Abstract
Cardiac arrest is the absence of a centrally palpable pulse and no respiratory effort in an unresponsive patient. This often-lethal medical condition affects hundreds of thousands of people in the United States alone every year. Immediate intervention is crucial to provide the patient with any chance of survival. Advanced cardiac life support (ACLS) is the cornerstone therapy for cardiac arrest. Increased awareness and proper identification of life-threatening arrhythmias is critical, as it may lead to prompt medical treatment and improved mortality. The use of focused echocardiography, during a cardiac arrest, has been a developing area of interest over the past several years. The specific aim of this literature review was to emphasize the role of a focused echocardiogram and the valuable information that can be provided during a cardiac arrest.
Cardiac arrest is a common occurrence, affecting greater than 300,000 Americans each year. 1 The American Heart Association estimated the occurrence of in-hospital cardiac arrests to be at 209,000 annually, with an adult survival rate reported to be approximately 24.8%. 1 A cardiac arrest results in a cessation of circulation that may be due to an underlying etiology and is often lethal. The cause of cardiac arrest can vary by population and age. 1 Medical providers attempt to quickly recognize the cause and administer appropriate treatment. 2 Despite attempts at intervention, only 10% of those who suffer from cardiac arrest will leave the hospital alive, and most are neurologically impaired. 1 Sonography has shown an impact on identifying reversible causes of cardiac arrest as well as on predicting survival. 3 Four cardiac rhythms are associated with cardiac arrest, which are categorized into shockable versus nonshockable rhythms.4,5 Advanced cardiac life support providers are trained to recognize these and proceed accordingly. Ventricular fibrillation (VF) and pulseless ventricular tachycardia (VT) are considered shockable rhythms. The treatment for these findings is directed toward reversing the electrical abnormality present by cardioversion, defibrillation, or pacing.4–6 Pulseless electrical activity (PEA) and asystole are in the nonshockable category. These are classified as nonelectrical cardiac arrests, and direction of treatment should be aimed at determining a reversible cause. 6 Proper identification of these rhythms can assist providers to determine a cause as well as how to promptly administer treatment.6,7 While echocardiography may not be a current standard at all facilities, the American Heart Association has recommended point-of-care ultrasound (POCUS) to evaluate for reversible causes of cardiac arrest. 8 The aim of this review is to highlight the benefits of a focused echocardiography during cardiac arrest as well as the challenges.
Echocardiography Benefits During Cardiac Arrest
Confirmation of Contractility
The confirmation of diagnosing cardiac arrest with echocardiography may invite skepticism to some medical professionals. The absence of a pulse and blood pressure can seem straightforward and simple in indicating cardiac arrest. Detection of cardiac output during cardiac arrest is generally performed by palpating central pulses and obtaining a blood pressure measurement. Both of these methods of assessment may not always be reliable. In fact, up to 45% of health care professionals are unable to accurately assess central pulses during a cardiac arrest. 6 The inability to palpate a distal pulse during cardiac arrest could be hindered by significant peripheral vascular disease. Pulseless electrical activity (PEA) as the presenting rhythm of cardiac arrest has been reported to account for up to 50% of these presentations. 5 Pulseless electrical activity is defined as any electrical cardiac activity expected to generate pulsatile circulation but with no palpable pulse.3–6 Asystole, in comparison, is identified by no detectable cardiac activity and no palpable pulse. Prognosis is much poorer when dealing with these nonshockable rhythms.2–5,7–10 A 2017 study on POCUS examinations revealed that 10% to 35% of asystolic patients have demonstrable cardiac contraction. Cardiac contraction can be defined as any visible movement of the myocardium, excluding movement of blood within the cardiac chambers or isolated valve movement. 9 Echocardiography has the ability to directly visualize the presence or absence of cardiac wall contractility.7,8–11 In addition, visualizing the left ventricle has the potential to diagnose a shockable rhythm when the electrocardiogram (ECG) is not ideal. 6 Multiple research studies have evaluated the use of echocardiography to distinguish between two types of pulseless electrical activity. The prevalence of pulseless electrical activity as the presenting rhythm in cardiac arrest varies between 17% and 50%. 4 “True” PEA occurs when there is primary electromechanical uncoupling resulting in complete dissociation of electrical depolarization from mechanical cardiac contractions. 9 Echocardiography in this scenario would demonstrate no detectable myocardial motion with “true” PEA cardiac arrest. 3 “Pseudo”-PEA is a less known entity that can appear electrically similar to the aforementioned diagnosis. With this, a significant pathologic event has occurred that impairs the ability of the heart to perfuse the body. However, the “pseudo”-PEA phenomenon can only be verified by actually visualizing the myocardium. Myocardial contractions are present but are so severely diminished that there is no detectable pulse or blood pressure. This phenomenon is still considered a cardiac arrest, and treatment does not necessarily change. 9 Both versions of PEA are nonshockable rhythms, and continued chest compressions are warranted. The consensus of studies has highlighted the potential benefits of making this determination during cardiac arrest. 12 Specifically, differentiating these can provide clues to treating reversible causes of cardiac arrest. “True” PEA likely results from hypoxia, acidosis, hyperkalemia, hypothermia, toxicity, and ischemia.9,11 “Pseudo”-PEA, however, can result from profound hypovolemia, pulmonary embolism, tension pneumothorax, cardiac tamponade, tachydysrhythmias, and advanced anaphylaxis.9,13 Regardless of the cause, these patients are all gravely ill and require life-saving interventions. Patients with “pseudo”-PEA have shown to have higher survival rates, compared to those with no detectable cardiac motion.4–7,9,11,12 When performed during cardiac arrest, the absence of cardiac activity harbors a significantly lower likelihood that a patient will achieve return of spontaneous circulation. 14 Only 45% of physicians correctly diagnose a lack of pulse in cardiac arrest without differentiating between true PEA and pseudo-PEA.3,14 The overall survival to discharge for PEA cardiac arrest is reportedly 13%. 5 The information reviewed did indicate that the determination of a “true” PEA scenario guided medical professionals to end resuscitative efforts sooner.2,3,5,7,9,10,14
Identifying Reversible Causes
Determining a cause of cardiac arrest is imperative to achieve successful return of spontaneous circulation (ROSC). Echocardiography can attempt to elucidate a reversible cause of cardiac arrest and expedite treatment.2,7 Reversible causes of cardiac arrest include hypovolemia, hydrogen ion buildup (acidosis), hypothermia, hyper/hypokalemia, thrombosis, toxins, tension pneumothorax, and tamponade.1–3,5,6,9 Only hypoxia, hypothermia, and hypo/hyperkalemia can be definitively diagnosed at the bedside using existing standard monitoring. The remainder of reversible causes depend on subjective clinical assessment, laboratory values, and/or imaging. 6 Thrombosis, tamponade, and hypovolemia are reversible causes that can potentially be detected by sonography.2,3,6,10,14 Rapid detection and management of the underlying cause can possibly improve patient outcomes.3,6,11 Although sonography is used by some providers to also evaluate for pneumothorax, this is beyond the scope of this review.
Thrombosis
Seventy percent of all cardiac arrests are caused by acute myocardial ischemia or massive pulmonary embolism. 4 Pulmonary embolism resulting in cardiac arrest is likely to be massive, with occlusion of more than two-thirds of the pulmonary vascular bed.2,3 Echocardiography is currently recommended for a suspected pulmonary embolism only in a patients too unstable to transfer to computerized tomography (CT).2,6 Cardiac arrest is an unstable time that can benefit from the use of focused echocardiography. The evaluation of right ventricular size and function can help diagnose and exclude the probability of pulmonary embolism. The absence of right ventricular enlargement or dysfunction effectively excludes pulmonary embolism as a cause for cardiac arrest. Conversely, if signs of right ventricular dysfunction with hemodynamic instability and clinical probability of pulmonary embolism are suspected, aggressive treatment is indicated. 2 There is also a possible but unlikely occasion when thrombus can be directly visualized in the pulmonary artery to confirm diagnosis. 2 Coronary artery thrombosis is a major cause of cardiac arrest in the Western world. In the context of cardiac arrest, regional wall motion abnormalities of the left ventricle are neither 100% specific nor sensitive for coronary artery thrombosis. 6 Echocardiography provides little value to a definitive diagnosis based on regional wall motion. Left ventricular free wall rupture is documented in 0.8% to 6.2% of patients after myocardial infarction and frequently results in sudden death due to tamponade. 6
Tamponade
The concern for cardiac tamponade is arguably the most frequent reason for an emergent echocardiogram to be requested. In the absence of trauma, cardiac tamponade is the most common cause of PEA cardiac arrest. 2 This is a life-threatening clinical syndrome caused by an increase of intrapericardial pressure, most commonly due to fluid. As intrapericardial pressure increases and begins to exceed the right heart pressure, parts of the chambers collapse and are unable to fill properly.2,6 Subsequently, this progressively diminishes cardiac output, resulting in an inadequate perfusion to the body. Although cardiac tamponade is considered a clinical diagnosis, this is not always possible in the periresuscitative state. 6 A clinical diagnosis of cardiac tamponade includes tachycardia (>100 bpm), hypotension (<100 mmHg systolic), pulsus paradoxus (>10 mmHg drop in blood pressure on inspiration), and jugular venous distention. 2 The clinical features of tamponade, Beck’s triad, include elevated jugular venous pressure, muffled heart sounds, and hypotension. 6 Cardiac arrest can provide obvious challenges in a clinical diagnosis of tamponade due to a lack of detectable blood pressure or heart rate. Criteria for a hemodynamically significant pericardial effusion on echocardiography also become irrelevant in a setting of cardiac arrest.2,6 However, the ability of sonography to detect the presence of pericardial effusion in a cardiac arrest setting should lead to consideration for immediate drainage. The American Heart Association (AHA) and American Society of Echocardiography (ASE) taskforces recommended that pericardiocentesis be performed under echocardiographic guidance, for success rates greater than 90%. 6 Success is dependent on the volume of effusion, location of effusion, and the operator experience. While major complications can occur, the use of echocardiography yields a significantly lower complication rate estimated at 1.2%.6,15 The availability of sonography at the bedside, during cardiac arrest, provides medical personnel quick access to perform image-guided pericardiocentesis, if warranted. 15
Hypovolemia
Hypovolemia is among the commonest causes of pulseless cardiac activity cardiac arrest.8,12 Echocardiography provides an advantage in visualizing the quantity of blood volume within the cardiac chambers.15,16 In a situation of penetrating trauma, there is often an obvious amount of blood volume loss externally. Victims of penetrating trauma are more likely to survive compared to those undergoing blunt traumatic arrests. 1 In blunt traumatic arrests, massive internal bleeding can be suspected by a lack of volume in the cardiac chambers. Left ventricular end-diastolic volume has been shown to correlate well with blood loss in both animal and human studies. 6 Other reasons, aside from trauma, can cause a hypovolemic-induced cardiac arrest to occur. Regardless of the origin of volume depletion, the need for aggressive volume resuscitation is warranted. Echocardiographic findings that can indicate severe hypovolemia include a small, hyperkinetic left ventricle; a left ventricular end-diastolic area of less than 5.5 cm2/m2; and/or a small inferior vena cava (IVC) with inspiratory collapse. 6 In the setting of cardiac arrest, the hyperkinetic left ventricle is not feasible, and therefore the diagnosis would rely solely on visualization of chamber sizes. 7
Prognostic Value
The possibility of using focused echocardiography to predict outcomes of cardiac arrest has become a growing area of interest. While no definitive standard has been established, the results of recent studies have been consistent. The area of interest has centered on determining the likelihood for a patient to achieve return of spontaneous circulation based on the presence of mechanical cardiac activity. One study indicated that the absence of cardiac activity harbors a significantly lower but not zero likelihood that a patient will experience return of spontaneous circulation.5,7,14,15 Another study similarly concluded that finding any coordinated cardiac activity confers a better outcome and should potentially be regarded as an indication to continue resuscitation. 6 The results of a study that focused on trauma-related cardiac arrest produced a negative predictive value approaching 100% for survival. 12 Likewise, in yet another study, the lack of cardiac activity showed nearly a 97% negative predictive value of death. 7 These consistent conclusions drawn from multiple studies indicate that echocardiography can be highly influential in predicting survival of cardiac arrest and can be used as a guide to end resuscitative efforts. However, echocardiography alone should not determine cessation of life-saving maneuvers.5,7,12,14 Detection of any cardiac contraction on the initial sonogram is of paramount prognostic importance. 10
Improved Outcomes
Although echocardiography has proven to be a valuable diagnostic tool, the effect on patient outcomes has yet to be established. The consensus from published articles is very optimistic. Rapid detection of reversible causes with prompt intervention can assume that patient survival will increase.7,17 One study stated, “Rapid detection and management of the cardiovascular complications can reverse the underlying cause, thus the possibility of patient survival will be increased.”
7
Another statement from the
Performance of a Focused Echocardiogram During Cardiac Arrest
Equipment
Obtaining good-quality images on a medically stable patient can often be challenging. The cardiac arrest environment consists of multiple health care professions performing critical tasks simultaneously. 3 These scenarios are not conducive to obtaining high-quality echocardiography images. In these situations, a small bedside ultrasound system can be very helpful, given the space provided. Advances in technology have allowed for the development of smaller sized diagnostic ultrasound systems that can be readily available at a lower cost compared to larger systems. However, smaller ultrasound equipment may not provide comparable image quality compared to larger units. Transducer technology may not be the same, screen resolution may be poorer, and these systems may have limited image optimization. 15 Despite the limitations, the capabilities needed for basic information during a cardiac arrest can be obtained with many of these smaller units. The ASE states, “Simplified operation, substantially smaller size, and cost have opened their potential use to non-traditional cardiac ultrasound users. The ease of operation for these small devices does not obviate the need for training, to acquire and interpret cardiac images.” 16 All ultrasound systems used for a focused echocardiogram should have a method for recording data onto a media format for offline reviewing and archiving. 15 Ultrasound systems with rapid imaging capabilities are crucial in these scenarios. 10 Systems should be capable of two-dimensional (2D), M-mode, color-flow imaging, spectral, and tissue Doppler. Although not all modalities will be used in each case, it is important to have them available. 16 Ideally, medical providers would have access to equipment with this feature to provide timely imaging in these circumstances.
Image Archival and Reporting
As previously mentioned, all equipment should have the capability to acquire and archive images.15,16 The ASE’s writing group recommended that all POCUS images performed to evaluate a symptomatic patient should be stored to a retrievable location. Ideally, images should be stored in the same location as comprehensive echocardiographic studies. All focused cardiac ultrasound examination results must be documented in the chart and/or the electronic medical record. Reporting parameters should include date and time of the examination; patient name, age, date of birth, and sex; medical record number; name of the person who performed and interpreted the study; and the examination findings. 16 These recommendations are not specific to a focused echocardiography during cardiac arrest but are directed to POCUS imaging in any circumstance. For this review, no information was available on image documentation and interpretation specific to a cardiac arrest scenario.
Operator Expertise
Performing a focused echocardiogram, in the cardiac arrest setting, is challenging, and the universal recommendation is that only providers with significant training fulfill that role.2,3,5,6,15,16–19 Cardiac sonographer training recommendations are well established for performing routine, comprehensive echocardiograms. 16 A consensus statement from the ASE stated that echocardiography is a very specialized field requiring specific training to become competent. Even in situations requiring a more limited imaging protocol, it is imperative for the operators to be highly skilled. However, there are no specific training requirements indicated to perform in this role.16,17 The ASE consensus statement on focused cardiac sonography states the following: “Although there are no specific training requirements offered, this document provides a framework from which the medical community can establish the criteria necessary to optimize the use of this exciting new technology.” 16 Experienced users could provide an additional benefit during a cardiac arrest, with multiple sonographic views being obtained during manual compressions. 2 This highlights the importance of a user who is exclusively trained in performing and interpreting echocardiograms. 10 Sonographers and physicians with level 2 or level 3 training in echocardiography are skilled to image without additional training. 16 A 2018 Brazilian study reported that 41.3% of physicians surveyed felt that their need for more training was the greatest barrier to becoming successful at echo. 18 The ASE/American College of Cardiology (ACC) policy expresses the importance of a skilled user with the following statement: “Cardiac sonography, even for a ‘quick look’ or ‘limited’ examination, requires substantial training to avoid diagnostic errors.” 20 Echocardiography images are highly user dependent, and the quality of information derived is directly related to the individual performing the examination. Even a skilled echocardiographer must be able to perform without interrupting the standard flow of resuscitation procedures. The ASE/ACC policy statement on echocardiography in emergency medicine suggests implementing an echocardiography laboratory “extender.” A person, already skilled in echocardiography, should be secured to improve time and efficiency during emergencies. An echocardiography lab extender can be of two varieties. A sonographer could perform in this role with previous clinical training that includes a minimum of 960 hours of hands-on experience, 960 hours of didactic training, and registry certification of competence in echocardiography. Alternatively, an echocardiographic laboratory extender can be a physician with level 1 training in echocardiography. This includes an echocardiographic program of a minimum of 3 months, and that entails 150 echocardiographic examinations with a wide variety of pathology. Training should occur in an accredited cardiovascular laboratory. 20 The ASE/ACC statement defines the role further by indicating the primary function of the extender is to acquire images and facilitate review. 20 In the setting of cardiac arrest, there is an obvious need for immediate results, which creates a critical circumstance. The ASE/ACC directly addressed the need for imaging during a cardiac arrest as follows: “Only in situations of dire emergency should the echocardiographic laboratory extender function alone to provide diagnostic information for clinical decisions; such occasions such as unavoidable emergent circumstances with a potentially lethal condition.” 20 The patient in cardiac arrest requires an immediate response with an exceptional level of imaging performance. Cardiac arrest is a lethal condition 100% of the time without proper intervention.
Uninterrupted CPR
It is imperative to know that echocardiography used during resuscitative efforts must not interfere with an advanced cardiac life support protocol.9,13 The operator must be comfortable in this situation and know when to image as well as when to step aside. Research has shown that the use of echocardiography can result in prolonged pauses in CPR if not used properly. 13 Recommendations to avoid this indicate that imaging needs to be performed in no more than 10 seconds.7,9 The operator must be aware of the importance of uninterrupted chest compressions and be able to perform as part of the cardiac arrest team. 2 The cardiac arrest team leader is responsible for coordinating tasks and ensuring guidelines are followed. 2 Pulse checks are rapidly performed and provide a small window of time for imaging to occur. The 10-second pulse check is a brief time without chest compressions while providers attempt to detect a central pulse and analyze underlying electrical activity. This is the ideal time for imaging to occur.2,21,22 This subcostal imaging has been shown to be the most beneficial imaging window in these situations. 2 Unfortunately, the variation in echocardiography skill levels can potentially hinder the success of using sonography during cardiac arrest. 6 Learning more about the process can provide valuable knowledge for echocardiographers on how to situate themselves in a very chaotic environment.
Echocardiography Examination Protocol
POCUS has been continually integrated in both the critical care and emergency department environments. Every major organ or system can be evaluated with POCUS.10,11 The continuing trend to integrate echocardiography early in the cardiac arrest scenario has prompted a variety of protocols to emerge.
23
While they may vary slightly in sequence, they all aim to improve the outcome for the patients. In 2017, the
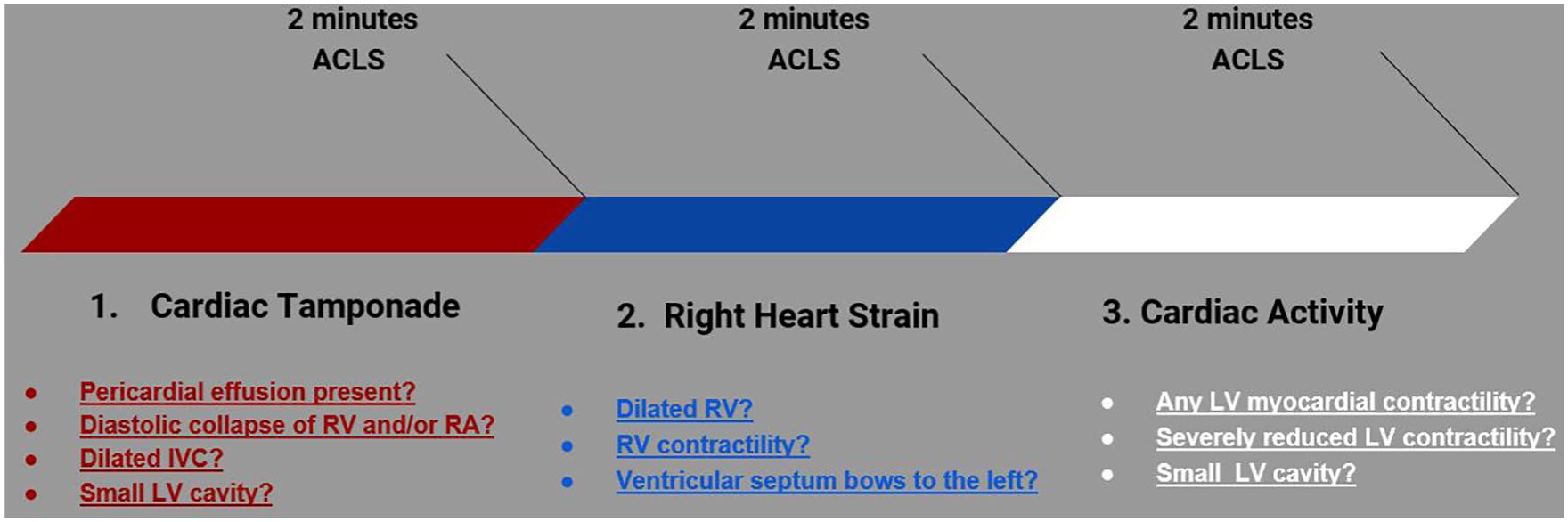
Cardiac arrest sonographic assessment components.
Conclusion
This review was aimed at providing evidence that supports that performance of a focused echocardiography during a cardiac arrest. The potential information that imaging brings in determining the underlying cause of cardiac arrest is well established in the literature. Understanding the possible etiologies of nonshockable, life-threatening cardiac rhythms can aid in quickly determining the etiology of cardiac arrest. Recognizing reversible causes of cardiac arrest expedites appropriate treatment, resulting in quicker, life-saving interventions. Echocardiography has shown to be highly accurate with determining prognosis of cardiac arrest. Providers can use this as an adjunct in the decision to extend or terminate resuscitative efforts. Effectively performing echocardiography during cardiac arrest demands a combination of exceptional technical ability along with a substantial level of clinical knowledge. Proper equipment with appropriate imaging capabilities is essential to the situation. The development of cardiac arrest echocardiography protocols has provided a framework for assessing reversible causes of cardiac arrest with minimal interruption in CPR. If these sonographic protocols are used in an efficient manner, there is minimal interruption in CPR. Future areas of research into the use of echocardiography during cardiac arrest should include determining the efficacy of chest compressions as well as validating the improvement in patient outcomes.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
