Abstract
Abstract
Background
The diagnosis of malignant lymphoma (ML) such as non-Hodgkin's lymphoma (NHL) and Hodgkin's lymphoma (HL) was mainly performed by morphological examination and gene analysis. There are only a few serum/plasma biomarkers such as lactate dehydrogenase and soluble interleukin-2 receptor α to diagnose ML. The classifications are various, and therefore the cell surface markers using flow cytometry or lymph node biopsy have been examined. It is difficult, however, to distinguish the two diseases, NHL and HL, from each other.
Methods
In order to identify the haematological malignancy-associated autoimmunoreactivity (autoantibodies) in patients' plasma, a novel proteomics-based approach using electrophoresis/mass spectrometry was applied. Solubilized proteins from a Burkitt's lymphoma cell line (Raji) were separated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis and Western blotting analysis, in which the plasma of individual patients with haematological malignancies was tested for primary antibodies, followed by visualization with anti-IgG antibody conjugated with horseradish peroxidase.
Results
Two proteins, L-plastin and α-enolase, capable of reacting with the antibodies in plasma of patients with NHL, were detected using matrix-assisted laser desorption ionization/time-of-flight mass spectrometry and tandem mass spectrometry. The rates of the detections of an anti L-plastin autoantibody were significantly higher: 0.84 (21/25) in patients with NHL; 0.00 (0/4) in HL; 0.38 (5/13) in autoimmune diseases; 0.20 (2/10) in leukaemia; and 0.13 (1/8) in healthy controls. In contrast, those of anti α-enolase antibody were not specific to NHL.
Conclusions
We first identified autoantibody against L-plastin in plasma of patients with NHL, suggesting that the autoantibody can be a new diagnostic biomarker for NHL.
Introduction
Malignant lymphoma (ML) is a lymphocyte-proliferating disease, being divided roughly into non-Hodgkin's lymphoma (NHL) 1 and Hodgkin's lymphoma (HL). 2 There are more than 30 types of NHLs differentiated by growth rate. About 85% of NHLs occur in B-lymphocytes. 3 NHL is a group of disease with abundant diversity. Generally, NHL involves the presence of cancerous cells in lymph nodes without specific localization. The pathological cause of NHL is still unclear; NHL is more than seven times as common as the other general type of lymphoma. The diagnosis of ML was mainly dependent on the morphological examination and gene analysis. The classifications are various, and therefore the examination of cell surface markers using flow cytometry or lymph node biopsy has been examined. It is difficult, however, to distinguish the two diseases, NHL and HL, from each other by conventional examinations.
There is substantial evidence for the human immune response to cancer in humans as demonstrated by the identification of autoimmunoreactivity, so called autoantibodies, against a number of intracellular and surface antigens in patients with various tumour types. 4 Brichory et al. 5 first reported that autoantibodies to Annexins I and II are useful biomarkers for the diagnosis of lung adenocarcinoma using a proteomics-based analysis. We have studied the autoantibodies in the plasma of patients with lung adenocarcinoma 6 and oesophageal cancer 7 using autoimmunoreactive detection and proteome analysis. Many researchers reported that the autoantibodies against nuclear components, phospholipids and plasma proteins were detected in the serum samples of patients with NHL. 8,9 Conversely, it was reported that patients with autoimmunity are prone to lymphoma. 10 Using the same proteomics-based technique, the identification of NHL-specific antigens and autoantibodies is useful for the establishment of a simple examination and an understanding of the pathological causes of NHL.
In the present study, we examined 71 plasma samples of patients with haematological malignancies to identify the autoantibody against proteins expressed in the Raji cell line established from Burkitt's lymphoma, being categorized as NHL. We found several positive spots on a membrane using a Western blot (WB)/enhanced chemiluminescence (ECL) detection system, and identified L-plastin as an NHL-specific spot, capable of reacting with these antibodies in the plasma of patients with NHL using matrix-assisted laser desorption ionization/time-of-flight mass spectrometry (MALDI-TOF/MS) and tandem mass spectrometry (MS/MS). These results suggest that the autoantibody against L-plastin can be a new diagnostic biomarker for NHL.
Materials and methods
Chemicals and materials
The total proteins derived from a human B-lymphoma cell line (Raji) were purchased from BioChain Institute Inc (Hayward, CA, USA). The total proteins dissolved in a buffer including HEPES (pH 7.9), magnesium chloride, potassium chloride, EDTA, sucrose, glycerol, sodium doxycholate, Nonidet P-40 and a cocktail of protease inhibitors were stored at −80°C until use. Plasma from 25 patients with NHL, 11 with myelodysplastic syndrome (MDS), four with HL, 10 with leukaemia (7 with acute lymphocytic and 3 with chronic myelogeneous leukaemia), 13 with autoimmune diseases and eight healthy subjects were analysed. Urea, glycine, sodium dodecyl sulphate (SDS), iodoacetamide, 3-{(3-cholamidopropyl)dimethyammonio}-1-propane-sulfonate hydrate (CHAPS), acrylamide, N, N′-methylene bisacrylamide, tris(hydroxymethyl)aminomethane (Tris), ammonium bicarbonate, glycerol, 2-mercaptoethanol, dithiothreitol, Tween 20, ammonium persulphate, phenylmethanesulfonyl fluoride (PMSF), Nonidet P-40, silver nitrate and EDTA were purchased from Nakalai Tesque (Kyoto, Japan); methanol, ethanol, acetonitrile, acetone, acetic acid, formic acid and distilled water from Merck (Darmstadt, Germany); 10–20% gradient slub-gel (9 cm long) from Daiichi Chemical Co. Ltd (Tokyo, Japan). The anti L-plastin (catalogue no. sc-16657) and anti α-enolase (catalogue no. sc-15343) polyclonal antibodies were purchased from Santa Cruz Biotech Inc (Santa Cruz, CA, USA). Anti L-plastin and anti α-enolase antibodies were raised against an internal region of human L-plastin and the amino acids 1–300 mapping of the N-terminus of human α-enolase, respectively.
Sodium dodecyl sulphate-polyacrylamide gel electrophoresis and Western blotting
Four micrograms of total proteins derived from the Raji cells (Lot no. A802144) were applied to each lane of SDS-PAGE (polyacrylamide gel electrophoresis) gels. Separated proteins were transferred to a membrane or visualized using silver staining. Proteins separated by SDS-PAGE were subjected to WB according to the method described previously. 6 The membrane was cut into individual lanes and the strips were incubated with the respective plasma at 1:1000 dilutions for 2 h at room temperature. After four washings, the strips were reacted with horseradish peroxidase-conjugated anti-human IgG (GE Healthcare Bioscience Co Ltd, UK) at 1:8000 dilutions for 1 h at room temperature. After washing, the strips were developed with an ECL system (ECL plus, GE Healthcare Biosciences). SDS-PAGE gel was stained with silver nitrite and the pieces of gel corresponding to WB positive bands were excised. Each piece of the gel band was treated with reduced alkylation, digestion and then extraction of digested peptides according to the preparation described previously. 6 The extracts were concentrated, followed by desalting in ZipTip C18 microcolumns (Millipore Co Ltd, Bedford, MA, USA).
Protein identification by mass spectrometry
The extracted peptides were loaded onto the MALDI target plate by mixing 1 μL of each solution with the same volume of a matrix solution that was freshly prepared every day by dissolving 0.3 g/mL of α-cyano-4-hydroxycinnamic acid (Wako Purified Reagent Co Ltd, Kyoto, Japan) in acetone–ethanol (1:1, v/v) solvent. Measurements were performed using an Ultraflex MALDI-TOF/TOF-MS (Brucker Daltonics, Germany). The conditions of MALDI-TOF/MS and MS/MS were the same as described in the manufacturer's instructions. The peptide mass fingerprint was used for protein identification from the tryptic fragment size using the Mascot Search engine based on the entire NCBIn and SwissProt protein databases.
Results
In this study, whole soluble proteins derived from Raji cells, which is an established cell line from a Burkitt's lymphoma categorized as a B lymphoma the same as NHL, were analysed as antigens to detect autoantibodies from the patients' plasma. When WB patterns were compared between the plasma from NHL (A–F) and HL (G–J) patients (Figure 1a), two positive bands were detected at positions 70 and 50 kDa; the 70 kDa band clearly existed with all the plasma of NHL patients (A–F), whereas the 50 kDa band was observed with the plasma of A, D and F. Neither bands were detected with the plasma of HL patients (G–J). These two bands were detected in the plasma of patients with autoimmune diseases at frequency of five of 13 for the 70 kDa band and four of 13 for the 50 kDa band (data not shown).
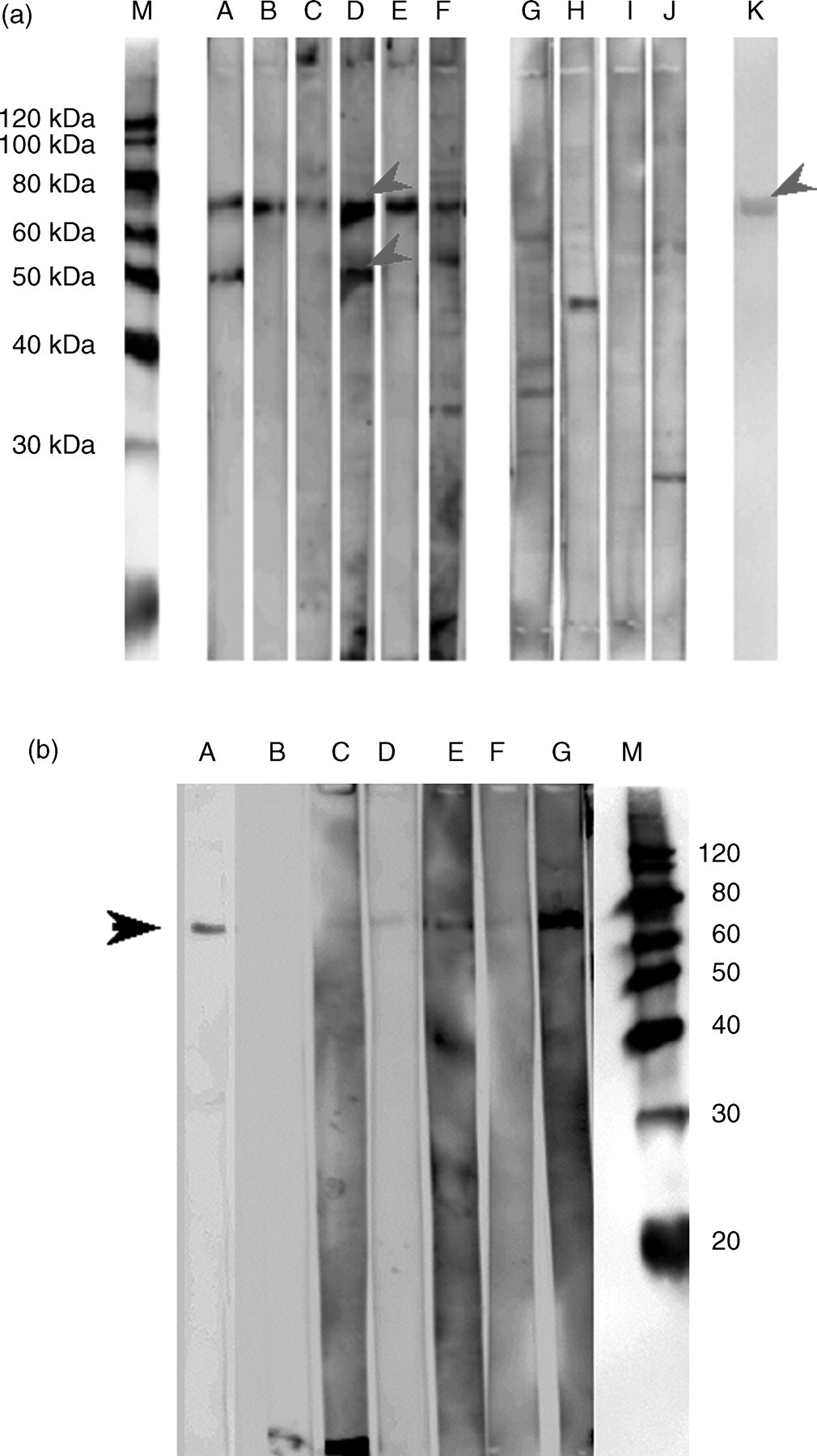
(a) Western blot (WB) patterns immunostained with the patients' plasma from non-Hodgkin's lymphoma (NHL) and Hodgkin's lymphoma (HL). Whole soluble proteins derived from Raji cells were subjected to sodium dodecyl sulphate-polyacrylamide gel electrophoresis/WB analysis. M, molecular weight marker; A–F, NHL; G–J, HL; K, polyclonal anti L-plastin antibody. Arrows indicate specific bands for NHL. (b) WB patterns immunostained with the patients' plasma from NHL, HL and polyclonal anti L-plastin antibody. The purified L-plastin derived from Raji cells by affinity chromatography was used as an antigen. M, molecular weight marker; A, polyclonal anti L-plastin antibody; B–G, NHL patients' plasma
The two bands in the silver-stained gels corresponding to those of WB-positive bands in NHL patients, band no. 1 for 70 kDa and band no. 2 for 50 kDa, were subjected to the proteomics-based analysis using MALDI-TOF/MS and MS/MS. The MS spectrum (peptide mass fingerprint) obtained from the protein in band no. 1 was shown in Figure 2a. Peaks numbered in the figure were identified as the m/z of tryptic peptides from L-plastin by their high score. Band no. 2 was confirmed as α-enolase with peptide mass fingerprints and MS/MS by their high score (Figure 2b). Using immunochemistry, 70 and 50 kDa bands were found to react with L-plastin antibodies (Figure 1a, lane k) and anti α-enolase (data not shown), respectively. In addition, on the WB analysis, the 70 kDa band was confirmed to be identical to L-plastin purified from Raji proteins by immunoaffinity chromatography (Figure 1b). As shown in Table 1, 21 of the 25 patients with NHL, and two of the 11 patients with MDS, five of 13 patients with autoimmune diseases and two of 10 patients with leukaemia showed positive WB bands for anti L-plastin (band no. 1). Eight of the 25 patients with NHL were positive for α-enolase antibodies (band no. 2). In addition, one of the eight healthy individuals was positive for both of these two antibodies. These results indicated that autoimmunoreactivity, namely autoantibody, against L-plastin was observed with NHL patients at higher frequency.
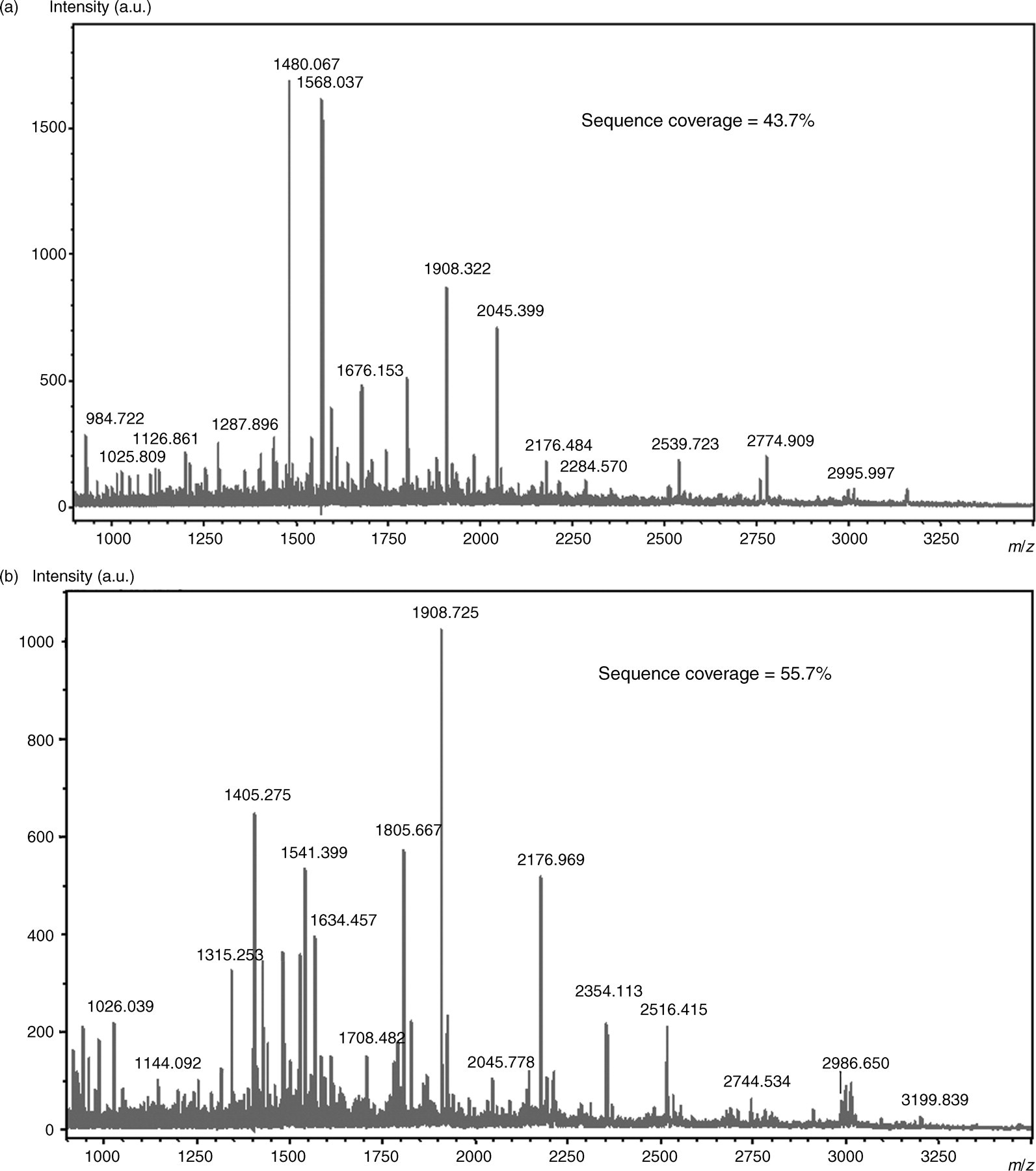
Matrix-assisted laser desorption ionization/time-of-flight-mass spectrometry (MALDI/TOF-MS) mass spectrum obtained from the two protein bands after trypsin digestion. (a) MALDI/TOF-MS spectra obtained from the protein, L-plastin, in band no. 1. (b) MALDI/TOF-MS spectra obtained from the protein, α-enolase, in band no. 2
Positive cases of the two autoantibodies against identified proteins
NHL, non-Hodgkin's lymphoma; MDS, myelodysplastic syndrome; HL, Hodgkin's lymphoma; ALL, acute lymphocytic leukaemia; CML, chronic myelogeneous leukaemia
Discussion
In this study, we examined 71 plasma samples of patients with haematological malignancies to identify haematological malignancy-associated autoantibodies (autoimmunoreactivity) in patients' plasma using electrophoresis/mass spectrometry. At high frequency, anti L-plastin and α-enolase autoantibodies were detected in patients' plasma of NHL.
Human plastin has three isoforms differing in the amino acid sequence. L-plastin is an actin-binding protein that predominantly exists in haematopoietic cells, and the L-form is expressed in the cytoplasm of leukocytes but is absent in normal cells. 11 The T-form is expressed in epithelial and mesenchymal cells derived from solid tissues, but neoplastic cells express both L- and T-forms; 12 the third isoform, I-plastin, is expressed in the small intestine, colon and kidney. 13 L-plastin has been described as a marker for many human cancer cells of non-haematopoietic origin. Lin et al. 14 reported that 68% of epithelial carcinomas and 53% of non-epithelial mesenchymal tumours examined expressed L-plastin. L-plastin plays an important role in leukocyte functions such as regulation of integrin-mediated leukocyte adhesion and activation, and is considered to be an effective marker for cancer.
Antibodies to tumour surface antigens or overexpressed antigens are often admitted, and these so-called autoantibodies can become tumour markers. Brichory et al. 5 have reported that autoantibodies against Annexins I and II are useful markers for the diagnosis of lung adenocarcinoma. We have also reported the corresponding antigens of the autoantibodies that exist in plasma from various tumour-bearing patients such as lung adenocarcinoma 6 and oesophageal cancer. 7
Neto et al. 15 found antibodies against T- and L-fimbrins, phosphorylated plastins in the serum samples of patients with systemic lupus erythematosus (SLE). Using an enzyme-linked immunosorbent assay (ELISA) method, the concentrations of anti T- and L-fimbrins in SLE patients were significantly higher than those of normal individuals. The production of autoantibodies against fimbrins in SLE patients' serum samples may be due to the destruction of fimbrin-containing cells, such as white blood cells, and epithelial and mesenchymal surfaces. In the present study, the autoantibody against L-plastin was also detected in various autoimmune diseases such as SLE and rheumatoid arthritis, but the frequency was not high (5/13 cases). In this study, we detected a WB band specific to the L-plastin antibody at high frequency in patients' plasma of NHL (21/25 cases) in comparison with the other haematological malignancies (1/8 cases). There were no reports concerning autoantibody against L-plastin in the plasma of patients with ML such as NHL. We speculate that the production of the autoantibody against L-plastin in the plasma of patients with NHL is triggered by the excessive production of L-plastin during the activation of B-lymphocytes or a shortened longevity of malignant B-lymphocytes, and so an immune response has induced autoantibody production. It has been suggested that the activation and multiplication of B-cells are the cause of these autoantibodies being produced. Although further investigation is needed to understand the function of these autoantibodies in vivo, L-plastin autoantibodies might be very useful as markers of B-cell proliferating diseases, such as NHL. Of course, a quantitative measurement of the autoantibody against L-plastin is needed for the efficient diagnosis of NHL. That is, establishing an ELISA method to detect the autoantibody to L-plastin may lead to a screening test with diagnostic value. The presence of the anti L-plastin antibody might be a decrease in the proliferation potency and metastasis ability of tumour cells because of a migration defect of the cells in NHL patients having the anti L-plastin antibody. Its presence also exhibits the possibility of becoming a marker for the prognosis of NHL.
The autoantibody to α-enolase was also detected in NHL. Previously, we detected the anti α-enolase autoantibody in the plasma of patients with lung adenocarcinoma. 5 It is not clear why autoantibodies against α-enolase and other proteins involved in cell functions are found in human plasma, but one possible mechanism of α-enolase autoantibody induction is as follows: in the lung adenocarcinoma patients, α-enolase was found to be massively expressed in cancer cells, and thus might have required the autoantibody to neutralize its side-effect. There have been many reports on the presence of α-enolase autoantibody in patients with various diseases, such as Hashimoto's encephalopathy, 16 cancer-associated retinopathy 17 and discoid lupus erythematosus. 18 Therefore, abnormally growing cancer cells may be a potential source of α-enolase as an autoantigen in various cancers.
In conclusion, we first identified a novel autoantibody against L-plastin in the plasma of patients with NHL. This autoantibody may constitute a new diagnostic biomarker for NHL.
Footnotes
Acknowledgement
This work was supported by a 2005–2006 Grant-in-Aid for Scientific Research (17590501) from the Ministry of Education, Science and Culture of Japan.
