Abstract
Abstract
The lowest observed adverse effect level for vitamin D, said to cause hypercalcaemia in normal adults, is officially 95 µg/day. Serum 25-hydroxyvitamin D (25[OH]D) concentrations associated with hypervitaminosis D remain undefined. Reported 25(OH)D concentrations resulting from prolonged excessive vitamin D3 intakes have exceeded 700 nmol/L. We report self-prescribed high dose of vitamin D3 over 5–6 years by two men. Subject 1 had been taking 100 μg/day for 3 years followed by 3 years of 200 μg/day. Serum 25(OH)D concentrations averaged 130 nmol/L while taking 100 μg/day of vitamin D3. While taking 200 μg/day of vitamin D3, mean serum 25(OH)D concentrations were 260 nmol/L with no hypercalcaemia or hypercalcuria over the 6 years of vitamin D3 intake. Subject 2 was a 39-year-old man diagnosed with multiple sclerosis. He initiated his own dose-escalation schedule. His vitamin D3 intake increased from 200 to 2200 μg/day over 4 years. The first evidence of a potential adverse effect was that urinary calcium:creatinine ratios showed an increasing trend, which preceded serum calcium concentrations above the reference range (2.2–2.6 mmol/L). His serum 25(OH)D concentration was 1126 nmol/L when total serum calcium reached 2.63 mmol/L. He stopped vitamin D3 supplementation at this point. Two months later, all biochemistry values were within reference ranges; serum 25(OH)D concentrations fell by about one-half, to 656 nmol/L. These results help to clarify the human response to higher intakes of vitamin D3. Close monitoring of biochemical responses confirmed that an increase in urinary calcium:creatinine ratio precedes hypercalcaemia as serum 25(OH)D concentrations rise.
Introduction
The relationship between serum 25-hydroxyvitamin D (25[OH]D) concentrations and adverse effects in humans remains poorly characterized. Excessive intakes of vitamin D are evident by hypercalcaemia, dehydration and tissue calcification. 1–3 Serum 25(OH)D concentrations causing adverse changes in calcium homeostasis, i.e. vitamin D intoxication, have been reported and exceed 500 nmol/L, 1,2,4 but these reports were isolated and not characterized by longitudinal follow-up during vitamin D3 intake.
The upper limit or guidance concentration for public safety are conservative at 25 μg/day in the UK 5 and 50 μg/day in the USA and Canada. 6 A recent review of the literature has advocated the use of 20–25 μg/day 7 of vitamin D to produce 25(OH)D concentrations >75 nmol/L required for skeletal health and fracture prevention. 8 Recent studies have demonstrated the safety of doses much higher, in the range of 100 μg (4000 IU)/day 2 and 250 μg (10,000 IU)/day. 9 Some have proposed that adults require 50 μg (2000 IU)/day to ensure adequate serum concentrations and to avoid deficiency. 10
The great majority of reported vitamin D toxicity cases are a result of overdosing with vitamin D2. Inadvertent excessive use of pharmaceutical doses of vitamin D is the most common cause of vitamin D toxicity. Only a few reports of vitamin D intoxication involve vitamin D3. In one study of eight patients, mean 25(OH)D concentrations were 731 nmol/L and the culprit, overdosed milk, contained 6146 μg (245,840 IU)/L of vitamin D3. 4 Contamination of table sugar with vitamin D3 has also resulted in vitamin D3 intoxication. A father and son displayed 25(OH)D concentrations of 1555 and 3700 nmol/L, respectively. 3 Investigation found that 1 g of table sugar contained 21.4 mg of vitamin D3. It was estimated that the father and son had been consuming 1.3 g of vitamin D3 per month for 7 months, or 42,000 μg/day. 3 A family suffered vitamin D intoxication as a result of unintentional cooking with a veterinary vitamin D3 concentrate, which contained cholecalciferol in peanut oil at 50,000 μg/g or 2,000,000 IU/g of oil. 1 The mean 25(OH)D concentrations of the 11 family members was 1162 ± 247 nmol/L. 1 Collectively, these results indicate that serum 25(OH)D concentrations need to exceed 700 nmol/L before vitamin D3 toxicity becomes evident. Here, we report on two men whose history support these data concerning toxicity of vitamin D3.
Methods
Two male subjects had obtained vitamin D3 from undisclosed sources and they had arranged for measurements of their own biochemical responses over time. After approval by the Mount Sinai Hospital Research Ethics Board, both subjects consented to a review and publication of their laboratory data.
Results were obtained from computer files of laboratory data. Serum and urine calcium and creatinine, as well as serum or plasma phosphate and parathyroid hormone (PTH) had been measured using the Roche Cobas Integra system (Basel, Switzerland). Serum 25(OH)D concentrations had been measured by the DiaSorin radioimmunoassay (Stillwater, MN, USA) method, with which samples showing high concentrations were diluted to ensure accuracy, by bringing the analysed concentration into the range of the calibration curve.
Case 1
Subject 1, a 56-year-old man of normal body mass index (BMI), reportedly healthy according to the last data obtained. He reported no intake of calcium but consumed about two glasses of milk daily along with other dairy products. He began taking vitamin D3 (cholecalciferol) in November 1999 at 100 μg (4000 IU)/day and in August 2002, he doubled this intake and has been taking 200 μg (8000 IU)/day of vitamin D3 to date. There were no changes in either serum or urinary measures of calcium, which remained within reference ranges (2.2–2.6 mmol/L and <1.0, respectively) (Figure 1). While taking 100 μg/day of vitamin D3, 25(OH)D concentrations averaged 130 nmol/L (range 101–176 nmol/L). With 200 μg/day of vitamin D3, 25(OH)D concentrations averaged 260 nmol/L (range 189–485 nmol/L). The high 25(OH)D concentration observed in June 2003 may be an anomaly, possibly due to analytical error; however, we did not have access to the sample for retesting.
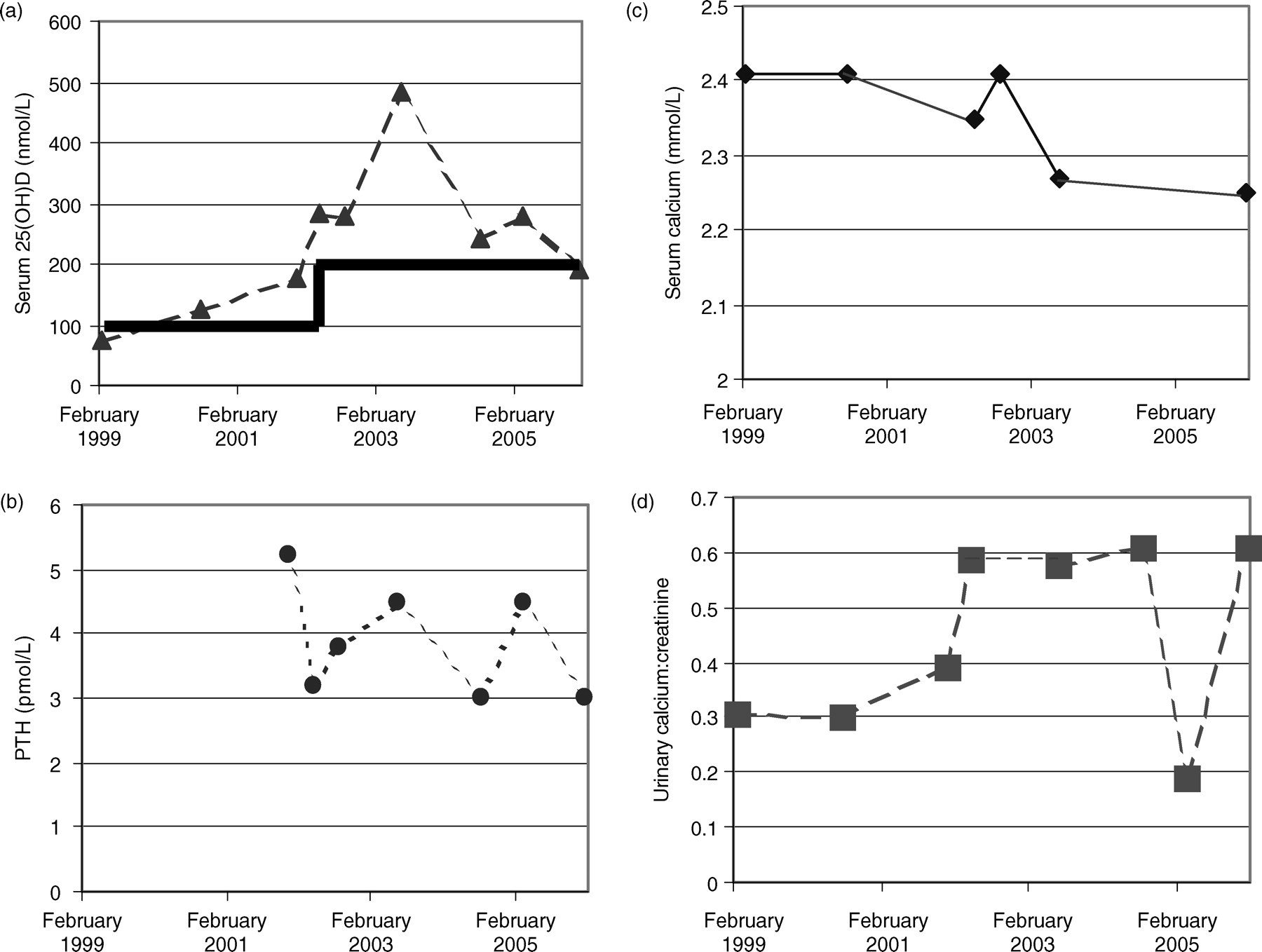
Patient 1: Biochemical parameters over 7 years of vitamin D3 supplementation at 100 and 200 μg/day (1999–2002 and 2002–2006, respectively). (a) Serum 25(OH)D; (b) serum parathyroid hormone (PTH); (c) serum calcium; (d) urinary calcium:creatinine. Laboratory decision point for hypercalcaemia >2.6 mmol/L. The heavy line in panel (a) indicates the reported daily dose of vitamin D3, initially 100 and later 200 μg/day
Case 2
Subject 2 was a 39-year-old man according to the last data reported. He had been diagnosed with multiple sclerosis (MS) in 1998, with symptoms of numbness in his extremities first appearing in early 1997. His BMI was normal, and he avoided dairy products. In 2000, the patient began investigating alternative therapies implicated in MS treatment and discussed such alternatives as vitamin B12, vitamin D, testosterone and medicinal marijuana with his treating neurologist.
After weighing the risks involved with each therapy, the subject first began taking daily injections of 500 mg of vitamin B12 (cyanocobalamin) in 2000, which continued until early in 2006 when this was increased to 1000 mg/day of methylcobalamin. Shortly after starting vitamin B12 injections, he began using medicinal marijuana as required in the evenings to relieve bladder and leg spasms. In 2000, this subject reported that he also started taking vitamin D3 at 25 μg (1000 IU)/day which has been steadily escalating over the past 6 years. In 2004, oral testosterone was started, in the form of testosterone undecanoate (Andriol®), at 100 mg/day. A testosterone gel (Androgel®) is also used by the patient at 120 mg/day, subsequently increasing to 160 mg/day in 2006. On alternate days depending on convenience, the patient uses either Androgel® or Andriol® for a daily dose of 120–160 mg of testosterone.
The status of this patient's MS has been relatively stable for the last few years and there has been no clear evidence of progression either clinically or radiologically since 2002.
The dose-escalation schedule of vitamin D3 reported by the subject was as reported in Table 1. From August 2003, when the patient reported he started taking 1200 μg (48,000 IU)/day until July 2004 when he reported he had increased the dose to 1600 μg (64,000 IU)/day; the actual doses of vitamin D3 that were taken have not been confirmed. The high-concentration solution of vitamin D3 was not consistent with the serum 25(OH)D response. In December 2004, he began taking 1800 μg (72,000 IU)/day. At his follow-up in April 2006, serum calcium exceeded 2.6 mmol/L and he discontinued the vitamin D3. Two months later, the 25(OH)D concentration had declined by approximately one-half, and serum and urine calcium values returned back to the reference range (Figure 2).
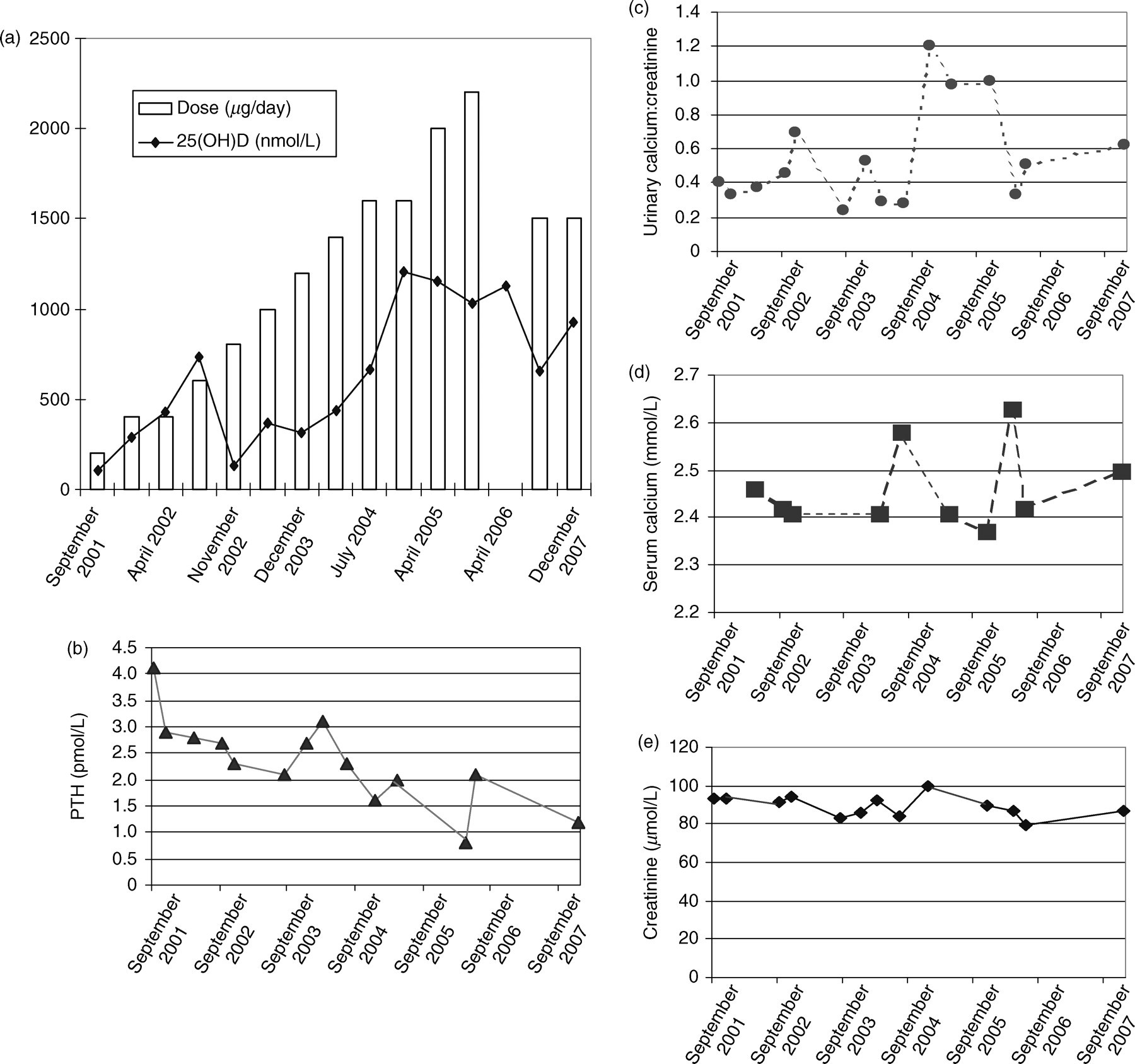
Patient 2: Biochemical responses to dose-escalation of vitamin D3 from 200 to 1600 μg/day over a 5-year period. (a) Vitamin D3 dose initiated and serum 25(OH)D; (b) serum parathyroid hormone (PTH); (c) urinary calcium:creatinine; (d) serum calcium; (e) serum creatinine. Laboratory decision point for hypercalcaemia >2.6 mmol/L
Vitamin D3 dose-escalation schedule for patient 2
*Actual concentration of vitamin D3 is unknown
In June 2006, subject 2 re-initiated vitamin D3 therapy with 1500 μg/day. In December 2006, his serum 25(OH)D was up to 925 nmol/L and measures of calcium were within reference ranges.
To the time of this writing, subject 2 has been maintaining the intake of vitamin D3 at 1500 μg (60,000 IU)/day, with this he takes daily injections of vitamin B12 (methylcobalamin) at 1000 mg/day, Aricept® at 10 mg/day (a treatment indicated for mild–moderate dementia intended to help improve his memory) and Andriol® (160 mg/day) or Androgel® (140 mg/day).
The dosage of vitamin D3 and concentrations of serum calcium, 25(OH)D, PTH as well as urinary calcium:creatinine ratios are shown in Figure 2. In December 2004, the urine calcium:creatinine ratio was 1.2; however, upon repeating it one week later it had been resolved and was 0.33. An increasing trend was seen in the calcium:creatinine ratio in the three visits preceding the rise in serum calcium concentrations. In April 2005, the calcium:creatinine ratio was 0.97 and by November 2005 it was 1.0. Serum calcium concentrations were within reference ranges on all but one occasion, including the timepoints when urinary calcium was seen to be increasing. In April 2006, serum ionized calcium was 1.37 mmol/L and total plasma calcium was 2.63 mmol/L. At this time, the patient was advised to stop taking vitamin D3. Concentrations of 25(OH)D up to 1126 nmol/L had been reached before evidence of hypercalcaemia. Two months after vitamin D3 supplementation was stopped, calcium values were within reference ranges. The patient again started taking vitamin D3 maintaining this at 1500 μg (60,000 IU)/day. Overall, serum PTH was significantly decreased from 4.1 in September 2001 to 2.1 pmol/L in June 2006 (reference range, 1.3–7.6 pmol/L).
Discussion
Both subjects achieved serum 25(OH)D concentrations far above what is considered minimally optimal (>75 nmol/L) by current standards. Although a specific serum 25(OH)D concentration associated with vitamin D toxicity has never been established, it was believed to be higher than physiological concentrations, i.e. >250 nmol/L. These two men demonstrate that concentrations far above this can be attained without causing hypercalcaemia in some individuals. By no means do we suggest that anyone undergo a similar treatment unsupervised as toxicity may result. Individuals taking high doses of vitamin D should consult a physician. As discussed above, cases of vitamin D3 toxicity in the literature have consistently shown that serum 25(OH)D concentrations >700 nmol/L are associated with toxicity. In subject 2, it was only once the serum 25(OH)D concentration exceeded 1000 nmol/L that any changes in serum calcium were seen. As well, subject 2 had been taking vitamin D3 doses that were 4–30 times the upper concentration of vitamin D3 intake, 40 μg (2000 IU)/day. Subject 2's 25(OH)D concentrations associated with hypercalcaemia are in agreement with the intakes seen in other reported cases of toxicity.
The urinary calcium:creatinine ratio may be an early indicator of eventual hypercalcaemia. Prior to the rise in serum calcium seen in April 2006 (ionized calcium 1.37 mmol/L and total calcium 2.63), urinary calcium excretion was showing an increasing trend. The urinary calcium:creatinine values for the two visits preceding the rise in serum calcium were 0.97 and 1.0. Close monitoring of biochemical responses confirmed that an increase in urinary calcium:creatinine ratio precedes hypercalcaemia as serum 25(OH)D concentrations rise.
It is interesting to note that the patient stopped taking vitamin D3 for 2 months (April–June 2006) and 25(OH)D concentrations decreased from 1126 to 656 nmol/L, indicating a half-life of ∼2 months.
The upper concentration, or guidance concentration, for vitamin D3 intake is intended to ensure that almost all of the general public will be safe from adverse effects. The mechanism causing vitamin D toxicity is thought to be relative saturation of vitamin D binding protein (DBP) in plasma. 1 This increases the ‘free’ concentration of 1,25(OH)2D hormone that stimulates intestinal calcium absorption and bone resorption. At lower 1,25(OH)2D concentrations, the effect is to promote bone mineralization. 11 Activity of each unit of 1-α-hydroxylase protein cannot be downregulated entirely, and the mass action of 25(OH)D as substrate continues to result in the production of 1,25(OH)2D. Further, clearance of 1,25(OH)2D cannot compensate fully for the production. 12 The increased concentrations of unbound circulating 1,25(OH)2D produced are responsible for hypercalcaemia. 1
Subject 1 was able to maintain dosing at 200 μg (8000 IU)/day of vitamin D3 and safely, with no change in calcium concentrations, over the long term (3 years). We speculate that the ability of subject 2 to reach serum 25(OH)D concentrations >1000 nmol/L, indicated by past reports as concentrations that should be toxic, prior to any evidence of hypercalcaemia may be related to the dose-escalation schedule that he followed. One possibility is that the gradual increase in circulating 25(OH)D concentrations may have allowed for increased concentrations of DBP to be generated and thus prevented the presence of unbound vitamin D metabolites. Unfortunately, samples were not available for measuring DBP in either subject.
Further studies, in the form of placebo-controlled trials, need to be conducted to determine the long-term safety of intakes of vitamin D3 in excess of 250 μg (10,000 IU)/day. As well, the effects of increasing doses on DBP concentrations or DBP polymorphisms may be considered as possibilities for the differences seen between individuals who do or do not become toxic with similar doses.
In summary, the two individuals presented here demonstrate that intakes of vitamin D3 that exceed the upper limit by at least four-fold are tolerated in some individuals without any effect on calcium concentrations. In the case of subject 2, intakes of vitamin D3 up to 30 times the upper limit were tolerated. This report also indicates that urinary calcium:creatinine ratios may be used as an early indicator for hypercalcaemia.
