Abstract
Objective:
The aim of this study was to assess the ultrasonographic appearance of the lower extremity arteries, after a femoropopliteal bypass surgery.
Materials and Methods:
Ninety patients, with atherosclerotic peripheral artery disease, underwent either autologous vein (30 subjects) or synthetic femoropopliteal bypass grafting, using an end-to-side (30 subjects) or end-to-end anastomotic technique. The outcomes were compared to 30 volunteers, who were recruited to serve as healthy controls. A lower extremity duplex ultrasonogram (DUS) was performed 1 year after treatment. The healthy volunteers underwent DUS at the point of being included in the study.
Results:
In healthy volunteers, the angle between the profunda femoris artery (deep femoral artery, PFA) and common femoral artery (CFA) did not exceed 30° of angulation. The diameter of the CFA, in the bifurcation area, was 9.8 ± 1.5 mm. The bypass patency rates, CFA diameter, PFA angle, and neointimal hyperplasia rates differed between the groups as presented.
Conclusion:
The results that were obtained may indicate the importance of bypass graft surveillance, using DUS, with precise analysis of the topography and geometry of the lower extremity arteries and vascular conduits, during the perioperative period. In addition, in this cohort, femoropopliteal bypass procedures were associated with an increase in the angle of the PFA from 30° to 80° of angulation. An increase in the diameter of the CFA in the area of the proximal anastomosis from 9.8 to 15.1 mm was also noted. In these patients, the saphenous vein bypasses were associated with minimal changes of the vascular geometry and better patency rates.
Keywords
Duplex ultrasonography (DUS) plays an important role in patients with peripheral artery disease in the preoperative and postoperative periods, due to the non-invasiveness of the technique, absence of significant radiation exposure, and the possibility of repeated studies.1,2 Assessment of the vascular anatomy and hemodynamics in patients who undergo lower extremity artery bypass surgery may help detect early re-stenosis and neointimal hyperplasia, which in turn may prevent graft thrombosis and limb loss. However, DUS is very operator dependent; therefore, it takes specific skills and expertise to make these evaluations. 3 Moreover, it is critically important to understand the technical aspects of each bypass procedure, atherosclerotic and post-procedural changes in vascular anatomy, which altogether may impair sonographic visualization.
Occlusion of the superficial femoral artery (SFA) is one of the most commonly encountered lesions in patients with atherosclerotic peripheral artery disease, occurring in more than 50% of patient cases.2,4 Femoropopliteal bypass grafting remains the most effective treatment option in subjects with chronic total occlusion of the SFA requiring revascularization. 5 There are two main techniques to perform anastomoses: “end-to-side” and “end-to-end.”2,6 The proximal anastomosis is usually performed over the orifice of the profunda femoris artery (deep femoral artery, PFA), the main vessel involved in collateral circulation in subjects with femoropopliteal lesions. During open bypass surgery, the proximal anastomosis is usually formed at an acute angle of approximately 60°, 6 which creates better conditions for blood flow due to a decrease in hemodynamic resistance and, accordingly, energy losses during the passage of blood from the native artery into the bypass. Therefore, it is believed that an end-to-end anastomosis may be a nearly perfect invention.2,5,6 Other important issues are positioning of the bypass graft and formation of the distal anastomosis. 6
This research was designed to address the following questions:
How would the type of proximal anastomosis change the diameter of the native artery, that is, CFA?
How would the types of the anastomosis and bypass graft affect the topography of the orifice of the PFA?
How would the type of open vascular access to the CFA bifurcation affect the performance of the proximal anastomosis?
Femoropopliteal bypass grafting does in fact change the topography of the orifice of PFA and can lead to an increase in the diameter of the common femoral artery (CFA), in the area of the proximal anastomosis. Moreover, the classical approach to the CFA bifurcation can be associated with possible injury to the inguinal lymph nodes. Saphenous vein bypass grafting is associated with better patency rates, which, nevertheless, does not reach 100% patency. 7
There have been several studies dedicated to the assessment of hemodynamically significant stenoses, in bypasses. The published evidence notes a decrease in the lumen diameter by more than 70%, specific changes in the spectral characteristics of blood flow, including an increase in the peak systolic velocity (PSV) at the site of stenosis by more than 2.5 times.3,8–10 At the same time, it is equally important to evaluate the anastomotic areas, large arterial branches, and collaterals, as well as surrounding tissues. Formation of neointimal hyperplasia, aneurysms in the anastomotic areas, or kinks in the bypass may affect short-term and long-term patency of the grafts. Taken altogether, evaluation of the ultrasonographic appearance of the lower extremity arteries after femoropopliteal bypass surgery is a challenging issue. Therefore, the aim of this study was to assess the ultrasonographic appearance of the lower extremity arteries, after femoropopliteal bypass surgery.
Materials and Methods
DUS was performed on 90 consented patients. In this cohort, there were 80 male patients who were an average age of 59 ± 5 years old. There were 10 female patients who were an average age of 56 ± 3 years. All patients had a stage IIb-III Fontaine chronic lower limb ischemia, due to atherosclerotic peripheral artery disease. Thirty patients underwent in situ above-knee femoropopliteal saphenous vein bypass. There were 30 patients, who had an above-knee femoropopliteal bypass grafting with an 8-mm synthetic graft with an end-to-side proximal anastomosis. There were 30 patients, who had an above-knee femoropopliteal bypass grafting with an 8-mm synthetic graft with an end-to-end proximal anastomosis. In addition, there were 30 healthy male volunteers, who were on average 60 ± 2 years old and were recruited to serve as control participants. All of the patients were treated by the same team of vascular surgeons. DUS was performed with an Esaote MyLab Alpha ultrasound equipment system (Esaote Europe B.V., Maastricht). The following types of transducers were used: a linear 3–13 MHz and a sector 3–5 MHz transducer. The researchers were certified sonographers and vascular surgeons with expertise in DUS. Lower extremity DUS was performed 1 year after treatment and when clinically necessary. The healthy volunteers underwent DUS at the time of being included in the study.
The following parameters were evaluated:
The patency of the bypass grafts, as determined by PSV,
Anatomy and geometry of the bypass grafts, including deformities and kinking,
Areas of proximal and distal anastomoses,
Anatomy of the adjacent soft tissues,
Anatomy and patency of the outflow arteries.
The data from this cohort study were statistically analyzed using both MS Excel 2016 and SPSS 26.0 v software.
Results
The results from this cohort study are provided in Table 1. In the group of healthy volunteers, the PFA arose from the posterior aspect of the CFA in 50% of cases, from the posterolateral aspect of the CFA in 47% of cases, and in 3% of cases (one patient), the PFA arose from the anterolateral aspect of the CFA in 50% of cases, from the posterolateral aspect of the CFA in 47% of cases, and in 3% of cases (one patient). The PFA arose from the anterolateral aspect of the CFA. The angle between the PFA and CFA did not exceed 30° of angulation in all patient cases, with 20° of angulation in 93.3% of cases and 30° of angulation in 6.7% of patient cases (see Figure 1). The diameter of the CFA in the bifurcation area was 9.8 ± 1.5 mm.
Results of Duplex Ultrasonography of the Lower Extremity Arteries in Patients Who Underwent Femoropopliteal Bypass Grafting and Healthy Volunteers.
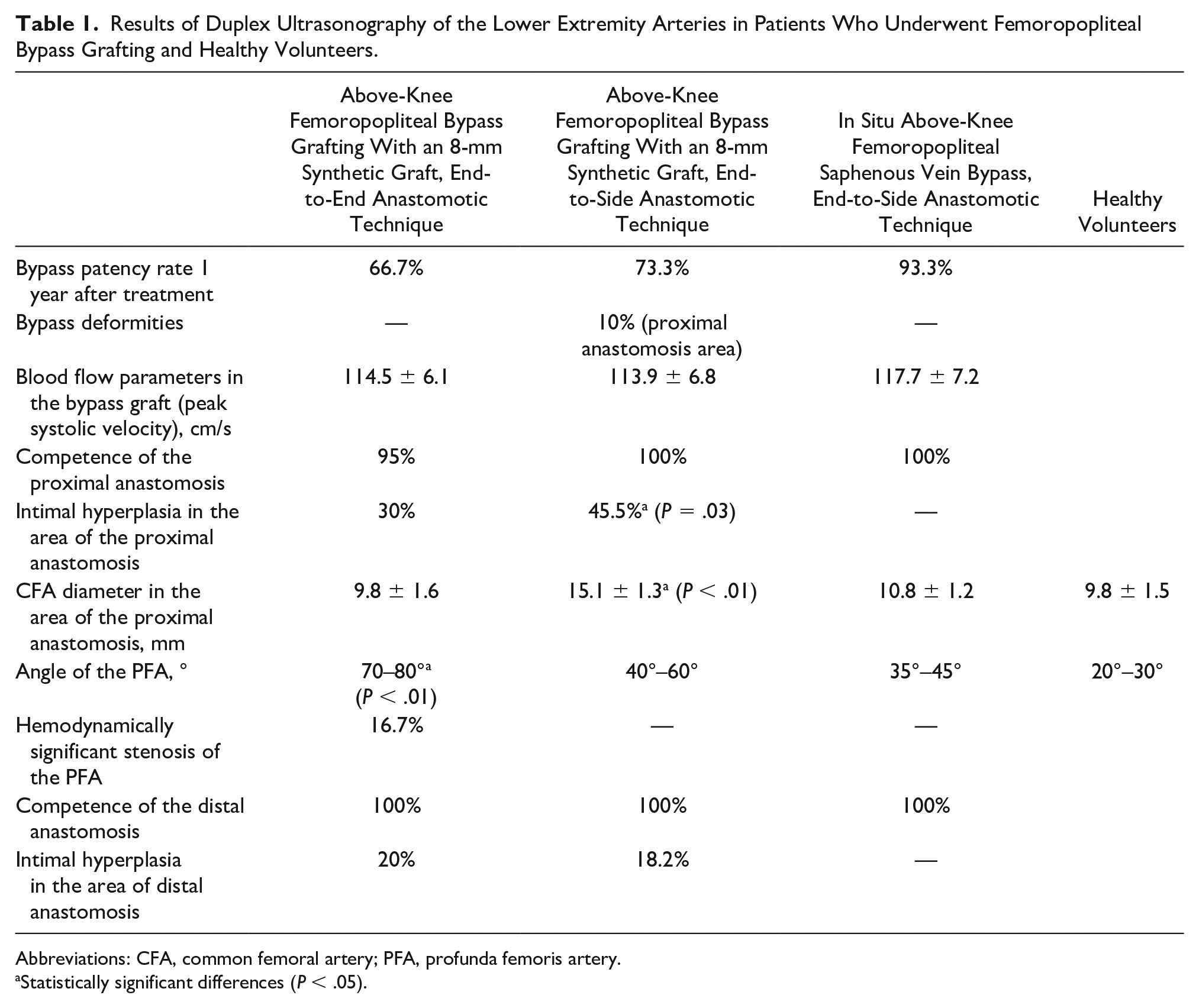
Abbreviations: CFA, common femoral artery; PFA, profunda femoris artery.
Statistically significant differences (P < .05).
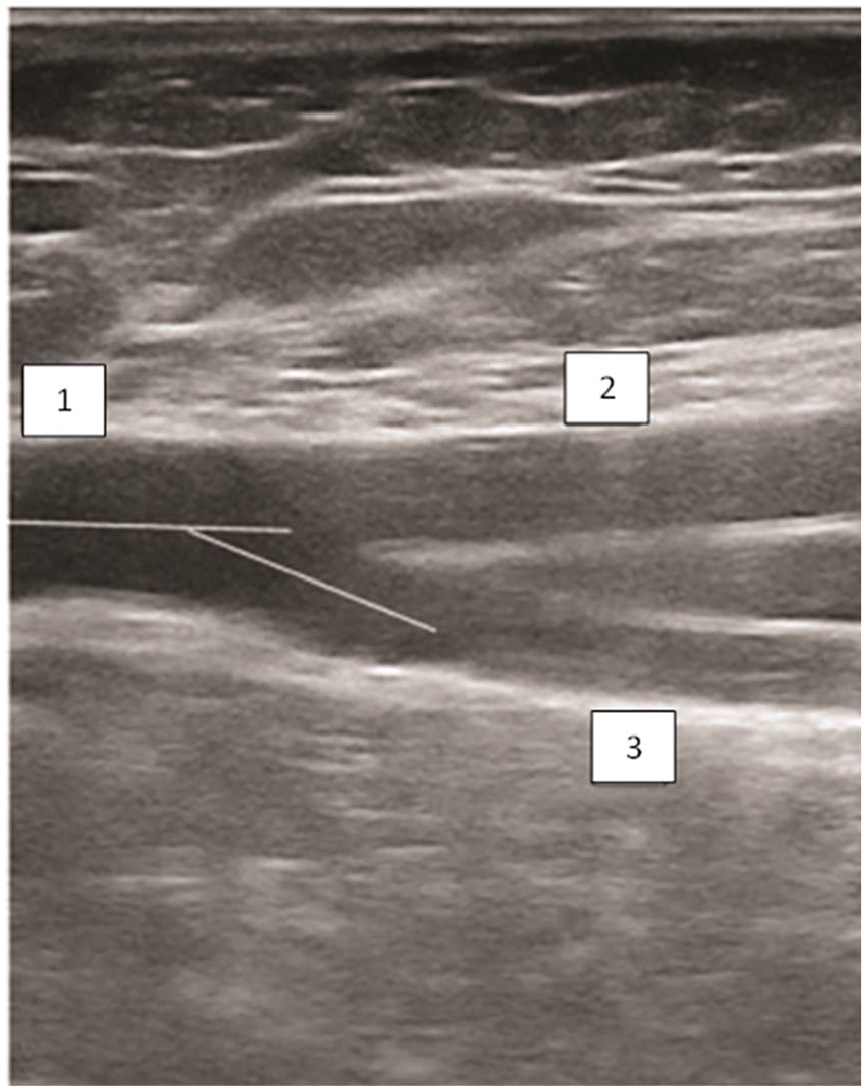
A sonogram that demonstrates a longitudinal section of the area of the common femoral artery (CFA) bifurcation in a healthy volunteer, in grayscale: (1) CFA; (2) femoral artery (FA); and (3) profunda femoris artery (PFA). The angle of the PFA is 20°.
In patients who underwent surgical treatment, PFA arose from the posterior aspect of the CFA in 60% of patient cases, and from the posterolateral aspect of the CFA in 40% of patient cases. The angle between the PFA and the CFA varied from 35° to 80° of angulation. The diameter of the CFA in the bifurcation area varied from 9.8 to 15.1 mm.
In patients who underwent in situ above-knee femoropopliteal saphenous vein bypass, the diameter of the CFA in the area of proximal anastomosis was 10.8 ± 1.2 mm. The angle of the PFA varied from 35° to 45° of angulation and in 20 patients it was 35°, in 7 subjects it was 40°, and in 3 patients it was 45° of angulation (see Figure 2). Autologous venous bypass graft was visualized as a tubular structure with isoechoic walls. The patency of the vein bypass grafts at 1 year was 93.3% (28 patients); PSV in the graft was 117.7 ± 7.2 cm/s. Although PSV in this cohort of patients was higher as compared to other groups, the differences were not statistically significant (P = .28). One patient was presented with an intact valve in the great saphenous vein in the upper third of the thigh, which was an equivalent of 50% stenosis (see Figure 3). DUS provided satisfactory imaging of both proximal and distal anastomoses with no signs of neointimal hyperplasia.
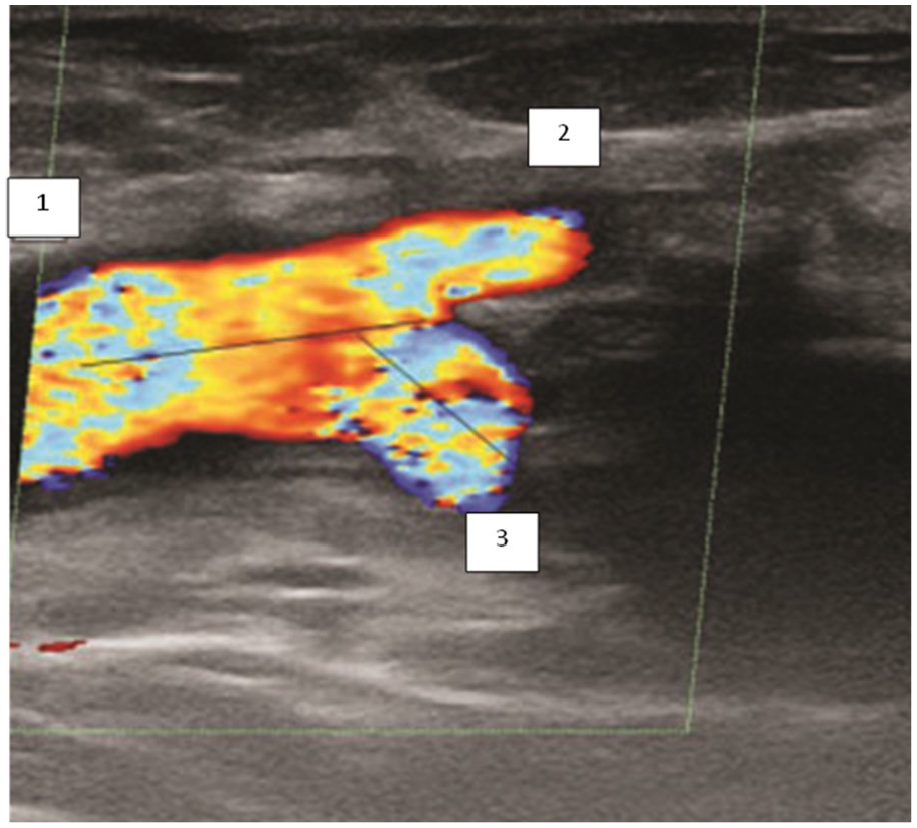
A sonogram that provides a longitudinal section of the common femoris artery (CFA) bifurcation area in a patient who underwent femoropopliteal saphenous vein bypass grafting: (1) CFA; (2) saphenous vein bypass graft; and (3) profunda femoris artery (PFA). The angle of the PFA is 45°.
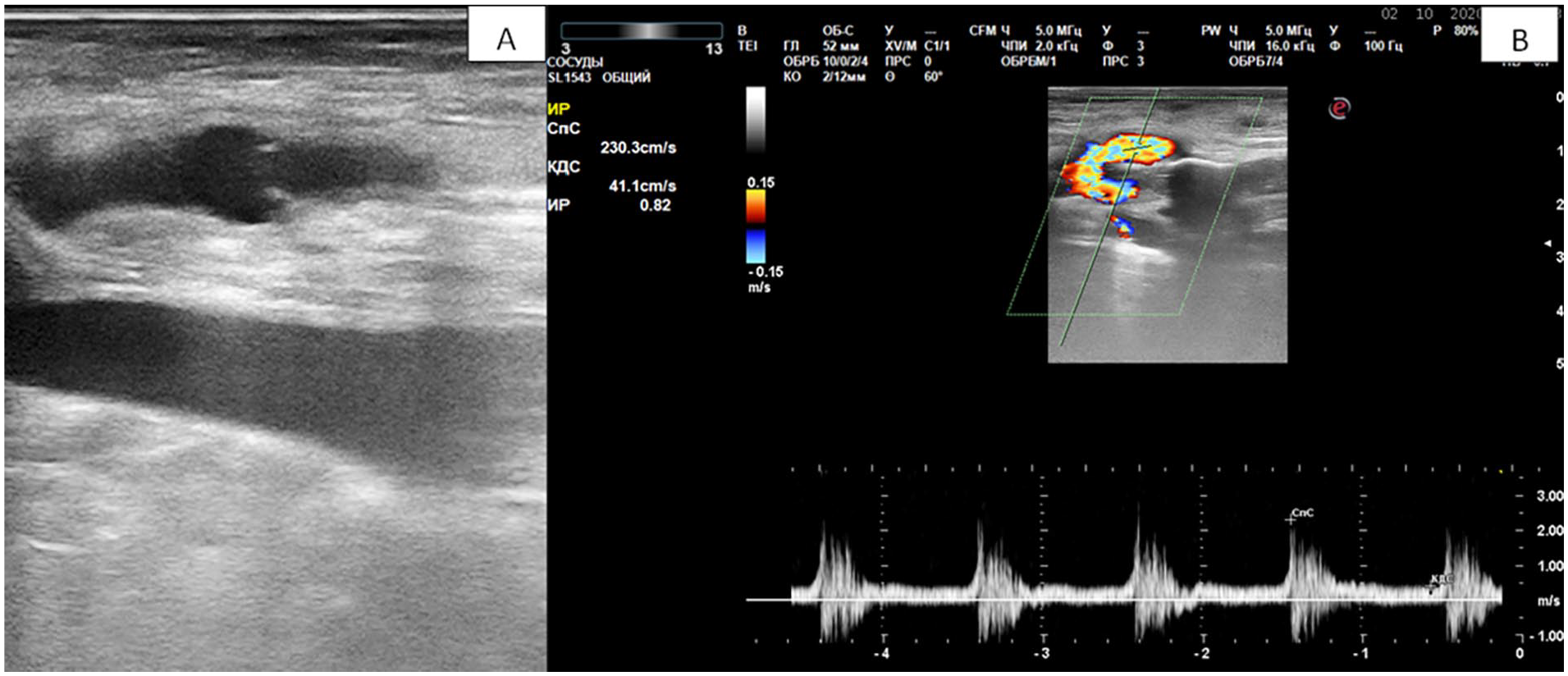
A sonogram that provides a longitudinal section of the saphenous vein bypass graft. An intact valve in the great saphenous vein in the upper third of the thigh: (A) Demonstrated with grayscale sonography and (B) Doppler.
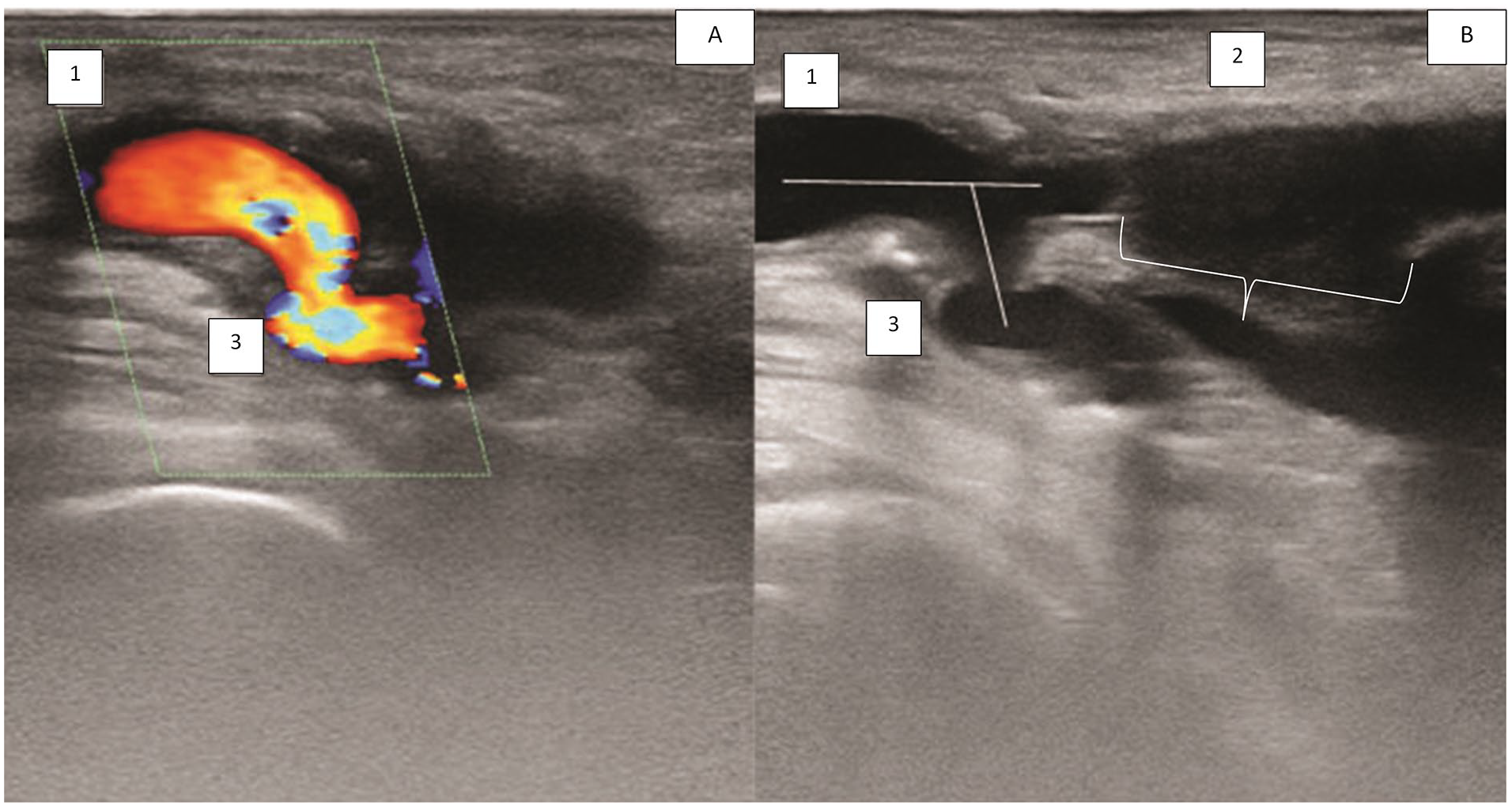
In patients who underwent above-knee femoropopliteal bypass grafting with an 8-mm synthetic graft with an end-to-side anastomotic technique, the diameter of the CFA in the area of the proximal anastomosis was 15.1 ± 1.3 mm (see Figures 4 –6). The angle of the PFA was 40° to 60° of angulation and 40° in 10 patients, 50° in 11 patients, and 60° of angulation in 9 participants (see Figures 4 and 6). Synthetic vascular grafts were visualized as tubular structures with hyperechoic walls, which made it possible to clearly distinguish them from the surrounding tissues. A distinctive feature of an end-to-side anastomosis was a longer connection with the CFA as compared to vein bypass grafts. In these types of anastomoses, it is possible to clearly distinguish the “heel” of anastomosis, which is a junction between the posterior semicircle of the bypass graft with the posterior wall of the native artery; the “toe” of anastomosis, which is a junction between the anterior semicircle of the bypass graft with the anterior wall of the native artery; as well as the “hood” of anastomosis, which is the anterior semicircle of the bypass graft (see Figures 4–6). It was due to the presence of the “hood” of anastomosis that the diameter of the CFA increased. DUS helped visualize turbulent blood flows in the majority of cases, both at the junction of the prosthetic bypass graft with the native femoral artery, and in the “heel” of anastomosis (see Figures 4 and 5), as compared to vein bypass grafts. Neointimal hyperplasia in the area of proximal anastomosis was detected in 45.5% of patient cases, which was statistically significantly higher as compared to patients treated with an end-to-end anastomotic technique (P = .03). Neointimal hyperplasia in the area of the distal anastomosis was detected in 18% of cases (see Figure 5). The patency of synthetic bypass grafts 1 year after treatment was 73.3% (22 patients); PSV in prosthetic bypass grafts was 113.9 ± 8.9 cm/s. Three out of eight patients with failed grafts were presented with graft compression in the proximal area (see Figures 7 and 8). Three patients required reconstruction of the proximal anastomosis due to the graft compression: by the enlarged lymph nodes in two patient cases, and a wound hematoma in one participant.
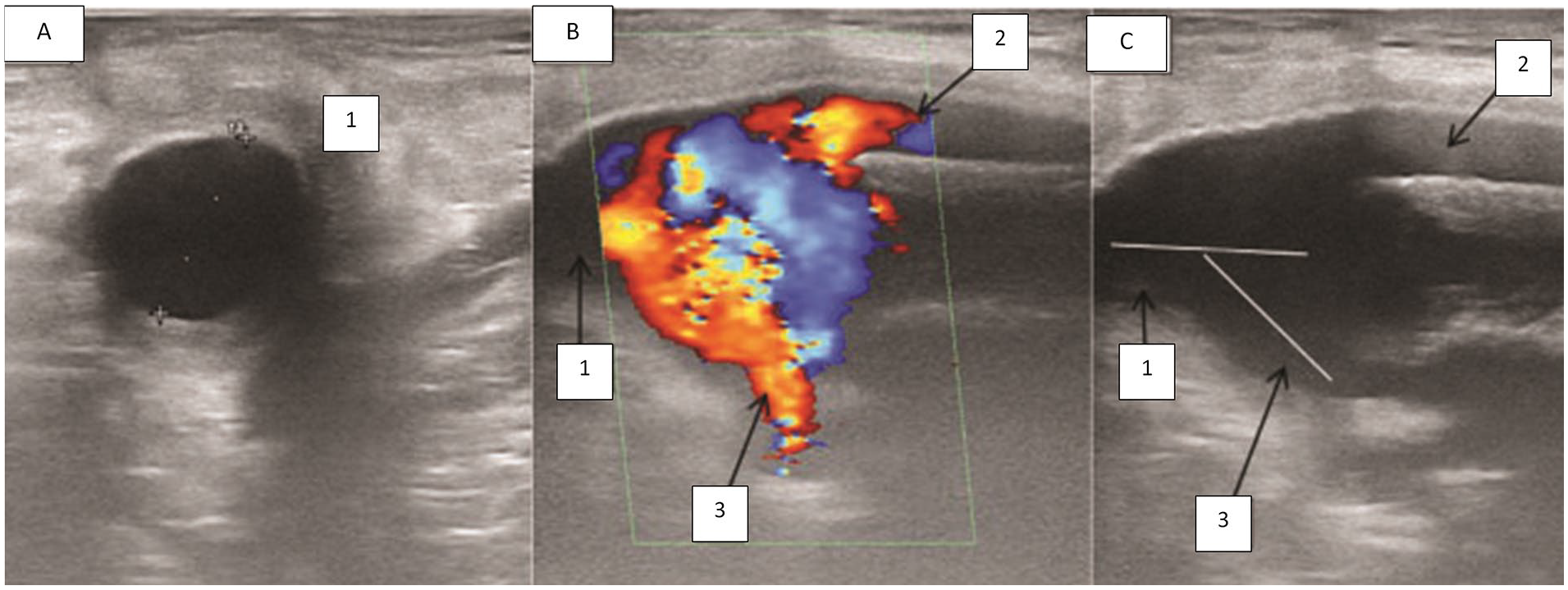
A sonogram that provides a section of the common femoral artery (CFA) bifurcation area in a patient who underwent femoropopliteal prosthetic bypass grafting using an end-to-side technique: (1) CFA; (2) prosthetic bypass graft; and (3) profunda femoris artery (PFA). The angle of the PFA is 40°. (A) Demonstrated with grayscale sonography, in longitudinal section; (B) demonstrated with color Doppler, in longitudinal section; and (C) demonstrated in grayscale sonography, in transverse section, at the inner diameter of the vessel lumen, which is 14.6 mm.
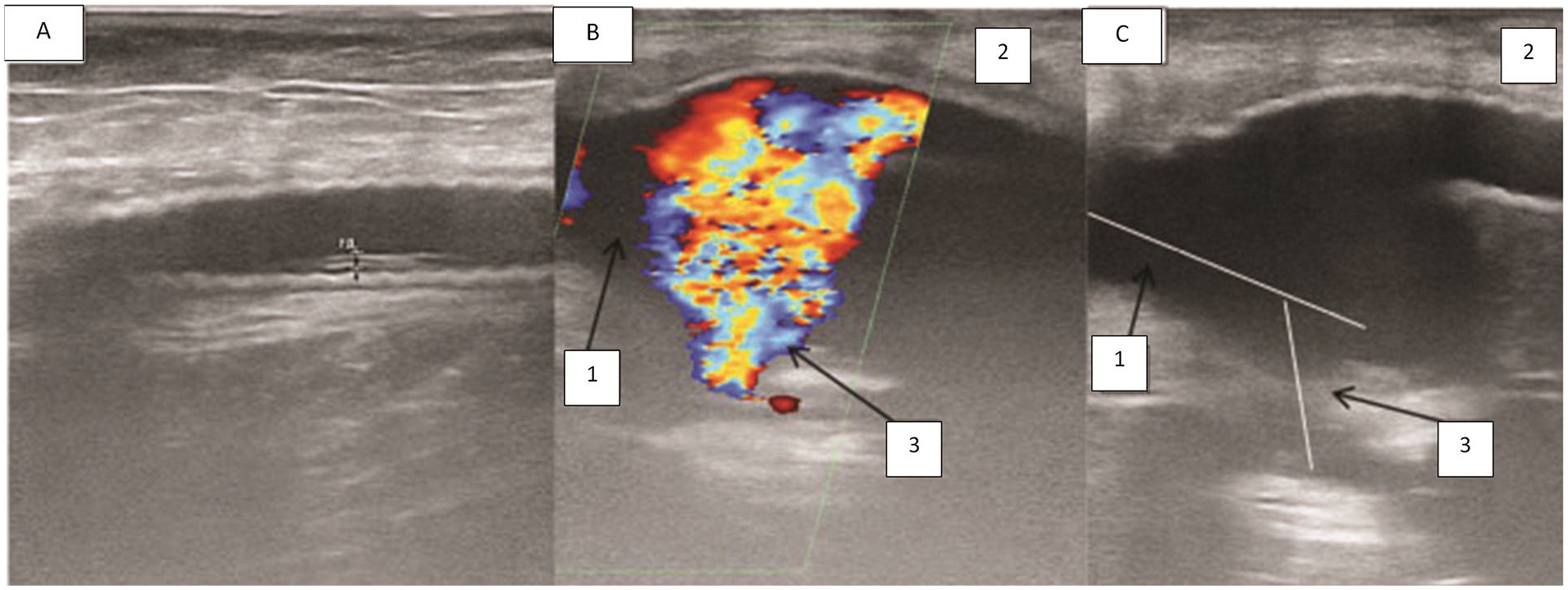
A sonogram that provides a longitudinal section of the common femoral artery (CFA) bifurcation area and prosthetic graft in a patient who underwent femoropopliteal bypass grafting using an end-to-side technique: (1) CFA; (2) prosthetic bypass graft; and (3) profunda femoris artery (PFA). The angle of the PFA is 60°. (A) Demonstrated with grayscale sonography, in longitudinal section; (B) demonstrated with color Doppler, in longitudinal section; and (C) demonstrated with grayscale sonography, in longitudinal section, neointimal hyperplasia in the lumen of the prosthetic bypass graft at the level of the upper third of the thigh.
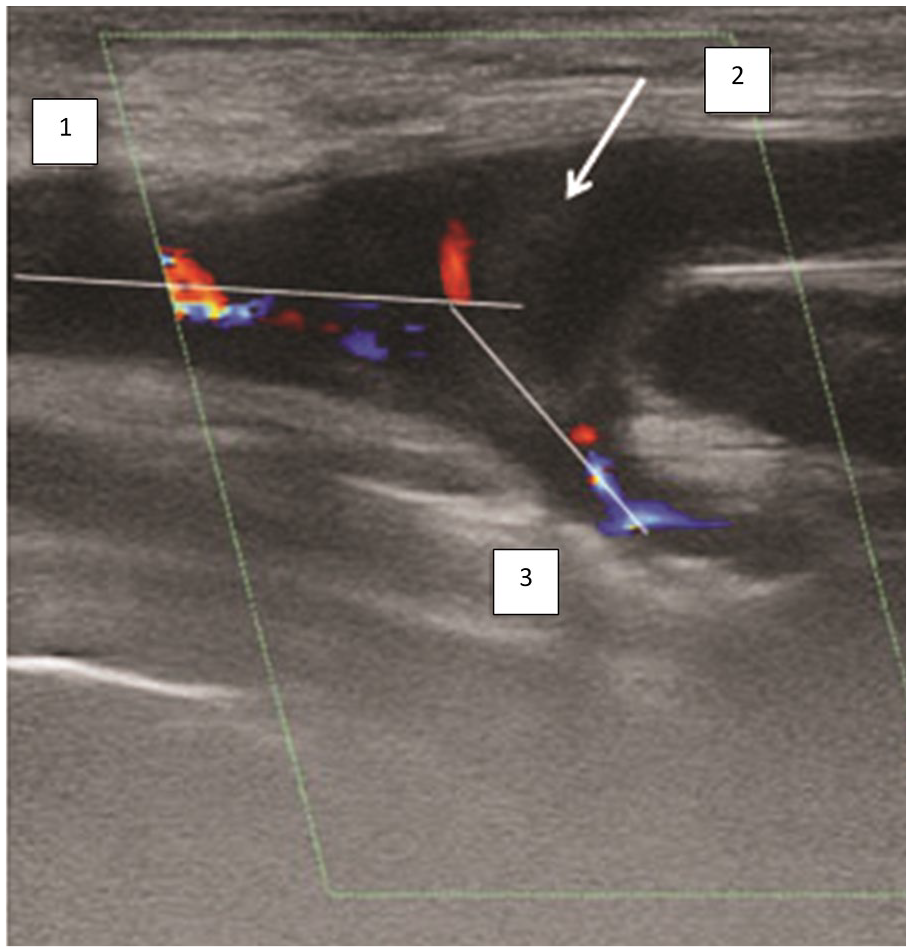
A sonogram that provides a longitudinal section of the common femoral artery (CFA) bifurcation area in a patient who underwent femoropopliteal bypass grafting with a synthetic graft (end-to-side anastomotic technique). Graft thrombosis (shown by an arrow): (1) CFA; (2) synthetic bypass graft; and (3) profunda femoris artery (PFA). The angulation of the distal femoral artery is 50°.
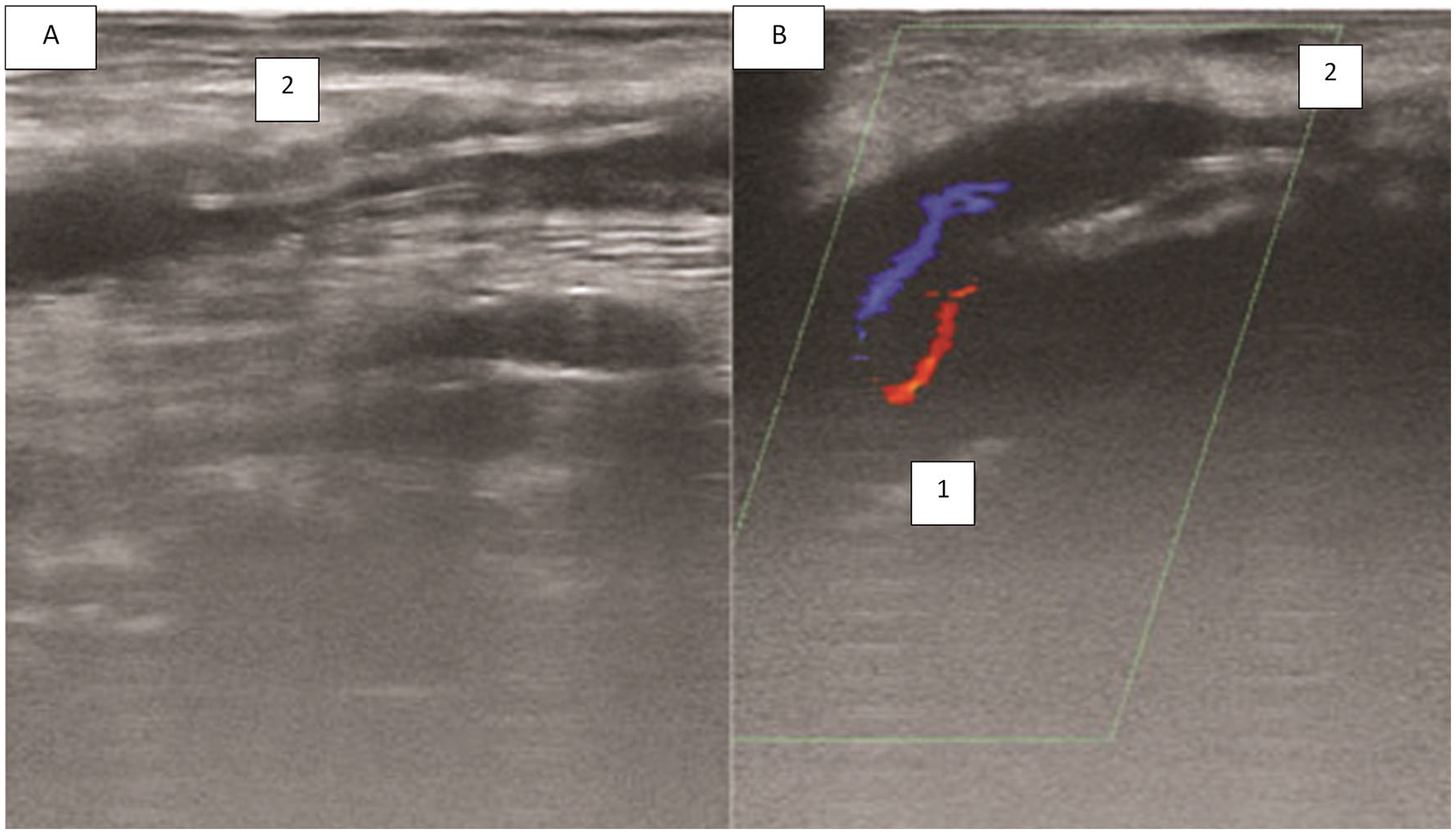
A sonogram that provides a longitudinal section of a compressed synthetic bypass graft: (1) common femoral artery (CFA) and (2) synthetic bypass graft. (A) Demonstrated with color Doppler, in a longitudinal section; and (B) demonstrated with grayscale sonography, in longitudinal section, compressed synthetic bypass graft, in the upper third of the thigh.
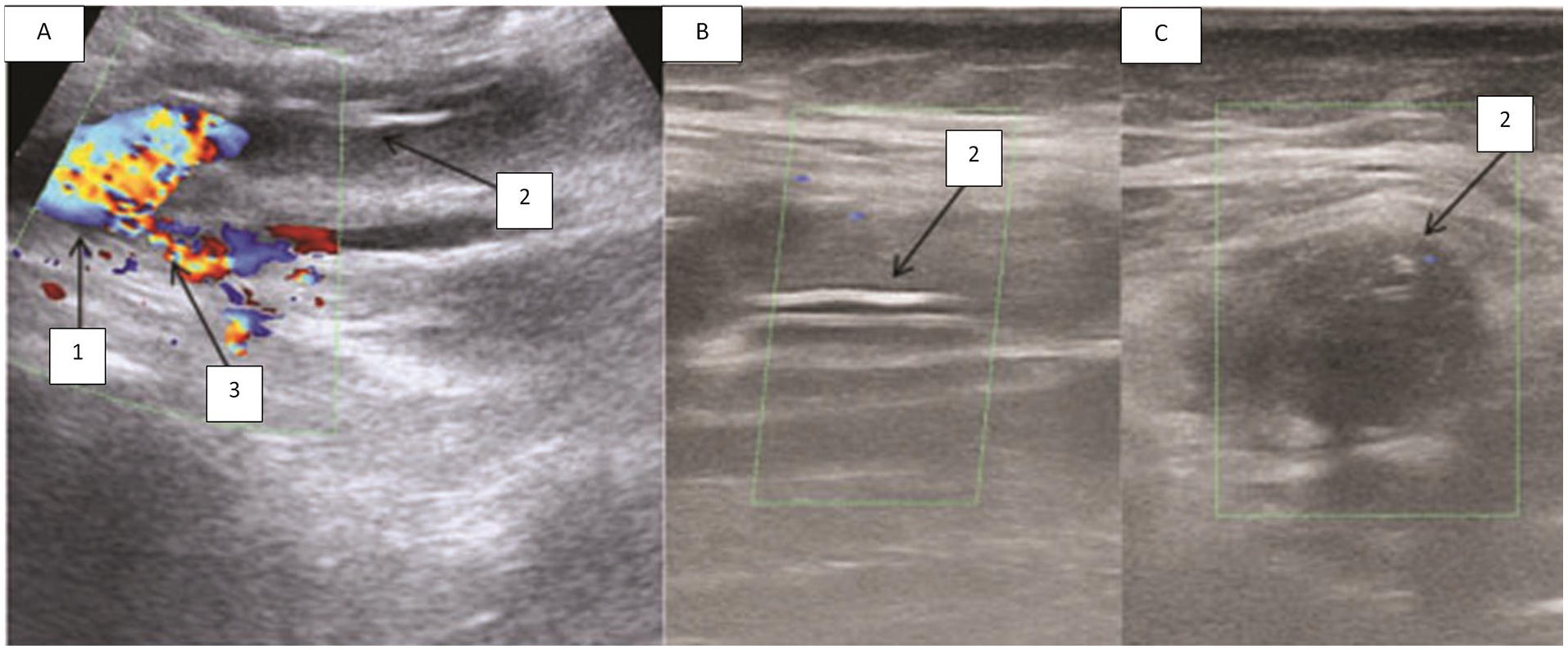
A sonogram that provides a view of the compressed synthetic bypass graft: (1) common femoral artery (CFA) and (2) synthetic bypass graft. (A) Demonstrated with grayscale sonography, in transverse section, compressed synthetic bypass graft (shown with an arrow); (B) demonstrated with grayscale sonography, in longitudinal section, compressed synthetic bypass graft (shown with an arrow); and (C) demonstrated with color Doppler, in longitudinal section, proximal anastomosis. (1) CFA; (2) synthetic bypass graft (compression shown with an arrow); and (3) profunda femoris artery (PFA).
In patients who underwent above-knee femoropopliteal bypass grafting with an 8-mm synthetic graft with an end-to-end anastomotic technique, the diameter of the CFA in the area of the proximal anastomosis was 9.8 ± 1.6 mm. The angle of the PFA was 70° to 80° of angulation and 70° in 22 patients, 75° in 4 patients, and 80° of angulation in 4 patients (see Figures 9 and 10). The increase in the PFA angle was statistically significant (P < .01). Patency rate at 1 year was 66.7% (20 patients); PSV was 111.8 ± 8.8 cm/s. The area of proximal anastomosis was easily visualized: hyperechoic walls of the bypass graft attached to the native artery resembling the shape of an ellipse. Turbulent blood flows were identified at the orifice of the PFA (see Figures 9 and 10). In 16.7% of patients, who underwent prosthetic bypass grafting using an end-to-end anastomotic technique, presented with a hemodynamically significant stenosis of the PFA. Neointimal hyperplasia in the area of the proximal anastomosis was detected in 30% of cases and in the area of the distal anastomosis in 20% of cases. One patient with a graft thrombosis was found to have a defect in the area of proximal anastomosis (see Figure 10).
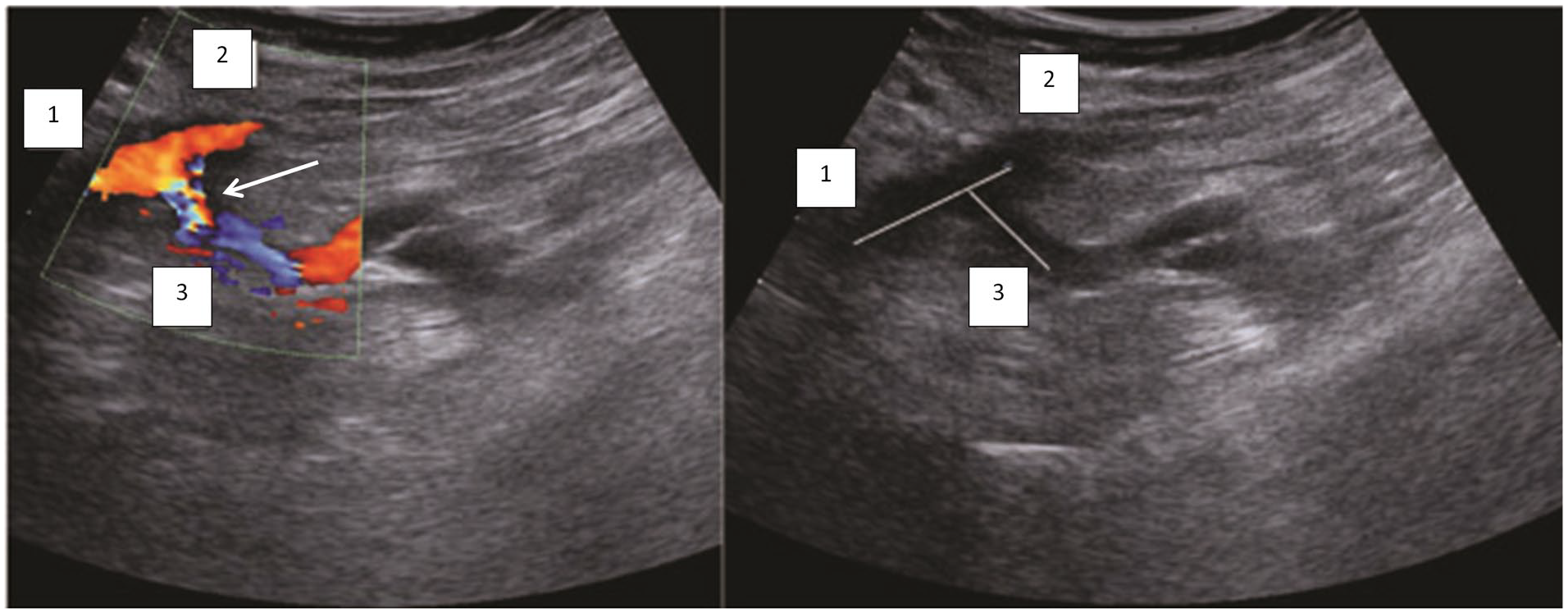
A sonogram that provides a longitudinal section of the common femoral artery (CFA) bifurcation area in a patient after femoropopliteal bypass graft using an end-to-end anastomotic technique: (1) CFA; (2) synthetic bypass graft; and (3) profunda femoris artery (PFA). The angulation of the PFA is 70°.
A sonogram that provides a longitudinal section of the common femoral artery (CFA) bifurcation area in a patient with thrombosis of the femoropopliteal bypass graft: (1) CFA; (2) synthetic bypass graft incompetent in the area of proximal anastomosis (a leaking defect), shown by a bracket); and (3) profunda femoris artery (PFA). The angulation of the PFA is 75°.
Discussion
A relatively constant relationship exists between anatomical structures, which has been described in the medical literature, but this can be quite variable, even in healthy subjects. Different pathologies as well as surgical interventions often result in further anatomical changes and defects. 11 When it comes to vascular bypass grafting, it is crucial to consider changes in the local hemodynamics.
There is an abundance of literature dedicated to the assessment of the geometry of vascular anastomoses and its relationship to vascular hemodynamics. Decreasing the angle of anastomosis reduces the blood flow disturbances in an antegrade direction; however, it is impossible to reduce the angle between the bypass graft and the native artery to less than 30°, without increasing the length of anastomosis. 6 Mathematical models show that there is always an area of separation of blood flows in anastomotic area; in addition, the blood flow may reverse or move in a circular direction toward the recipient artery or bypass. There are especially “weak” areas such as the “toe” and the “heel” of anastomoses, where the separation of blood flows is most noticeable, while the shear forces are small, and the speed of flow is unstable. These hemodynamic factors are associated with platelet adhesion, endothelial injury, and intimal hyperplasia, which may have significant impact on the patency of bypass grafts. Therefore, it would be sensible to decrease the angle of anastomosis to 15°. 2 However, as shown in this study, attempts to perform an end-to-side anastomosis with the minimum possible angle lead to an increase in the diameter of the CFA in the area of proximal anastomosis and, accordingly, increases the tension within the vascular wall, which may further lead to the development of anastomotic aneurysm. Physically, this has been described by Laplace’s law. 2 Mikati et al. 12 reported that the approximate timing of aneurysmal transformation of the proximal anastomosis was between 5 and 10 years after bypass surgery. Interestingly, the rates of formation of intimal hyperplasia in the area of proximal anastomoses were higher in patients after femoropopliteal bypass grafting using an end-to-end technique. At the same time, at a 1-year follow-up period, such findings had no impact on local hemodynamics; however, further observations are required.
Another issue to be considered is that during femoropopliteal bypass procedures, the proximal anastomosis is usually directly adjacent to the orifice of the PFA, which complicates local hemodynamics in the bifurcation area.13,14 When PFA is represented by two arterial trunks, hemodynamics become even more complicated.15,16 Moreover, it is possible that the technical aspects of dissection of CFA bifurcation, especially PFA, during the open procedures may also adversely affect hemodynamics in the area of proximal anastomosis.
Voloshin et al. 17 analyzed the results of femoral profundoplasty in terms of hemodynamic effects and reported that the angle of the PFA and overall diminished blood flow velocities characteristic of atherosclerosis played the key role in the development of PFA stenosis, as well as an optimal PFA angle of less than 35°. In this study, the angle of the PFA in healthy volunteers was less or equal to 30° of angulation. In patients who underwent in situ above-knee femoropopliteal saphenous vein bypass, the angle of the PFA was within 35° to 45° of angulation. In subjects who underwent prosthetic above-knee femoropopliteal bypass grafting with an end-to-end anastomotic technique, the angle of the PFA was from 70° to 80° of angulation. This may be explained by the technical aspects of performing the proximal anastomosis as well as the possible excessive tension and/or stretching of the prosthetic grafts, when performing the distal anastomosis. 6 An indirect confirmation of this fact was the discovery of a leak in the proximal anastomosis in a patient with a prosthetic graft thrombosis. Interestingly, a hemodynamically significant stenosis of the PFA at a 1-year follow-up period was observed only in patients who underwent prosthetic femoropopliteal bypass grafting using an end-to-end anastomotic technique, that is, a technique, which implied the greatest changes in the angle of the PFA.
Open access to the CFA bifurcation may result in lymphatic vessel or lymph node injury, which may later have a detrimental effect on the graft patency as seen in two cases; therefore, it is advisable to perform the skin incision more laterally. 11 Optimal hemostasis is also critically important as hematomas may result in bypass deformity and compromise blood flow.
Limitations
The major limitation of this study was the research design and the convenient sample of patients recruited. In addition, there were a rather small number of observations, which were performed retrospectively, and a rather short annual postoperative follow-up period. This study needs to be continued and as such added information could be presented at both the 3- and 5-year follow-up period.
Conclusion
These cohort results demonstrated the possible importance of bypass graft surveillance using DUS with precise analysis of the topography and geometry of the lower extremity arteries and vascular conduits in the perioperative period. In this cohort, the femoropopliteal bypass procedures were associated with an increase in the angle of origin of the PFA from 30° to 80° of angulation. It was also noteworthy that this cohort had increase in the diameter of the CFA in the area of the proximal anastomosis from 9.8 to 15.1 mm. In this group, the saphenous vein bypasses were associated with minimal changes of the vascular geometry and better patency rates.
Footnotes
Correction (October 2023):
The Ethics Approval section has been updated with the IRB number.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grant of the President of the Russian Federation (ID number MK-1214.2022.3).
Ethics Approval
The study has been approved by the Local ethical committee of the Ryazan State Medical University (N7).
Informed Consent
Informed consent was obtained from the patients for publication.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study.
Trial Registration
The trial required no specific registration.
