Abstract
Pancreaticoduodenal artery aneurysm (PDAA) is a rare disease that is recognized as an intra-abdominal hemorrhage differential diagnosis. This case study reports on a ruptured PDAA case in which ultrasonography (US) proved useful for the initial diagnosis. A 60-year-old man presented to the emergency department with pain in the epigastrium and back. An abdominal US performed at presentation suggested a PDAA and intraperitoneal hemorrhage/retroperitoneal hematoma. In addition, a contrast-enhanced computed tomogram (CT) revealed a hematoma extending from the upper abdomen to the pelvic floor and an aneurysmal pancreaticoduodenal artery arcade dilatation. Furthermore, emergency abdominal angiography findings revealed bleeding from a posterior-inferior PDAA, and coil embolization was performed. Postoperative progress was good, and the postoperative CT at 7 days showed no rebleeding signs and/or enlargement or new PDAA development. Therefore, the patient was discharged from the hospital 14 days after embolization.
Pancreaticoduodenal artery aneurysm (PDAA) is often asymptomatic and is identified incidentally on diagnostic imaging, such as a computed tomogram (CT) or magnetic resonance imaging (MRI) study, in surveillance of other diseases. However, ruptured aneurysms cause fatal hemorrhage and thus require early diagnosis and treatment. 1 This study reports on a PDAA rupture that was diagnosed using ultrasonography (US) and treated via endovascular therapy.
Although there are many PDAA reports in previous literature, there are a limited number of studies diagnosed by US. Therefore, the literature on patients with PDAA was reviewed, and its clinical and imaging features were summarized. A literature search was performed to identify all case reports and clinical reviews on PDAA and US. A combination of medical subheadings and free text searches was used. Particularly, the following terms were used for literature searches: “pancreaticoduodenal artery aneurysm” and “ultrasound.” Databases, including MEDLINE, were reviewed to search for studies reported from January 2012 to July 2022, and nine cases in which US was performed at the time of PDAA diagnosis were considered for review. PDAA’s clinical features and imaging diagnosis based on these reports were summarized.
Case Report
A 60-year-old male patient presented to the emergency department with epigastric and back pain that had worsened over the previous day. He had no medical history of abdominal trauma, surgery, acute or chronic pancreatitis, or peptic ulcers.
During the physical examination, his vital signs were as follows: temperature, 35.5°C; heart rate, 57 beats per minute; and blood pressure, 87/49 mmHg. Moreover, he was in a state of shock, and his abdomen was flat and tender. His laboratory test results revealed elevated levels of C-reactive protein (3.45 mg/dL) and a slightly low hematocrit (39.1%), but his white blood cell count (5.7 × 103/µL), red blood cell count (428 × 104/µL), and hemoglobin level (13.0 g/dL) were normal.
At the time of presentation, the US examination revealed a turbid intraperitoneal free fluid collection around the liver on right hypochondriac longitudinal scanning (see Figure 1). On the epigastric transverse view, while scanning, a borderline indistinct hypoechoic lesion and a 12-mm-diameter cystic lesion were observed between the duodenum (descending to horizontal portion) and inferior vena cava (see Figure 2). Color Doppler imaging findings revealed turbulent blood flow inside the cystic lesion. In addition, continuous blood flow signals were visualized from both sides of the cystic lesion toward the celiac and superior mesenteric arteries (see Figure 3). No blood flow signal was present within the hypoechoic lesion. Other findings in the surrounding organs, such as traumatic changes or neoplastic lesions indicative of hemorrhage, were not noted. Therefore, intraperitoneal hemorrhage and retroperitoneal hematoma associated with PDAA rupture were suspected.
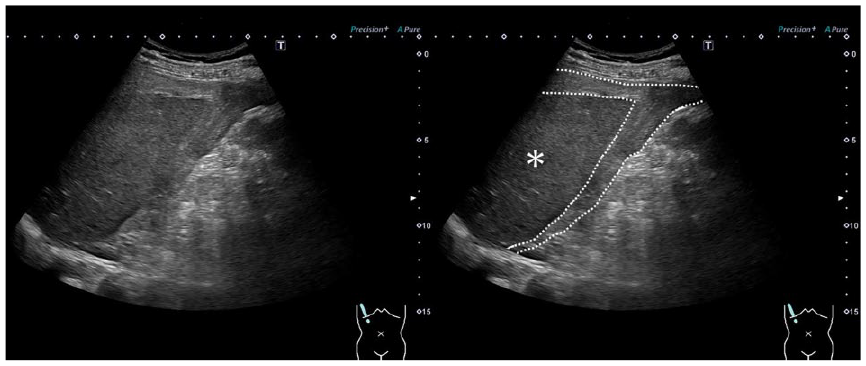
A sonographic longitudinal view which demonstrates turbid intraperitoneal free fluid collection (dotted circle) around the liver (asterisk) in the right hypochondrium.
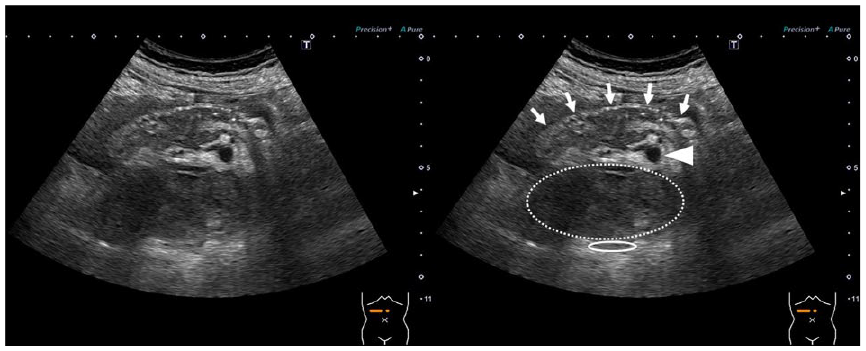
A sonographic transverse view that demonstrates a borderline indistinct hypoechoic lesion (dotted circle) and a 12-mm-diameter cystic lesion (arrowhead) that were observed between the duodenum (descending portion to horizontal portion) (arrow) and the inferior vena cava (circle), in the epigastrium.
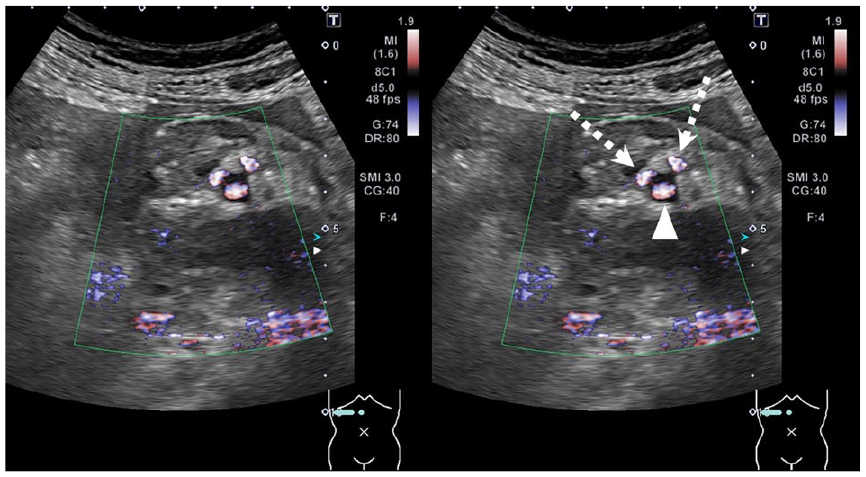
A color Doppler image that revealed turbulent blood flow inside the cystic lesion (arrowhead). Moreover, continuous blood flow signals were visualized from both sides of the cystic lesion toward the celiac and superior mesenteric arteries (dotted arrow).
Contrast-enhanced CT revealed a pale high-density area extending from around the duodenum to the pelvis, as well as aneurysmal dilatations in the pancreaticoduodenal artery arcade (see Figure 4A and 4B). No extravasation of the contrast medium was noted on the examination.
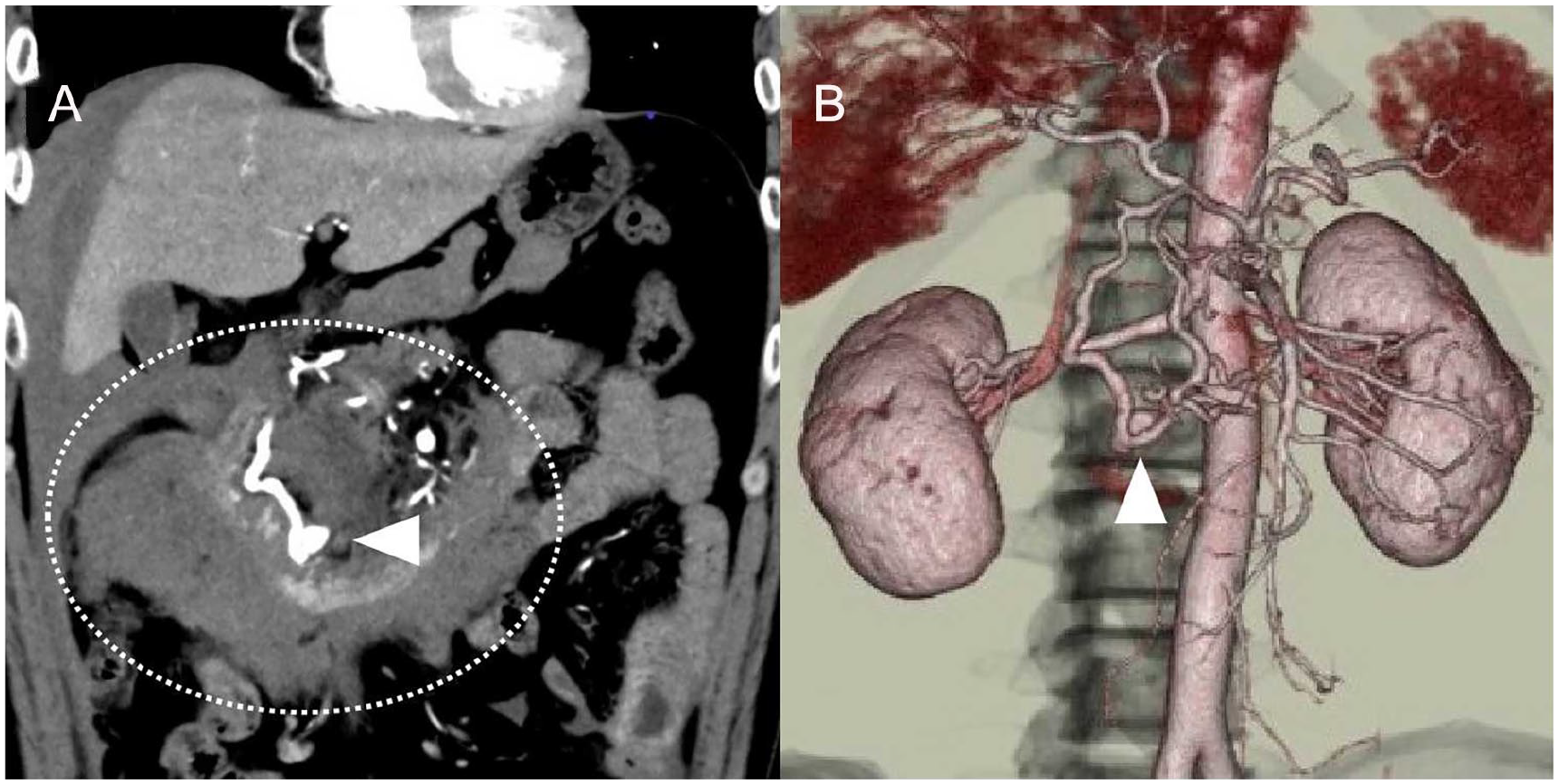
A computed tomogram (CT) that shows a high-density area extending from around the duodenum to the pelvis (dotted circle), and aneurysmal dilation in the pancreatoduodenal artery arcade (arrowhead). (A) In the coronal plane and (B) a three-dimensional computed tomographic view.
An emergency angiography was performed to determine the definitive diagnosis and treatment. The diameter of the blood vessels was slightly irregular, and there was stenosis at the celiac axis. Notably, an aneurysm and hemorrhage were observed from the posterior-inferior pancreaticoduodenal artery, and as a result, a coil embolization was performed (see Figure 5A and 5B).
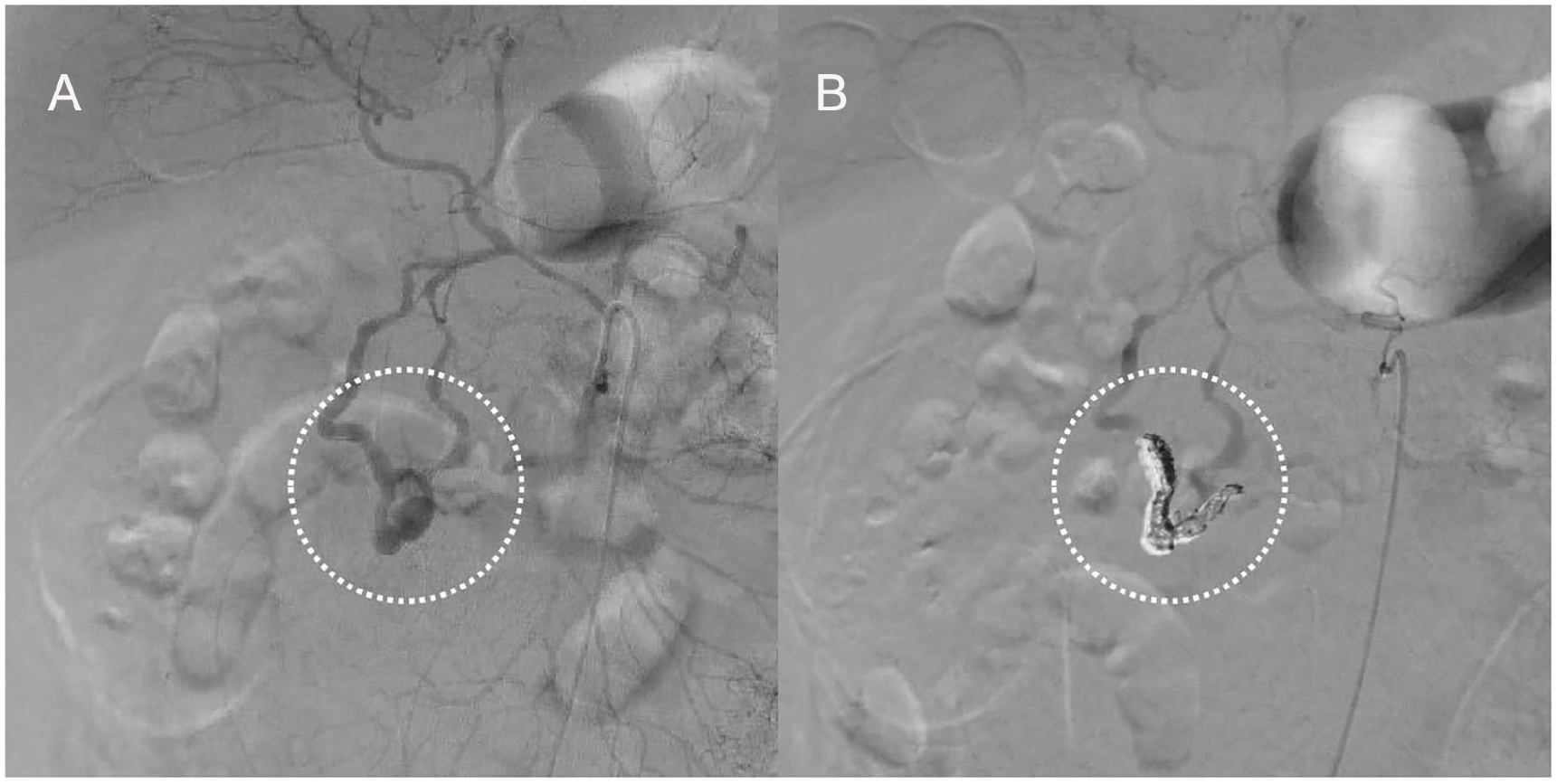
(A) An angiogram which demonstrates an aneurysm that was observed in the posterior-inferior pancreaticoduodenal artery (dotted circle), and (B) an image documents that a coil embolization was performed (dotted circle).
The postoperative progress of the patient was good, and the findings of the CT, performed on the seventh postoperative day, indicated no signs of rebleeding and/or enlargement or new PDAA development. Therefore, the patient was discharged from the hospital after 14 days of embolization. Notably, the subsequent CT examinations have revealed no recurrence of the aneurysm, over the last 3 years.
Discussion
Visceral aneurysms account for 0.1% to 0.2% of all aneurysms, with splenic, hepatic, and superior mesenteric artery aneurysms being the most common, in that order. Of these, PDAA is extremely rare, accounting for approximately 2% of all visceral aneurysms. 8 In particular, these aneurysms can be divided into two types: true aneurysms, which are associated with increased blood flow from the superior mesenteric artery because of stenosis or occlusion of the celiac axis, segmental arterial tunica media fusion, and atherosclerosis, 9 and pseudoaneurysms, which occur secondary to infection, trauma, or pancreatitis. 1
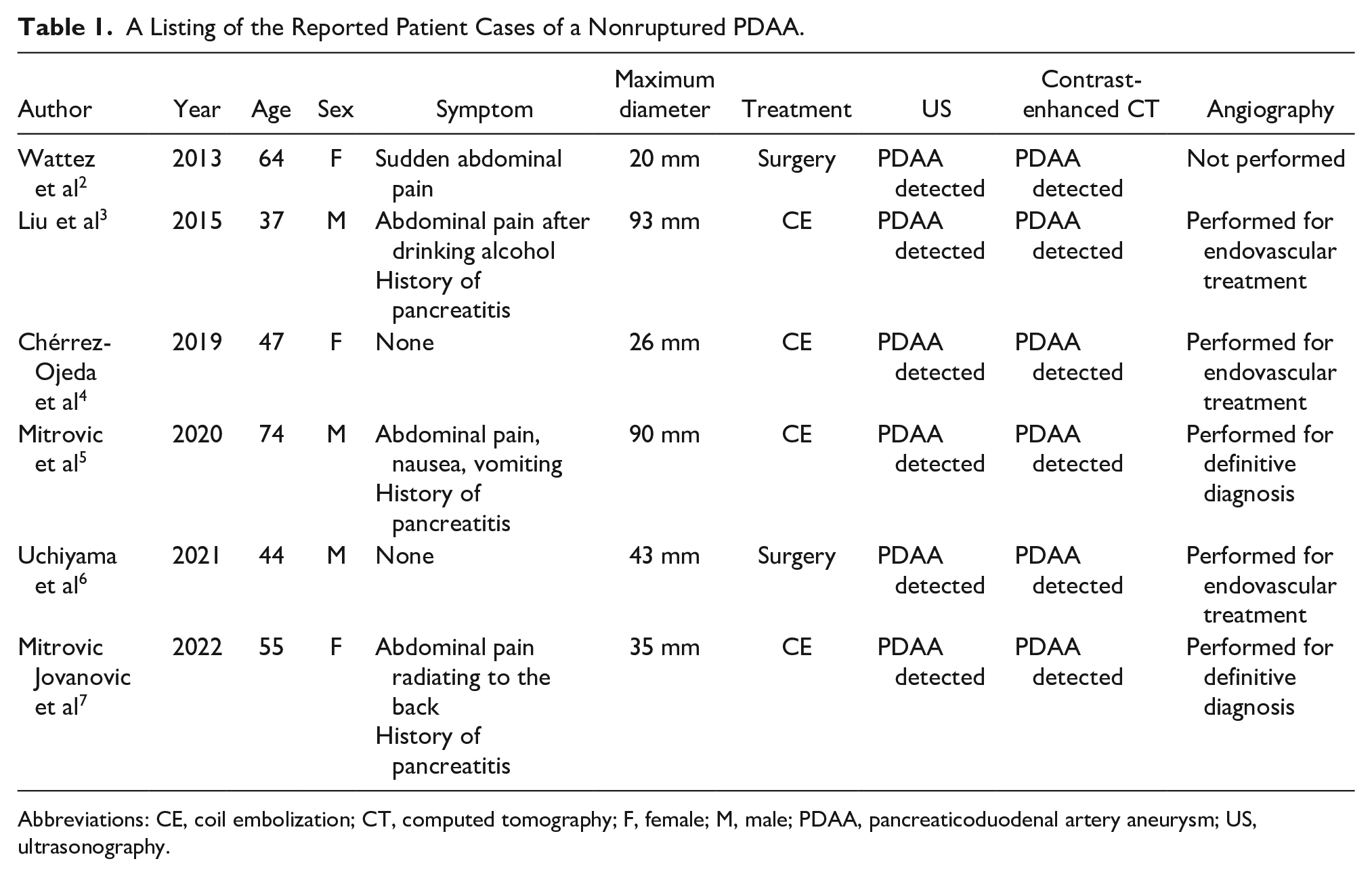
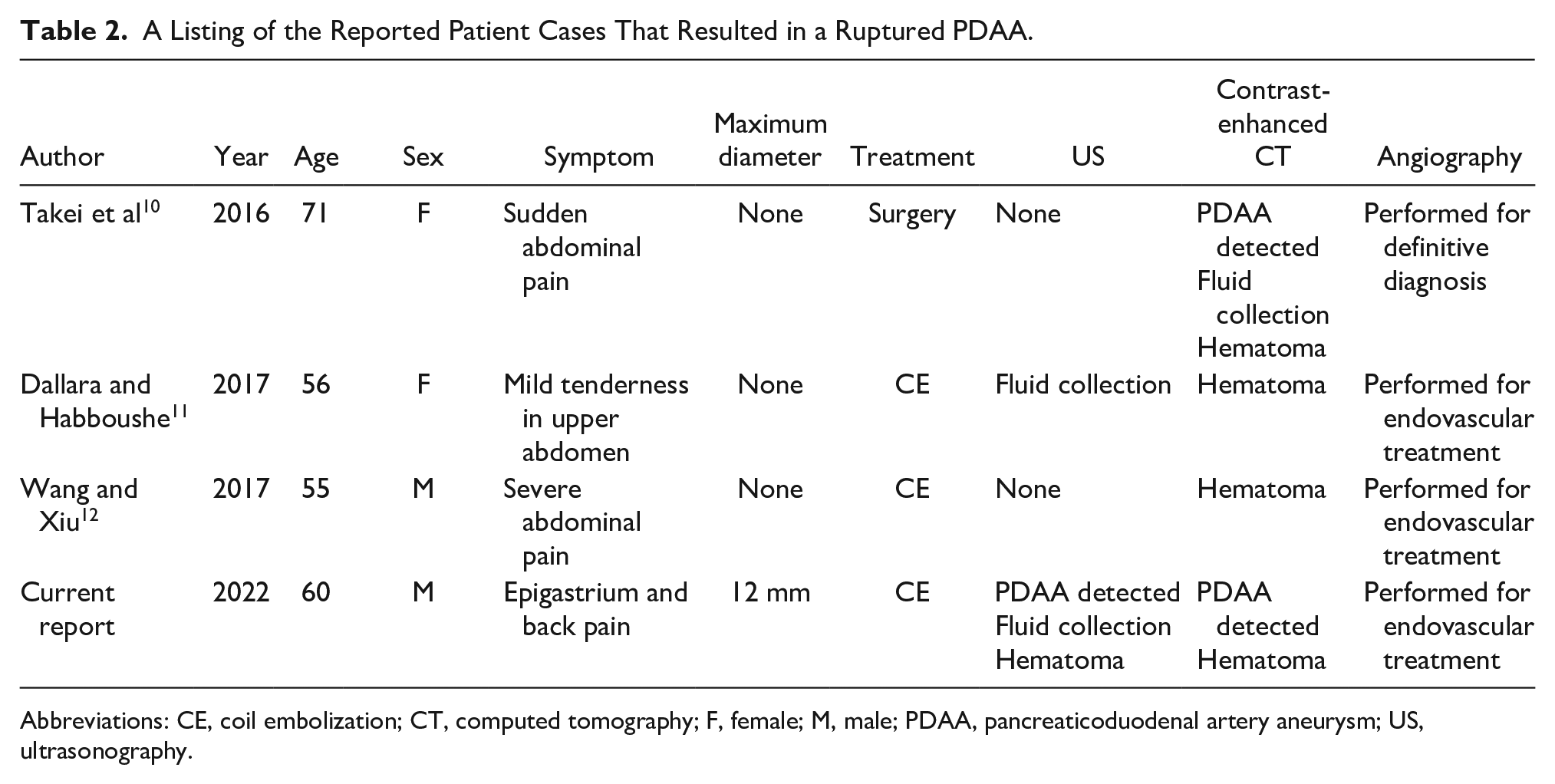
Generally, US, CT, and angiography are used for diagnostic imaging of PDAA. In the past 10 years, nine cases, excluding the present case, have been reported wherein US was performed at the time of diagnosis (based on English language publications). In the present study, the patients were classified into the nonruptured aneurysm group (n = 6) (see Table 1) and the ruptured aneurysm group (n = 4, including the present case; see Table 2).
A Listing of the Reported Patient Cases of a Nonruptured PDAA.
Abbreviations: CE, coil embolization; CT, computed tomography; F, female; M, male; PDAA, pancreaticoduodenal artery aneurysm; US, ultrasonography.
A Listing of the Reported Patient Cases That Resulted in a Ruptured PDAA.
Abbreviations: CE, coil embolization; CT, computed tomography; F, female; M, male; PDAA, pancreaticoduodenal artery aneurysm; US, ultrasonography.
Regarding the symptoms, among the six cases in the nonruptured aneurysm group,2–7 two were asymptomatic.4,6 Moreover, three of four patients with abdominal pain were considered to have pancreatitis-related abdominal pain.3,5,7 Notably, abdominal pain was present in all cases in the ruptured aneurysm group.10–12 In addition, except for one, 11 all patients had severe symptoms, such as sudden or severe abdominal pain. The present case also had severe abdominal pain that had worsened over the previous day.
US findings from previous studies indicated that the size of the nonruptured aneurysms ranged from 20 to 93 mm in maximum diameter. The US findings in one case 6 were not detailed, but all other cases were depicted as cystic masses with blood flow signals.2–5,7 PDAA was diagnosed in three of the cases during pancreatitis follow-up owing to the appearance of a blood flow signal on color Doppler inside a large hypoechoic cystic mass suggestive of a pancreatic pseudocyst or walled-off necrosis.3,5,7 On the contrary, the size was not noted in all cases in the ruptured aneurysm group. In addition, no aneurysmal findings were observed in any case, except for the present case, although the US findings in one case revealed fluid collection suggestive of intraperitoneal hemorrhage. 11 In the present case, the US examination revealed a cystic mass with a diameter of 12 mm, which was smaller than that of nonruptured aneurysms. Moreover, this mass was adjacent to a hypoechoic image suggestive of retroperitoneal hematoma. In addition to internal blood flow signals identified by color Doppler, real-time continuity with the surrounding vessels was traced, which provided a clue to suspect PDAA. With regard the PDAA size, previous studies on current management of PDAA reported that ruptured aneurysms were smaller than nonruptured aneurysms as all nonruptured aneurysms had a diameter of ≥10 mm, whereas ruptured aneurysms were almost always ≤10 mm in diameter, 13 and the present case also had a smaller aneurysm compared with the nonruptured group. For the nonruptured aneurysm group, the US results indicated the presence of an aneurysm, and contrast-enhanced CT was performed in all six cases to understand the detailed pathophysiology. In addition, angiography was performed mainly for endovascular treatment, except for two cases.5,7 However, none of the cases of rupture were detected using US, except for the present case. Dallara et al. reported that focused assessment with sonography for trauma (FAST) can diagnose intraperitoneal hemorrhage, but lesions that bleed into the retroperitoneum may not be detected by FAST. 11 Wang et al. reported that because of the patient’s body habitus and bowel gas, there were no positive signs on sonography. 12
Based on previous studies, in the diagnosis of PDAA, US is considered useful in detecting cases of nonruptured aneurysms but diagnosing ruptured aneurysms by US may be difficult. However, in the present case, the retroperitoneal hematoma and bleeding extending into the abdominal cavity obliterated the gaseous image of the gastrointestinal tract around the lesion, allowing the aneurysm to be clearly identified.
Conclusion
Contrast-enhanced CT is considered the gold standard in the diagnosis of PDAA rupture. However, US requires no special preparation, is easy to operate compared with other imaging methods, and is relatively simple to perform. In addition to the evaluation of intraperitoneal hemorrhage, the newer high-resolution color Doppler imaging enables the assessment of aneurysms and adjacent fine vascular geometry without contrast media. Therefore, US should be considered a useful diagnostic tool for the initial diagnosis of PDAA, which requires early diagnosis and treatment.
Footnotes
Correction (February 2023):
The in-text citations for Tables 1 and 2 have been moved, and Figures 1, 2, and 3 have been replaced with the correct figures.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
