Abstract
Congenital heart disease (CHD) is the existence of structural heart problems since the formation of the heart prebirth. There are different categories of CHD, all having their own unique frequency and severity characteristics. One type of CHD is a ventricular septal defect (VSD), more specifically outlet VSD. A VSD is the most common congenital heart defect; however, their presentation can be quite complex. Color and spectral Doppler transthoracic echocardiography (TTE) are valuable tools used in the diagnosis of VSDs. The often-challenging presentation of these defects requires sonographers to confirm their existence through the parasternal long-axis, parasternal short-axis, apical four chamber, apical three chamber, and apical five chamber scanning windows. Although VSDs are the most common congenital heart defect, their wide-ranging uniqueness and location along the interventricular septum (IVS) to assess all areas along the IVS on all exams, specifically those that demonstrate left-to-right shunting. The objective of this case report was to stress the importance of multi-window assessment during the sonographer’s interrogation of outlet VSDs, due to the potential implications on adjacent anatomy and the potential for surgical intervention.
Noninvasive cardiovascular imaging, specifically transthoracic echocardiography (TTE), has persistently evolved in a manner to transform patient care since the first reported use of sonography in 1954. 1 TTE has been a steadfast tool in the diagnosis and surveillance of structural heart disease patients, specifically ventricular septal defects (VSD). 2 These left-to-right shunts can be categorized as either acquired or congenital. VSDs can be acquired through trauma, injury during surgery, and myocardial infarction (MI). 3
Case Report
The case presented is of a 36-year-old woman who presented to the echocardiography lab for preoperative clearance. Upon initial evaluation from the ordering provider, a systolic murmur was auscultated, which promoted noninvasive evaluation by two-dimensional (2D) transthoracic echocardiography (TTE). The patient’s blood pressure was documented at 107/56 and she was in sinus rhythm, during her TTE appointment. All the cardiac images were acquired using a Philips Epiq ultrasound equipment system (Bothell, Washington, USA) and the sonographer utilized the lab’s complete TTE protocol. The 2D TTE demonstrated left ventricular systolic function, size, and structure with no evidence of diastolic dysfunction.
Upon interrogation of the parasternal long-axis (PLAX), a small left-to-right shunt was visualized as the sonographer strategically displayed color Doppler imaging (CDI) over the aortic root and the basilar septum (See Figure 1). A continuous wave (CW) Doppler through the defect demonstrated a velocity of 5.7 m/s (See Figure 2). Further assessment during parasternal short-axis imaging revealed a similar appearing flow near the right coronary cusp at the 2 o’clock position (See Figure 3). Additional CW Doppler imaging uncovered a higher velocity when compared with the PLAX at 5.9 m/s (See Figure 4). Because different types of VSDs can be excluded with additional imaging windows, the sonographer continued to sample through the apical-five chamber and subcostal five-chamber views. In addition, the sonographer included a label on the image, to alert the reading physician that he recognized the shunt while completing his full, sweeping assessment (See Figures 5 and 6). The patient was referred to a cardiovascular surgeon for surveillance and risk stratification for potential closure.
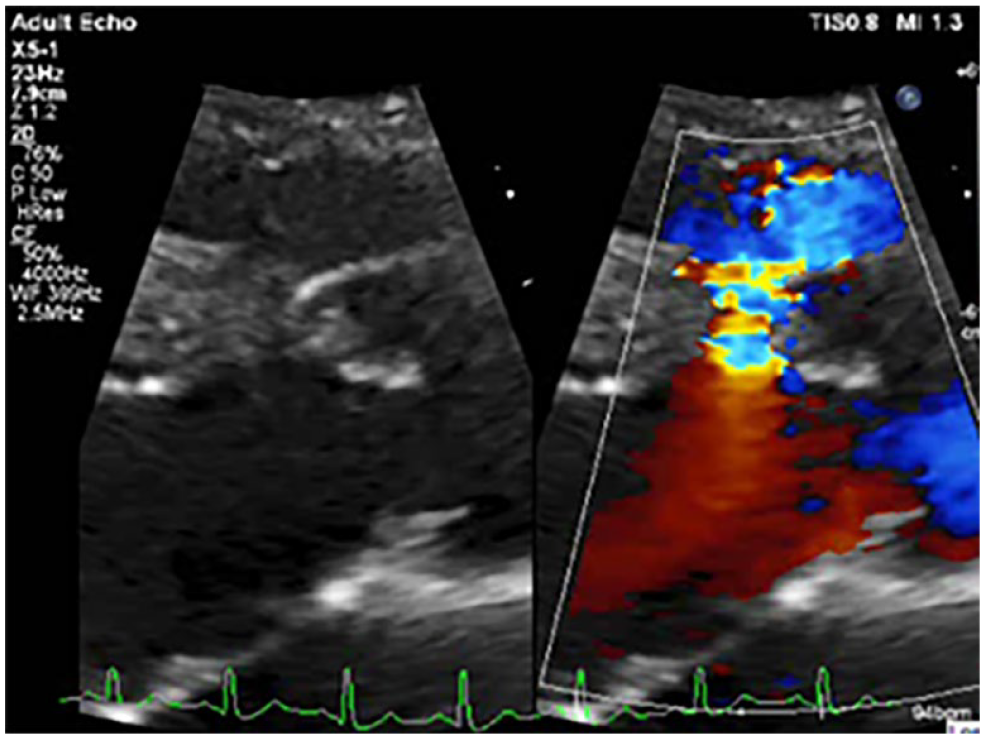
A zoomed PLAX view with color Doppler demonstrating a left-to-right shunt near the AV. AV, aortic valve; PLAX, parasternal long-axis.
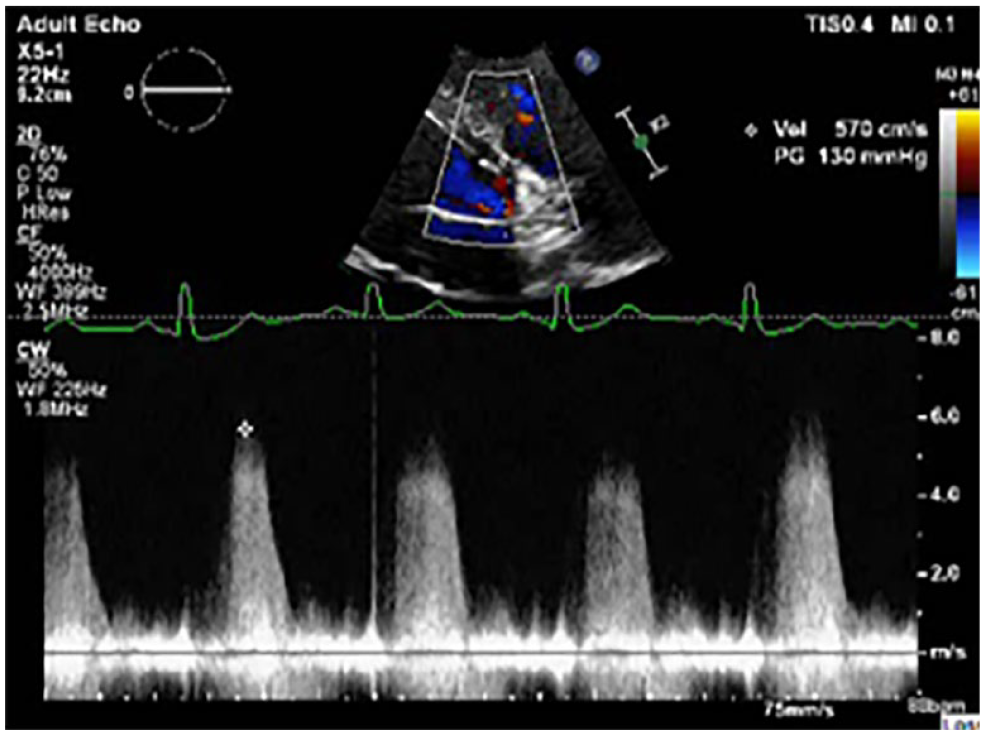
An example of the continuous wave spectral Doppler assessing a left-to-right shunt with a velocity of 5.7m/s.
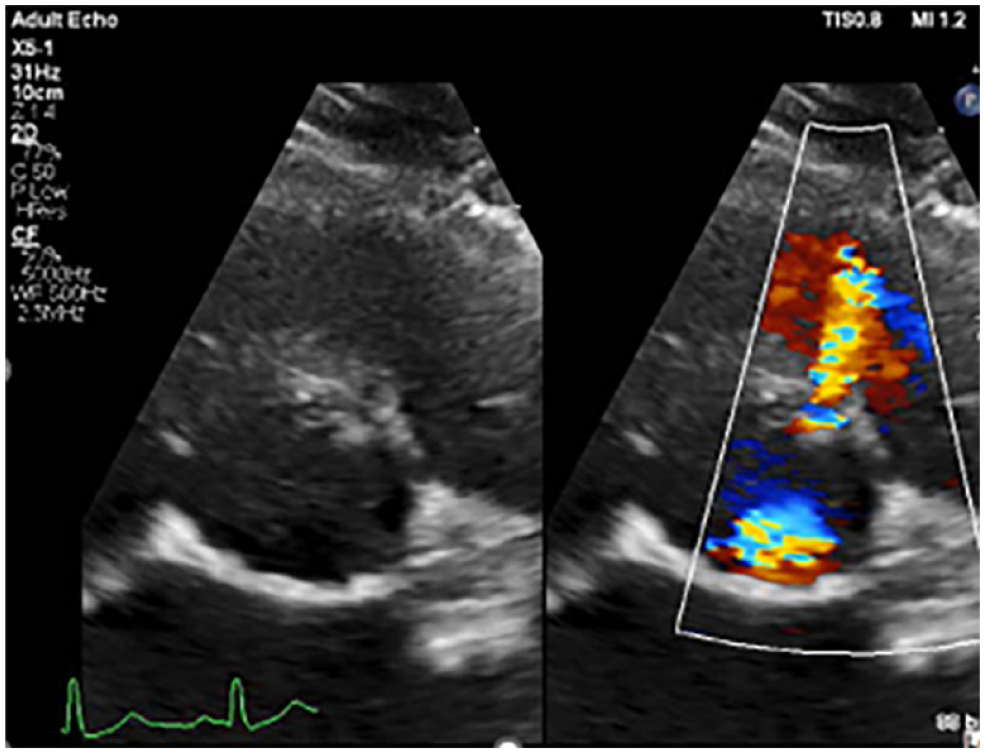
Additional imaging of a suspected VSD with a zoomed PSAX view with color Doppler demonstrating a left-to-right shunt near the AV in the RVOT. AV, aortic valve; PSAX, parasternal short-axis; RVOT, right ventricular outflow tract; VSD, ventricular septal defect.
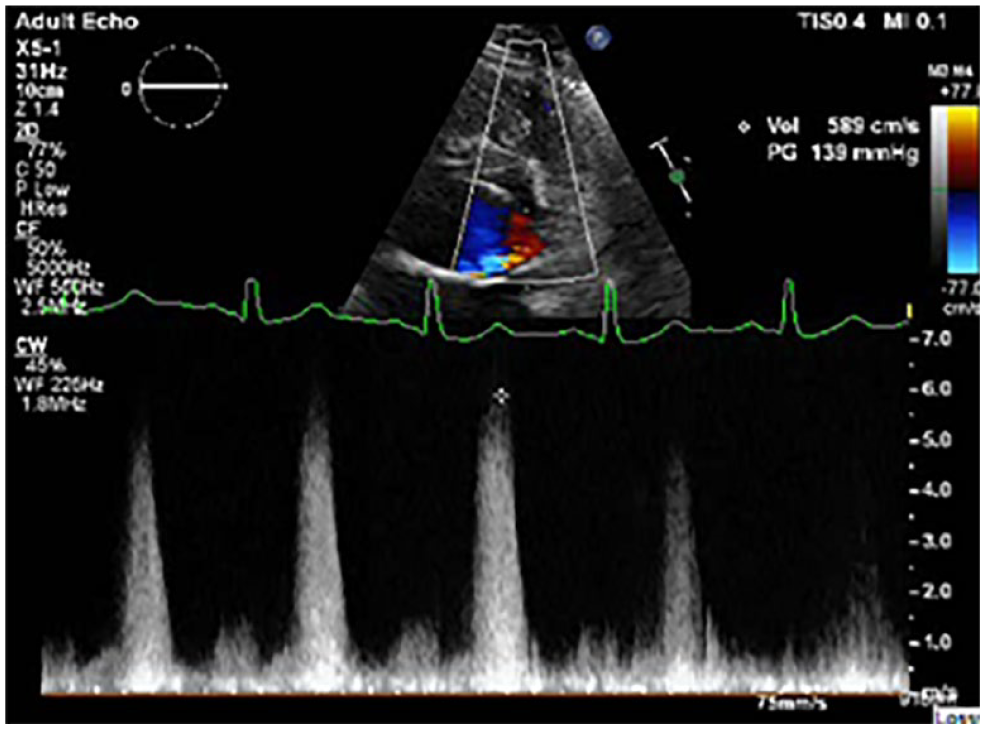
An example of continuous wave spectral Doppler assessing a left-to-right shunt with a velocity of 5.89 m/s. Due to the position of the heart and sampling, a high velocity was achieved when compared with the PLAX. PLAX, parasternal long-axis.
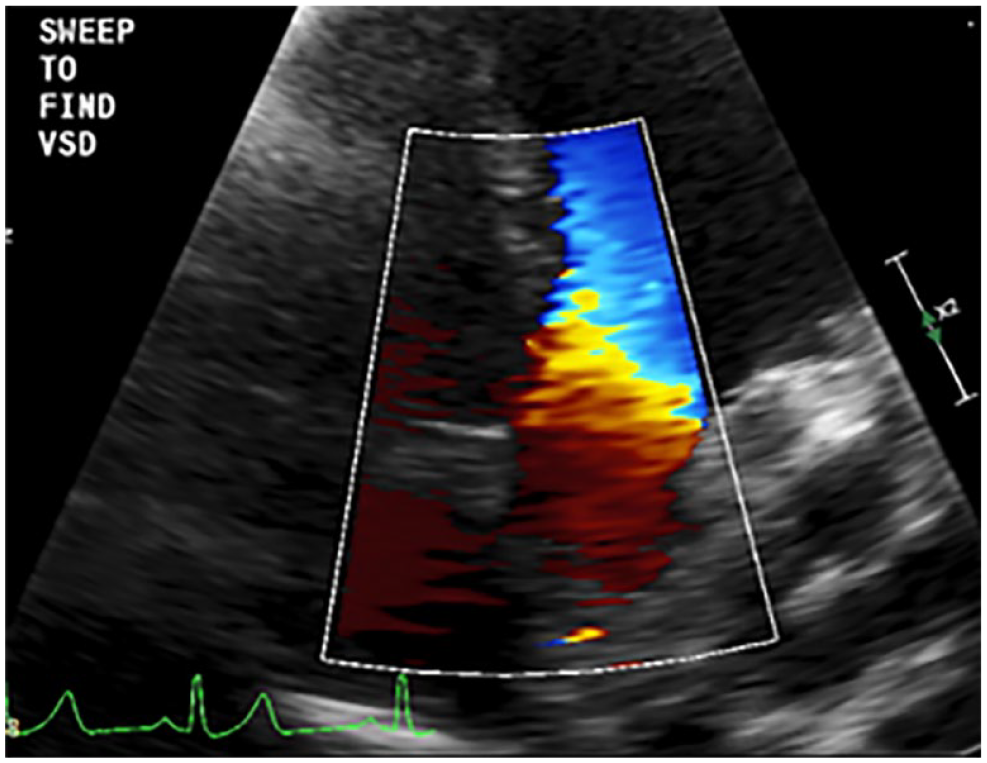
An apical five-chamber view demonstrating the sonographer sweeping through multiple windows to accurately characterize the suspected supracristal VSD. VSD, ventricular septal defect.
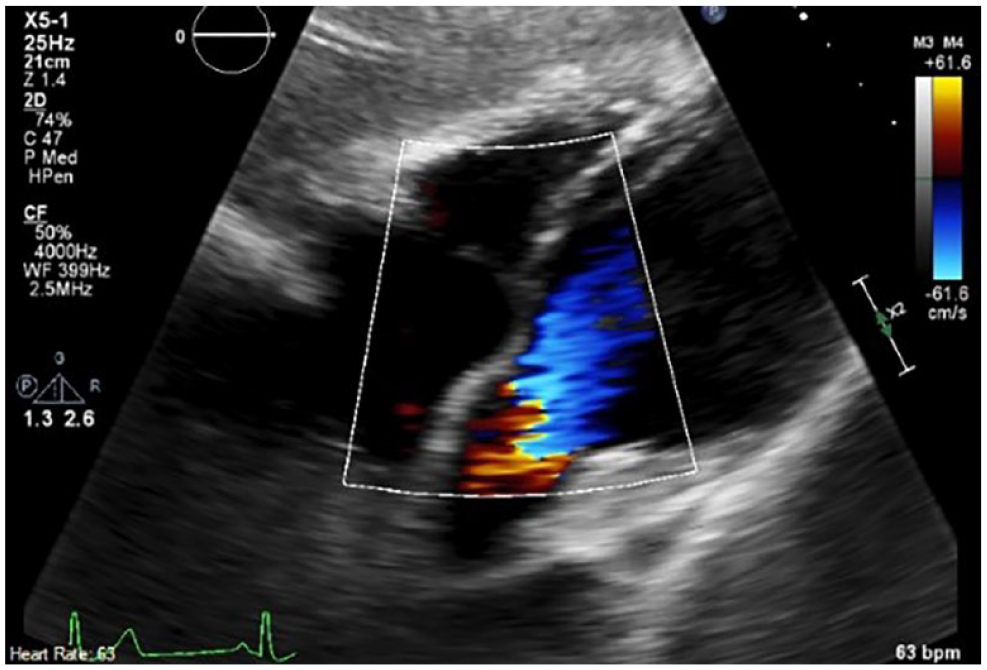
A subcostal imaging demonstrating the sonographer sweeping through multiple echocardiographic windows to fully assess a suspected supracristal VSD. VSD, ventricular septal defect.
Discussion
Ventricular septal defects are the most common form of congenital heart disease in children and the second most common in adults.4 –6 VSDs have a wide range of appearances and can be located across all areas of the ventricular septum. These left-to-right shunts are created due to early fetal morphogenesis failure of the membranous, trabecular, infundibular, atrioventricular, and inlet areas of the ventricular septum.7,8 Spontaneous closure can occur, but this is highly dependent upon age, the type, and size of the VSD. 9 When unable to close naturally, the resulting bi-ventricular communications are classified into four types of VSDs: outlet, membranous, inlet, and trabecular. 5 There has been historical debate on how each VSD phenotype is categorized. The best practice consensus, from the International Society for Nomenclature of Pediatric and Congenital Heart Disease, created a system of combining both geographic system and border approach, with adjacent anatomical anatomy. 8 Regarding the present case findings on TTE, Figure 7A and B demonstrated the possible locations of VSDs along the IVS, as seen in the PLAX, parasternal short-axis (PSAX), apical four chamber, apical five chamber, and apical three chamber echocardiographic windows. As previously discussed, this case highlights an outlet VSD which was located below the aortic valve (AV) and above the crista supraventricularis.
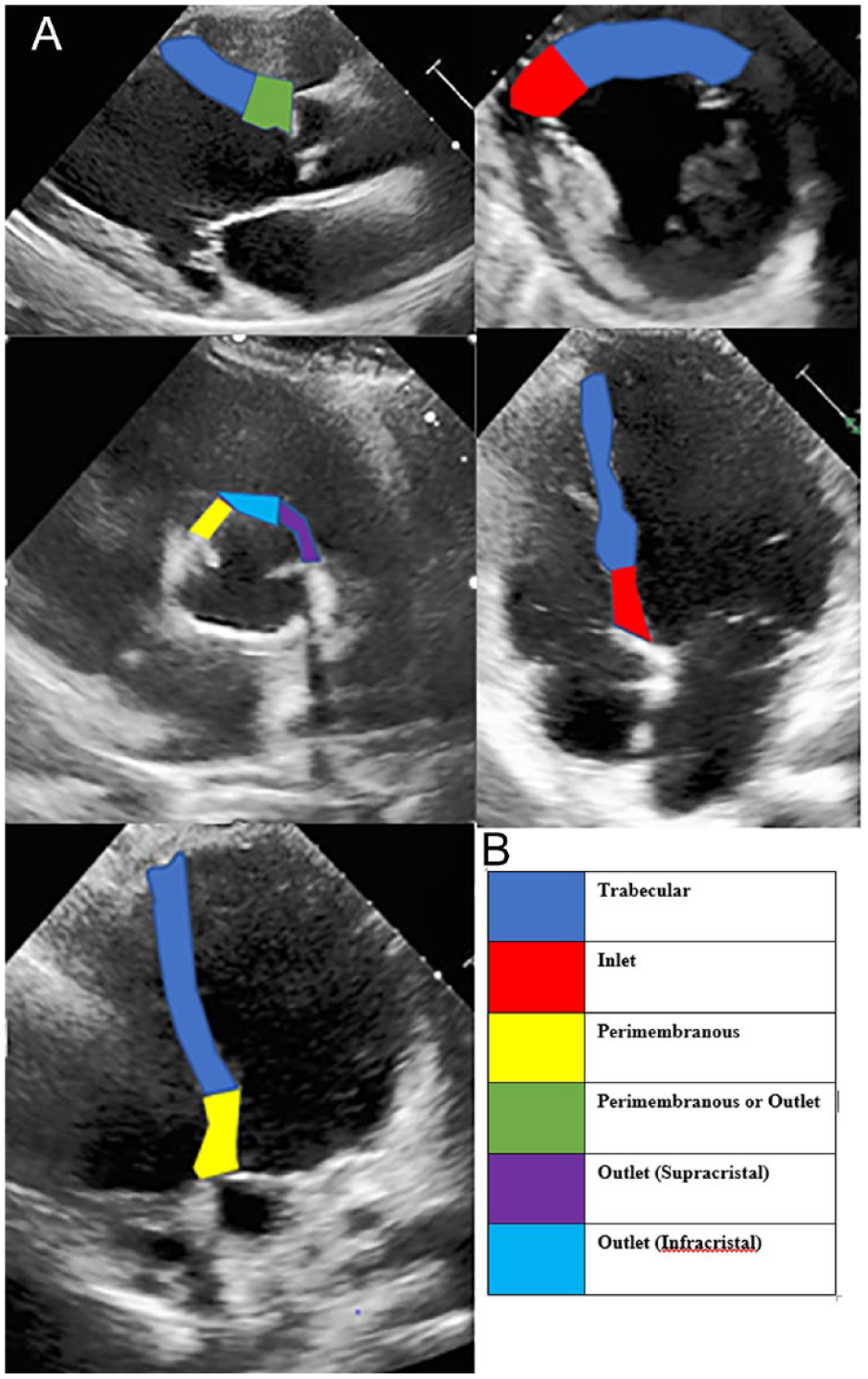
(A) The echocardiographic scanning windows demonstrating LV wall segment territories and the associated VSD with that territory. (B) Color coding table depicting the associated VSD with its location across the ventricular septum. LV, left ventricular; VSD, ventricular septal defect.
The location of outlet VSDs, upon initial inspection in the PLAX, is like that of perimembranous VSDs. Because of this similarity, sonographers should continue to sweep/sample CDI across all parts of the ventricular septum in multiple windows to facilitate accurate characterization of a true outlet VSDs. All complete TTE examinations should include in an exhaustive practice of utilizing CDI of the left ventricular outflow tract (LVOT) and AV, however, when outlet VSDs are suspected, greater scrutiny must be placed on interrogating the PLAX, PSAX, apical five-chamber, and apical long-axis view windows. 10 Special attention should continue to be paid to the color gain settings as different flows may require certain adjustments. In addition to comprehensive CDI in multiple scanning windows, CW should be conducted in multiple windows through the VSD to ensure that sonographers are obtaining the most accurate gradient. As seen in this patient case, CW was taken from both the PSAX and PLAX, which demonstrated different gradients. This practice of multiple window assessment is critical in defining the true characteristics of VSDs and associated hemodynamics as the most desired angle to sample CW may not always be the first window where the VSD is visualized.
This case report emphasizes this type of outlet VSD due to the associated pathology and need for potential surgical correction. VSDs are usually classified as an outlet VSD when the primary location is in close proximity to the pulmonary valve (PV). These defects open directly into the RV outlet and are subclassified further into perimembranous outlet and muscular outlet defects. 8 Although close to the PV, a common finding of outlet VSDs is associated aortic regurgitation (AR) and aortic valve prolapse.11 –13 In a 2019 retrospective study conducted by Kumari et al, the medical records of 190 patients aged 18 and younger were reviewed for their VSD diagnosis. Aortic valve prolapse and AR were present in 28.9% and 23.2% of patients, respectively. 9 In patients specifically with an outlet VSD, AR was present in 83.3% of patients. These outlet VSDs are located just under the aortic valve and maintain the potential to cause the right coronary cusp to flow into the defect by a Venturi effect. 14
Although the AR in this report was mild, these findings further stress the need for sonographers to fully quantify AR using ASE recommended techniques such as vena contracta, flow convergence, jet height, pressure half-time, descending aorta assessment, regurgitant volume, regurgitant fraction, and effective regurgitant orifice area in the presence of VSDs. 15 Thorough assessment of the degree of AR in these patients facilitates the management of these types of VSDs. In fact, many labs and current literature recommend serial TTE when outlet VSDs are discovered, as AR is an indication for surgery. When caught late, AR can continue to progress postintervention due to longstanding shunt exposure on the aortic cusps.16,17 Early detection of mild AR and resulting surgical intervention has demonstrated to spare the AV and need for a second operation.
In addition, VSD area indexed to body surface area (BSA) and Qp:Qs are also valuable measurements that can help to risk stratify patients. 18 Although 2D TTE VSD indexed area is a useful tool, there can be limitations due to the shape of the VSD and hemodynamic changes during the cardiac cycle. Three-dimensional (3D) TTE, when available, displays increased visualization and assessment of the maximal diameter of a VSD.19,20 Underassessment and mischaracterization of VSDs, specifically outlet VSDs and peripheral aortic valve anatomy, can lead to missed pathology that would require surgical VSD closure or device closure intervention.21,22 In addition to aortic valve prolapse and AR, a sinus of Valsalva aneurysm (SOVA) is another condition that can be caused by an outlet VSD that compromises the integrity of the ventricular septal wall.23,24
Conclusion
The interrogation and assessment of adult congenital heart disease, specifically outlet VSDs, is a high-level responsibility of sonographers which maintains implications on patient prognosis. Due to the location and proximity to the aortic valve of these types of VSDs, it is crucial that sonographers understand the importance of multi-window assessment, being through in our pursuit to document pathology for interpreting cardiologists. Because associated valve repair/replacement is common with outlet VSDs, it is the sonographer’s duty to provide a complete echocardiogram to allow for accurate classification of these defects. If additional scanning windows were not acquired after the PLAX at the time of the TTE during this case study, the shunt may have been misrepresented as a different type of VSD and surgical planning would have been altered. This aim of this case report was to demonstrate the magnitude of comprehensively evaluating suspecting areas of left-to-right shunting to illustrate the characteristics of these shunts for potential intervention.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because this was a case report which in which the findings were analyzed retrospectively. The case study did not affect/alter the management in any way.
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
