Abstract
Objective:
During pregnancy, sonographic measurements of subcutaneous adipose tissue may be a reliable diagnostic predictor of gestational diabetes mellitus (GDM) and fetal metabolism. A sonographic view of the fetal abdominal circumference can be used to measure the fetal anterior abdominal wall thickness (AAWT), which is straightforward and simple. Consequently, in this study, the aim was to evaluate whether the fetal AAWT value could be used as a diagnostic indicator of GDM, thus preventing the adverse neonatal and maternal outcomes thereof, including shoulder dystocia, neonatal hypoxia, and injury to the birth canal, secondary to fetal macrosomia.
Materials and Methods:
This prospective cross-sectional study was conducted at a hospital in Lahore, Pakistan, over a 9-month period. A total of 176 female participants in their third trimester of pregnancy were included in this study. The participants were assigned to two cohorts: a cohort of 88 participants diagnosed with GDM and a control cohort of 88 participants without GDM. An independent sample t test, analysis of variance, and chi-square test were used to compare the characteristics between the two cohorts of participants. A final analysis was performed to determine the strength of the correlations between the estimated fetal weight (EFW) and AAWT values.
Results:
The GDM and control cohorts had mean fetal AAWT values of 12.65 ± 1.276 and 6.389 ± 1.503, respectively. Thus, a statistically significant difference in the fetal AAWT values was observed between the GDM and control cohorts. Furthermore, a statistically significant positive correlation between the EFW and AAWT values was observed (P = .01).
Conclusion:
This study highlighted that women with GDM have a considerably higher fetal AAWT values than those without GDM. In addition, the findings revealed that the AAWT value was correlated with the EFW value. Consequently, an increased fetal AAWT value can potentially be utilized as a predictive indicator for those pregnant women at risk of developing GDM, thus preventing adverse maternal and fetal outcomes.
Keywords
During the 20th century, pregnant women with diabetes mellitus (DM) had less favorable results than those of the present day. During the 1990s, abnormally high prevalence rates of poor fetal outcomes and neonatal mortalities occurred in pregnant women diagnosed with gestational diabetes mellitus (GDM) who later developed type 2 DM. 1 The initial description of GDM, which appeared in the 1950s, described the disease as a temporary maternal ailment that adversely affected fetal outcomes and resolved after delivery. 2 In 1998, the Fourth International Workshop—Conference on Gestational Diabetes introduced the current definition of GDM. Thus, GDM is characterized by variable degrees of glucose intolerance, beginning, or initially observed during pregnancy. 3 Moreover, GDM can be defined as a hyperglycemic condition, arising from insufficient insulin delivery required to meet tissue needs and achieve normal blood glucose regulation. 4 At present, one of the most typical gestational-related medical conditions, with a prevalence of 4%–14%, is GDM. 5
Numerous factors, including subclinical inflammation, an increase in placental hormones, decreased adiponectin secretion, and excessive lipolysis, contribute to considerable insulin resistance in the muscle, liver, and adipose tissue in women with GDM. 6
Insulin levels are commonly comparable between women with and without a history of GDM, suggesting that the inadequate insulin secretion inherently characteristic of GDM is predominantly limited to the gestational period. 7 GDM is associated with hypertensive disorders, and the onset of preeclampsia is thrice more likely to result in preterm deliveries. 8 Cesarean sections and induced births are more common in women with GDM. 9 Preeclampsia affects pregnant women who are obese, with poorly controlled insulin-treated GDM more frequently than pregnant women who are obese, with well-controlled, insulin-treated GDM. 10 Regardless of the treatment and glycemic control achieved in women with GDM, women who are overweight and obese with concomitant GDM have a twofold to threefold higher prevalence of chronic hypertension. 11 The most crucial and commonly employed diagnostic tool in the obstetric management of diabetic pregnancies is sonography. Moreover, in diabetic pregnancies, sonographic measurements of subcutaneous adipose tissue may be a reliable diagnostic predictor of the fetal metabolic state. 12 Sonography can be utilized to measure subcutaneous measurements longitudinally, thus detecting differences and potentially disease processes. To identify excessive fetal adipose deposition and assess the level of maternal glucose in pregnancies with GDM, the availability of reference values for fetal subcutaneous fat sonographic measurements may offer therapeutically helpful diagnostic information. 13 Furthermore, the fetal subcutaneous tissue thickness is a better predictor of maternal glucose control than the ambulatory glucose profile. 14 Evidently, several studies regarding subcutaneous and visceral abdominal fat thickness and GDM have been published; however, the optimal diagnostic methods utilizing sonographic measurements to predict GDM have not been studied or validated.
A typical sonographic view of the fetal abdominal circumference can be used to measure the fetal anterior abdominal wall thickness (AAWT), which is straightforward and simple. Considerable variations in the fetal AAWT have been found between pregnant women with and without DM. Assessing the fetal AAWT value appears to be a valuable diagnostic tool for GDM. Thus, the clinical relevance of this parameter during prenatal care should be further elucidated. In this study, the aim was to evaluate whether the fetal AAWT value could be utilized as a diagnostic indicator of GDM, thus preventing the adverse neonatal and maternal outcomes thereof, including shoulder dystocia, neonatal hypoxia, and injury to the birth canal, secondary to fetal macrosomia.
Materials and Methods
Ethical Approval and Informed Consent
Ethical approval was granted by the Research Ethics Committee of the University of Lahore (IRB#REC-UOL-/195-07/2022). Informed consent was obtained from all study participants for their participation in the study, for completion of questionnaires and data sheets, and for publication of their de-identified data. This study was conducted in accordance with the principles of the Declaration of Helsinki.
Study Design
This prospective cross-sectional study was conducted by the radiology department of a hospital in Lahore, Pakistan, over a 9-month period. A total of 176 female participants in their third trimester of pregnancy were included in this study. The participants were assigned to two cohorts: a cohort of 88 participants diagnosed with GDM and a control cohort of 88 participants without GDM. Because this was a research study, involving the selection of participants available around a particular location, our hospital, convenience sampling was used. The inclusion criteria were as follows: (1) pregnant participants in their early third trimester, which was 27–32 weeks of pregnancy; (2) a diagnosis of GDM had to be made; and (3) pregnant participants with either a previous history of GDM or healthy pregnancies without diagnostic complications.
The exclusion criteria for this study were as follows: (1) those in their first and second trimesters of pregnancy and (2) those with current morbidities apart from GDM.
Data Collection
Data were collected using methods, such as questionnaires and data sheets. After the assessment of the participants, they were openly questioned regarding their age, parity, and family history. A sonographic method was used to measure fetal AAWT. A Toshiba Xario ultrasound equipment system (Toshiba Medical Systems Corporation, Tochigi, Japan) equipped with a curvilinear transducer, with a frequency range of 3–5 MHz was used to evaluate the participants in the supine position. The transducer was placed perpendicular to the floor and parallel to the longitudinal axis of the participant. The fetal AAWT was measured at the fetal abdominal circumference. Measurements of the AAWT, amniotic fluid index, and estimated fetal weight (EFW) were recorded. Pregnant participants at risk of GDM underwent additional screening using an oral glucose tolerance test (OGTT). The process involved the initial testing of the fasting plasma glucose levels. Subsequently, a 100-g oral glucose solution was administered, and after administration, the plasma glucose levels were measured at 1-, 2-, and 3-hour intervals. GDM was diagnosed when participants exhibited abnormal values of at least two of these measurements. A diagnosis of GDM based on the blood glucose levels was used as a reference standard for the comparative analyses.
Statistical Analyses
Descriptive data were presented using graphs, tables, and charts. Descriptive frequencies were used to present qualitative data. Conversely, means and standard deviations were used to convey quantitative data. An independent sample t test, analysis of variance, and chi-square test were used to compare the characteristics between the two cohorts of participants. A final analysis was performed to determine the strength of the correlation between the EFW and AAWT values. Collected data were analyzed using IBM SPSS Statistics version 25.0 (IBM Corp., Armonk, New York). Statistical significance, selected a priori, was set at P < .05.
Results
The demographic characteristics encompassing both cohorts of participants included an age range of 16–46 years, with a mean age and standard deviation of 32.13 and 6.94 years, respectively. Furthermore, box plots have been provided to demonstrate the distribution of data and their apparent normality (see Figure 1).
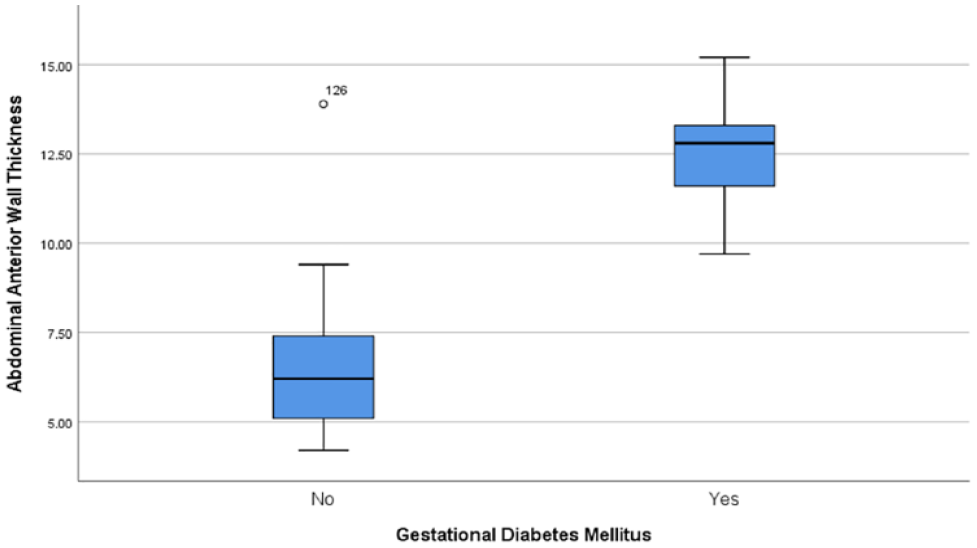
The box plot of the distribution of the cohort data, based on the fetal abdominal anterior wall thickness and maternal gestational diabetes.
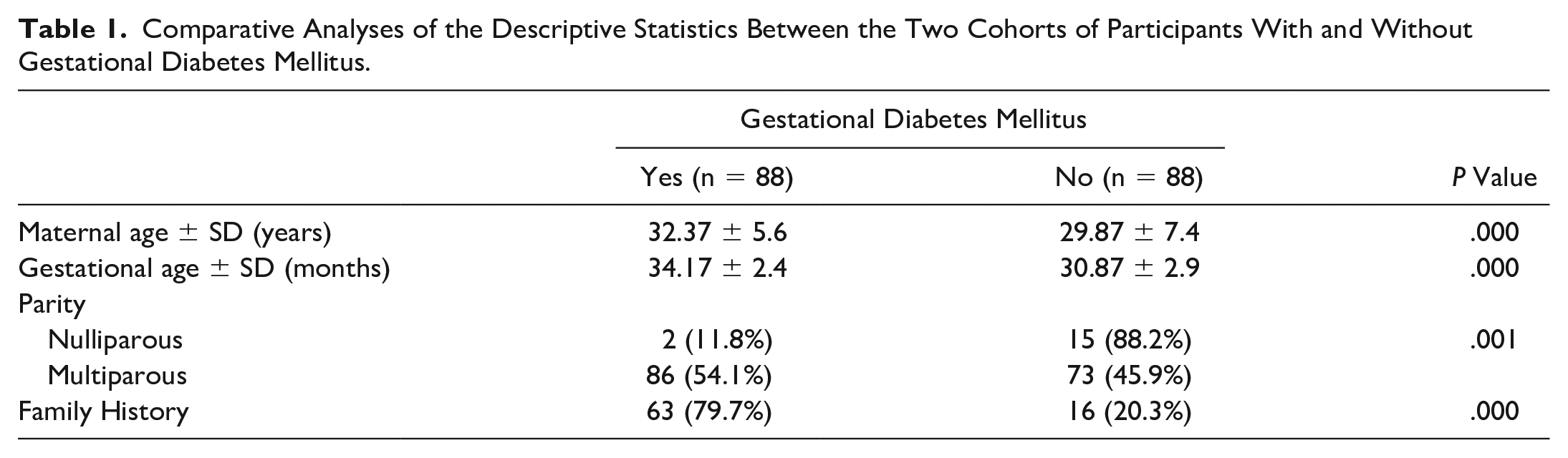
In addition, a statistically significant correlation was observed between GDM and various maternal factors collected from the interviews. Notably, these factors included the maternal and gestational age, parity, and family history, all of which suggested a potential impact on the occurrence of GDM (see Table 1).
Comparative Analyses of the Descriptive Statistics Between the Two Cohorts of Participants With and Without Gestational Diabetes Mellitus.
The GDM and control cohorts had mean fetal AAWT values of 12.65 ± 1.276 and 6.389 ± 1.503, respectively. Thus, a statistically significant difference in the fetal AAWT values was observed between the GDM and control cohorts.
The mean EFW values of the GDM and control cohorts were 2582.00 ± 577.363 and 1711.88 ± 563.599, respectively (see Table 2, Figures 2 & 3). Therefore, statistically significant differences based on EFW were observed between the GDM and control cohorts.
Comparative Analyses of the Descriptive Statistics Regarding the Fetal Anterior Abdominal Wall Thickness Between the Two Cohorts of Participants With and Without Gestational Diabetes Mellitus.
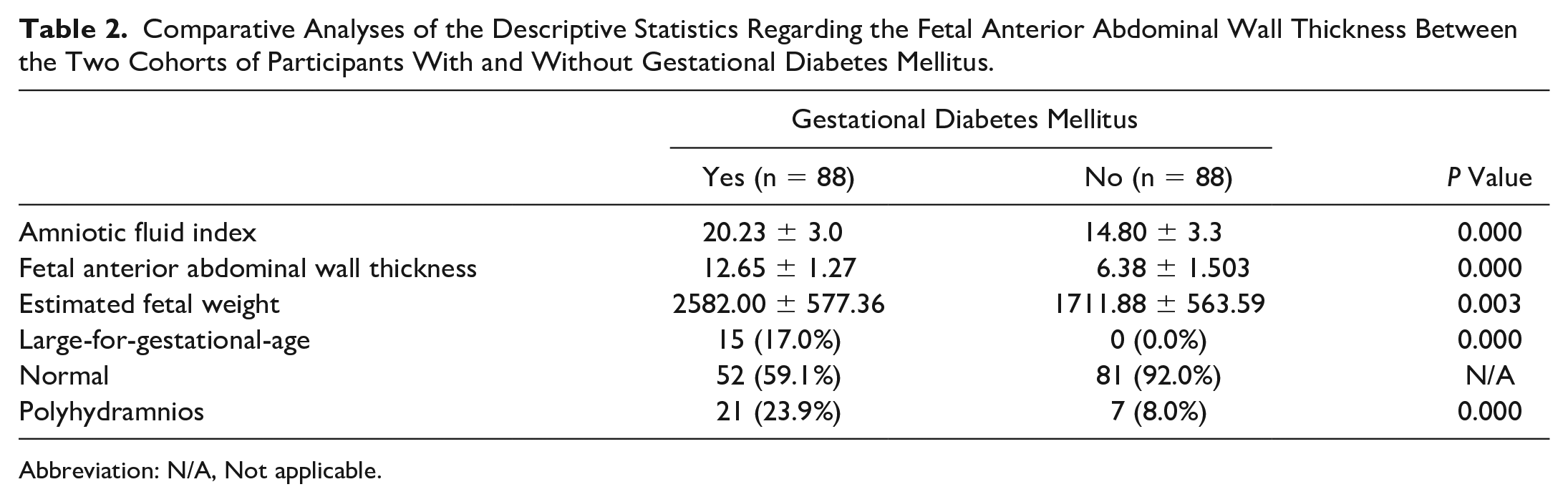
Abbreviation: N/A, Not applicable.
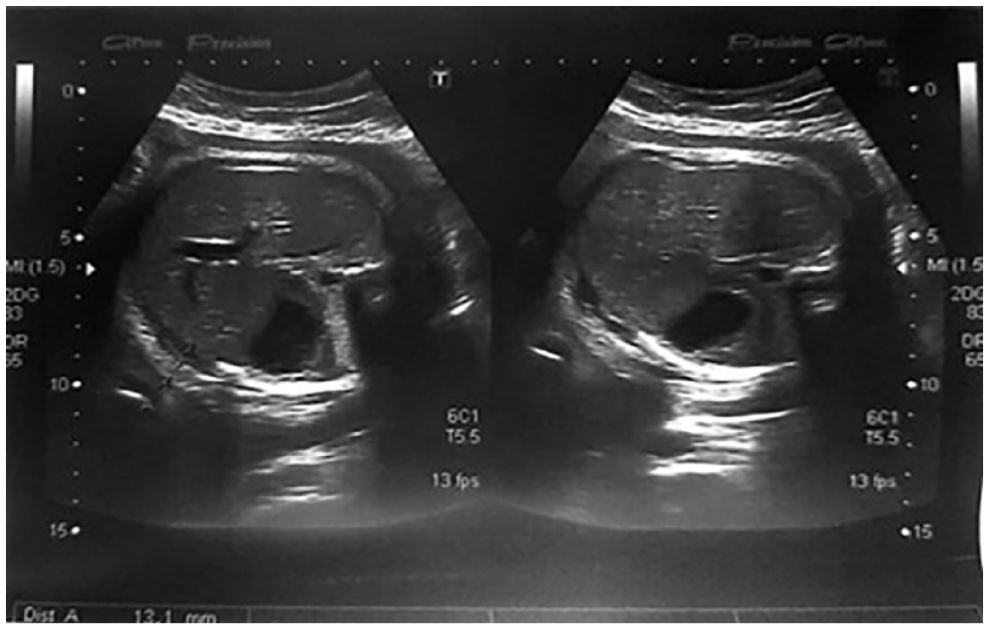
A transverse sonographic fetal view revealing an increased anterior abdominal wall thickness of 13.1 mm in pregnant participants at 33 weeks of gestation, diagnosed with gestational diabetes.
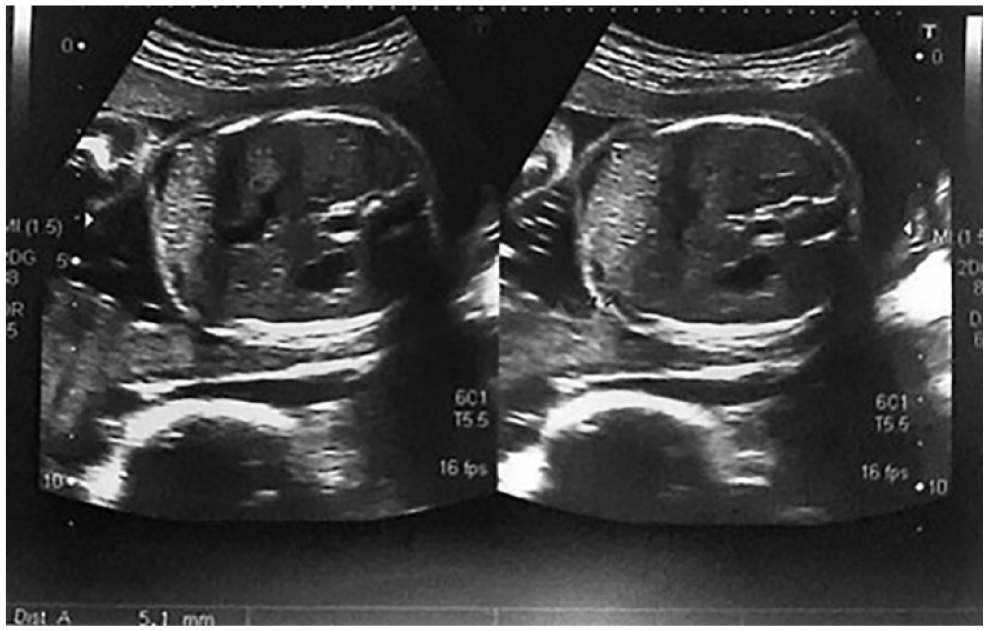
A transverse sonographic fetal view revealing a normal anterior abdominal wall thickness of 6.1 mm in pregnant participants at 33 weeks of gestation, without gestational diabetes.
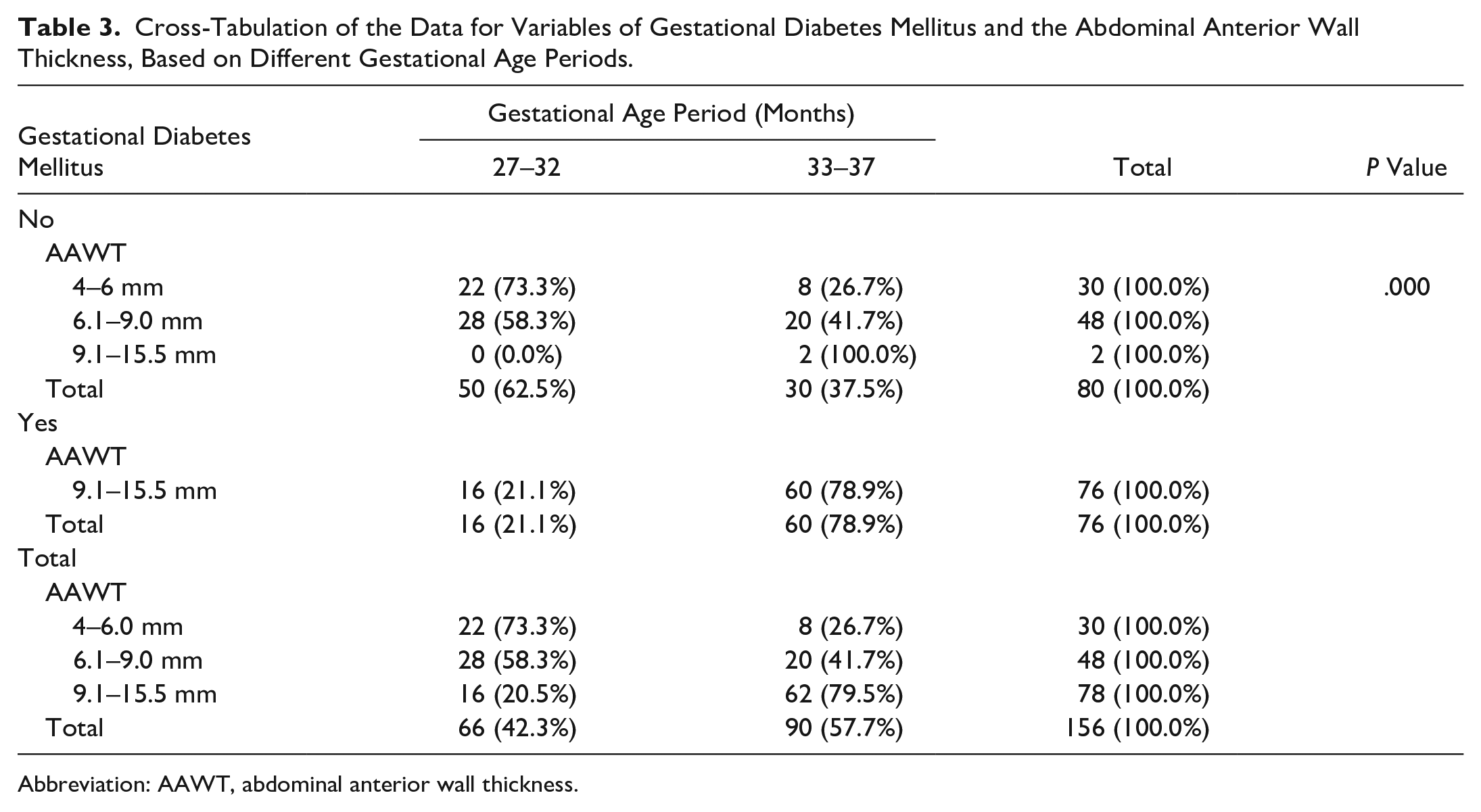
In the control cohort, 73.3% and 26.7% of the participants had AAWT values within the range of 4–6 mm, during the gestational age periods of 27–32 and 33–37 weeks, respectively. Furthermore, 41.7% and 100% of the participants in this cohort had AAWT values within the ranges of 6.1–9 and 9.1–15.5 mm, respectively, during the gestational age period of 33–37 weeks.
In the GDM cohort, 21.1% and 78.9% of participants had AAWT values within the range of 9.1–15.5 mm, during the gestational age periods of 27–32 and 33–37 weeks, respectively.
The highest percentage of AAWT values at 79.5% and 78.9% was within the range of 9.1–15.5 mm and was observed overall and in the GDM cohort, respectively, during the gestational age period of 33–37 weeks (see Table 3).
Cross-Tabulation of the Data for Variables of Gestational Diabetes Mellitus and the Abdominal Anterior Wall Thickness, Based on Different Gestational Age Periods.
Abbreviation: AAWT, abdominal anterior wall thickness.
Furthermore, a statistically significant positive correlation between the EFW and AAWT values was observed (P = .01) (see Table 4).
Strength of the Correlations Between the Estimated Fetal Weight and Anterior Abdominal Wall Thickness.
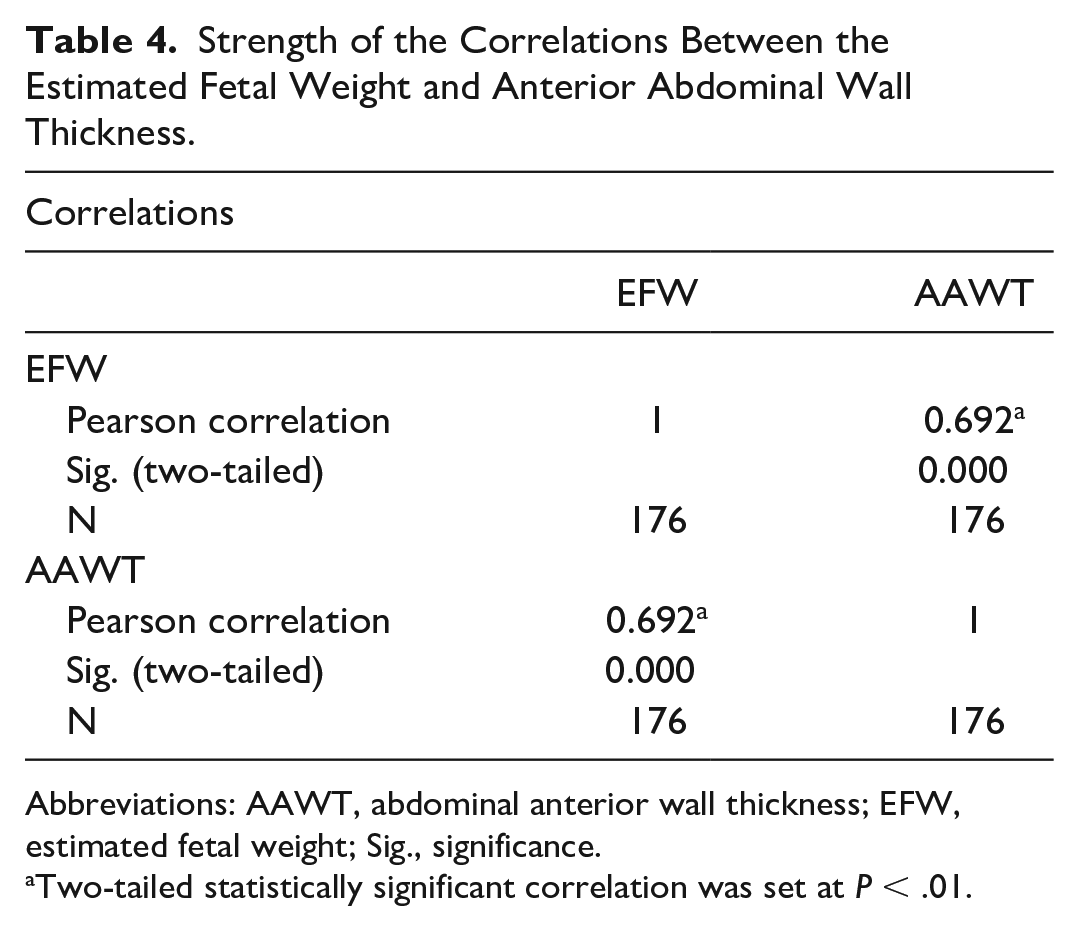
Abbreviations: AAWT, abdominal anterior wall thickness; EFW, estimated fetal weight; Sig., significance.
Two-tailed statistically significant correlation was set at P < .01.
Discussion
In this cross-sectional study, a comparative analysis of the fetal AAWT values between participants with and without GDM was conducted. Findings of this study revealed a higher fetal AAWT value in the GDM cohort than that in the control cohort.
Comparison With the Findings of Previous Studies
In a similar investigation, Aksoy et al. assessed the efficacy of sonography in measuring the fetal AAWT value in 48 singleton pregnant women with an impaired 50-g OGTT, however with a normal 100-g OGTT. Their findings highlighted that an increased AAWT can be detected in pregnant women with an impaired 50-g OGTT. 15
In addition, a study by Gardeil et al. 16 demonstrated that assessing the thickness of the abdominal subcutaneous adipose tissue was an easy-to-use method, for predicting fetal growth restriction and large-for-gestational-age (LGA) fetuses. These findings are consistent with those of this study, which highlighted that fetal AAWT was considerably higher in pregnant participants with GDM than that in those without GDM.
In a study by Ali et al., sonography was used to evaluate the AAWT prior to delivery in both GDM and non-GDM pregnancies. Their findings revealed that the GDM pregnancies have larger sonographic AAWT, EFW, and actual birth weight values than those of the non-GDM pregnancies, as measured by a neonatologist. 17 Therefore, their findings are consistent with the findings of this study.
Aberrant fetal growth in both nondiabetic and diabetic pregnant women has been correlated with an increased fetal AAWT value. The AAWT values have been found to increase in both GDM and non-GDM pregnancies, as the gestational age progresses; the fetuses of pregnant women with GDM have considerably higher AAWT values, during the third trimester. Moreover, pregnant women with GDM have significantly more fetal fat than that of pregnant women without GDM, throughout the first trimester of pregnancy. 18
The findings of a study by Tantanasis et al. have revealed that evaluating the AAWT and EFW values using a noninvasive approach, such as sonography, may help to better detect GDM and reduce any potential morbidities resulting from a missed diagnosis. Furthermore, these sonographic measurements may be beneficial in cases of pregnant women not tolerating the OGTT or in those who have received prior inadequate follow-up treatment. 19 Once again; the findings of this study support those of this earlier study, regarding the AAWT value being higher in pregnant women with GDM than that in those without GDM.
The AAWT value has been found to be considerably greater in GDM pregnancies, particularly when other routine fetal biometrics possess equivalent values, at the time of GDM screening. This suggests that the AAWT value can potentially serve as an early sonographic indication of GDM. 20
In addition, neonates deemed LGA had considerably higher AAWT values at each prenatal measurement time point during the gestational period. However, only women who have GDM with LGA births have been shown to have significantly increased AAWT values at each time point of 28–30, 32–34, and 36–38 weeks. Conversely, women who have GDM with LGA births only have significantly increased AAWT values at 36–38 weeks. Thus, LGA neonates born to mothers with GDM may be identified as early as 28–30 weeks of gestational age. Consequently, an increase in the AAWT value can potentially be utilized as a predictor of a higher risk for LGA births in women with GDM. 21
Limitations
The major limitations of this study were its study design and convenience sampling of participants. Because these participants were assigned to cohorts, this preexperimental design was limited by challenges to internal and external validity. Furthermore, to improve the generalizability of GDM predictors in clinical practice, further external validation studies are recommended. 22 The results of this study are unique to these pregnant participants and their fetuses, and generalizability to other study populations is less feasible. In addition, the accuracy of sonographic measures, such as the AAWT value, could be hindered because the examinations were conducted during the third trimester, when pregnant women tend to be obese. The sample size of 88 participants within each cohort was small, thus limiting the statistical power and effect.
Conclusion
This study highlighted that pregnant women with GDM have considerably higher fetal AAWT values than those without GDM. Moreover, the findings revealed that the AAWT and EFW values were correlated with each other. Thus, an increased fetal AAWT value can potentially be utilized as a noninvasive parameter for identifying pregnancies at risk of developing GDM, thereby preventing maternal and neonatal adverse events through timely intervention. These findings need to be replicated and externally validated in larger and other populations to further advance the clinical relevance of the fetal AAWT value, as a diagnostic indicator for GDM.
Footnotes
Ethics Approval
Ethical approval for this study was obtained from the Research Ethics Committee of the University of Lahore (IRB#REC-UOL-/195-07/2022).
Informed Consent
Informed consent was obtained from all study participants for their participation in the study, for completion of questionnaires and data sheets, and for publication of their de-identified data. This study was conducted in accordance with the principles of the Declaration of Helsinki.
Animal Welfare
Guidelines for humane animal treatment did not apply to this study because no animals were used during the study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
