagent blanks for both channels have been omitted for clarity; (1) D-lactate channel; (2) sample blank channel
Analytical performance
Two patient serum pools were prepared to study the precision, linearity and recovery of the D-lactate assay. The pool used to assess the intra- and inter-assay imprecision was spiked with D-lactate at a final concentration of 46 and 91 μmol/L plus the endogenous D-lactate present in the pool. The inter-assay imprecision was assessed by measuring aliquots of the pool for 18 days and included seven calibration curves. The pool used to study the linearity and recovery was spiked with D-lactate over the range of 60–1800 μmol/L. The linearity and recovery experiments were done in triplicate using new reagent and calibration curve each time.
Sample stability
Three pools of plasma from blood collected on lithium heparin, plasma from blood collected on fluoride/oxalate and serum, were made with spare patients' samples. D-lactate was added at a final concentration of 240 μmol/L plus the endogenous D-lactate present in the pools. Aliquots of these pools were kept at 4°C and room temperature (22°C) for 0 h, 12 h, 24 h and 48 h, and then frozen. These samples were analysed together in the same batch to avoid reagent and calibration variability.
Ultrafiltration procedure
In samples with a high activity of L-LDH and in the presence of L-lactate, the use of a sample blank correction leads to under-estimation of plasma D-lactate concentrations due to the differences in the affinity of L-LDH and D-LDH for the co-factor NAD+. Under these circumstances, 300 μL of sample has to be ultrafiltered centrifuging at 10,000
Reference ranges
Spare plasma samples, collected over a six-month period from patients seen at Birmingham Children's Hospital, were used to formulate reference intervals for plasma D-lactate in our paediatric population. Inclusion criteria were applied based on: (1) samples from patients seen in our outpatient department or from the community; (2) no clinics or ward attendance during the previous and following 21 days of sample collection; (3) no biochemical abnormality in the liver or kidney profile, and C-reactive protein lower than 10 mg/L. The samples were batched and analysed on three different days to include reagent and calibration variability.
Results
Analytical performance
The intra-assay imprecision at D-lactate concentrations of 17, 60 and 96 μmol/L was 5.1, 3.1 and 2.6%, respectively, and the inter-assay imprecision at D-lactate concentrations of 20, 64 and 105 μmol/L was 13.3, 6.9 and 5.4%. The inter-assay imprecision of the D-lactate assay when the ultrafiltration procedure was used at D-lactate concentrations of 27, 81, 130 μmol/L was 10.6, 7.0 and 4.8%, respectively. The mean recovery for the D-lactate assay (Figure 3a) was 95% (range 88–103%) and for the ultrafiltration procedure (Figure 3b) it was 111% (range 102–116%). The assay was linear up to 1800 µmol/L.
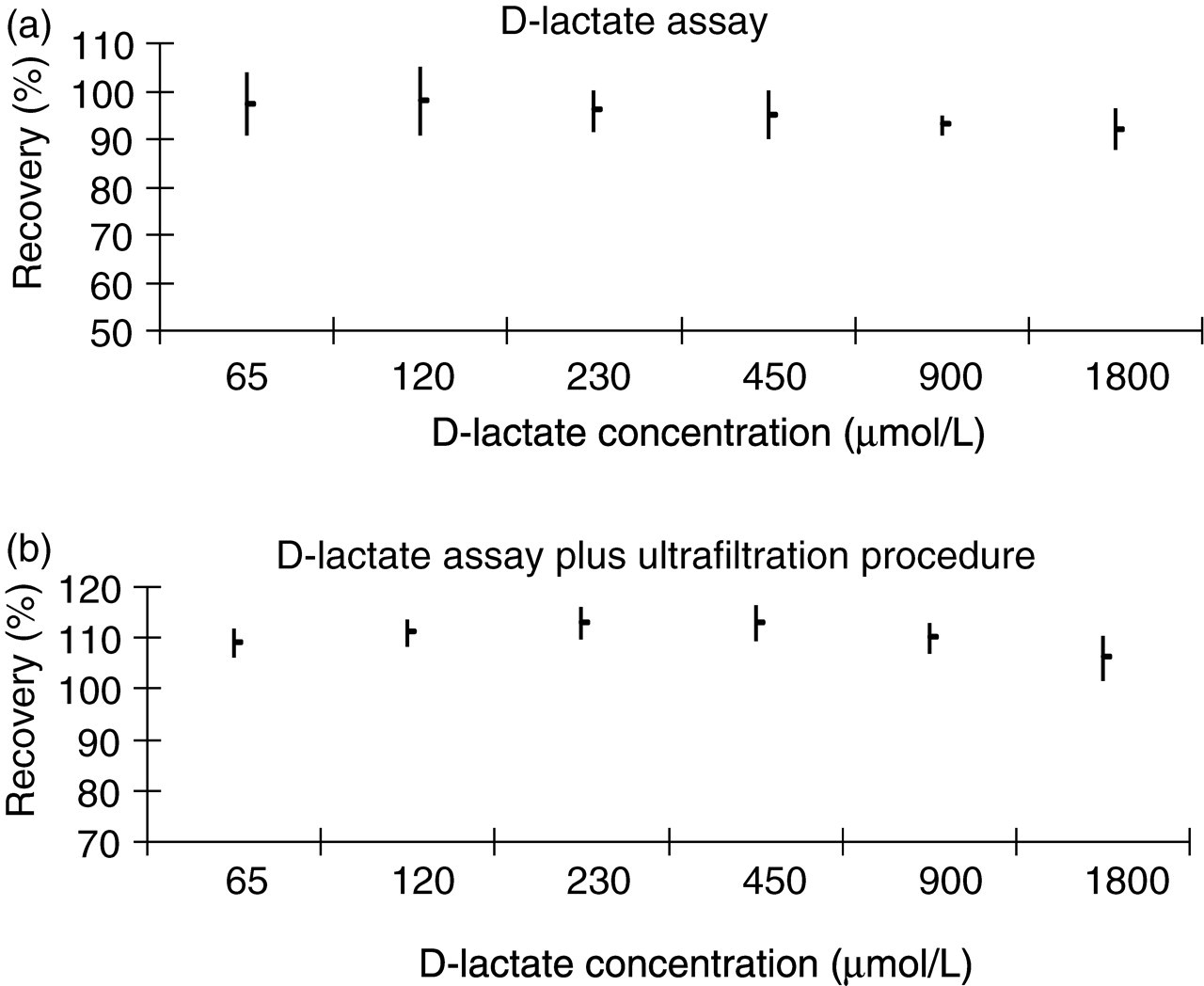
Recovery experiment. (a) D-lactate assay, (b) D-lactate assay plus ultrafiltration procedure. The points represent the mean recovery for serum D-lactate and the lines the standard deviation of the recovery (n = 3)
Sample storage and stability
No differences in D-lactate concentrations (<3% compared with the concentration of D-lactate at 0 h) were observed when serum/plasma was kept at 4°C or room temperature (22°C) for up to 48 h.
Interference by L-lactate dehydrogenase and L-lactate
The recovery of the D-lactate assay, in the presence of a constant concentration of L-lactate, decreases with increasing activities of L-LDH (Figure 4). The recovery was calculated against the D-lactate concentration on an aliquot of the pool not spiked with L-LDH. This effect was not observed in the plasma ultrafiltrate, as the majority of the enzyme was removed. The results for the experiment are summarized in Table 1. Table 1A illustrates the effect of increasing activities of L-LDH on the measurement of plasma D-lactate. The plasma D-lactate ranged from 341 μmol/L (100% recovery) when the L-LDH activity was 284 U/L to 154 μmol/L (45% recovery) when the L-LDH activity was 10,266 U/L. The plasma D-lactate concentration was calculated by subtracting the concentration of the sample blank channel (SBC) that accounts only for the non-specific transformation of NAD+ to NADH from the concentration of the D-lactate channel (D-LC) that measures both the D-lactate in the sample plus the non-specific transformation. Table 1B shows a non-significant effect in the recovery of plasma D-lactate with increasing activities of L-LDH when the ultrafiltration procedure was performed before the analysis. The sample blank was always below 50 μmol/L indicating that the interference was removed with the ultrafiltration procedure and the recovery of plasma D-lactate was greater than 90% for all experimental values.
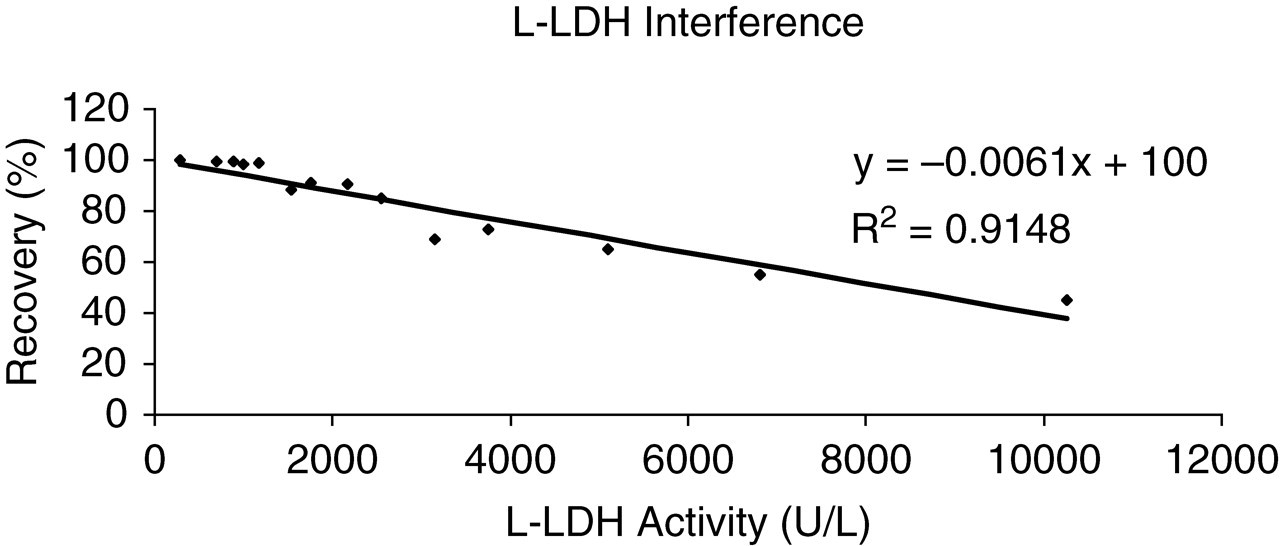
Effect of increasing activities of L-lactate dehydrogenase (L-LDH) on the recovery of plasma D-lactate concentration. The plasma L-lactate concentration was 4.0 mmol/L
Summary of the L-lactate dehydrogenase (L-LDH) interference experiment. (A) D-lactate assay, (B) D-lactate assay plus ultrafiltration procedure
Part of the original data has been omitted for clarity
D-LC, D-lactate channel; SBC, sample blank channel
D-LC, SBC and D-lactate are expressed in μmol/L
Reference ranges for plasma D-lactate
The results obtained for our ‘non-diseased’ paediatric population have been summarized in Table 2. No difference based on gender or age was observed when using the analysis of variance between groups. Due to the small number of data for patients below three years of age, the statistical analysis was done by splitting the population into two groups (0.4–12 years and 12–17 years, respectively). The D-lactate concentration showed a non-Gaussian distribution and the upper limit of normal was calculated based on the 95th percentile of the distribution and was found to be 19 μmol/L. All the original data are presented in Table 2 and was used to calculate the means and standard deviations.
Distribution of plasma D-lactate concentrations (μmol/L) in our hospital-based paediatric population
Discussion
Analytical performance
The assay of plasma D-lactate shows a good analytical performance and is able to assist clinicians in a variety of disorders where D-lactate might be elevated. The low inter-assay imprecision at a concentration of 105 μmol/L (5.4%) allows effective monitoring of patients with mild elevations of plasma D-lactate. The assay was linear up to 1800 μmol/L and an automatic dilution was programmed for samples with a D-lactate concentration greater than this value. This dilution extended the dynamic range up to 18,000 μmol/L, making this assay useful for either, a variety of conditions where a marginal increase of plasma D-lactate might occur such as, in patients with septic shock where the mean D-lactate concentration after 24 h of admission for non-survivors was 240 μmol/L, 6 or for conditions such as, ‘D-lactic acidosis syndrome’ where the plasma D-lactate concentration might be as high as 8000 μmol/L. 16
The mean recovery for the D-lactate assay (95%) over the range of plasma D-lactate concentration studied (50–1800 μmol/L) permits its use as a routine procedure for the quantification of D-lactate. The mean recovery for the D-lactate assay when the ultrafiltration procedure was used (111%) was acceptable and reflected a small concentration of the solutes of lower molecular weight in the ultrafiltrate. If results from both methods need to be compared, a factor of 0.84 can be applied to the D-lactate results obtained by the ultrafiltration procedure, to take into account the difference in recovery of both methods.
The sample volume could be a problem in some circumstances when the ultrafiltration is needed (300 μl), particularly when dealing with paediatric samples. However, the number of samples requiring ultrafiltration will be minimal and in these situations a sample dilution up to four times would be possible to assess mild elevations of D-lactate ( >200 μmol/L).
Serum or plasma samples collected on lithium heparin or oxalate/fluoride were stable at room temperature for at least 48 h after centrifugation and separation from cells, making them suitable for transport if samples from other hospitals need to be analysed.
Sample blanking and ultrafiltration procedure
The use of a sample blank is mandatory in order to avoid an overestimation of the D-lactate concentration due to the non-specific transformation of NAD+ to NADH due to the presence of oxido-reductases in the sample. Table 1 shows the importance of blanking the sample. The sample blank concentrations ranged from 52–935 μmol/L and the concentrations of plasma D-lactate if the sample blank is not performed would range from 393–1089 μmol/L; up to 300% increase compared with D-lactate concentration in this pool (360 μmol/L). This effect will be particularly important when the assay is used to assess mild elevations of D-lactate. Therefore, a sample blank was performed in all samples and was subtracted to calculate the final concentration of D-lactate.
On the other hand, when the activity of L-LDH in the sample was greatly elevated, the use of a sample blank leads to an underestimation of the plasma D-lactate. Figure 4 shows how increasing activities of L-LDH affect the assessment of plasma D-lactate concentration. The linear regression of the points plotted in the graph and set to intercept the 100% recovery, illustrates an average decrease of 0.6% in recovery for every 100 U/L of the enzyme L-LDH. Table 1A shows that with increasing activities of L-LDH (284–10,266 U/L), the SBC increases in concentration to a greater extent than the D-LC does. This fact points out that the slopes for the sample blanks are greater than for the samples and explains the origin of the underestimation in the assessment of plasma D-lactate when a sample blank is processed. The explanation of this situation was described in detail by Marti et al. 12 The Michaelis constant (KM) of D-LDH for NAD+ was reported to be 17-fold higher than of L-LDH indicating that the former has a lower affinity for NAD+. This difference in affinity of D-LDH and L-LDH for NAD+ explains why the slopes of the samples are lower than their respective sample blanks and explains also why the recovery decreases with increasing activities of L-LDH. In our assay, the final D-LDH in the reaction vessel is 250,000 U/L and most of the co-factor NAD+ is bound to D-LDH, which is clearly present in excess. Under these circumstances the oxidation of L-lactate catalysed by L-LDH is limited by the presence of the co-factor NAD+. When a sample blank is performed no D-LDH is present in the final reaction and the limiting factor for the chemical reaction is not the co-factor NAD+ but the concentration of L-lactate in the sample. This is the rationale because, the contribution to the non-specific transformation of NAD+ to NADH due to the effect of L-LDH and L-lactate is less important in the D-LC than in the SBC. This discrepancy is more significant when the activity of L-LDH is greater because its contribution to the final signal is also more important. Table 3 shows the importance of this effect in a patient who was biochemically monitored for several days. Six samples were sent to our laboratory with L-LDH activities and L-lactate concentrations ranging respectively from 1638 to 7351 U/L and from 1.16 to 3.55 mmol/L. The D-lactate calculated using a sample blank gave negative results in six out of seven samples making it impossible to assess the plasma D-lactate in this patient. Even the positive result obtained was clearly lower than the one obtained with the ultrafiltration procedure. When the ultrafiltration procedure was performed before analysis all samples provided a positive result and it was possible to assess the plasma D-lactate on the patient.
Assessment of serum D-lactate in a patient with increasing activities of L-lactate dehydrogenase (L-LDH)
D-LC, D-lactate channel; SBC, sample blank channel; D-LU, D-lactate ultrafiltration
D-LC, SBC, D-lactate and D-LU are expressed in μmol/L. L-lactate is expressed in mmol/L
The value of L-LDH activity above which it is advisable to use the ultrafiltration device was calculated by linear regression of the recovery against the L-LDH activity, and it was estimated to be 1500 IU/L. Below this value the recovery was greater than 90%, and we concluded it was not necessary to use the ultrafiltration procedure. If the concentration of L-lactate was greater than 4.0 mmol/L, the interference of L-LDH in the assessment of D-lactate might be important even at lower L-LDH activities. Fortunately, plasma samples with L-LDH greater than 1500 U/L and L-lactate greater than 4.0 mmol/L will be minimal and the use of the ultrafiltration devices will not usually be required. A different situation would be the assessment of D-lactate in biological fluids such as pleural or ascitic fluids where the activity of L-LDH and the concentration of L-lactate are frequently elevated. In this situation the use of the ultrafiltration procedure would be necessary in most of the samples. In patients with acute liver failure and severe cell damage, the release of NADH dehydrogenases from the mitochondria into the circulation could potentially affect our assay and lead to an underestimation of plasma D-lactate concentration when blanking the sample. However, the group of patients to whom the assay will be addressed, rarely present with acute liver failure. We propose the following algorithm (Figure 5) to process the samples arriving at our laboratory for the assessment of plasma D-lactate.
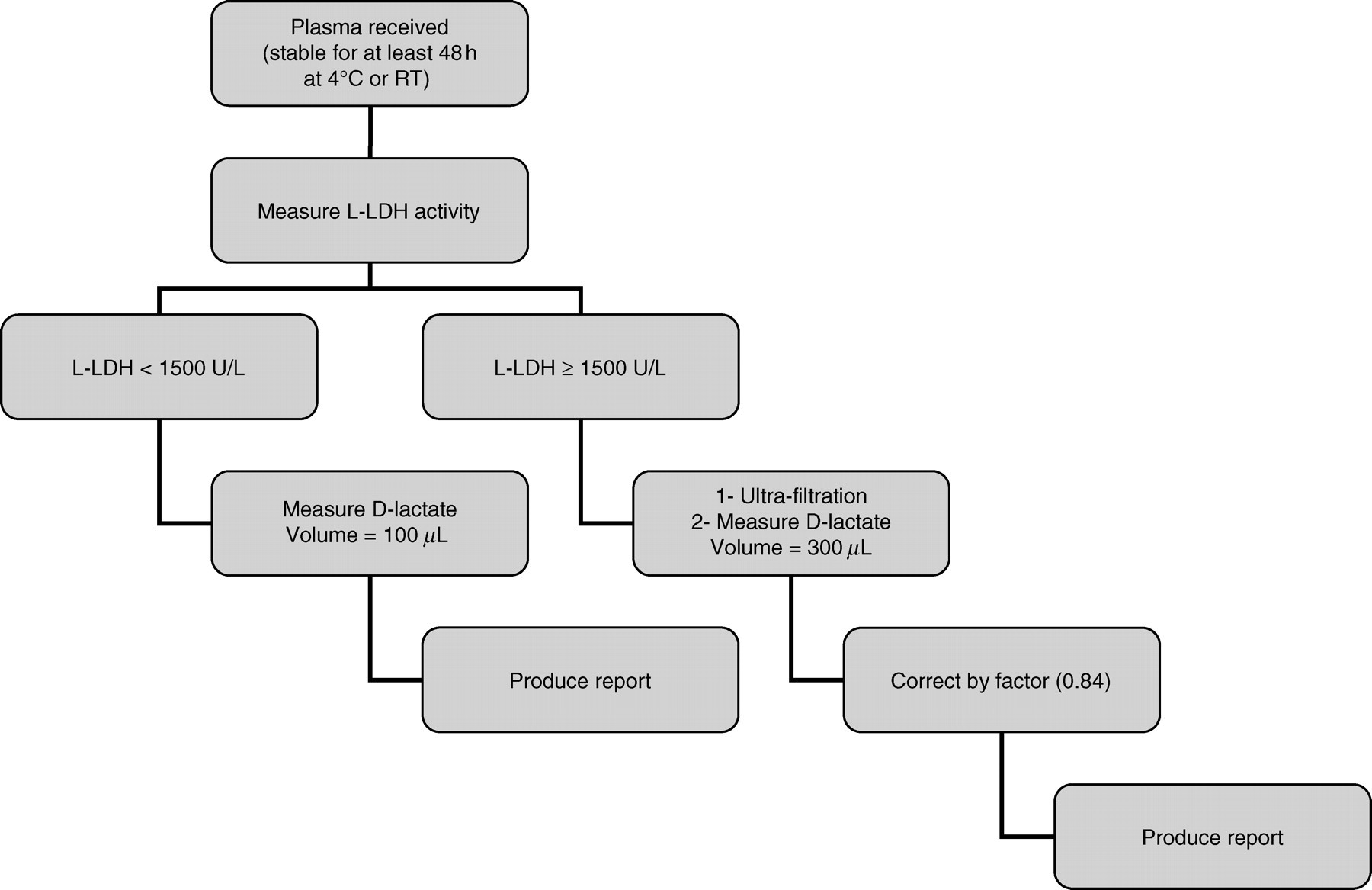
Proposed algorithm to process samples arriving at our laboratory for assessment of D-lactate
Reference ranges
Several reference values have been reported for plasma D-lactate in healthy individuals ranging from 11–70 nmol/L. 10 The mean D-lactate in human blood from healthy donors has been reported to range from 10 μmol/L using an endpoint enzymatic fluorescence method to 30 μmol/L using a high-performance liquid chromatography method with absorbance detection. 13 In both cases, the numbers of healthy individuals tested were six and seven, respectively. In a recent case report of D-lactic acidosis secondary to short bowel syndrome in a young boy the normal reference range for plasma D-lactate concentration was stated to be lower than 200 μmol/L. 17 This disparity encouraged us to formulate reference values for plasma D-lactate for our paediatric population based on our assay. The knowledge of reference values for plasma D-lactate can be informative in those situations where D-lactate might be moderately elevated such as in patients suspected of intestinal ischaemia and gut failure, for the diagnosis of appendicitis or in patients suspected of NEC. However, in order to study the usefulness of the plasma D-lactate as a biochemical marker in these conditions, it will be necessary to study the diagnostic sensitivity and specificity of this marker using appropriate control groups of ‘diseased patients’ who could present a clinical challenge for the diagnosis of the conditions cited above. On the other hand, the establishment of a suitable reference population is important to formulate reference values, but it is difficult for ethical reasons and in general is time consuming. The use of hospital or primary health-care-based population to formulate reference values has been reported in the literature. 18 We are aware of the limitations of its use, such as including a relatively large number of diseased patients. Thus, the application of certain inclusion/exclusion criteria to select ‘non-diseased individuals’ is essential. The first selection criterion to identify ‘non-diseased individuals’ was the use of patients from our outpatient department or from the community. This criterion allowed us to select, within our hospital paediatric population, those more likely to be ‘healthy’. The second inclusion criteria based on no clinics or ward attendance during the previous and following 21 days permitted the exclusion of patients expected to be sick. We believed that ill patients would be admitted to hospital or at least would probably be seen again in the next few days. The last criterion of no abnormality in the liver or kidney profile and low C-reactive protein allowed us to exclude patients with disorders in these organs as both are involved in the metabolism and excretion of D-lactate and to exclude patients likely to have a systemic infection/inflammation. These exclusion/inclusion criteria together with the inclusion of a ‘relatively high’ number of patients (n = 126) make this approach useful to formulate reference values in our paediatric population for plasma D-lactate. The mean of the plasma D-lactate was 11 μmol/L and the upper limit of the reference range was calculated using the 95th percentile of the distribution and resulted to be 19 μmol/L. The distribution of plasma D-lactate concentration showed a non-Gaussian distribution probably due to the likely inclusion of some ‘non-healthy’ individuals. No statistical difference based on gender or age was observed when plasma D-lactate means were compared.
We have not verified our results against a reference method for D-lactate and a bias in our assay could be possible. However, this bias cannot be substantial as the mean D-lactate concentration for our paediatric population is very close to previous reports 13 and the recovery experiment showed a good performance over the range studied.
Conclusion
In summary, we offer a reliable and automated method for the assessment of serum/plasma D-lactate concentration and with a further minimal validation its use might be extended to other biological fluids such as pleural, peritoneal or urine samples. The assay was used to formulate paediatric reference ranges – 95th percentile of plasma D-lactate was equal to 19 μmol/L – and will be used to assist clinicians to evaluate ‘D-lactate toxicity’ in patients with a variety of clinical conditions.
Footnotes
ACKNOWLEDGEMENTS
We acknowledge Birmingham Children's Hospital Charities for their award of a grant to establish an assay to measure plasma D-lactate. We also acknowledge Dr George Gray from Birmingham Children's Hospital and Mr Craig Webster from Birmingham Heartlands Hospital for reviewing the manuscript and for their constructive comments. A special thanks also to Dr Segura from Vall d'Hebron Hospital (Barcelona) for her comments about D-lactate assays.
