Abstract
Objective:
There is limited evidence available regarding intermittent tibial artery compressibility in the published literature. This investigation looks to determine the existence of intermittent tibial artery compressibility, in the noninvasive vascular laboratory.
Materials and Methods:
Consecutive patients underwent noninvasive arterial physiologic testing. An ankle-brachial index (ABI) ≥1.40 was defined as noncompressible and classified into 4 subgroups: (1) noncompressible single occurrence (NC-S), (2) noncompressible multiple occurrence (NC-M), (3) compressible to noncompressible (C-NC), and (4) intermittently compressible (I-C) ABI. Normal patients were compared to these noncompressible subgroups. Selective risk factors were compared in the C-NC and I-C ABI patient groups.
Results:
Of the 13 881 patients that were evaluated, 89% had compressible tibial arteries and 11% were noncompressible. Intermittent tibial artery compressibility was present in 11.3% of the patients, with noncompressible arteries. There were 718 patients from the NC-M, C-NC, and I-C subgroupings that had 3014 follow-up exams, over mean duration of 38 ± 27.7 months. The C-NC and I-C subgroup patients averaged 3.3 and 3.6 calcification risk factors, respectively.
Conclusion:
A subset of patients with intermittent tibial artery compressibility was confirmed in the noninvasive vascular laboratory. Additional research is needed to substantiate these findings and investigate whether intermittent compressibility may be marker for increased cardiovascular risk.
Keywords
The ankle-brachial index (ABI) has been the most used noninvasive method for diagnosing peripheral arterial disease (PAD). Normal ABI values range between ≥0.90 and <1.40. Noncompressible tibial arteries and an elevated ABI value of ≥1.40 (i.e., noncompressible ABI) are historically linked with medial arterial calcification (MAC).1,2 A noncompressible ABI is associated with aging and becomes accelerated in chronic kidney disease, diabetes, atherosclerosis, and cardiovascular disease. 2 Noncompressible tibial arteries falsely normalize and underestimate ABI severity in patients with PAD.
The toe-brachial index (TBI) is advocated in noncompressible tibial arteries because digital arteries are less affected by arterial wall calcification.1,3 Guidelines for the management and treatment of lower extremity PAD, recommend the use of TBI in patients with an ABI ≥1.40.1,4,5 In the host institution, a subset of noncompressible ABI patients was found to have intermittent tibial artery compressibility. The purpose of this investigation was to ascertain the prevalence and implications for surveillance of patients identified with intermittent tibial artery compressibility in the vascular laboratory.
Materials and Methods
This study was based on the retrospective review of all lower extremity arterial physiologic examinations performed in an Intersocietal Accreditation Commission (IAC) accredited vascular laboratory, between June 2011 and December 2022. The vascular laboratory reporting database, Medstreaming (Fivos Health, West Lebanon, NH), was the repository for all arterial physiologic examination data. The Epic electronic medical record system (Epic Systems Corporation, Verona, WI) was the reference source for selected patient group’s risk factors. The study was approved by the hospital’s institutional review board (IRB) and informed consent was not needed (IRB# 23-048).
Patients with an ABI of <1.40 were considered to have compressible tibial arteries. Patients with a formal diagnostic report that included sonographer findings or interpretive statements of “noncompressible,” “can-not-be-occluded,” (CNO), or “falsely elevated” in conjunction with a posterior tibial (PT) and/or dorsalis pedis (DP) ABI of ≥1.40 or systolic pressure >255 mmHg were considered to have noncompressible tibial arteries. Patients without ankle pressures or ABI were excluded from the analysis.
All patients had lower extremity segmental arterial pulse-volume recording (PVR) waveforms in the thigh, calf and ankle, and continuous-wave (CW) Doppler waveforms of the PT and DP arteries, after 5 minutes of rest, in the supine position. Systolic pressures in the brachial, PT and DP arteries were performed with appropriately sized pressure cuffs using standard ABI protocol with the higher of the right and left brachial pressure readings used as the denominator. 1 Compressible ABI patients were compared to noncompressible ABI patients and were divided into 4 groups:
Noncompressible ABI—single exam occurrence (NC-S).
Noncompressible ABI—multiple exam occurrence (NC-M).
Compressible to noncompressible ABI—initial period tibial artery compressibility, followed by noncompressibility (C-NC).
Intermittently compressible ABI—discontinuous periods of tibial artery compressibility and noncompressibility (I-C).
Comparisons for number of diagnostic exams and patients, age and gender were performed for all patient groups. The NC-M, C-NC and I-C ABI subgroups were assessed to decide the number and duration of surveillance exams. The C-NC and I-C groups underwent further review for the presence of select arterial calcification risk factors: hypertension, diabetes, chronic kidney disease, coronary artery disease (CAD) and PAD. Fisher’s exact test and unpaired t-test, using QuickCalcs GraphPad software, was used to compare compressible and noncompressible patient groups. The statistical significance was defined and set as p < .05.
Results
A total of 27 023 lower extremity arterial physiologic exams were performed, between June 1, 2011, and December 31, 2022. Excluding examinations where there was a lack of specified sonographer findings, physician interpretive statements, or ABI data, resulted in 23 320 examinations and 13 881 patients, to comprise the final analysis. The prevalence of noncompressible ABI and intermittent ABI compressibility are displayed in Table 1. The prevalence of noncompressible ABI represented 11% of all patients and 16% of all examinations in our study cohort. Intermittent tibial artery compressibility represented 27.4% of all noncompressible ABI exams and 11.3% of all noncompressible ABI patients.
Exams and patients with compressible and non-compressible ABI.
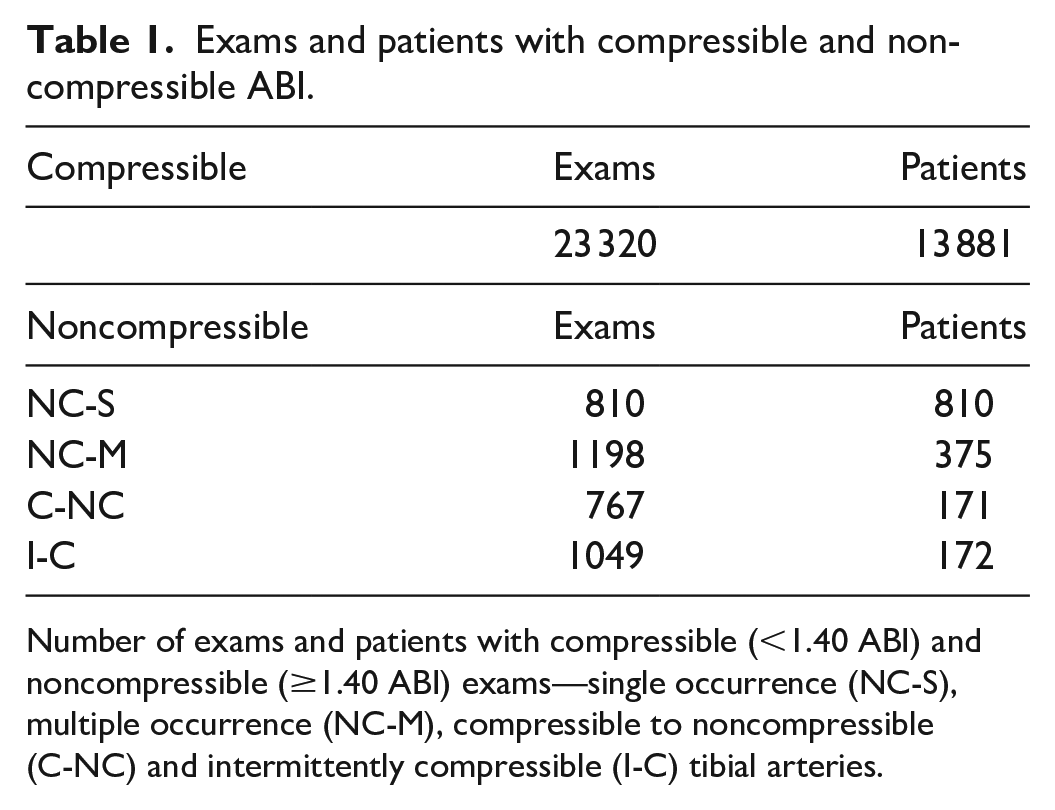
Number of exams and patients with compressible (<1.40 ABI) and noncompressible (≥1.40 ABI) exams—single occurrence (NC-S), multiple occurrence (NC-M), compressible to noncompressible (C-NC) and intermittently compressible (I-C) tibial arteries.
Age and Gender
The average age and gender of patients in the compressible and noncompressible ABI groups are presented in Table 2. Patients with compressible tibial arteries were significantly younger than the noncompressible ABI groups (p < .009). NC-S patients were significantly older than NC-M and I-C patients (p < .002). No significant differences in patient age were noted between those in the NC-M and C-NC (p = .468), NC-M and I-C (p = .269), and C-NC and I-C group comparisons (p < .110).
Age, and gender of patients with compressible and noncompressible ABI.
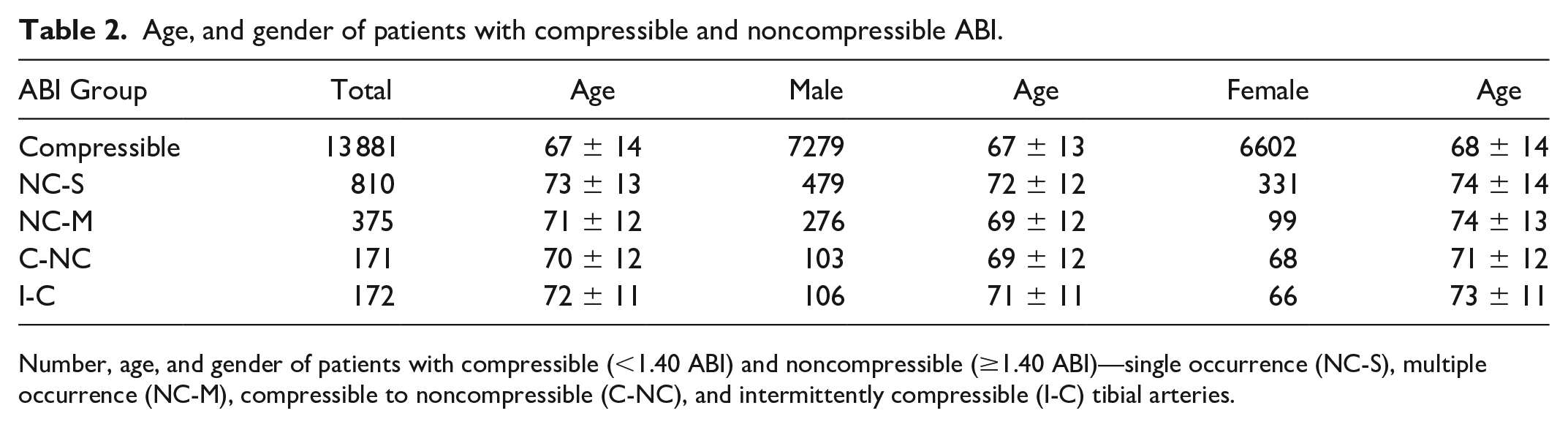
Number, age, and gender of patients with compressible (<1.40 ABI) and noncompressible (≥1.40 ABI)—single occurrence (NC-S), multiple occurrence (NC-M), compressible to noncompressible (C-NC), and intermittently compressible (I-C) tibial arteries.
There were significantly more males in the compressible artery group than in the NC-S, NC-M, and I-C groups (p < .020). There were borderline significant differences in males in the C-NC group (p < .051). The NC-S group had significantly more males than females than the NC-M group (p < .000). No significant gender differences were noted between NC-S and C-NC, and NC-S and I-C group comparisons, p = .032 and p = .270, respectively. No significant gender differences were noted between the compressible to noncompressible and the intermittently compressible ABI group comparisons, p = .024.
Surveillance Exams
There were 3014 follow-up exams performed on a total of 718 NC-M, C-NC, and I-C patients. The average number of surveillance exams and the duration of the surveillance for NC-M, C-NC, and I-C groups are presented in Table 3. The NC-M patients had significantly fewer surveillance exams than the C-NC and I-C groups (p < .000).
Average surveillance exams and duration of surveillance in noncompressible multiple occurrence, compressible to noncompressible and intermittently compressible ABI groups.
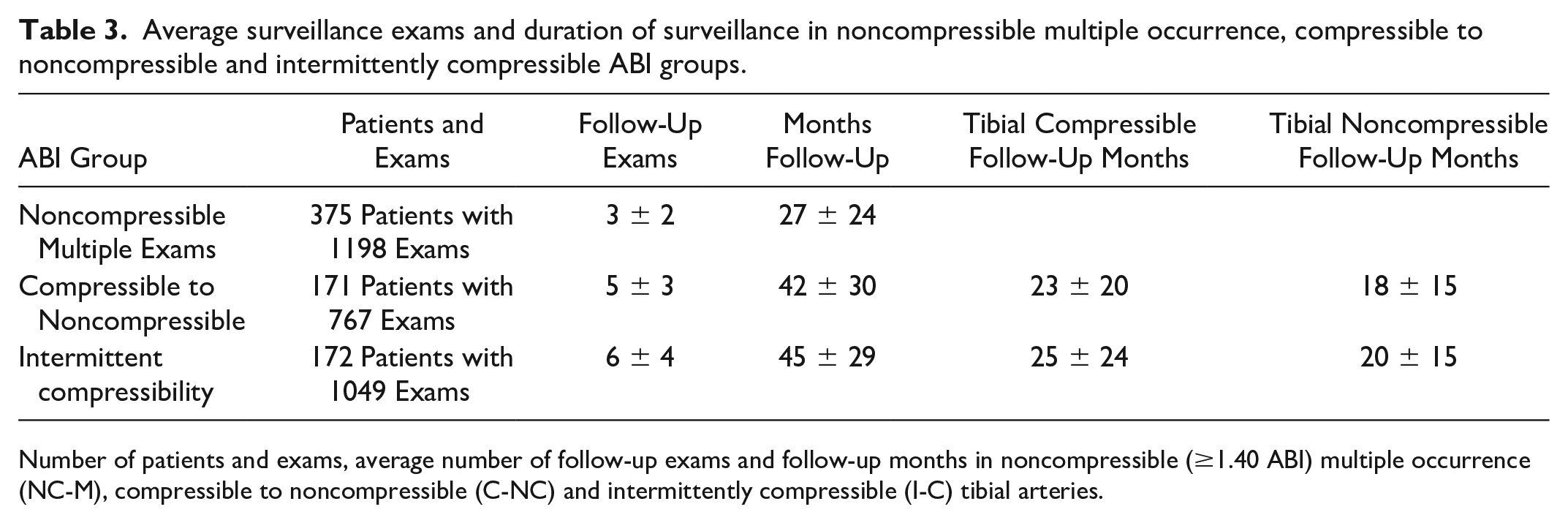
Number of patients and exams, average number of follow-up exams and follow-up months in noncompressible (≥1.40 ABI) multiple occurrence (NC-M), compressible to noncompressible (C-NC) and intermittently compressible (I-C) tibial arteries.
The I-C patients had significantly more surveillance exams than the C-NC group (p < .000). No significant differences in the duration of surveillance exams were noted between the C-NC and I-C groups (p < .056); and no significant differences in the interval of tibial artery compressibility (p = .058) and noncompressibility (p = .258) were noted.
Calcification Risk Factor Comparisons
Selective arterial calcification risk factor comparisons for the C-NC and I-C ABI patient groups are displayed in Table 4. The C-NC patients averaged 3.3 and I-C patients averaged 3.6. There were no statistically significant differences in the selected risk factors: hypertension (p = .080), diabetes (p = .280), chronic kidney disease (p = .094), CAD (p = .474), and PAD (p = .237). Hypertension was the most common and chronic kidney disease was the least common risk factor in both groups.
Calcification risk factors in compressible to noncompressible and intermittently compressible ABI groups.
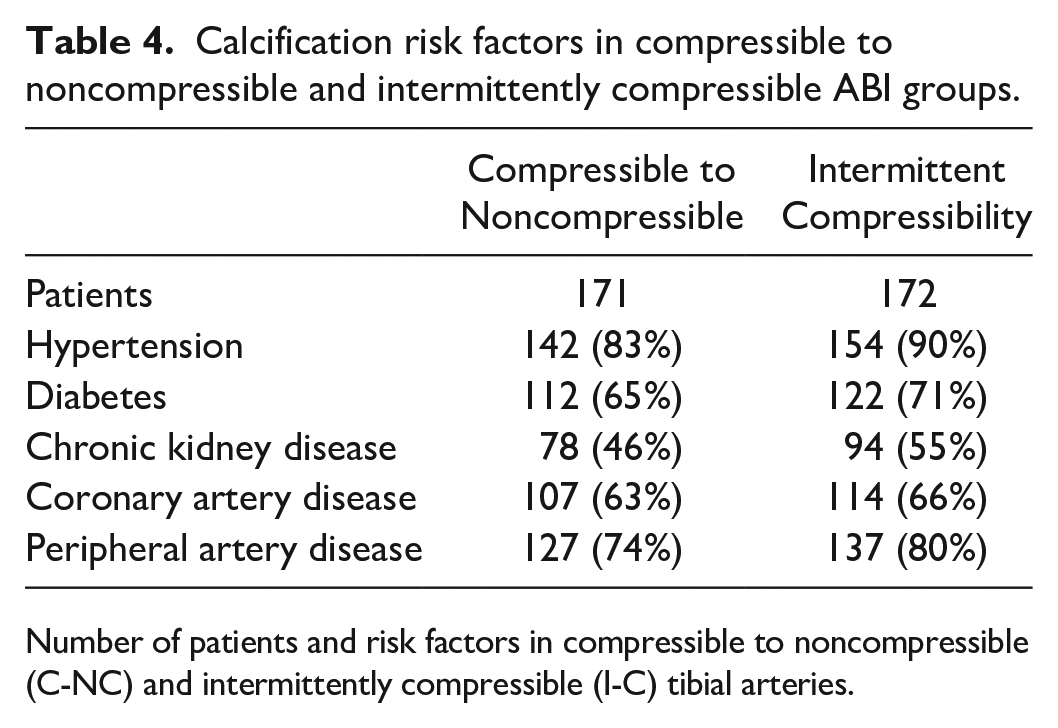
Number of patients and risk factors in compressible to noncompressible (C-NC) and intermittently compressible (I-C) tibial arteries.
Discussion
A retrospective review of lower extremity arterial physiologic exams, referred to the vascular laboratory, found noncompressible tibial arteries (≥1.40) in 11% of the patients, as compared to an reported incidence of 4.5 to 24%.6–9 In addition, this study verified the presence of a subset of ABI patients with intermittent tibial artery compressibility. Intermittent tibial artery compressibility represented 11.3% of all noncompressible patients and 27.4% of all noncompressible exams. The prevalence of noncompressible and intermittent compressible ABI patients would be invariably greater with a lower ABI threshold for denoting vascular calcification (≥1.30).9,10
Peripheral arterial disease and arterial calcification risk factors observed in previous investigations,6,9–13 was present in both of the noncompressible and intermittently compressible ABI groups. The incidence of diabetes, hypertension, CAD, and chronic kidney disease, in these groups, however, was 10 to 16% higher than percentages reported in the literature. 2
Intermittent tibial artery compressibility adds another variable to ABI and TBI recommendations, in the vascular laboratory. Current guidelines describe multiple ABI categories: ≤0.90; 0.91 to 0.99, 1.00 to 1.39, noncompressible (≥1.40),1,5 as well as alternative categories for borderline high (1.31 to 1.39) and noncompressible ABI (≥1.30).2,9 TBI is typically recommended when the ABI is ≥1.40.1,3–5
A noncompressible ABI has been historically linked to medial arterial calcification; however, the pathogenesis of MAC remains uncertain.10,14–16 The etiology of intermittent compressibility may be equally ambiguous. Patients with multiple calcification risk factors, (e.g., hypertension, diabetes, and chronic kidney disease), should be designated calcification susceptible at the onset of noninvasive arterial testing, in the vascular laboratory. Waveform consensus literature and research17,18 has suggested Doppler waveform analysis of the pedal arteries, to help mitigate ABI/TBI testing limitations and aid in the standardization of patient surveillance.
To help regulate the diagnosis and surveillance in calcification susceptible patients, this work would suggest that the initial noninvasive PAD evaluation include ABI, TBI with continuous-wave (CW) Doppler, disease severity waveform analysis of the PT, and DP arteries. Other investigators have endorsed additional or alternative noninvasive methods such as segmental pressures, treadmill exercise, transcutaneous oxygen pressure (TcPO2), pedal Doppler waveforms, pulse volume recordings, and duplex sonography.3,4,7,9,10,19–23 However, the addition of one or more of these diagnostic techniques may result in reimbursement coding conflicts or be time prohibitive in the initial noninvasive evaluation.
Limitations
This retrospective clinical study is limited due to the research design and has threats to internal and external validity. It also was based on a patient sample composed by convenience. A strength of this study is the size of the patient cohort. Conversely, however, such a large study group limited any analysis for the predilection of noncompressibility or intermittent compressibility in 1 tibial artery over the other.
Another limitation was the partial review of calcification risk factors; however, tibial artery calcification risk factors have been addressed extensively in previous investigations.2,8,11,19–21,24
Conclusion
This retrospective cohort study helped to confirm a subset of vascular laboratory patients with intermittent tibial artery compressibility, which was characterized by noncompressible PT or DP ABI (≥1.40) and converted to ≤1.40 during 1 or more surveillance examinations. Additional research is needed to substantiate these diagnostic findings and investigate whether intermittent compressibility may be marker for increased cardiovascular risk.
Footnotes
Acknowledgements
This paper would not have been possible without the assistance of Marsha Neumyer, BS, RVT, FSDMS, FSVU, FAIUM, International Director Vascular Diagnostic Education Services Harrisburg, PA. We are grateful for the support and encouragement of the Jobst Vascular Institute and research department of the ProMedica Healthcare System and Toledo Hospital and the Jobst Vascular Laboratory sonographers, physicians, and administrative staff.
Ethics Approval
Ethical approval for this study was waived by the hospital’s institutional review board (IRB# 23-048).
Informed Consent
Informed consent was not sought for the present study because all case data was de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
