Abstract
Comprehension of cardiac anatomy is critical for health care providers and allied health professionals in the pursuit of an accurate and timely patient diagnosis. Many times, when these anomalies are discovered, they are incidental findings from an asymptomatic patient during cardiovascular imaging. One specific anomaly which requires keen attention to detail and critical decision-making abilities is a persistent left superior vena cava (PLSVC). On a transthoracic echocardiogram (TTE), PLSVCs are discovered with the addition of agitated saline, commonly known as bubble studies. Because these anomalies are left-sided, it is critical for the sonographer and IV (intravenous)-trained staff to administer agitated saline through an IV on the left arm. Although rare, PLSVC may correlate with other cardiac pathologies, thus indicating the magnitude of a sonographer’s attention to surrounding anatomy (such as a dilated coronary sinus) and the necessity of appropriate IV location. This case study demonstrated the significance of a strategically thinking sonographer while diagnosing a PLSVC on TTE with agitated saline, which could have been missed if the IV line were not initiated on the left arm.
Persistent left superior vena cava (PLSVC) is a rare cardiac anomaly that occurs in approximately 0.3% to 0.5% of patients, with an otherwise normal heart1,2 and is the most frequently discovered thoracic venous variant. A transthoracic echocardiogram (TTE) with agitated saline (bubble study) is a common route for diagnosis of these congenital venous variants. Because of the rarity of these anomalies and the steps needed for accurate detection by TTE, sonographer decision-making plays a key role in not only these cases, but in all diagnostic tests performed. The case provided describes how critical thinking by sonographers can play a key role in the detection of rare findings, such as PLSVC. This kind of anomaly may have otherwise been missed if a dilated coronary sinus (CS) was not detected and correlated with the need for a left-sided intravenous (IV) line.
Case Report
This was a 54-year-old Caucasian woman with hypothyroidism and a medical history of atrial septal defect (ASD), bicuspid aortic valve (AV), and coarctation of the aorta, all of which were repaired. She later developed atrial fibrillation (AF) and had a history of several atrial ablations and cardioversions. Her AV was replaced with a bioprosthesis, and her total arch was replaced in 2019 with a 21 mm Edwards Resilia. In addition to these interventions, a maze procedure and left atrial appendage clip were also performed. She had obstructive sleep apnea and persistent atrial flutter, and on direct admission for small bowel obstruction, possible need for surgical support, heart failure with preserved ejection fraction (HFpEF), and pulmonary hypertension.
A 2D TTE was ordered for an assessment of the ASD repair viability. The standard care protocol was followed with an agitated saline bubble study to assess possible atrial septal shunting. The 2D TTE demonstrated a normal left ventricular volume with normal variant false chords and mild concentric left ventricular hypertrophy (LVH). The diastolic function was indeterminate due to the presence of AF. The left ventricular ejection fraction by Simpson’s bi-plane was 67%, and an indexed left ventricular outflow tract (LVOT) stroke volume of 58 mL/m2. The surgical aortic valve replacement (SAVR) was in place and well seated with a max peak gradient of 28 mm Hg, a mean peak gradient of 16 mm Hg, and an AV velocity ratio of 0.60. The left and right atrium were severely dilated, and after the injection of agitated saline with and without Valsalva, no intracardiac shunting was noted. Micro-bubbles administered intravenously (IV) through the left arm demonstrated a PLSVC entering the dilated CS.
During the assessment of the cardiac chambers and structures, the sonographer noticed a dilated CS, and when a new IV site was being accessed by nursing staff, the sonographer requested to have the IV placed in the left arm by the sonographer. In the outpatient setting, the sonographers start their IVs, preferably in the left arm, for incidental chances of finding PLSVC.3,4 This can be difficult in the hospital setting as IVs are placed at noticeably accessible sites by nursing staff, and sonographers are subject to use what is available. The nurse was agreeable and assisted the sonographer with the bubble study. This TTE bubble study demonstrated a PLSVC by visualizing bubbles entering the dilated CS, seen adjacent to the left atrium, then to the right atrium, and finally into the right ventricle (See Figure 1). After the injection of agitated saline with and without a Valsalva maneuver, no intracardiac or pulmonary shunting was noted. Noticing the dilated CS, early capture with cine recording was essential and demonstrated a keen attention to detail. A subsequent chest computed tomogram (CT) and radiograph were performed demonstrating a left-sided vas catheter tip lying within a left-sided SVC (See Figures 2 and 3).
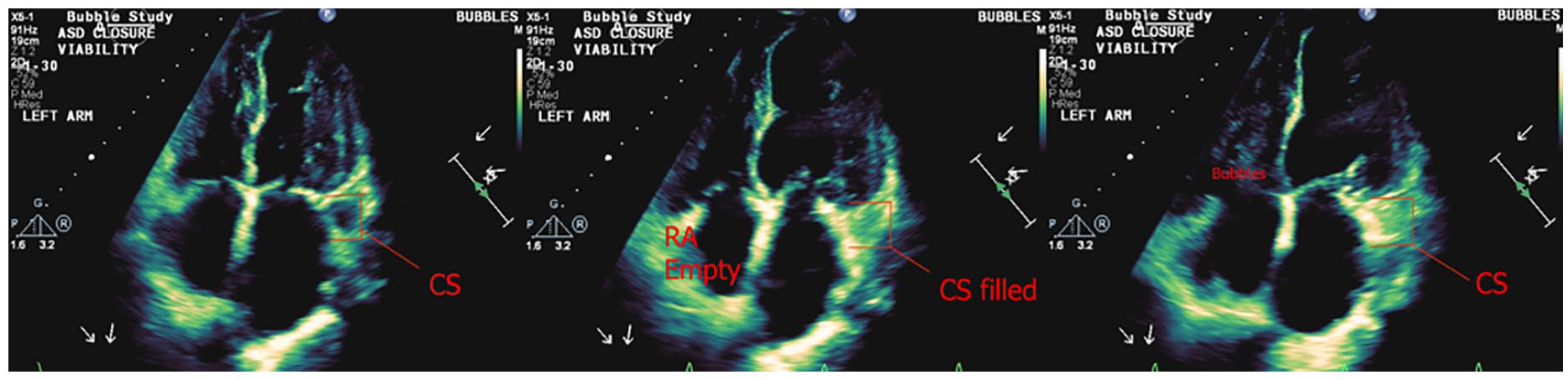
A 2D transthoracic echocardiogram demonstrated a visual representation of the sequence of bubbles entering the CS before the right heart, indicating a PLSVC.
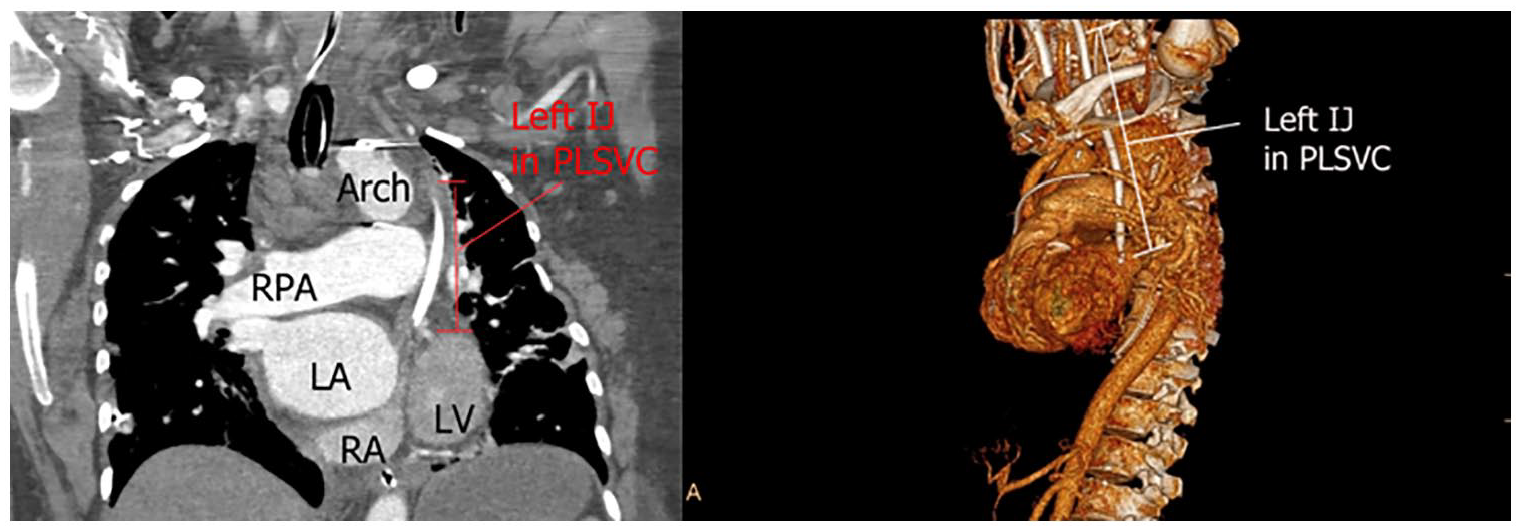
The computed chest tomographic image on the left demonstrates a left internal jugular venous central line, which is described as terminating at the CS. The image on the right is showing a 3D reconstruction of the ascending aorta and illustrating the left internal jugular in the PLSVC.
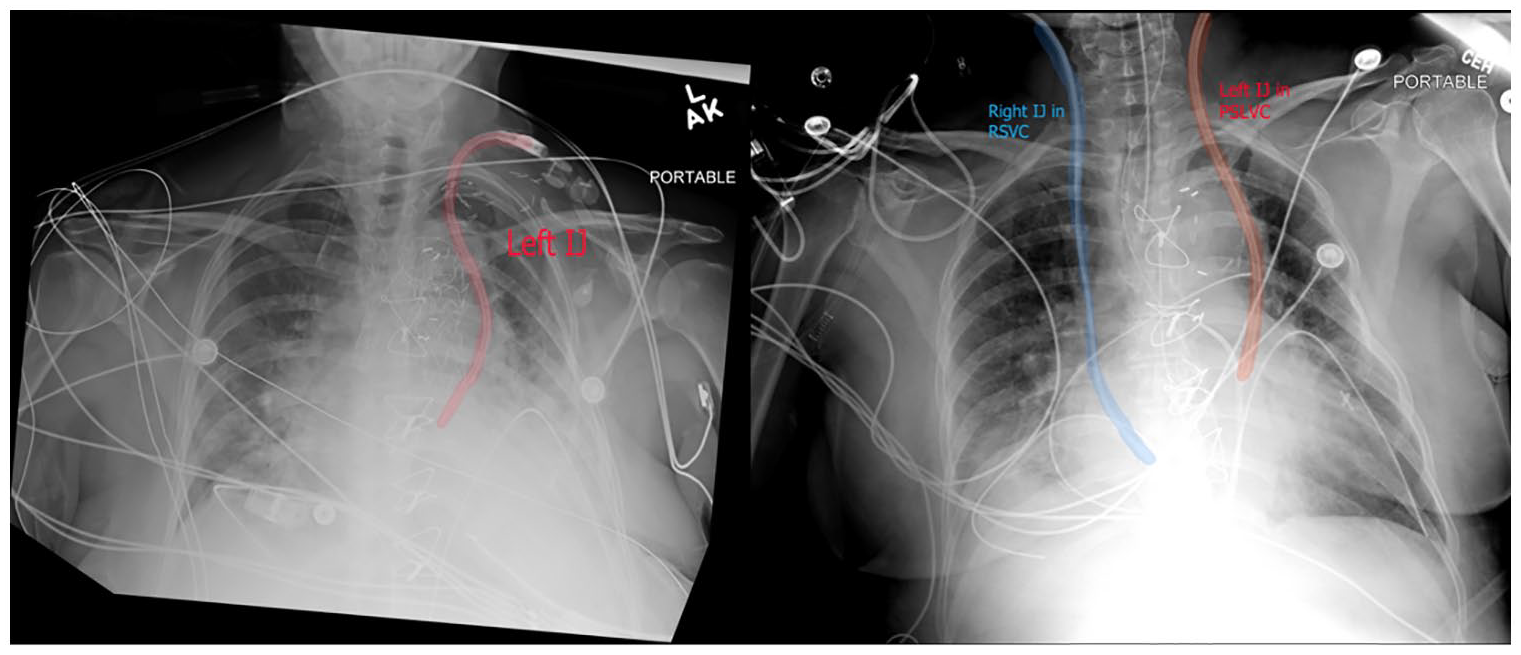
The patient’s chest radiograph is provided and demonstrates a left-sided vascular catheter tip lying within a left-sided SVC.
A reasonable question arises as this case study develops: why was the presence of the PLSV not already described in the patient’s chart or known to the medical care providers? Typically, sonographers rely on the most recent H&P or the latest cardiac notes, neither of which contain any reference to the PLSV. Surprisingly, there was no mention of a PLSV in the transfer notes or the initial procedural H&P. The first mention of a possible PLSVC was from a routine chest x-ray confirming the placement of a left Internal Jugular (IJ) and then a STAT CT scan, neither of which are common reports read by sonographers. A deeper interpretation may be a need for a more robust history and physical reporting system that cross-references multiple care providers reporting.
Discussion
In the diagnosis of cardiac shunts, TTEs with agitated saline (bubble study) can be conducted bedside by sonographers in the outpatient and inpatient setting. Some studies have concluded that transesophageal echocardiography (TEE) with agitated saline plays a significant role in diagnosis and can be the imaging tool of choice when evaluating PLSVC.5,6 When PLSVCs are suspected with a dilated CS due to volume overload, 7 it is paramount that trained staff administer agitated saline via the left arm, and not the right, when the opportunity persists. In a PLSVC, bubbles will first appear in the CS before filling the right heart.8 –10 Whereas in normal venous anatomy with a left-sided IV, bubbles will appear in the right heart first. Complementary modalities to evaluate a PLSVC along with TTE are contrast-enhanced CT, magnetic resonance imaging (MRI), and invasive angiography.11,12 With an increased reliance of CT and MRI for cardiac imaging techniques in the quest for proper cardiac management, PLSVC are more frequently being discovered. 13
The hemodynamic implications of this anomaly are typically minimal; however, these incidental findings are important to forward to interventionists as their presence may make some procedures challenging to perform.5,14 –16 One challenging intervention in the presence of a PLSVC is pacemaker implantation. 17 A case study by Totorean et al. reports on an unknown PLSVC at the time of lead implantation, which deemed traditional transvenous implantation not feasible. 18 In addition to the implications on interventional procedures, PLSVCs predispose patients to clinical complications such as arrhythmias, embolism, decreased exercise tolerance, chest pain, and cyanosis.19,20
Because these thoracic abnormalities are generally discovered incidentally and patients may be asymptomatic at the time of their intervention, 21 maintaining focus on all anatomy during TTE is paramount in a patient’s care continuum. As sonographers, we possess a decision-making ability that could potentially change the outcomes of our patients. Asserting awareness of PLSVC and its anatomy will facilitate quality care and avert exiting potential of complications in neonatal and adult populations. 22
Although TTE is a common procedure performed in noninvasive laboratories, the addition of an agitated saline study is not always ordered in conjunction with each test. However, department administrators must ensure that supplies and best practice procedures are available in these echocardiography laboratories. The apparatus used for the bubble study consists of two 10-mL syringes connected by a three-way stopcock—one syringe containing 8 mL of saline and 1 mL of room air (9 mL) (Mitchell, Takaya). In contrast, the other syringe is empty and depressed to 0 mL, leaving no available volume, and the stopcock connecting the two facilitates the mechanical mixing process and subsequent delivery. This setup is secured to the patient’s left arm IV, ensuring a closed system for the adequate agitation of the contrast elements and their rapid infusion in the venous system.3,4
For a quality agitated saline mixture, the bubble study combines 1 mL of room air and 8 mL of saline solution in a single syringe (Mitchell, Takaya). To this mixture, 1 mL of the patient’s venous blood is drawn and added, totaling 10 mL of unprepared contrast (Takaya). We then subjected the total contents to vigorous agitation through the operation of the syringes connected by the three-way stopcock (turned off to the patient during preparation), resulting in a homogeneous 10 mL volume of agitated saline contrast. This agitated mixture is essential for enhancing the visualization of cardiac shunting. 3
The recommended echocardiographic window during administration of agitated saline is in the assessment of a PLSVC the apical four-chamber (A4C). However, the initial inquiry of a dilated CS can be visualized from the Parasternal long axis (PLAX). Although not required during assessment of a PLSVC, a Valsalva maneuver should be performed from at least one window to induce shunting across the interventricular septum (IVS). This can be performed by the patient coughing or tightening/flexing of his or her abdominal muscles, sometimes referred to as “bearing down.” Timing and acquisition of images during the administration of agitated saline is an important step in PLSVC assessment. In a prospective capture sequence, sonographers should turn the beat number to 20 and begin acquisition a cardiac cycle or two before the agitated saline reaches the right heart. 3 In the presence of a PLSVC, as mentioned earlier, the CS will fill before the right heart. While the traditional bubble study portion is performed in the A4C, sonographers and cardiologists can better visualize the sequence of bubble opacification of the CS before the right heart while utilizing m-mode from the PLAX due to its superior time resolution.3,23 From the window, the proximal portion of the m-mode cursor should be aligned from the top of the sector through the right ventricular outflow tract (RVOT) and ending distally through the CS. From this window, it is also important to note the differential diagnosis of a pericardial effusion, atrioventricular groove abscess, and can be confused for adjacent anatomy such as the descending aorta. 23
Conclusion
As the evolution of sonography expands with new technology and interventions, our duties as sonographers are becoming increasingly important. The role of the cardiac sonographer often requires balancing many different responsibilities during the patient examination: from generating examination quality, best practice imaging protocols in the pursuit of patient diagnosis, to being a patient’s advocate and realizing that sonographers can be held accountable to many different practice standards. This unique case study demonstrated a sonographer’s decision-making ability during a TTE bubble study and knowing how to pivot amid abnormal findings during the test. Because the sonographer noticed the dilated CS and administered agitated saline via a left-sided IV, it enabled the cardiologist to identify a venous anomaly that may potentially contribute to other cardiac complications and posed a challenge for other interventions needed by a patient. By failing to administer agitated saline in the left arm and utilizing the right arm, a PLSVC would have potentially been missed and changed the course of care for this patient.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because this was a case report in which the findings were analyzed retrospectively. The case study did not affect/alter the management in any way.
Informed Consent
Informed consent was not sought for the present study because all case data was de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
