Abstract
With advances in technology and applications, the utility of echocardiography is progressively increasing. Developed to assess acute and chronic cardiac pathology, echocardiography is a steadfast component in a physician’s imaging arsenal to detect cardiovascular disease more effectively and efficiently. Specifically, assessment of the pericardial cavity is conducted with echocardiography in routine and emergent scenarios, due to its rapid availability. Cardiac tamponade, which is the large accumulation of pericardial fluid, is life-threatening if not discovered immediately. Sonographers are paramount in assessing cardiac tamponade because they are at the forefront of a patient’s initial encounter. Cardiac tamponade can be an intimidating finding to even the most experienced sonographer. Therefore, familiarity with this pathology, what to expect, and how to assess is vital in ensuring all the information is acquired for interpreting cardiologists. This case study highlights cardiac tamponade and the sonographers’ role during initial discovery and surveillance, as demonstrated in this patient case.
Transthoracic echocardiography (TTE) is a primary tool in evaluating and assessing pericardial disease.1,2 A pericardial effusion refers to fluid accumulation in the pericardial sac between the epicardium (visceral layer) and the parietal layer of the serous pericardium. 3 The parietal layer and the fibrous pericardium comprise the fibrous double-walled sac surrounding the heart in the mediastinal space. The amount of fluid found in this cavity can range from physiologic to severe amounts with tamponade physiology. Early detection and serial echocardiography are paramount in preventing further cardiovascular deterioration, due to cardiac tamponade, 4 as it can lead to death. Sonographers play a key diagnostic role in providing patients with best practices and timely cardiac care. This is facilitated by the sonographer’s understanding the echocardiographic presentation of cardiac tamponade patient symptoms, providing expedient results to the provider, and the necessity of standardized serial echocardiography.
Case Report
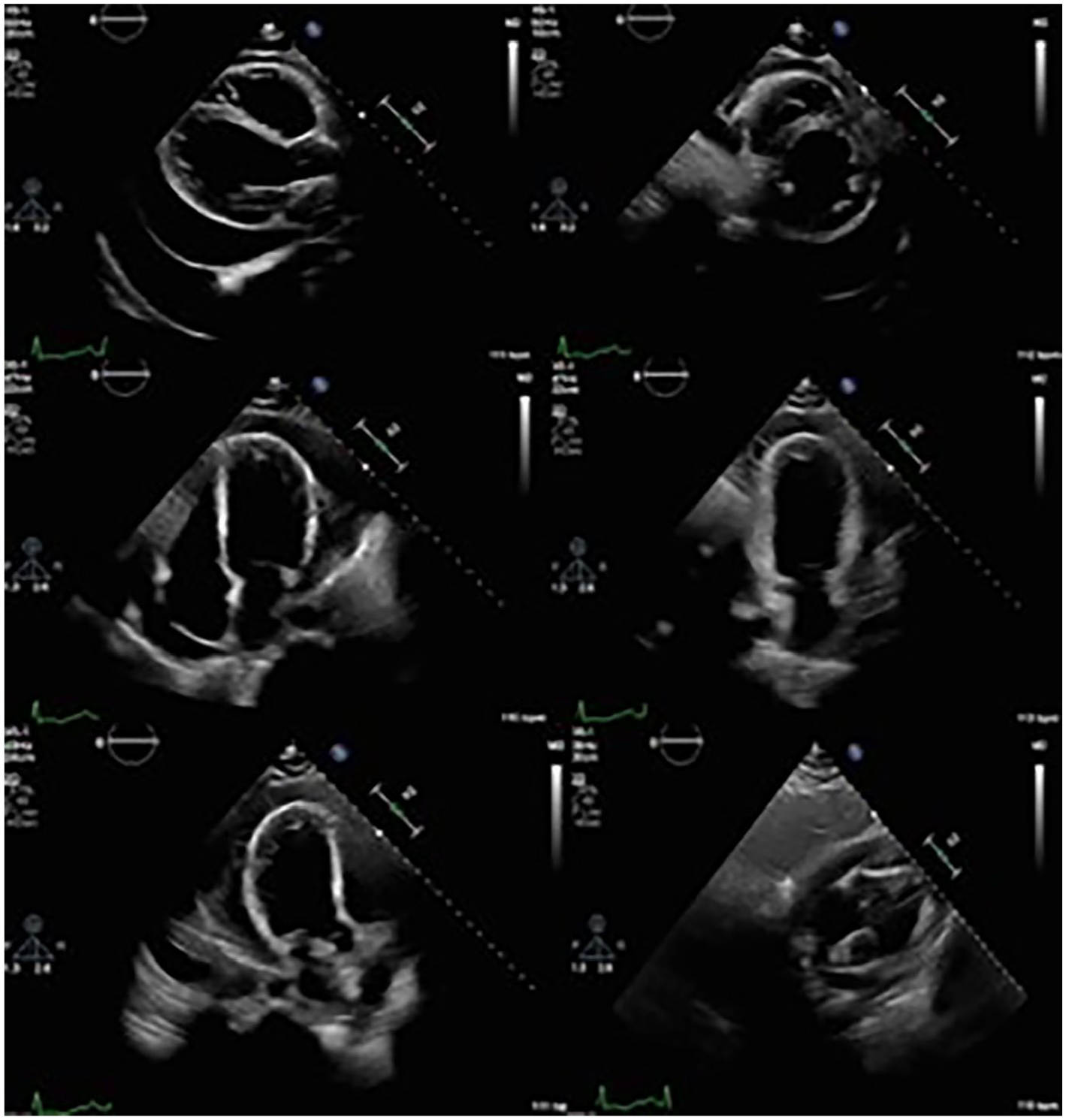
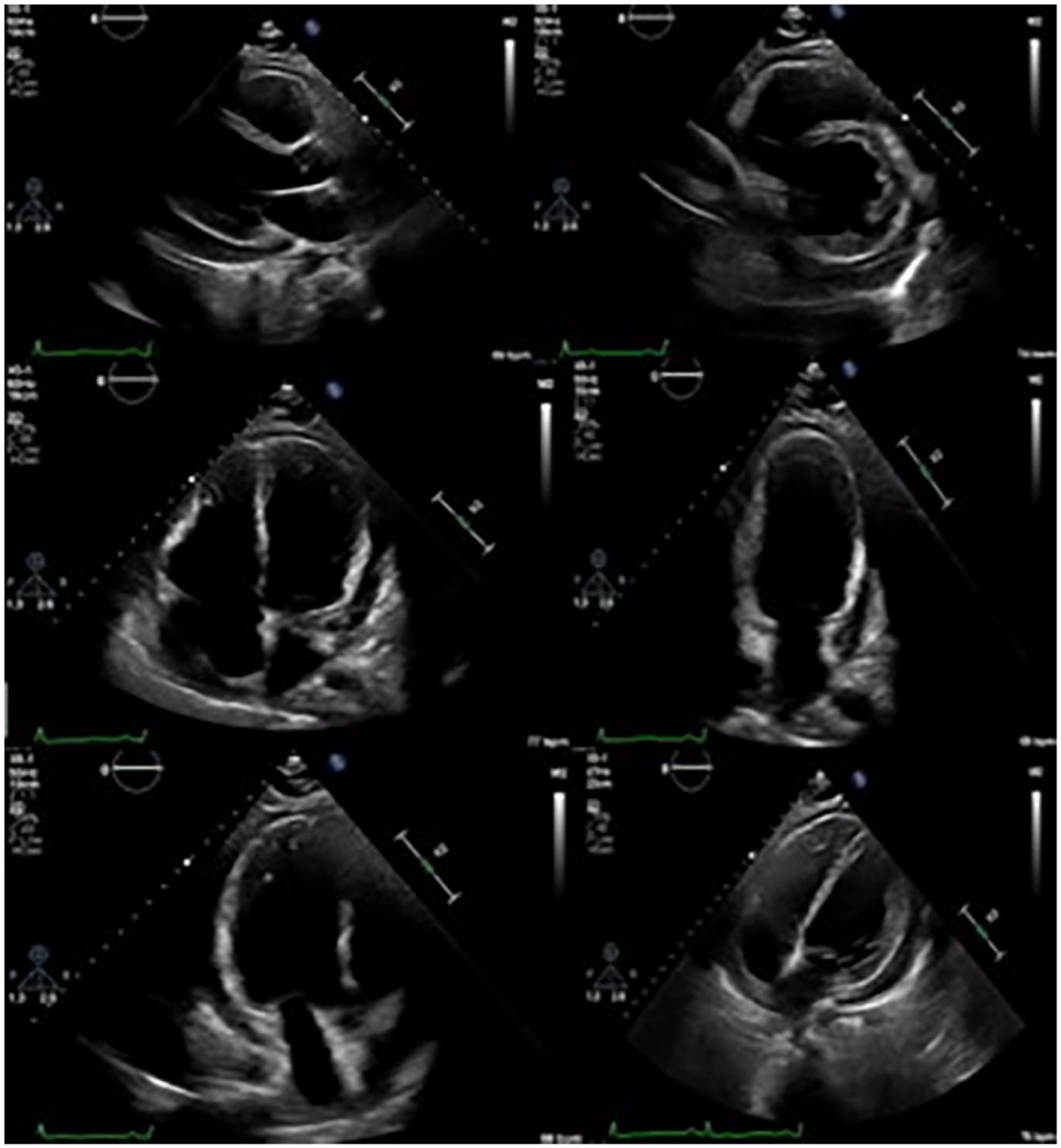
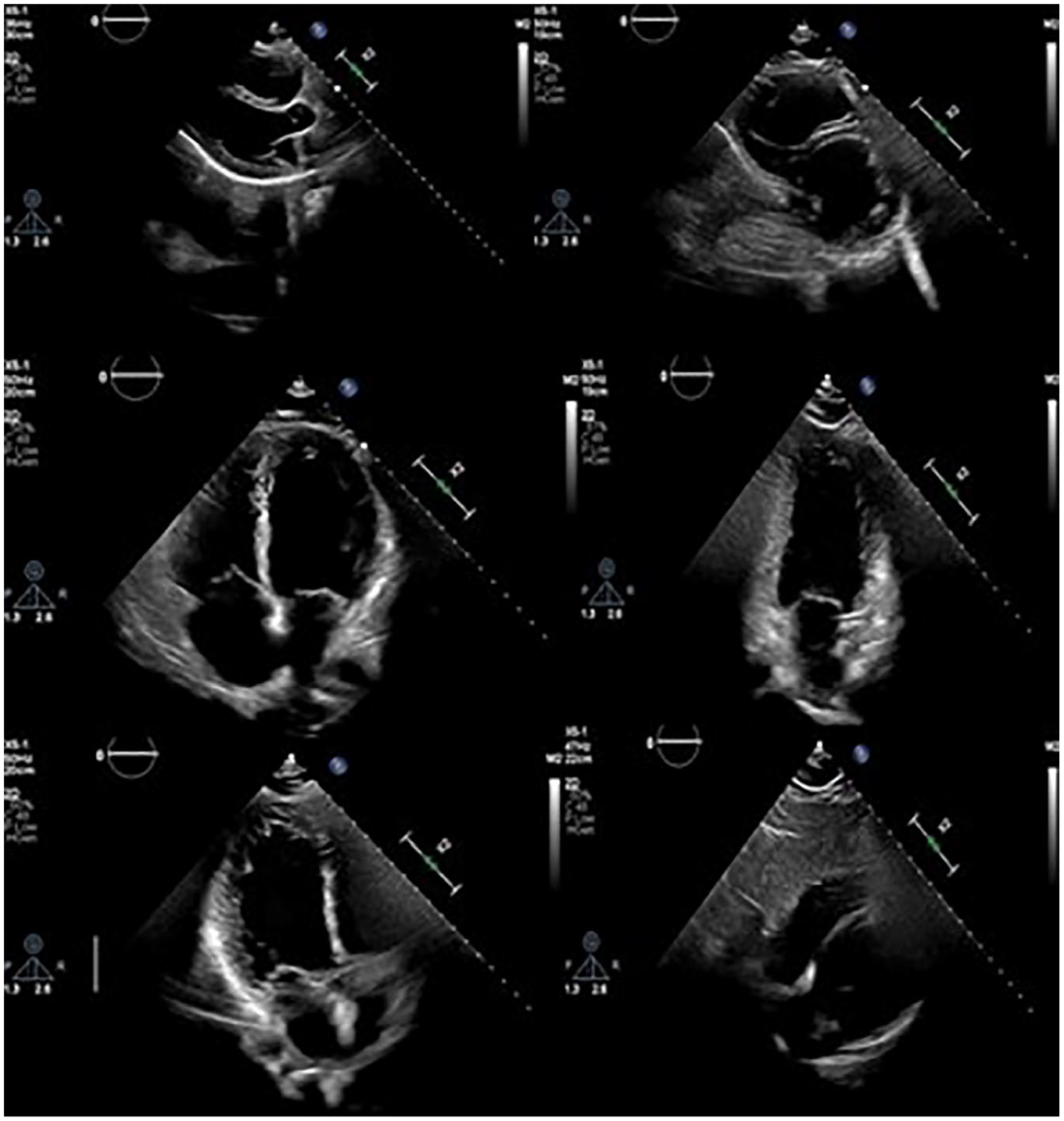
A 23-year-old man presented to the emergency department (ED) with hematemesis, diarrhea, leg swelling, and proteinuria. His blood pressure (BP) was 133/89, and a heart rate (HR) of 108 bpm (beats per minute). The attending physician ordered a complete formal TTE due to these initial findings and his symptoms. The TTE demonstrated normal left ventricular size and function with a visually normal left ventricular ejection fraction (LVEF), due to limited visualization. A large circumferential pericardial effusion (with septations) was apparent, measuring 5 cm (See Figure 1). Diastolic collapse of the right ventricle (RV) and respiratory variation across the mitral valve (MV) were consistent with cardiac tamponade physiology (See Figures 2 and 3). The inferior vena cava (IVC) was measured as dilated with <50% inspiratory collapse with an estimated right atrial (RA) pressure of 15 mm Hg (See Figure 4). Due to the patient’s symptoms and echocardiography findings, based on recommended measurements from Mitchell et al, 5 the sonographer immediately escalated the patient’s findings, to the primary care team for pericardiocentesis. If complete TTE studies are delayed, due to patient symptoms and diagnostic findings, sonographers can annotate a statement on their screen like, “exam halted to notify physician of critical finding,” to expedite the patient’s care. This practice is important for sonographers to implement as time is of the essence in these scenarios and transparency with the interpreting physician is crucial.
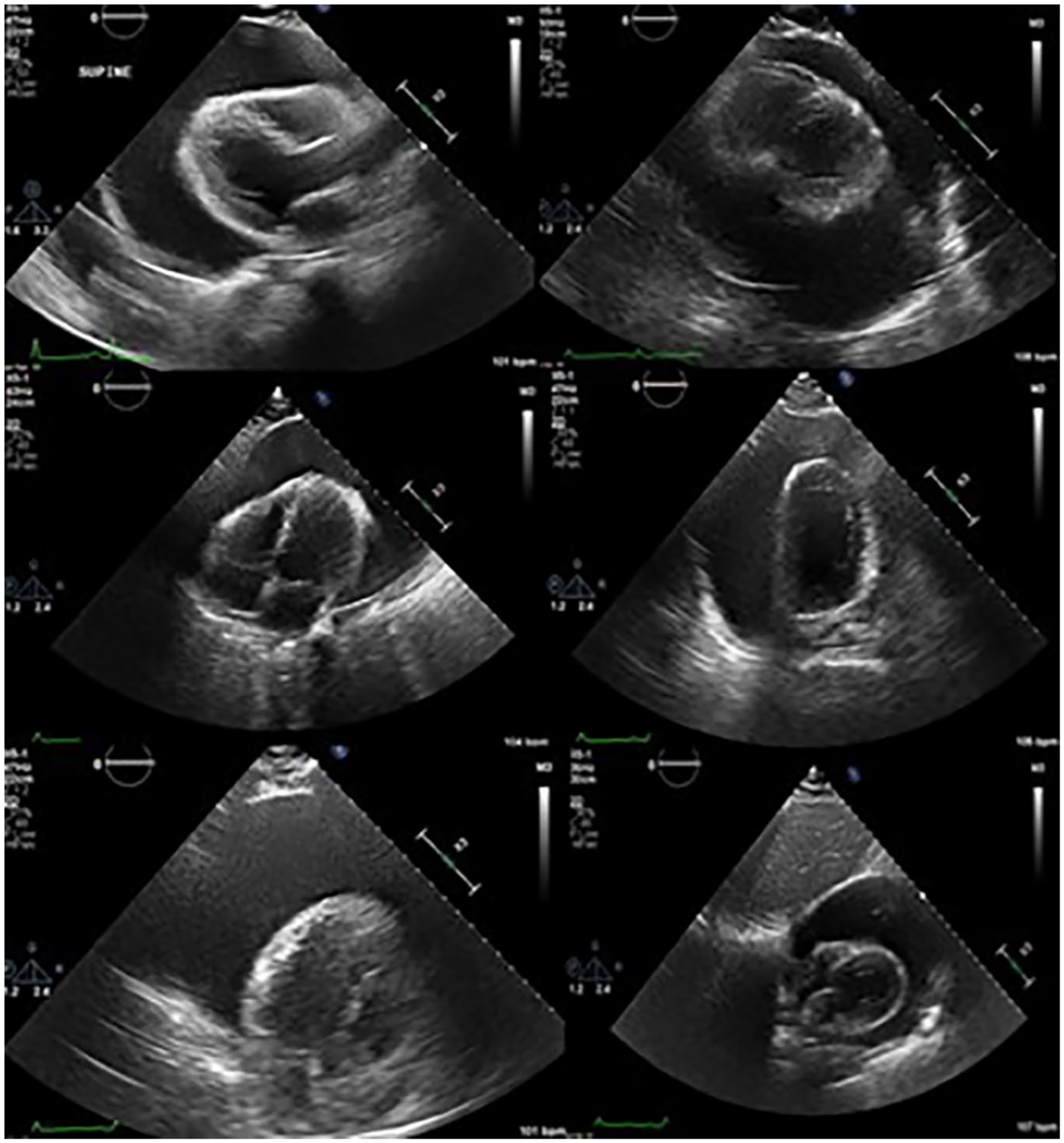
A 2D echocardiographic assessment is provided that demonstrated cardiac tamponade in the PLAX, SAX, AP4 chamber, AP2 chamber, AP3 chamber, and subcostal SAX windows.
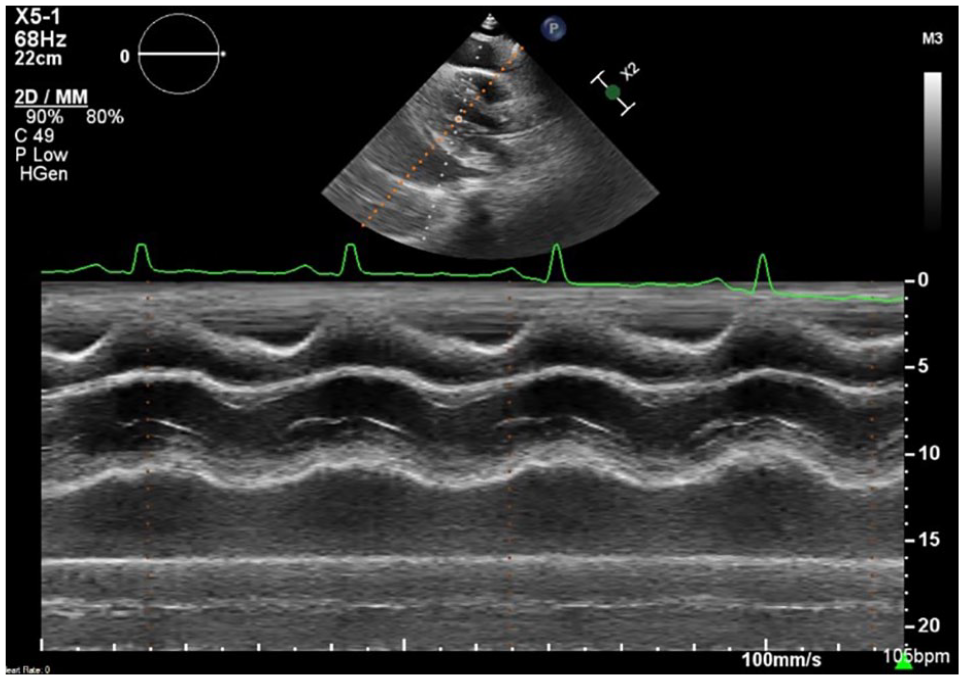
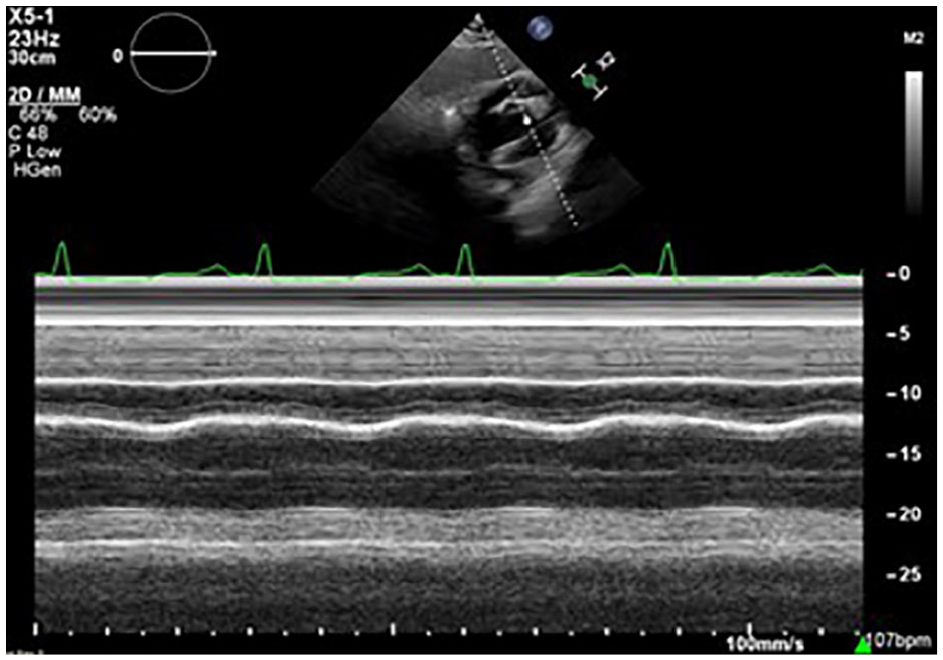
A PLAX view allowed for utilizing M-mode through the RV demonstrating diastolic collapse caused by significant fluid accumulation in the pericardial sac.
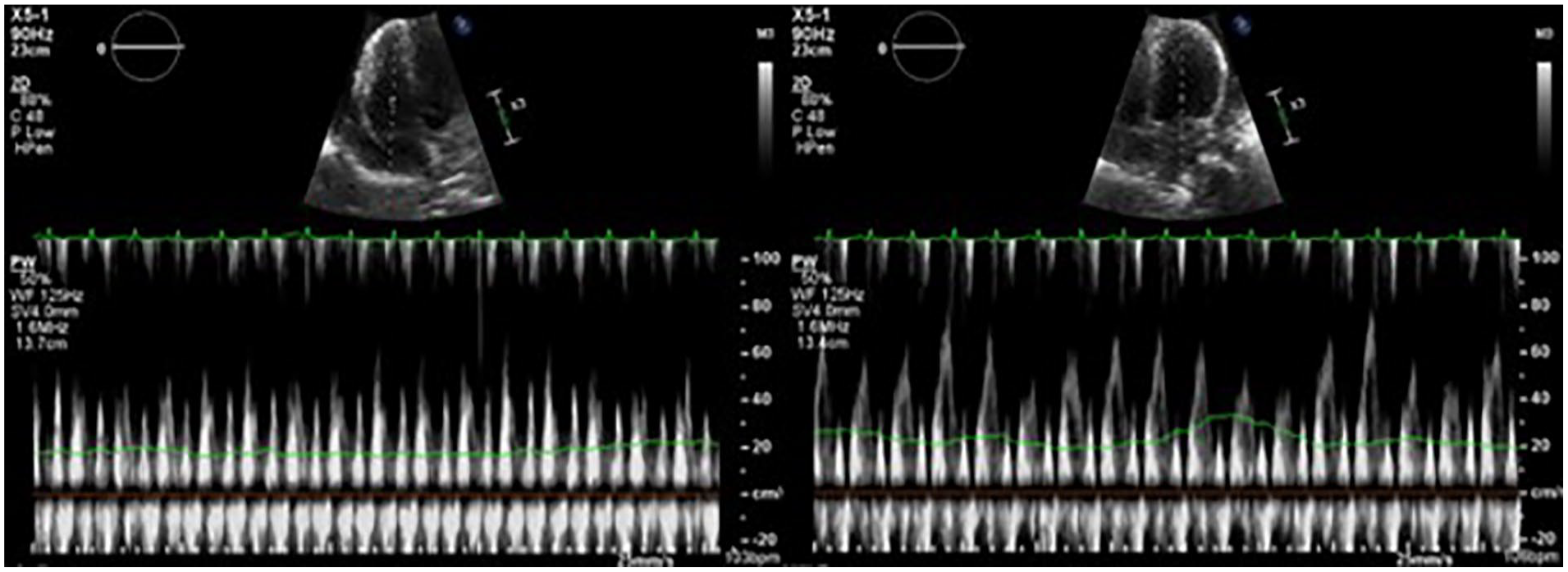
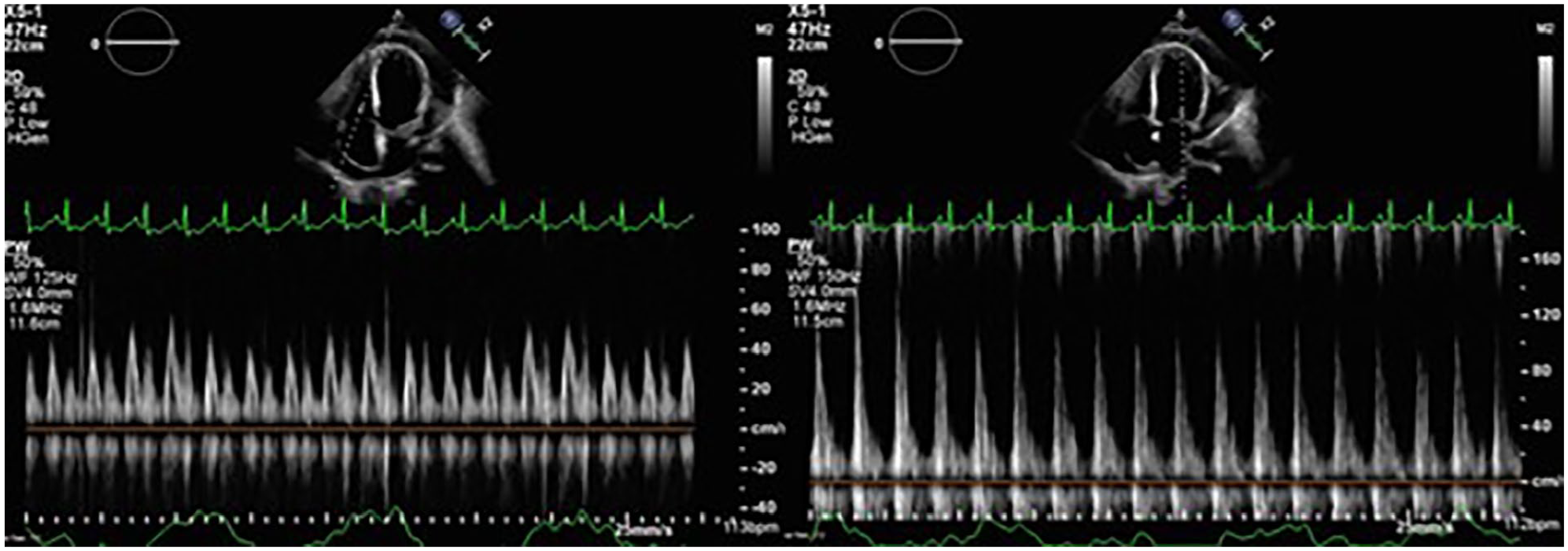
The MV and TV inflow velocities were taken from the apical 4 chamber window, with associated respiratory variation, which indicated cardiac tamponade physiology.
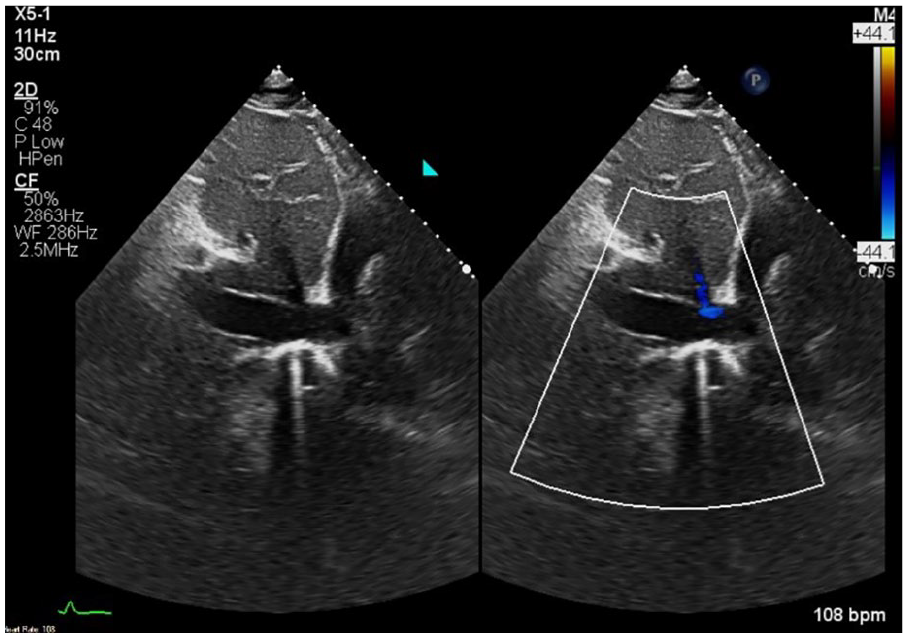
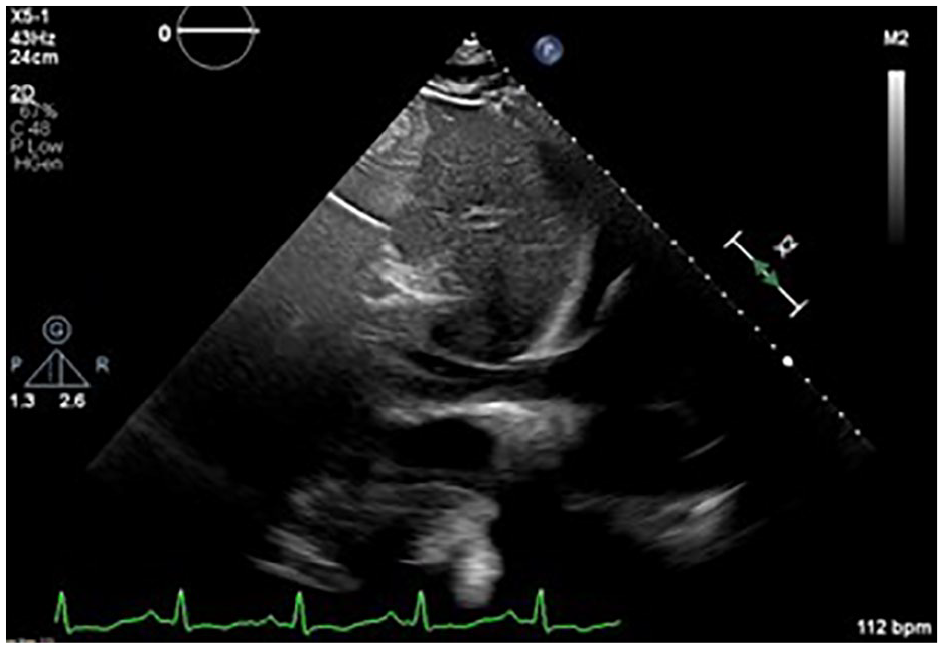
Subcostal window demonstrating a dilated IVC with <50% inspiratory collapse with an estimated right atrial (RA) pressure of 15 mm Hg.
After expedited delivery of results to the ordering providers, the primary care team transferred the patient to the cardiac catheterization laboratory. A emergency pericardiocentesis with fluoroscopy and sonography guidance removed 1.5 L of bilious colored fluid. For comparison, the pericardial sac usually contains 20 mL of fluid which facilitates cardiac motion. 6 After the successful pericardiocentesis, the interventional cardiologist sutured a pericardial drain, which is vital for preventing fluid reaccumulation. 7
Two days later, a limited post-procedure repeated TTE demonstrated a normal LV size with global hypokinesis and an LVEF of 43% by Simpson’s biplane. The sonogram showed a dilated RV with mildly reduced function and a large loculated pericardial effusion. The pericardial effusion measured 2.7 cm, more prominently visualized posteriorly; however, no signs of tamponade physiology were evident (See Figure 5). TV and MV inflow patterns revealed a mildly exaggerated respirophasic variation that aligned within the range of constriction (See Figure 6). The TTE report noted that the IVC as dilated with >50% inspiratory collapse, consistent with an RA pressure of 8 mm Hg (See Figure 7). The diastolic collapse of the RV was no longer persistent (See Figure 8), but due to potential constriction concerns, reasonable analysis required cardiac magnetic resonance imaging (cMRI). The cMRI results revealed a large pericardial effusion with septal flattening during inspiration (suggestive of ventricular interdependence) and mild diffuse thickening of the pericardium with enhancement. The diagnostic findings from the TTE and cMRI were consistent with a constrictive-effusive pericarditis pattern.
A limited post-procedure follow-up TTE demonstrated global hypokinesis, a dilated RV, and a large loculated pericardial effusion.
The MV and TV inflow patterns revealed a mildly exaggerated respirophasic variation that aligns within the range of constriction.
A subcostal window demonstrated a dilated IVC with >50% inspiratory collapse, consistent with an RA pressure of 8 mm Hg.
The M-mode tracing completed through the subcostal window demonstrated resolution of diastolic collapse of the RV.
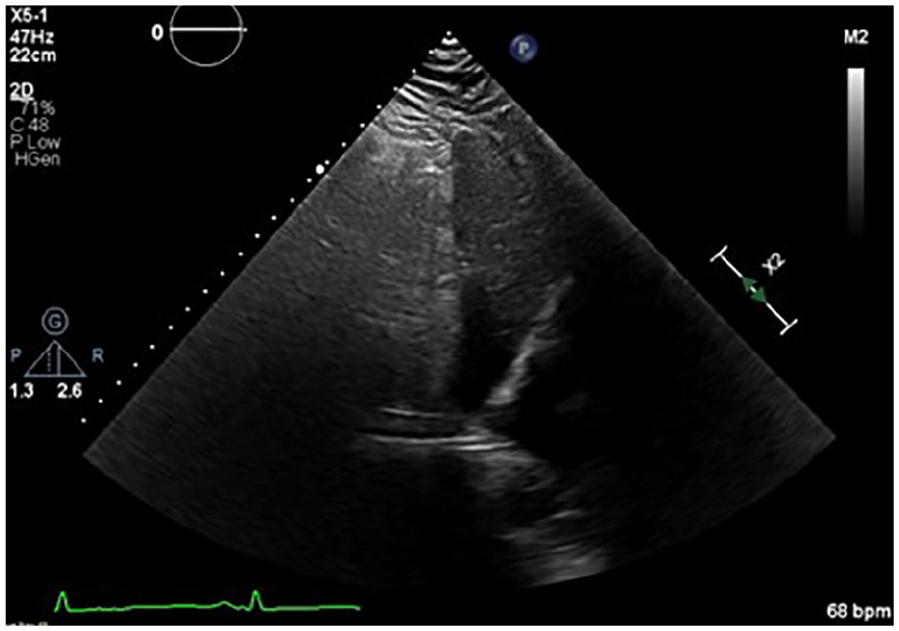
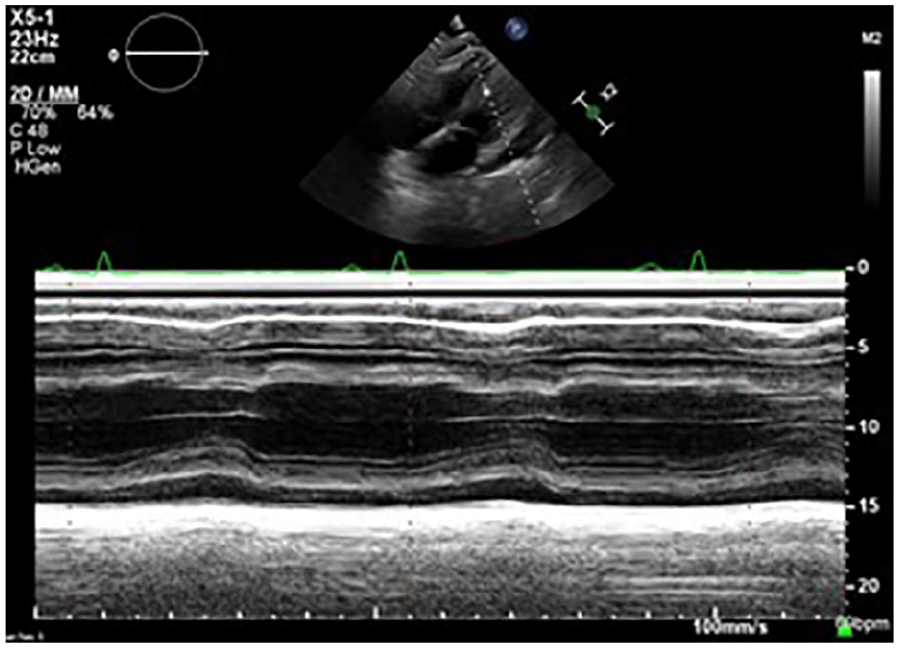
On the sixth-day post-procedure, an interval TTE was performed for possible repeat pericardiocentesis, demonstrating a dilated, hypokinetic RV and global LV hypokinesis with an LVEF of 45% to 50%. A small-to-moderate, loculated pericardial effusion measuring 1.2 cm was evident, predominantly posterior (See Figure 9). The IVC was normal, with >50% inspiratory collapse, consistent with normal RA pressure (See Figure 10). There was no RV diastolic collapse or respirophasic variation across the MV and TV (See Figures 11 and 12). The findings on this TTE demonstrated no signs of cardiac tamponade physiology. A final interval TTE, 2 months later, demonstrated complete resolution of pericardial effusion and removal of pericardial drain (See Figure 13). Normalization of RV and LV function returned; however, echocardiographic findings, consistent with known constrictive physiology, remained. These will be discussed later in this case study. It is also important to note that each interval TTE performed was ordered as a limited exam.
An interval TTE demonstrated a dilated, hypokinetic RV and global LV hypokinesis with a small to moderate, loculated pericardial effusion.
A subcostal window demonstrating a normal IVC size with >50% inspiratory collapse, which is consistent with a normal RA pressure.
The M-mode tracing revealed no evidence of RV diastolic collapse.
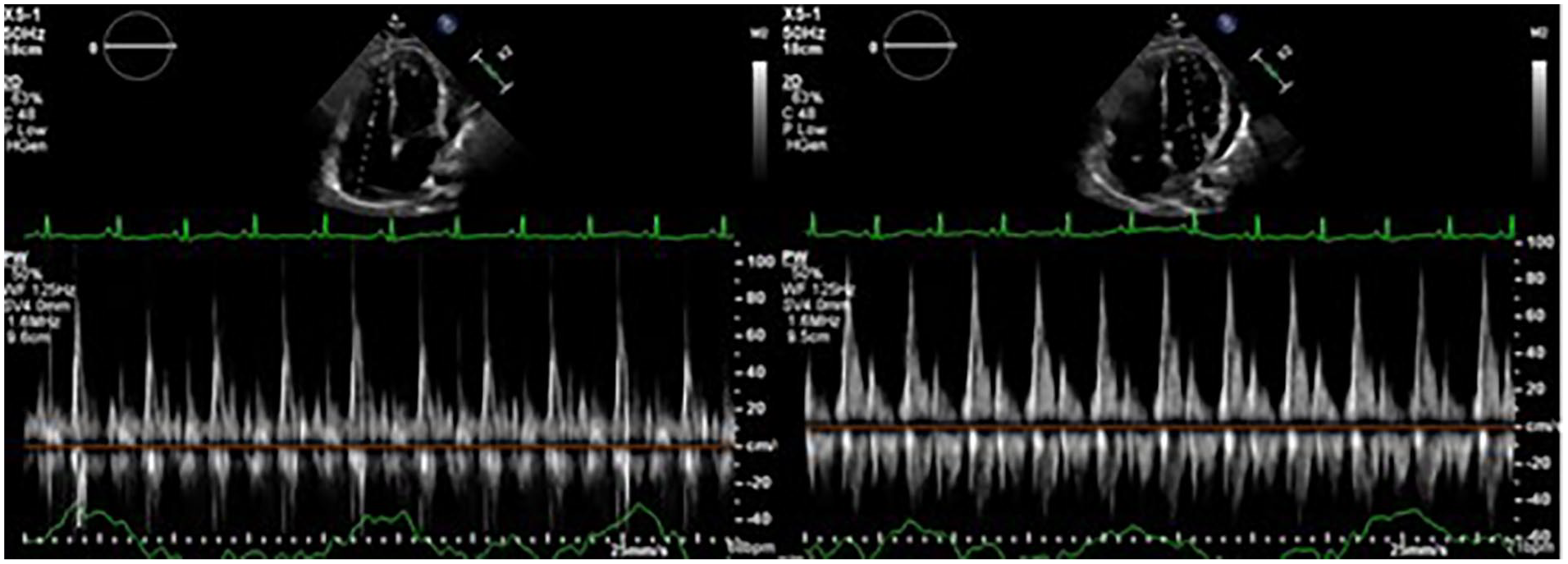
The MV and TV inflow patterns demonstrating no evidence of respiratory variation.
A final interval TTE which demonstrated normalization of RV and LV function and complete resolution of pericardial effusion.
Discussion
Healthcare professionals have exponentially applied sonography, to triage patients, in critical care setting. 8 One specific emergency condition that needs immediate intervention is cardiac tamponade. Providers consider cardiac tamponade when a history and clinical review, specifically Beck’s triad (hypotension, jugular venous distension, and muffled heart sounds), qualifies such a possible crisis.9,10 In this case study however, the patient’s care team did not provide a comment on the presence of these clinical signs. Although symptoms are like cardiogenic or obstructive shock,11,12 echocardiography plays a vital role in the confirmation of patients with cardiac tamponade. Cardiac sonography is utilized when diagnosing tamponade, as a guidance tool during pericardiocentesis, and for surveillance, during the post-pericardiocentesis period, with serial echocardiography. This case study demonstrates a practical application of echocardiography throughout the patient’s care continuum, in addition to a quick tip sheet to reference in the presence of cardiac tamponade (See Table 1).
A Quick Diagnostic Tip Sheet Is Provided for Sonographers, as a Reference for Imaging Patients With a Suspected Tamponade.

Beck’s triad has at least one sign that keen sonographers can utilize while preparing to start the exam: blood pressure recorded by the sonographer at the time of the study and any study they perform. Noting hypotension on a demographic page or annotating it on the display screen equips physicians with more data to manage the patient. Reading the most recent history and physical can answer two additional diagnostic questions: is there noted jugular venous distension, and did the physician hear muffled heart sounds? Knowing some or all of Beck’s triad will help determine the appropriate acoustic window to start the exam.
In the presence of tamponade, cardiac hemodynamics will dictate echocardiographic findings. 13 Even when beginning the echocardiogram in the parasternal long-axis window, if there is significant pericardial effusion visible, which is not a fail-safe predictor of hemodynamic compromise, 14 and hypotension, as the exam commences. This should trigger a move to subcostal window for quick assessment of the RV free-wall and IVC, as well as escalating these findings through a lab-set protocol. My expediting these findings, to the appropriate interpreting physician, saves time as physicians can compare the patient’s clinical symptoms with echocardiographic findings, while notifying health care teams and departments of the patient’s status, as the sonographer completes the formal echocardiogram. The judgment on where and how to perform a pericardiocentesis is based on the status of the patient and the availability of diagnostic imaging techniques. 15
The initial formal echocardiogram allows the sonographer and interpreting physician to compare structural and physiologic changes, post-pericardiocentesis. Improvements in the IVC (diameter, collapsibility, and respiratory variations), as well as measuring the fractional area change, RV base, and mid-ventricle using an RV-focused view, quantifies structural and physiologic changes. Doppler of the RV inflow, outflow, and right ventricular systolic pressure (RVSP) will also demonstrate improvement in the right ventricle quantitative values as the hemodynamic burdens brought on by excessive pericardial fluid build-up resolve. Presenting to the ED with the physical symptoms noted above, fails to direct a cardiac sonographer to suspect pericardial effusion before starting the exam. Peripheral edema could suggest the possibility of some level of heart failure. Thus, reviewing the patient’s most recent history and physical may reduce the initial surprise of detecting a 5-cm effusion, filling the pericardial space, during the initial exam.
Obtaining high-quality M-mode images of the RV free wall, from any tomographic plane where the heart and free wall are visible, as well as using angle adjustments, provides systolic and diastolic motion imagery through the cardiac cycle. The visualization of external pressure compressing the free wall toward the interventricular septum, in diastole, argues a case for hemodynamic compromise. 16 Adding respiratory tracings to long sweep pulse wave Doppler interrogations of the mitral and tricuspid valves further shows the heart struggle against the increasing external pressure from accumulating fluid in the pericardium. It may also indicate an inability to maintain adequate low pressure, during diastole, to move blood from atria to ventricle. 17
Demonstrating a notable pericardial effusion, respiratory variations in the ventricle inflow Doppler, and diastolic collapse of the RV free wall, is indicative of tamponade. 18 Imaging the IVC and RV from the subcostal window has proven to be most valuable in relaying findings to the interpreting physician and care team. Often, the first concern raised by the physician, when a sonographer escalates this type of case, is to describe the blood pressure and IVC. Blood pressure, IVC dilation, and a lack of respiratory variations when tamponade is suspected speaks to the urgency of the patient’s situation.
The intent of repeating a limited TTE, after pericardiocentesis, assists in the continue care of the patient. Following an imaging protocol looking for improvements in hemodynamic and cardiac function, can uncover other physiologic conditions, such as constrictive pericarditis, a rare finding that overlaps other syndromes affecting the pericardium. 19 Residual pericardial effusion requires the sonographer to investigate the possibility of tamponade signs. The current case study patient had improved LV inflow waveform with respiratory tracing as well as a collapsing IVC, and resolution of diastolic collapse of the RV free wall. Therefore, these diagnostic results indicated an improvement in hemodynamics.
A limited TTE, for effusion, may not require tissue Doppler imaging (TDI) of the mitral valve or M-mode of the interventricular septum, especially when providers expect notable improvements. The RV inflow and persistent septal bounce demonstrated the possibility of constrictive pericarditis, which limits the diastolic filling of the ventricles due to a noncompliant, thickened pericardium. 20 Additionally, constrictive pericarditis is distinguished clinically by Kussmaul’s sign, is more chronic in nature compared to tamponade, and is treated medically. In some cases, an improvement of the LV inflow may cause a rushed sonographer to skip RV inflow or minimally optimize the waveform for interpretation. This case study indicated an elevated RA pressure by having exaggerated respirophasic variations and a dilated IVC. Careful attention must be given to the extent of IVC collapsibility using M-mode as overestimation can occur if the vessel moves out of the imaging plane. 21 Sonographers must recognize the correlation between the ordered examination and how the technical components will confidently answer the diagnostic questions, based on a limited study.
Reading cMRI reports and comparing the interpretations with TTE reports helps to build an understanding of the heart, its intricate structures, hemodynamics, and surrounding anatomy. This is in line with reviewing a patient’s recent cardiology clinic note or history and physical to better answer the question being posed, as well as the reason for the exam. Having several echocardiograms prior to discharge, including a cMRI that confirmed constrictive effusive pericarditis (due to the remaining effusion and thickened pericardium), allows a sonographer to note the changes that the heart undergoes, during one hospital admission. The final echocardiogram, which shows resolution of hemodynamic compromise and the pericardial effusion, demonstrated signs of constrictive physiology that healthcare professionals can medically manage in an outpatient setting. 22 These findings (in addition to the resolution of effusion) included a persistent septal wall motion abnormality, E/A > 1, and a mitral annular E’ > 8 cm/s. The Hepatic Vein (HV) was not assessed during the patient’s last TTE.
Conclusion
Cardiac tamponade is a serious medical emergency in which a large volume of fluid accumulates in the pericardial sac, thus preventing the heart from filling and pumping effectively. The diagnostic finding of this severe pathology depends on a cardiac sonographer’s swift assessment and rapid notification to the patient care teams. Pericardiocentesis is the traditional method of draining the accumulated fluid, which was demonstrated in the presented case. This case study underscores the important diagnostic role that echocardiography plays in three different phases, in the management of a patient diagnosed with cardiac tamponade. Appropriately recognizing and assessing this pathology, expediting results to care teams, and assisting during/post pericardiocentesis exhibits the impactful role that the echocardiographer has in a patients’ outcomes.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was not sought for the present study because only anonymous information was used in this report.
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
