Abstract
Influence of the concentration (0–1×10−3M) of Mo(IV) compounds on the electroless deposition of Co–Mo–P alloy from alkaline citrate bath was investigated. The changes of the plating potential, mass of the deposits and hydrogen evolution as well as composition and morphology of the deposits with the concentration of additives were determined. Similar behaviour was observed for all compounds. Increase in the
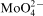 concentration in the bath was accompanied by inhibition of Co–Mo–P deposition and only traces of deposits were obtained at 1×10−3M Mo(VI). Molybdenum content in the deposits was low and oxygen was detected in the layers produced at 5×10−4 and 1×10−3M Mo(VI). Such changes corresponded to a gradual evolution of the morphology of the deposits from needle to fine grained structure, but Na2MoO4 produced deposits of the best quality. Adsorption of
concentration in the bath was accompanied by inhibition of Co–Mo–P deposition and only traces of deposits were obtained at 1×10−3M Mo(VI). Molybdenum content in the deposits was low and oxygen was detected in the layers produced at 5×10−4 and 1×10−3M Mo(VI). Such changes corresponded to a gradual evolution of the morphology of the deposits from needle to fine grained structure, but Na2MoO4 produced deposits of the best quality. Adsorption of
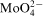 ions at the metal/electrolyte interface was proposed as responsible for phenomena observed in the Co–Mo–P system.
ions at the metal/electrolyte interface was proposed as responsible for phenomena observed in the Co–Mo–P system.
Get full access to this article
View all access options for this article.
