Abstract
Abstract
Background
Abnormal cell membrane composition and functional cobalamin (vitamin B12) deficiency was reported in schizophrenic individuals. We aimed to investigate the relationship between cobalamin state and cell membrane composition in patients with schizophrenia.
Methods
Malondialdehyde (MDA), cholesterol, and phospholipid classes in the erythrocyte membranes of 18 schizophrenic and 20 healthy individuals of the same age and sex distribution were determined. Serum vitamin B12, plasma total homocysteine, serum folate, and urine methylmalonic acid (uMMA) concentrations were measured in both groups.
Results
In the schizophrenic group, uMMA, membrane MDA, membrane cholesterol, membrane phosphatidylinositol concentrations were significantly higher and the membrane phosphatidylserine concentrations were lower than the control group values. In schizophrenic individuals, uMMA concentrations have a significant positive correlation with membrane MDA and a negative correlation with membrane cholesterol concentrations (P < 0.05). The negative correlation of uMMA with membrane cholesterol concentrations may be a biological response to the increased membrane rigidity. Also, a free radical elevation related to the elevated uMMA concentrations in the erythrocyte membrane, might reflect the role of methylmalonic acid (MMA) in membrane damage.
Conclusion
Our present findings suggest that, functional vitamin B12 deficiency representing itself by MMA elevations in schizophrenic individuals could damage cell membrane.
Introduction
The association of schizophrenia and cell membrane abnormalities was originally suggested by Feldberg 1 and Horrobin, 2 and is currently known as the membrane hypothesis of schizophrenia. This hypothesis is based on the important role that lipids and fatty acids play in the structural integrity of cellular membranes and their actions as messengers in the cell-signalling system. Altered membrane phospholipid metabolism has been considered as the pathophysiological basis of schizophrenia. 3 Several studies have reported variable changes in the concentrations of membrane phospholipids in erythrocytes. 4–6 The composition and balance of these molecules in the brain have been shown to be critical for the proper development and functioning of the nervous system. 7 Relatively small changes in membrane lipid structure can lead to successive changes in tertiary protein structure and alterations in the functions of a wide variety of membrane-bound and membrane-associated proteins. Apart from this structural role, lipid derivatives form many of the second messenger signalling systems in neurons and also regulate the behaviour of other signalling systems.
In schizophrenic individuals, reduced essential fatty acid concentrations in the membrane phospholipids were found to be associated with an increase in lipid peroxidation products. 8 The low concentrations of arachidonic acid and docosahexanoic acid in red cell membranes in some schizophrenics 9–11 and the failure of niacin flushing, 12 indicating a lack of arachidonic acid for prostaglandin D2 formation, are consistent with increased activity of phospholipase A2. Also, increased membrane cholesterol content in biomembranes has been shown to reduce membrane fluidity. 13
Cobalamin has a long and controversial history of association with psychiatric illnesses although the evidence detailing these relationships remains incomplete. 14–16 Recently, it has become clear that neuropsychiatric symptoms often precede other evidence of a deficiency. 17,18 Deficiency of this vitamin is reported to play a causal role in the development of psychiatric and neurological illnesses such as methylmalonic acidaemia and subacute combined degeneration of the spinal cord, even when the serum concentration of the vitamin is in the reference range. Therefore, it is important to understand the mode of action of cobalamin, which acts as a coenzyme in two important reactions: first, in the synthesis of methionine from homocysteine in the form of methylcobalamin and secondly, in the conversion of methylmalonyl CoA to succinyl CoA in the form of deoxyadenosylcobalamin, which is a key step in fatty acid metabolism. Deficiency of any of these ‘active’ forms of cobalamin may play a role in the onset of schizophrenia. The absence of deoxyadenosylcobalamin causes accumulation of abnormal lipids and an increase in methylmalonic acid (MMA) and homocysteine, which are the substrates for the reactions catalysed by the active cobalamin forms, may cause increased free radicals leading to membrane damage. Methylmalonyl CoA, in excess, is converted to MMA, which leads to mitochondrial complex II inhibition, and hence the accumulation of free radicals. Therefore, a functional vitamin B12 deficiency may be one of the factors contributing to the cell membrane abnormalities in schizophrenia. The urine methylmalonic acid (uMMA) concentration is currently used as a preferred indicator of functional vitamin B12 status, because the elevated serum or uMMA concentrations represent the intracellular metabolic change that is significantly related to the vitamin B12 as described above.
Previous studies have shown cell membrane abnormalities and a functional vitamin B12 (cobalamin) deficiency in schizophrenia. However, data defining the relationship between cobalamin metabolism and cell membrane alterations in psychiatric disorders do not exist. In this study, our aim was to investigate a possible relationship between abnormal cell membrane composition and the functional vitamin B12 status in schizophrenia. Therefore, we evaluated cell membrane abnormalities by determining free radicals (malondialdehyde [MDA]), cholesterol and the individual phospholipid content of the cell membrane in schizophrenic and healthy individuals. To reveal a possible correlation, we also assessed cobalamin metabolism of both groups by measuring uMMA, serum vitamin B12, folate, and plasma total homocysteine (tHcy) concentrations.
Materials and methods
Reagents
Tris, 1,1,3,3-tetraethoxypropane, diethyl 2-thiobarbituric acid (DETBA), triethylamine, disodium EDTA, individual phospholipids, potassium hydroxide, and cupric nitrate were from Sigma Chemical Co (St Louis, MO, USA); triethanolamine and disodium phosphate were from MERCK (Darmstadt, Germany); ethanol, phosphoric acid, n-butanol, glacial acetic acid, methanol, n-hexane, 2-propanol, acetonitrile, perchloric acid, and HCl were from JT Baker (Deverter, The Netherlands). All chemicals were in the analytical grade.
Subjects
Eighteen chronic-medicated schizophrenic patients (10 men, 8 women; mean age ± SD: 31 ± 7 years) who were diagnosed as schizophrenic according to the Diagnostic and Statistical Manual of Mental Disorders (DSM IV) criteria, were selected from the patients admitted to the Department of Psychiatry and 20 healthy individuals (10 men, 10 women; mean age ± SD: 30 ± 8 years) were selected as a control group. The study was approved by the local ethics committee and written informed consent was obtained from patients and the control group. The schizophrenic patients had been taking atypical antipsychotics (for at least 5 years [mean ± SD: 11 ± 2.5 years]). The Positive and Negative Syndrome Scale 19 was used to derive subscale scores for positive, negative, and general psychopathology symptoms. Positive and negative symptom scores of patients were similar. In addition, the weekly fish, white meat, and red meat consumption of each schizophrenic and healthy individual was determined by a questionnaire. None of them were vegetarian or had poor nutritional diet. Patients and controls were having similar diet compositions. The control and patient groups were also in the same sex and age distribution. None of the patients had received either vitamin B12 or folate supplementation before or after the study. The blood samples of both groups were collected via vacutainer tubes and K3EDTA-containing tubes. Plasma and serum were separated, and serum vitamin B12 and serum folate were determined on the same day. The remaining serum and plasma were stored as portions at −70°C. The erythrocytes separated from plasma were washed three times with isotonic saline and stored at −70°C until the day of use. Early morning fasting urine samples were also collected from patients and analysed for MMA concentrations on the same day.
Determination of erythrocyte membrane phospholipid composition
The quantification of phospholipids in the erythrocyte membrane was performed using the high-performance liquid chromatography (HPLC) method described by Mawatari and Murakami. 20 One volume of packed erythrocytes was haemolysed in 40 volumes of hypotonic buffer (10 mmol/L Tris–HCl, pH 7.4) and centrifuged at 25,000 g for 20 min at +4°C. This procedure was repeated four times for washing membranes.
Lipid extraction
Lipids were extracted with chloroform and methanol. 21 The membrane pellet was suspended in 2 mL methanol by sonication and vigorous mixing and then 4 mL of chloroform was added. The chloroform–methanol suspension was left at room temperature for 60 min and then 1.2 mL of water was added to the solution. After vigorous mixing, the mixture was centrifuged at 2000 g for 5 min. The chloroform layer was transferred to another tube and was dried under a stream of nitrogen gas.
Phospholipid separation by high-performance liquid chromatography
The dried total lipids were dissolved in 200 μL of n-hexane:2-propanol (3:1, v/v) and were filtered through a 0.5 μm filter before injecting 5–10 μL onto the HP Agilent HPLC system (Agilent Technologies, Palo Alto, CA, USA). Separation of phospholipid classes from the total lipid extracts was achieved using a Bondesil-NH2 column (300 × 4.6 mm). The mobile phase was acetonitrile/methanol/0.2% triethylamine (67:22:11, v/v/v). The triethylamine solution was adjusted to pH 4.0 using o-phosphoric acid. The flow rate was 0.8 mL/min and the column temperature was maintained at 40°C. Phospholipid classes were detected at 210 nm using an ultraviolet (UV) detector.
Determination of erythrocyte membrane malondialdehyde
MDA was measured using the method reported by Conti et al. 22 Briefly, 50 μL of membrane suspension, 1 mL of (1,3-diethyl-2-thiobarbituric acid) DETBA solution (10 mol/L, pH 7.0), 100 μL of EDTA (18.75 mmol/L), and 100 μL of H3PO4 (3 mmol/L) were added into a test tube and vortexed for 30 s. After 45 min incubation at 95°C in the waterbath, the tubes were cooled in ice immediately. Five millilitre of n-butanol was added to the mixture and vigorously mixed for 30 s. After centrifugation in 1500 g for 10 min, the butanol layer was used for fluorometric measurements (scanning range: 500–600 nm; speed: very slow; delta λ: 14 nm).
Determination of erythrocyte membrane cholesterol concentrations
Membrane suspensions were analysed for cholesterol content using the enzymatic photometric cholesterol determination method 23 in the biochemical autoanalyser. The protein concentrations of membranes were determined using the method reported by Lowry et al. 24 The membrane cholesterol concentrations were normalized by dividing with the membrane protein concentrations.
Urine methylmalonic acid determination
The uMMA measurements were based on a modified photometric method using a scanning spectrophotometer (Philips UV/VIS scanning spectrophotometer, PU 8700 series, UK).
18
The photometric uMMA determination method is based on the reaction between MMA and diazotized p-nitroaniline. One millilitre of urine sample was added to a column containing strongly basic anion exchanger resin. After washing with 30 mL of distilled water, MMA was eluted using 10 mL of 0.1 mol/L HCl. The acid eluant from the column (1 mL) was mixed with 1.5 mL of 1.0 mmol/L acetate buffer and 1.5 mL of cold diazo reagent, heated to 37°C in a waterbath for 30 min and 1 mL of 3 mmol/L NaOH was added. After an additional incubation at 37°C for 30 min, the samples and standards were read against reagent blank in the scanning spectrophotometer. The results were obtained at 620 nm (A
620), 570 nm (A
570), and 670 nm (A
670) and corrected absorbance (A) was calculated using the formula:
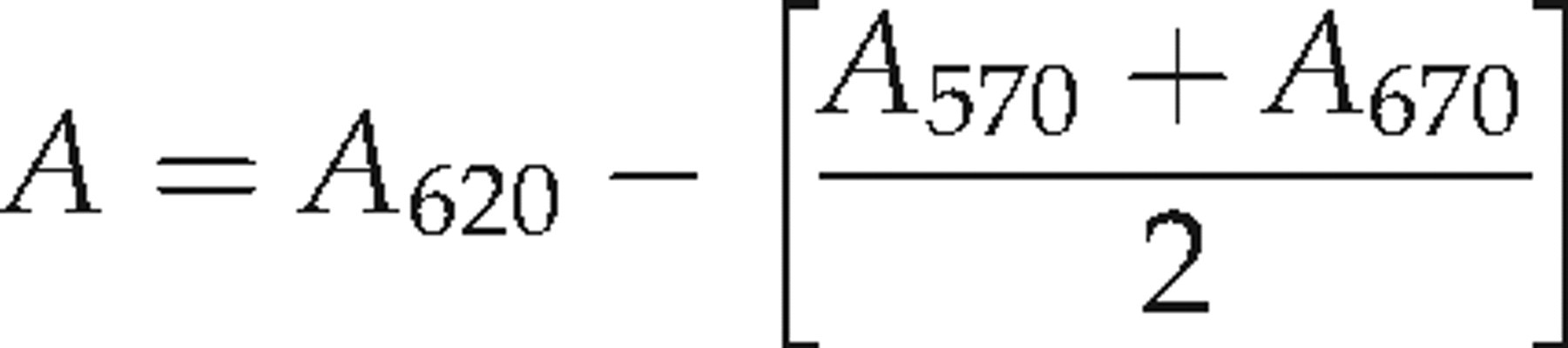
Other laboratory analyses
Folate and vitamin B12 measurements were measured using a microparticle enzyme immunoassay method on the Abbott Axsym System Automated Immunoassay Analyzer (Abbott Laboratories, Abbott Park, IL, USA). The tHcy concentrations were measured on the HP Agilent 1100 HPLC system using an HPLC kit (Chromsystems GmbH, Munich, Germany) system.
Statistical analysis
The distributions of all variables were tested using normality plots and the Kolmogorov-Smirnov test, which quantifies the discrepancy between data distribution and ideal Gaussian distribution. The data passed the normality tests and comparisons of each group's laboratory parameters were determined using the Student's t-test. The relationships between laboratory parameters were evaluated by Pearson's correlation coefficient. All statistical calculations were performed by SPSS Inc, Chicago, IL, USA, for Windows (version 11.0) software.
Results
Cobalamin status parameters
The mean ± SD values and the statistical difference of parameters used in the assessment of cobalamin status of patient and control groups are listed in Table 1. Although the tHcy, serum vitamin B12, and folate concentrations of schizophrenic individuals were at the same concentrations as the control group, the uMMA concentrations were found to be higher than the control group (P < 0.01). This implies that there was a functional (tissue level) cobalamin deficiency and this condition manifested itself by elevated uMMA in schizophrenic patients. uMMA, which was the only elevated marker, showed a positive correlation with plasma tHcy concentrations both in the control and in the patient groups (r = 0.445; P < 0.05 and r = 0.631; P < 0.01, respectively). No other cobalamin status parameter correlated with the others.
Statistical differences (P values) of mean ± standard deviation values of laboratory parameters between patient and control groups
PC, phosphatidylcholine; SM, sphingomyelin; PE, phosphatidylethanolamin; PI, phosphatidylinositol; PS, phosphatidylserine; UMMC, urine methylmalonic acid; THCY, total homocysteine
Factors affecting cell membrane composition
Erythrocyte membrane MDA, phosphatidylinositol (PI) and cholesterol concentrations were found to be higher than the control group values (P < 0.01), whereas the amounts of phosphatidylserine (PS) in schizophrenic patients were lower than the control group (P < 0.05) (Table 1). The phospholipid compositions of erythrocyte membranes were defined as the percent values of individual phospholipids in the total phospholipid concentration. The elevated membrane cholesterol and decreased PS concentrations may cause reduced membrane fluidity. PI was negatively correlated with PS and phosphatidylethanolamine (r = −0.680, P < 0.01 and r = −0514, P < 0.05, respectively).
Effect of cobalamin deficiency on the membrane composition
In schizophrenic individuals, elevated uMMA concentrations were found to be positively correlated (r = 0.534, P < 0.05) with elevated membrane MDA concentrations indicating increased lipid peroxidation in the cell membrane. uMMA concentrations also showed negative correlation with elevated membrane cholesterol concentrations (r = 0.489, P < 0.05). Although MDA concentrations were negatively correlated with serum folate and plasma tHcy concentrations in the patient group, folate and tHcy concentrations were not different from those in the control group.
Discussion
In this study, we detected important changes in the erythrocyte membranes of schizophrenic patients. One of the most important findings is the significantly elevated membrane cholesterol concentration in the schizophrenic group. Cholesterol is an essential component of mammalian cells. It is found in high concentrations in the plasma membrane, and in low concentrations in intracellular membranes. 26 Several membrane receptor proteins have been reported to depend on cholesterol for their functional activities. 27,28 Membrane cholesterol elevation may cause significant changes to membrane functions and decrease fluidity. 13 Mahadik et al. 29 reported increased membrane cholesterol contents of fibroblasts of schizophrenic individuals. These data support the hypothesis that schizophrenia is associated with disordered membrane lipid metabolism, and that this predates the onset of psychosis.
In a recent study, 30 abnormal cholesterol processing was reported in Alzheimer's disease patient's fibroblasts. Similarly, the elevated cholesterol concentrations of cell membranes in the schizophrenic group could reflect a dysregulation of cholesterol processing, which may contribute to the pathogenesis of schizophrenia. The independency of elevated cholesterol concentrations in membranes from the dietary features support the idea of altered endogenous biochemical mechanisms in cholesterol metabolism. Interestingly, there was a negative correlation between uMMA and membrane cholesterol. The elevated cholesterol level in the cell membrane could be a preventive mechanism against elevated MMA concentrations, which was reported to be an inhibitor of the mitochondrial respiratory chain. 31
The second important finding is the presence of elevated amounts of free radicals (MDA) in the erythrocyte membranes of schizophrenics. MMA elevations can increase MDA concentrations by inhibiting the mitochondrial respiratory chain. We found a positive correlation between MDA and uMMA that could explain the elevated free radicals in the erythrocyte membrane. This increase in free radicals could damage the membrane, particularly polyunsaturated fatty acids, or intracellular components. The last product of fatty acid catabolism, acetyl CoA, can be diverted to cholesterol synthesis and can cause membrane cholesterol elevations. Thus, the elevated MMA could be a trigger for membrane alterations.
In a recently reported study, 32 the amyloid-β protein, which plays a major role in the pathogenesis of Alzheimer's disease, was found to be a possible protective molecule against lipid peroxidation. Similarly, increased free radicals in the schizophrenic group may be responsible for cognitive abnormalities via elevated amyloid-β protein. Recent neuropathological studies have demonstrated that late-onset schizophrenic patients exhibit a restricted limbic tauopathy, glial tangles (thorn-shaped astrocytes) and scarce amyloid deposition. 33,34
The significantly elevated PI and reduced PS concentrations of the schizophrenic group may be the cause of changes in membrane functions and in the second messenger system. In particular, elevated PI amounts previously reported to be present both in the brain 35 and in the platelets 36,37 of schizophrenic individuals demonstrate the hyperactivity of the PI-signalling system in schizophrenia. Jope et al. 38 have recently demonstrated selective increases in PI-signalling activity and Gα0 concentrations in the postmortem brain from schizophrenic subjects. The reduced arachidonic acid content in red blood cell membranes of schizophrenic individuals was also previously reported. 8,9 Because elevated MMA concentrations could also be related to branched chain fatty acids being incorporated into membrane phospholipids, 39 analysis of the fatty acids in the erythrocyte membranes associated with MMA elevations of schizophrenic individuals should also be further investigated.
The functional vitamin B12 deficiency that is frequently seen in neuropsychiatric disorders was shown in the schizophrenic group with significantly elevated uMMA concentrations. Interestingly, tHcy concentrations of the patient group were not elevated. Although Levine et al. 40 reported dramatically elevated tHcy concentrations in 193 Israeli schizophrenic subjects compared with controls, the differences between these groups were almost entirely attributable to men 50 years of age and younger. However, Virgos et al. 41 found no difference in tHcy concentrations between 210 Spanish schizophrenic psychiatric inpatients compared with 218 controls. Susser et al. 42 compared the concentrations of homocysteine of schizophrenic subjects and normal controls, stratified by folate concentrations, and found that increased homocysteine was only observed in the low folate cases. Therefore, the selected patient group in this study having normal folate concentrations and consisting of relatively younger subjects could explain why tHcy concentrations were normal.
In the cell, vitamin B12 as a coenzyme should be converted to methylcobalamin and deoxyadenosylcobalamin in order to be active. The conversion mechanisms could not be understood yet. The cell membrane may have a function in this biological process. Thus, the reason of selectively elevated uMMA concentrations of the schizophrenic group can be the altered membrane compositions of these patients. The relationships between uMMA and membrane alterations in this study are the first findings that show the dynamic relation of cobalamin metabolism and cell membrane functions.
Methylcobalamin which is one of the active forms of cobalamin, has a critical role as a regulator of methylation reactions; particularly phospholipid methylation, via methionine synthase. Thus, if methylcobalamin concentrations were reduced, lower methionine synthase activity would lead to the accumulation of S-adenosylmethionine, an inhibitor of methylation reactions. Phospholipid methylation is considered to be a very robust process 43 and impaired methylation would lower membrane fluidity and could lead to secondary effects. Thus, concentration of cobalamin derivatives including methylcobalamin and adenosylcobalamin in the cell should be further analysed in schizophrenic individuals. Glutathionylcobalamin was suggested as an important intermediate during the synthesis of methylcobalamin and deoxyadenosylcobalamin. 44 The increased oxidative stress related to the functional cobalamin deficiency could reflect a limitation in glutathione availability for the synthesis of active forms of cobalamin.
In summary the important findings of this study are:
There was an elevated membrane cholesterol composition in the schizophrenic group; In the schizophrenic group, the composition of membrane phospholipids was different and elevated PI and reduced PS concentrations were detected. Decreases in PS concentrations are important due to the change they cause in the dynamic structure of the membrane; There was a functional cobalamin deficiency that expresses itself with elevated uMMA concentrations in schizophrenic patients. This selective increase of MMA in cobalamin deficiency could depend on membrane alterations; There was also an increased free radical concentration in the membranes of schizophrenics. This increase showed high correlation with uMMA.
