Abstract
Objective:
A pediatric upper abdominal pain evaluation can be challenging due to the broad differential diagnoses. The aim of this study was to evaluate the relationship between abdominal sonography and gold-standard endoscopy findings in a cohort of pediatric patients.
Materials and Methods:
In this cross-sectional study, 40 children undergoing abdominal sonography and upper gastrointestinal endoscopy were evaluated. Sonographic measurements included wall thicknesses, and anatomical dimensions were compared with endoscopic findings that were categorized as esophageal, gastric, or duodenal lesions.
Results:
Of the 40 patients (7.8 ± 4.6 years; 57.5% male), common sonographic abnormalities included increased gastric body (75%) and cardia (57.5%) wall thickness and increased abdominal esophageal length (75%). Endoscopic findings revealed esophageal lesions in 35%, gastric lesions in 32.5%, and duodenal lesions in 20% of patients. No statistically significant associations were found between any sonographic parameters, which included wall thickness, pyloric length, or the angle of His, as well as the presence of esophageal (p = .12–1.00), gastric (p = .20–1.00), or duodenal lesions (p = .31–1.00). Similarly, clinical variables such as age, symptom duration, and prior medication use showed no significant correlation with endoscopic abnormalities (p > .05).
Conclusion:
In this cohort of children, abdominal sonography demonstrated limited diagnostic value in detecting mucosal gastrointestinal lesions for those with upper abdominal pain. This may suggest that abdominal sonography should not replace endoscopic evaluation.
Keywords
The clinical evaluation of upper abdominal pain in pediatric patients presents a diagnostic challenge due to the wide range of possible underlying causes. These may vary from benign functional gastrointestinal (GI) disorders to more serious conditions requiring prompt medical or surgical intervention.1,2 A prompt and accurate diagnosis is essential to choose suitable treatment plans and avoid unnecessary interventions and even invasive procedures like endoscopy.3,4 Thus, improving diagnostic pathways is crucial for enhancing the efficiency of clinical practice in pediatric gastroenterology.3,5
In recent years, sonography has become an increasingly valuable diagnostic tool. This imaging technique provides a noninvasive, nonionizing method to visualize abdominal structures, making it especially beneficial for children who need repeated evaluations.2,6 Its safety record and diagnostic effectiveness have contributed to its widespread use as a primary investigation for children experiencing upper abdominal pain. Sonography is capable of identifying a range of conditions, including cholelithiasis, appendicitis, and intussusception, which might otherwise require more invasive procedures.2,7
Nonetheless, endoscopy remains the diagnostic gold standard in the evaluation of GI diseases, in children. Despite being inherently invasive, endoscopy allows direct visualization of mucosal surfaces and facilitates interventions such as biopsies, which are essential for diagnosing conditions like inflammatory bowel disease, GI bleeding, or suspected neoplasia.3,8
Despite the widespread use of both abdominal sonography and endoscopy in pediatric gastroenterology, there remains a lack of comprehensive assessments exploring the relationship between the findings of these two diagnostic techniques and their respective or complementary roles in clinical practice. To address this gap, the aim of this study was to perform a comparative evaluation between sonographic findings and endoscopic outcomes in pediatric patients with upper abdominal pain, to clarify their diagnostic value and inform more effective, less invasive clinical decision-making.
Materials and Methods
This was a cross-sectional, observational study conducted at a children’s hospital in Iran, for approximately 13 months. Ethical approval for the study was obtained from the ethics committee at Mashhad University of Medical Sciences (IR.MUMS.REC.1401.495). Prior to being entered into the study, informed consent was obtained from the parents or legal guardians of all participants, once the objectives of the study were clearly explained.
The inclusion criteria were set as children with upper abdominal pain who underwent both abdominal sonography and upper GI endoscopy, as part of their diagnostic evaluation. The exclusion criteria included patients with previously diagnosed GI diseases, those with incomplete clinical or imaging data, or those in whom endoscopy was not feasible due to clinical contraindications.
Clinical and Demographic Data Collection
For each patient, demographic characteristics (e.g., age, sex), clinical presentation (e.g., symptom onset time, presence of associated symptoms such as nausea, vomiting, or weight loss), and medication history (e.g., use of H2 receptor blockers and proton pump inhibitors [PPIs]) were recorded using a standardized data collection form completed at the time of admission or outpatient evaluation.
Sonographic Assessment
The same qualified radiologist performed abdominal sonography of all the pediatric patients. All examinations were performed using a WS80A ultrasound equipment system (Samsung Medison, Republic of Korea). A CA1-7A curved array transducer was used for abdominal imaging, and a high-frequency L3-12A linear transducer was applied in younger or thinner children, when higher resolution was required. Patients fasted for at least 6 hours before each examination. Examinations were performed in the supine position, with additional oblique or left lateral decubitus views obtained when necessary. Each parameter was measured in both longitudinal and transverse imaging planes, using electronic calipers placed from the inner to the outer echogenic layers of the GI wall. Two measurements were obtained for each parameter, and the average was recorded. The following parameters were assessed:
Gastric cardia wall thickness (e.g., normal: ≤ 4 mm).
Gastric body wall thickness (e.g., normal: ≤ 3 mm).
Distal esophageal wall thickness (e.g., normal: ≤ 3.2 mm).
Abdominal esophageal length (e.g., normal: ≥ 30 mm).
Pyloric length (e.g., normal: ≤ 15 mm).
Angle of His (e.g., normal: 138 degrees).
Those measurements exceeding or falling below standard thresholds were considered abnormal. The interpretation of diagnostic findings was anonymized for those interpreting the endoscopic results. Representative sonographic images, from the study protocol, are provided (See Figure 1).
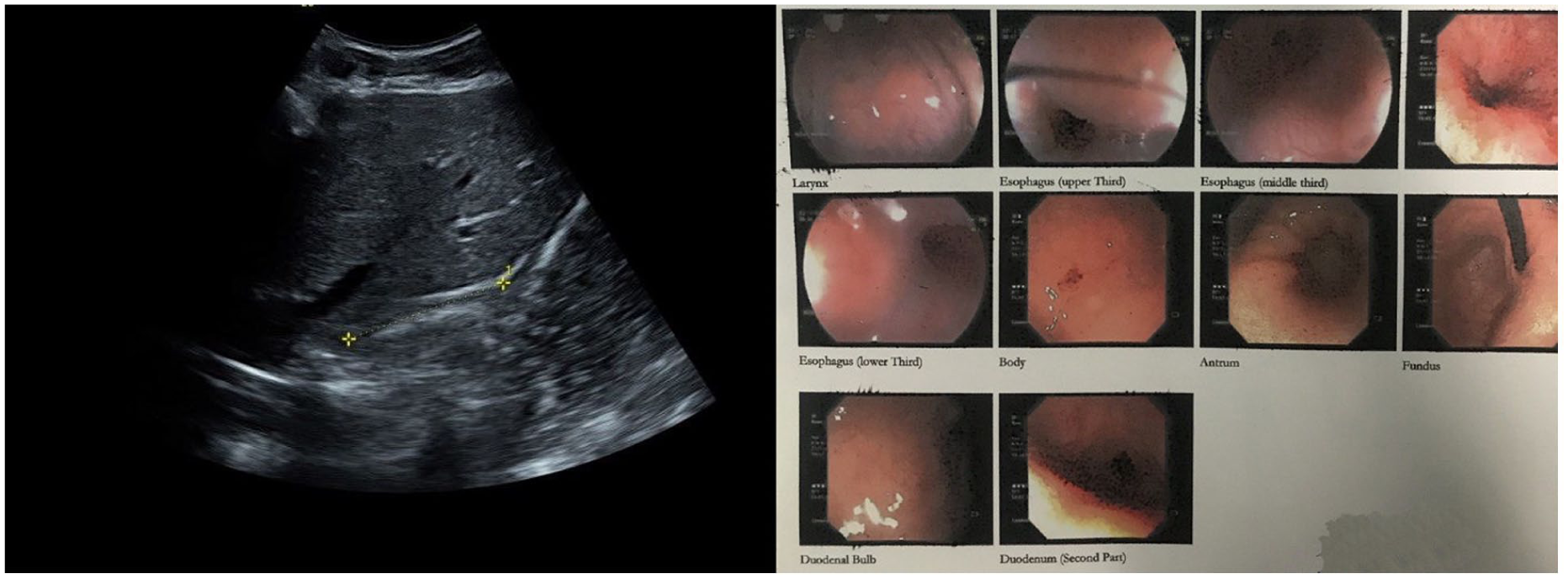
Representative abdominal sonographic and endoscopic images are provided that were obtained during the study protocol (Left: Transabdominal sonogram demonstrating a normal intra-abdominal esophageal length in a child with a complaint of upper abdominal pain. Right: Sequential endoscopic views from the same patient, representing a ring-like pattern in the middle esophagus and a severe stricture in the lower esophageal sphincter, and otherwise normal findings).
Endoscopic Evaluation
Upper GI endoscopy was performed by an experienced pediatric gastroenterologist, following standard sedation protocols. The procedure involved direct visualization of the esophagus, stomach, and duodenum. Lesions were recorded based on macroscopic appearances, categorized as esophageal, gastric, or duodenal abnormalities. Representative endoscopic images from the same diagnostic protocol are provided (See Figure 1).
Statistical Analysis
Data analysis was performed using SPSS statistical software (v23, IBM, Armonk, New York). Continuous variables were expressed as mean ± standard deviation (SD), and categorical variables as frequencies and percentages. Fisher’s exact test was used to assess the association between the presence of endoscopic lesions and clinical or sonographic parameters. A p-value < .05 was set a priori and considered statistically significant.
Results
A total of 40 pediatric patients, presenting with upper abdominal pain, were included in this study. The mean age of the patients was 7.8 ± 4.6 years, comprising 23 males (57.5%) and 17 females (42.5%). Based on Figure 2, the majority of the children fell into two age groups: 6–12 months (38%) and 2–6 months (32%). In 60% of cases, the symptoms had an acute onset. The most frequently reported accompanying clinical feature was nausea and vomiting, observed in 55% of patients. Figure 3 provides the medication history of these patients, and it is evident that only a small proportion of the children had a history of using acid-suppressive agents: 8% reported prior use of H2 receptor blockers (H2Bs), and 28% had a history of PPI consumption.
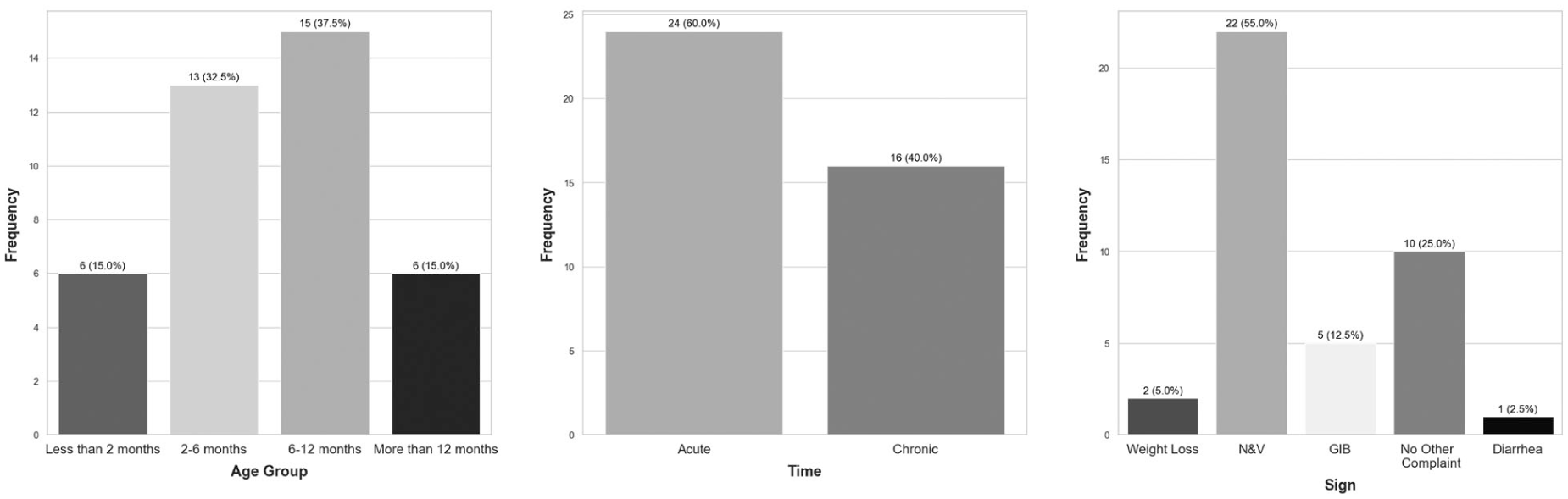
A bar chart is provided of the distribution of age groups, associated symptoms, and symptom onset characteristics in this pediatric cohort.
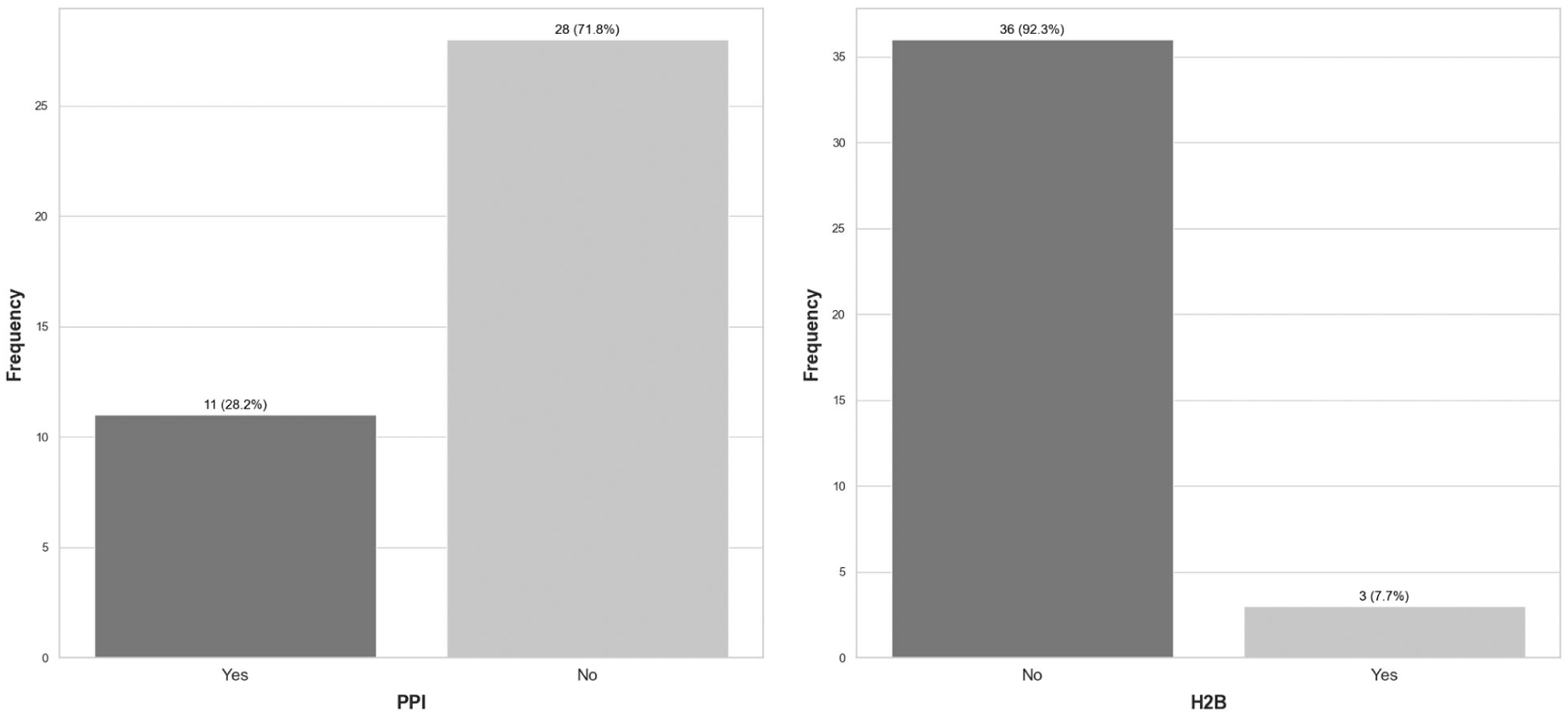
A bar chart provides a depiction of acid-suppressive medication history, specific to this pediatric cohort.
Sonographic evaluation findings depicted in Figure 4 revealed abnormal findings in several anatomical parameters. Increased gastric cardia wall thickness was observed in 57.5% of the children, and increased gastric body wall thickness was present in 75%. Abnormal distal esophageal wall thickness was identified in 65%, and increased abdominal esophageal length was found in 75% of the cases. In contrast, most patients exhibited normal measurements for pyloric length (92.5%) and the gastroesophageal junction angle (i.e., angle of His), which was within normal limits in 77.5% of the children.
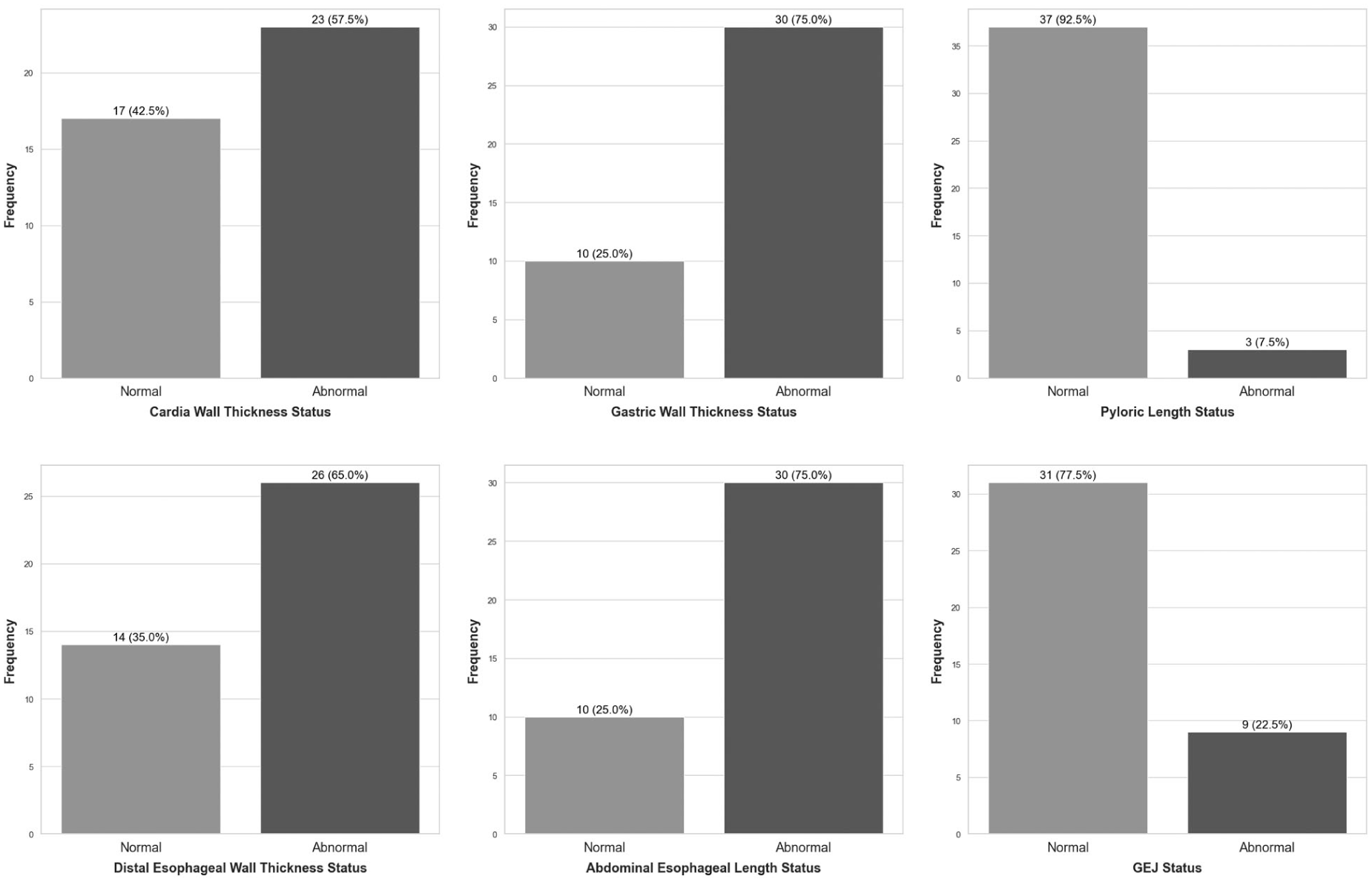
A bar chart illustrates the sonographic findings, within this pediatric cohort.
Esophageal Lesions
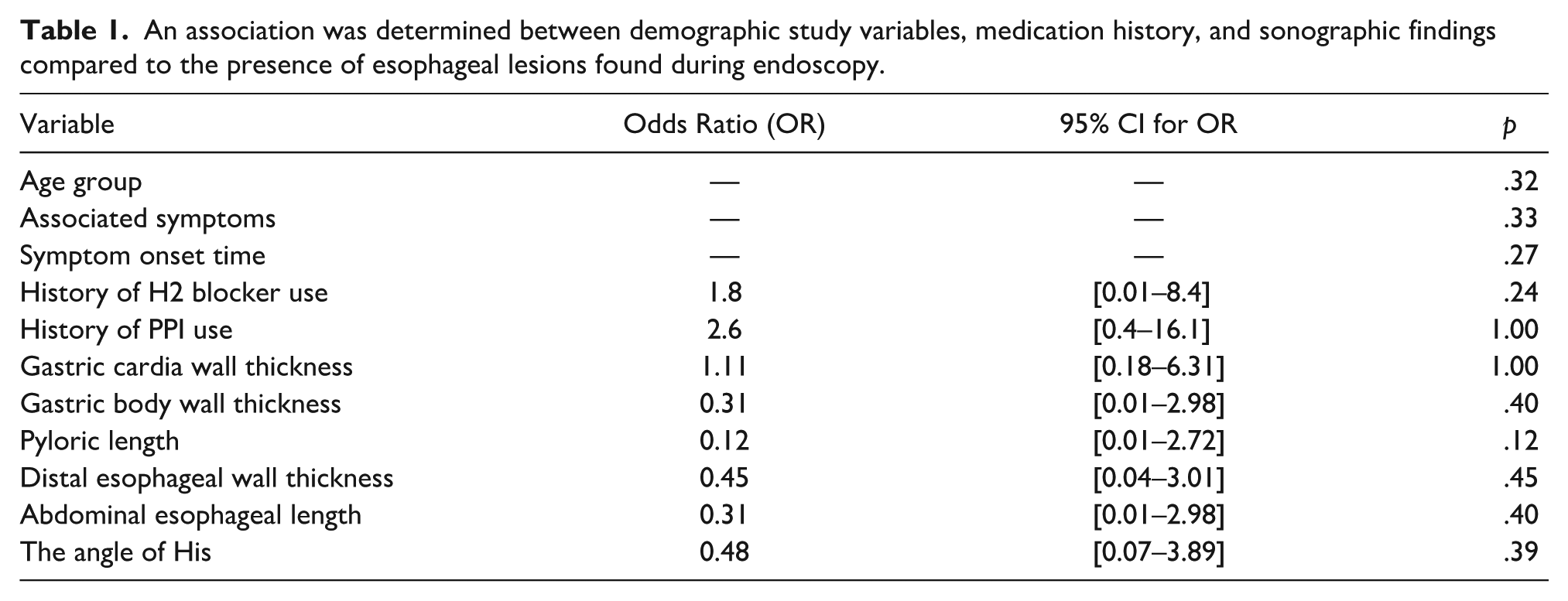
The Fisher’s exact test was used to examine associations between clinical and sonographic variables and the presence of esophageal lesions identified via endoscopy. Table 1 indicates that no statistically significant association was found between esophageal lesions and age group (p = .32), presence of associated symptoms (p = .33), or symptom onset time (p = .27). Similarly, the use of H2 blockers (p = .24) or PPIs (p = 1.00) was not significantly related to esophageal lesions. Sonographic parameters, including gastric cardia wall thickness (p = 1.00), gastric body wall thickness (p = .40), pyloric length (p = .12), distal esophageal wall thickness (p = .45), abdominal esophageal length (p = .40), and the angle of His (p = .39), also showed no significant association with the presence of esophageal abnormalities.
An association was determined between demographic study variables, medication history, and sonographic findings compared to the presence of esophageal lesions found during endoscopy.
Gastric Lesions
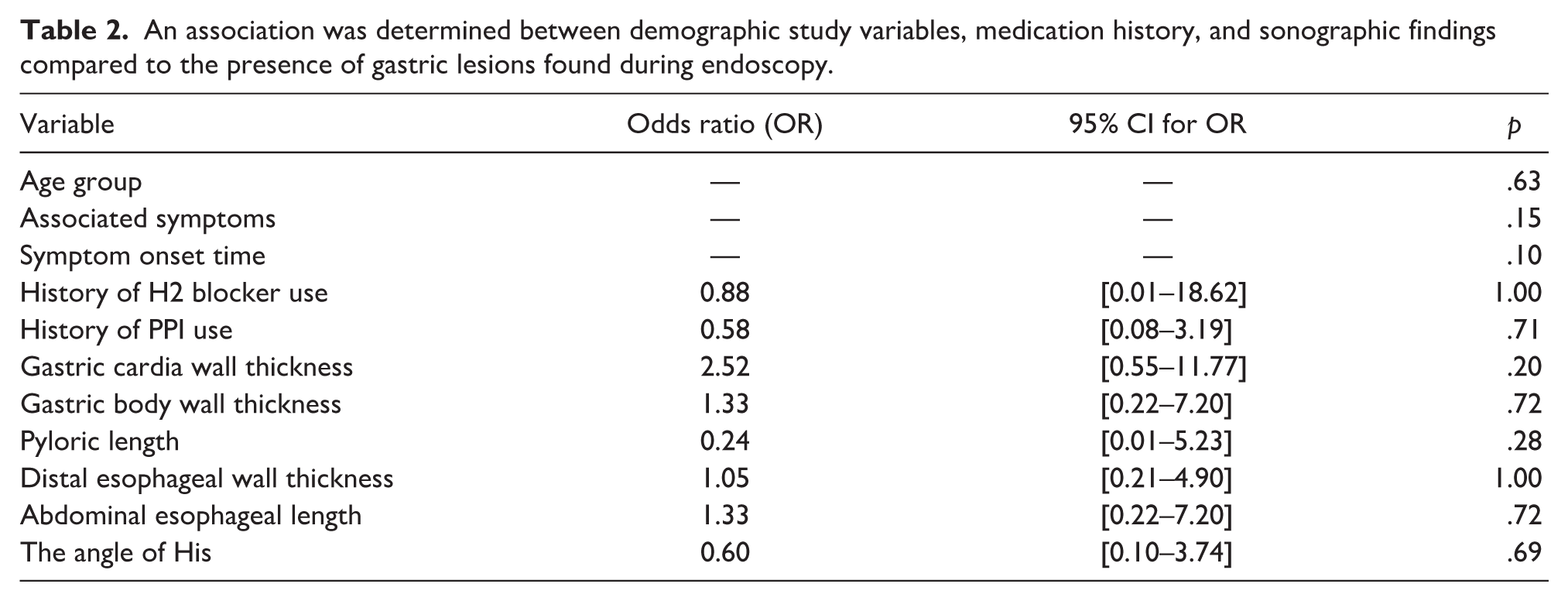
According to Table 2, when assessing the presence of gastric lesions, no significant associations were observed with age group (p = .63), associated symptoms (p = .15), or symptom onset time (p = .10). Neither prior use of H2 blockers (p = 1.00) nor PPIs (p = .71) was significantly related to gastric lesions. Likewise, none of the sonographic findings, including gastric cardia wall thickness (p = .20), gastric body wall thickness (p = .72), pyloric length (p = .28), distal esophageal wall thickness (p = 1.00), abdominal esophageal length (p = .72), and angle of His (p = .69), were found to be significantly associated.
An association was determined between demographic study variables, medication history, and sonographic findings compared to the presence of gastric lesions found during endoscopy.
Duodenal Lesions
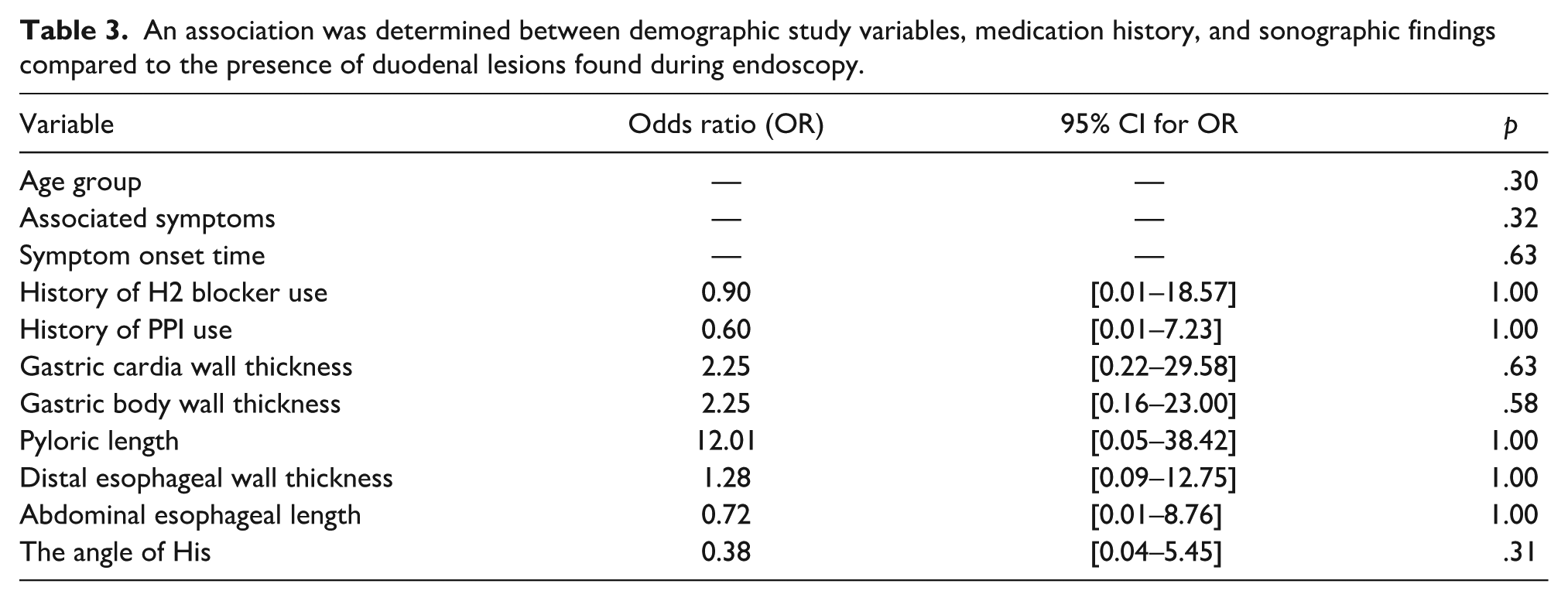
Similarly, Table 3 findings provide no statistically significant relationship between duodenal lesions and age group (p = .30), associated symptoms (p = .32), or duration of symptoms (p = .63). The history of H2 blocker (p = 1.00) or PPI use (p = 1.00) was not associated with duodenal findings. In addition, sonographic parameters such as gastric cardia wall thickness (p = .63), gastric body wall thickness (p = .58), pyloric length (p = 1.00), distal esophageal wall thickness (p = 1.00), abdominal esophageal length (p = 1.00), and angle of His (p = .31) showed no significant associations.
An association was determined between demographic study variables, medication history, and sonographic findings compared to the presence of duodenal lesions found during endoscopy.
Discussion
This study assessed the diagnostic performance of abdominal sonography compared with endoscopy, in a cohort of pediatric patients, presenting with upper abdominal pain. The results showed no statistically significant association between abnormal sonographic measurements, such as gastric or esophageal wall thickness, and the presence of mucosal lesions identified through endoscopy. The findings with this pediatric cohort would suggest that routine abdominal sonography, as applied here, may provide only limited sensitivity for detecting upper GI lesions, particularly when relying solely on direct anatomical measurements.
The clinical evaluation of upper abdominal pain in children poses considerable diagnostic challenges due to the wide range of differential diagnoses, from benign functional disorders to significant organic pathologies.9,10 Although sonography is routinely employed as a primary imaging technique due to its noninvasive nature, nonionizing radiation, and accessibility, its diagnostic correlation with endoscopic findings remains unclear.11-13
The results are consistent with earlier studies that have questioned the standalone utility of conventional sonography in diagnosing mucosal GI disease. Imran et al. reported an accuracy of less than 50% when evaluating upper abdominal pain in patients using sonography. 14 Lim et al. reported a sensitivity of 65% and specificity of 91% for detecting duodenal ulcers based on a duodenal wall thickness threshold of >5 mm. While these findings underscore the high specificity of sonography, the relatively low sensitivity suggests that a normal sonogram cannot reliably exclude significant pathology, particularly in early or subtle mucosal disease. 15 Another study by Lee et al., 16 which focused on the role of transabdominal sonography, in diagnosing pediatric peptic ulcer disease, reported sensitivity rates of 66.7% for gastric ulcers and 38.9% for duodenal ulcers, which are comparable to the relatively modest sensitivity.
Numerous studies demonstrated that conventional sonograms lack the precision of endoscopy in the diagnosis of upper GI lesions. Therefore, several studies aimed to analyze the combination of adjuvant anatomic markers and imaging modifications to enhance sonography’s capability in the evaluation of GI tract disorders. Comparatively, Hosokawa et al. retrospectively assessed the diagnostic accuracy of direct and indirect sonographic findings in 244 pediatric patients undergoing endoscopy. They reported that direct signs, such as increased gastroduodenal wall thickness, had a sensitivity of 60.0% and a specificity of 98.1%, while indirect signs, such as surrounding inflammatory changes and lymphadenopathy, had a higher sensitivity of 80.0% and a specificity of 97.2%. 17 This was consistent with the findings, as the results indicated that wall thickening alone is an unreliable marker of peptic ulcer in children. However, unlike Hosokawa et al., the present study did not systematically assess indirect sonographic features, which limits the ability to fully evaluate the diagnostic potential of ultrasonography in this context.
In addition, there is constantly increasing evidence regarding the role of endoscopic ultrasonography (EUS) in the evaluation of pediatric upper abdominal pain, particularly pancreatobiliary system disorders.18,19 Even though invasive, EUS combines the benefits of both upper endoscopy and abdominal sonography, making it demonstrate diagnostic and therapeutic advantages over transabdominal sonography. 20 Studies have shown that EUS has equivalent or even superior performance compared with combined upper endoscopy and transabdominal sonography and is also more advantageous regarding the cost-effectiveness. 21 It is found that EUS was more sensitive than transabdominal sonography in diagnosing pancreaticobiliary disorders. 22 However, other findings highlight the limitation of EUS in the evaluation of appendicitis or genitourinary diseases, as well as its invasive nature and inability to provide a rapid initial examination. 21 Furthermore, findings from a randomized controlled trial comparing combined upper endoscopy and EUS with upper endoscopy and transabdominal sonography to determine the etiology of upper abdominal pain revealed no statistically significant difference between the two approaches. The diagnostic rates were 64% and 62%, respectively, necessitating the need for further studies to support the superiority of EUS over transabdominal sonography for the examination of upper abdominal pain. 20
A recent study by Zhang et al. evaluated the performance of GI contrast-enhanced ultrasonography (CEUS) versus routine sonography and gastroscopy in diagnosing peptic ulcers in children. In their cohort of 43 symptomatic children, routine sonography detected only 8 of 17 gastroscopically confirmed ulcers, with a diagnostic concordance rate of 79.1% and significant discrepancy compared to endoscopy. In contrast, CEUS achieved a much higher concordance rate of 95.3%, identifying 15 of the 17 ulcers, with no statistically significant difference from gastroscopy. These findings reinforce the limitations of conventional ultrasound in diagnosing mucosal lesions, consistent with the present study, which also demonstrated poor correlation between sonographic wall thickening and endoscopic mucosal pathology. However, Zhang et al. provide compelling evidence that CEUS, through its ability to delineate ulcer morphology, location, and perfusion in real-time, may significantly improve diagnostic yield. The CEUS was not utilized in this study, and the absence of a contrast agent may partially explain the low sensitivity of current ultrasound assessments. Thus, while both studies affirm that standard sonography alone is insufficiently accurate for mucosal disease detection, Zhang et al. 23 suggest that CEUS could serve as a valuable non-invasive adjunct, potentially bridging the diagnostic gap between sonography and endoscopy.
Limitations
The major limitation was related to the study design with threats to internal and external validity. In addition, the selection of participants was by convenience, which resulted in a relatively small sample size, which limits statistical power and generalizability. In addition, sonographic interpretation is inherently operator-dependent, and although all sonograms were performed by an experienced radiologist, inter-observer variability could not be assessed. Finally, histopathological confirmation was not uniformly performed for all endoscopic findings, which may have impacted diagnostic accuracy. Future studies with larger, multicenter cohorts and standardized ultrasound protocols are warranted to better define the role of sonographic findings in pediatric GI diagnostics. In addition, exploring advanced imaging techniques such as contrast-enhanced ultrasound, endoscopic ultrasound (EUS), or elastography may offer greater specificity for detecting mucosal and submucosal pathology.
Conclusion
In summary, transabdominal sonography offers several benefits, including noninvasiveness, affordability, non-ionizing radiation, accessibility, and suitability for initial evaluation in children. However, the results of this study may indicate a limited concordance between sonographic findings and endoscopic lesions. The present cohort study results indicated that transabdominal sonography alone is insufficient for definitive diagnosis of upper abdominal pain. Upper endoscopy remains the standard for definitive evaluation, while EUS may provide improved diagnostic accuracy in selected pediatric conditions, which warrants further investigation.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval for this study was obtained from the local ethics committee of Mashhad University of Medical Sciences (ethics registry number: IR.MUMS.REC.1401.495).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
