Abstract
Objective:
The aim of this study was to assess the topography of the femoral and popliteal vessels focusing mainly on the venous variants, in patients with chronic venous disease (CVD).
Materials and Methods:
Duplex ultrasonography (DUS) was performed on 200 patients with varicose veins (VVs) and 253 patients with postthrombotic syndrome (PTS). In addition, an anatomical dissection was performed on 30 amputated lower extremities, without signs of CVD.
Results:
Duplication of the femoral vein (FV) was found in 14% of patients with VVs. Multiple FV trunks were identified in 42% of patients with PTS. One patient had a developmental anomaly with a hypoplastic FV and dilated deep FV. Two trunks of the popliteal vein (PV) below the knee level were detected in 83.3% of cases during anatomical dissection, in 87.5% of cases in patients with VVs, and in 90.1% of cases in PTS subjects during DUS. In 98.2% of cases, popliteal artery (PA) had a typical bifurcation. One patient was presented with a duplication of PA above the level of the knee. In 1.8% of cases, the PA was divided into an anterior tibial and peroneal artery.
Conclusion:
This study found variant anatomy in the FV and PV, which is frequent, with major changes occurring in patients with PTS. Variant anatomy of the PA was also found and is considered uncommon, occurring in less than 2% of cases.
The femoral artery (FA) and femoral vein (FV) are typically represented by single trunks of arteries and veins. 1 Topographically, three parts are distinguished in the superficial femoral vessels: (1) within the apex of the femoral triangle, where the vein lies medial and deeper than the artery; (2) the area in front of the sartorius muscle, where the vein is also located deeper than the artery; and (3) the area located within the adductor canal, where the vein lies lateral to the artery. The popliteal vessels, as well as the femoral ones, are normally represented by single trunks of the popliteal artery (PA) and popliteal vein (PV). Topographically, the popliteal vessels are subdivided into three segments: P1—from the point of exit from the adductor canal to the upper edge of the patella, P2—to the level of the knee, and P3—to the level of the bifurcation of the PA/confluence of the anterior and posterior tibial veins into the PV. 2 In clinical practice, they are divided into two sections: above the level of the knee, where the PA goes superficially and on the lateral side of the PA, and below the level of the knee, where the PV comes along the medial side of the artery.
At the same time, there are data in the literature on the duplication of the femoral and popliteal vessels.3–5 Whereas artery duplication is an extremely rare developmental anomaly,6,7 FV duplication can occur with a frequency of up to 55%, 7 and PV duplication with a frequency ranging from 2% to 42%.4,7 Conversely, variant anatomy of the femoral or popliteal vessels may add difficulties during surgical treatment. Arguably, this creates more options for the possible use of additional FV or PV trunks as conduits for bypass procedures or permanent vascular access.8–10
The objective of this research study was to evaluate the femoral and popliteal vessel topography, with special attention to the venous variants, in patients with chronic venous disease (CVD).
Materials and Methods
Duplex ultrasonography (DUS), of the femoral and popliteal vessels, was conducted in 200 patients, which provided 200 lower extremities with varicose veins (VVs), of the lower extremities (VVs). Likewise, there were 253 patients, which provided 302 lower extremities, with postthrombotic syndrome (PTS). The study did not include patients with a history of surgical interventions performed on the lower extremity arteries or veins as well as subjects with arteriovenous malformations.
DUS was performed with an Esaote MyLab Alpha ultrasound equipment. Two types of transducers were used, a linear 3- to 13-MHz transducer and a sector 3- to 5-MHz transducer. The researchers were board-certified sonographers, with expertise in vascular disorders. The DUS study was performed during the morning. DUS of the femoral vessels was performed in supine position, while the popliteal vessels were assessed in prone position.
Anatomical dissection of 30 amputated lower extremities, derived from patients without CVD, was additionally performed. Before dissection, the venous system was prefilled with blue latex, through a large saphenous vein, isolated at the level of the medial malleolus. The topography of the vessels of the femoral-popliteal segment, which differed from the classical description, was considered to be variant anatomy.
The topography and variant anatomy of superficial veins, nerves, tendons, and muscles were not considered in the study.
Results
Among patients with VVs, 120 subjects were female and were on average 50 ± 12 years old. There were 65 females who had a mesomorph body type, 35 had an endomorph body type, and 20 had an ectomorph body type. There were 80 male patients with VVs and were on average 48 ± 15 years old. Among the VV group, there were 48 patients that had a mesomorph body type, 25 had an endomorph body type, and 7 patients had an ectomorph body type.
Among patients with PTS, there were 123 females and were on average 47 ± 14 years old. There were 80 females who had a mesomorph body type, 38 had an endomorph body type, 5 patients had an ectomorph body type. In this cohort, there were 130 male patients and on average they were 49 ± 10 years old. In this cohort, there were 75 patients who had a mesomorph body type, 42 had an endomorph body type, and 13 had an ectomorph body type. In 90 patients with PTS, deep vein obstruction prevailed, and in 163 patients, there was deep vein reflux. The median time from the acute deep vein thrombosis was 7 ± 1.2 years ago.
The following anatomical variants were identified: duplication/triplication of FV with or without transposition relative to FA, high division of the PA into the anterior tibial artery and the posterior tibial artery, division of the PA into anterior tibial and peroneal arteries, and duplication of PV (Tables 1 and 2).
The Incidence of Variant Anatomy in the Femoral Vein, Based on the Current Study.
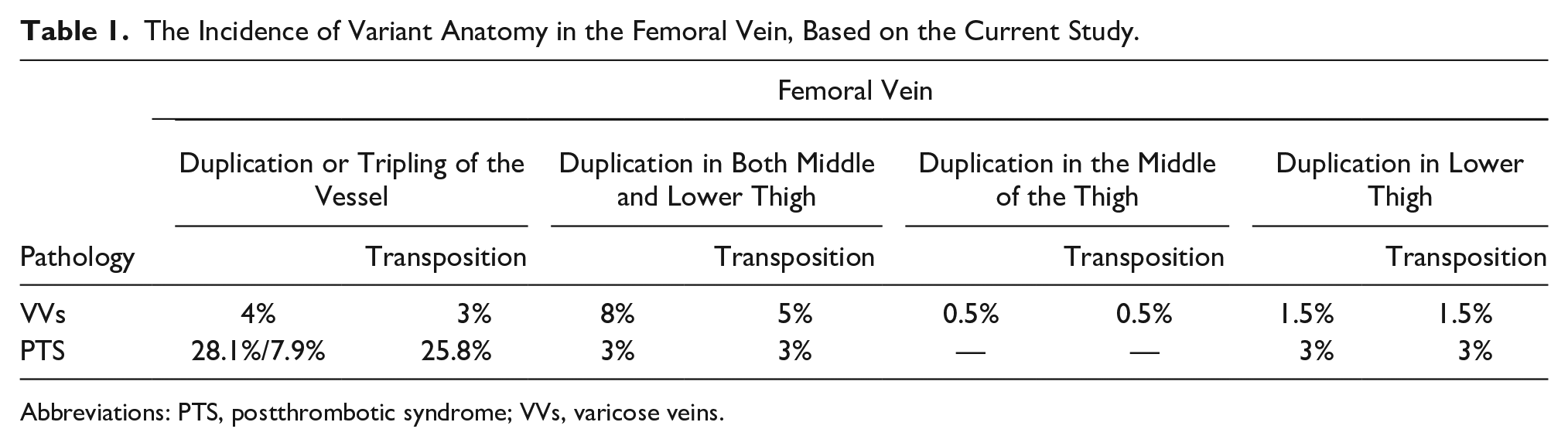
Abbreviations: PTS, postthrombotic syndrome; VVs, varicose veins.
The Variant Anatomy of the Popliteal Vein.
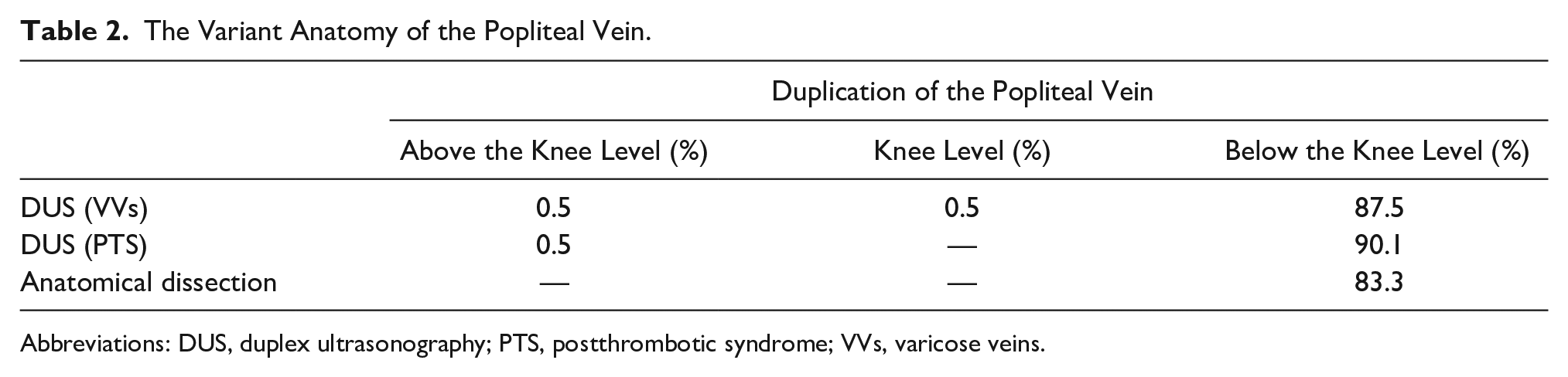
Abbreviations: DUS, duplex ultrasonography; PTS, postthrombotic syndrome; VVs, varicose veins.
In patients with VVs, FV in most cases was represented by one trunk and one or two companion (satellite) veins (Figure 1). Duplication of FV was detected only in 14% of cases (Figures 2 and 3). Of these, 10% had a FV transposition (Figures 4 and 5). In patients with PTS, the incidence of multiple trunks of FV was up to 42.1%. Moreover, in 7.9% of the cases, there was a triplication of the FV. One patient with VVs was diagnosed with a FV hypoplasia with a dilated deep femoral vein (DFV). There was no variant anatomy of FA according to these study findings.
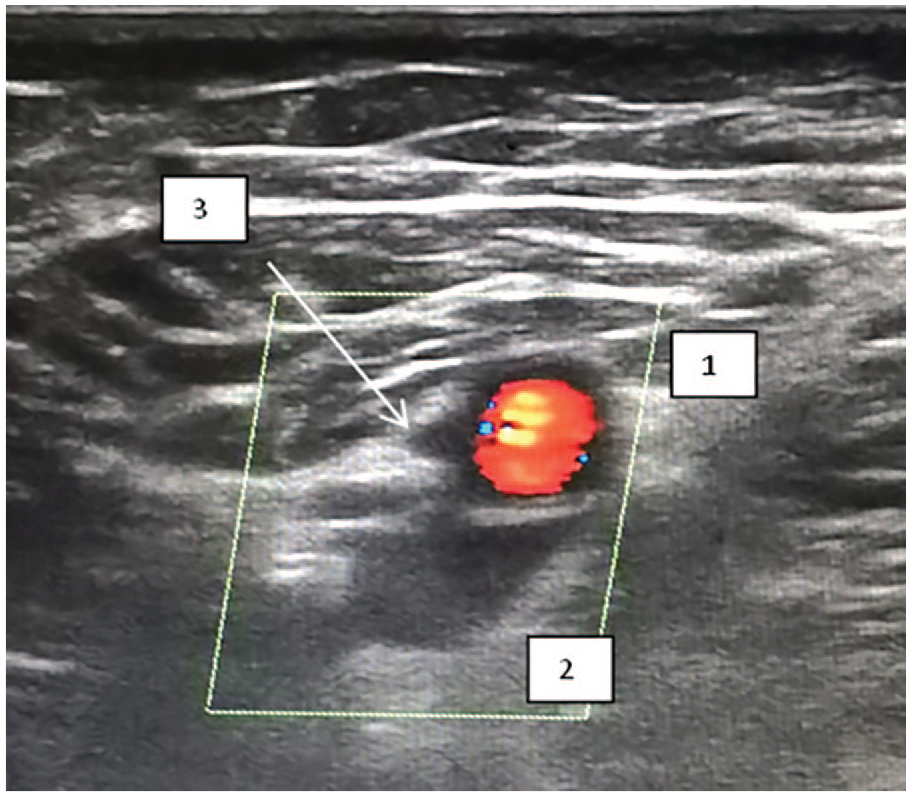
A sample sonogram that provides a transverse section of the FA and FV, in the lower third of the thigh. Note that 1 is the FA, 2 is the FV, and 3 is the satellite vein. FA, femoral artery; FV, femoral vein.
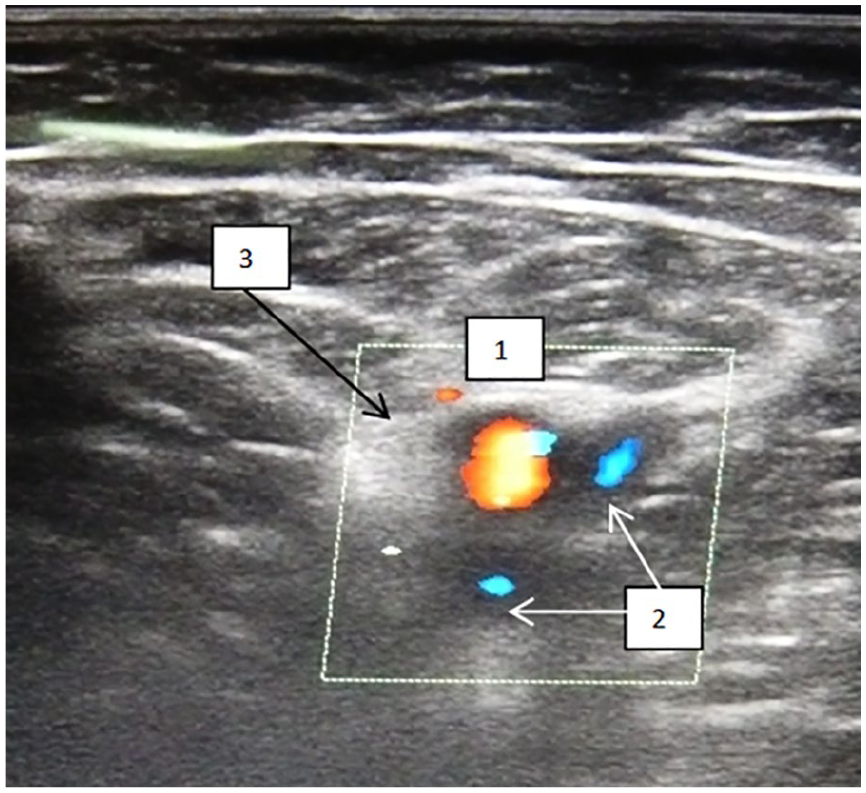
A sample sonogram that provides a transverse section of the FA and two trunks of the FV, in the middle third of the thigh. Note that 1 is the FA, 2 is the FV, and 3 is the satellite vein. FA, femoral artery; FV, femoral vein.
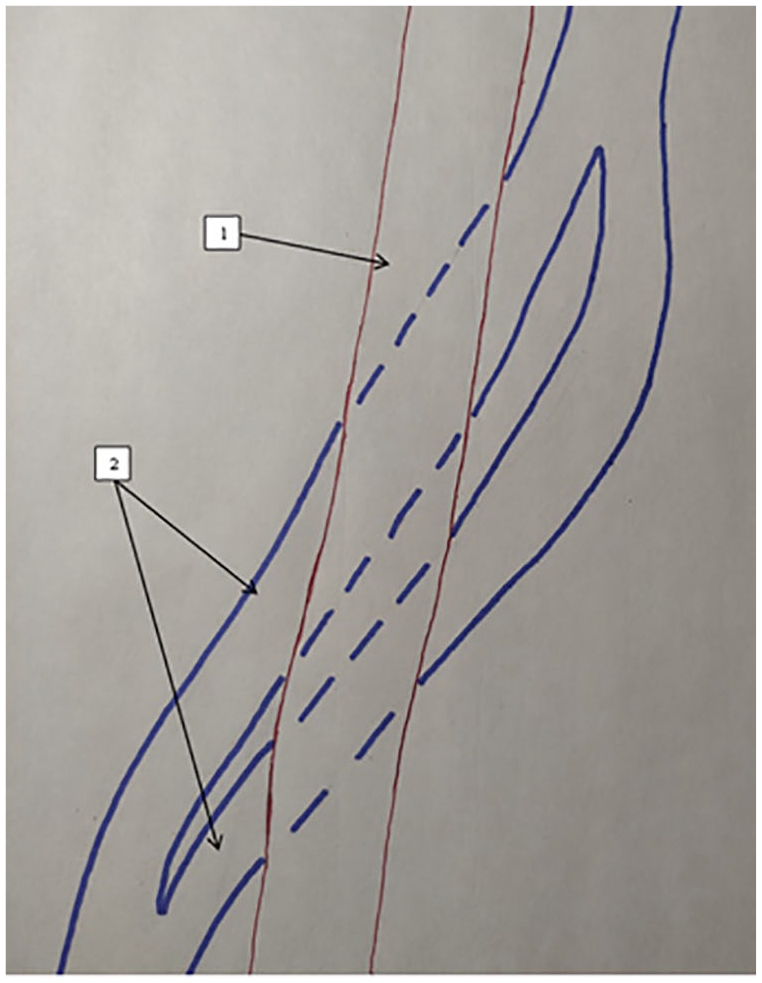
Topography of the superficial FA as related to the topography of the duplicated superficial FV, without transposition. Note that 1 is the FA and 2 is the FV. FA, femoral artery; FV, femoral vein.
A sample sonogram that shows the femoral vessels, in the lower third of the thigh. Note that 1 is the FA and 2 is the duplication of FV, with occlusion of the main trunk (on the left) and distal compression (on the right) revealing blood flow through the additional trunk. FA, femoral artery; FV, femoral vein.
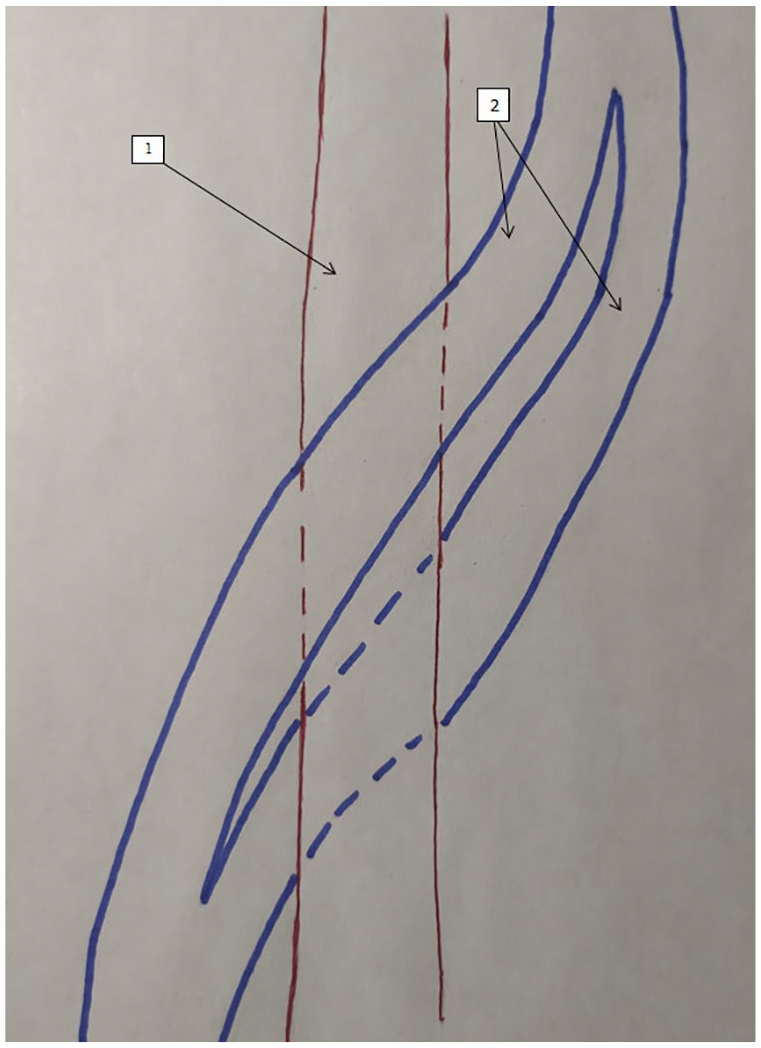
Topography of the FA, as related to the topography of the duplicated FV with transposition. Note that 1 is the FA and 2 is the FV. FA, femoral artery; FV, femoral vein.
However, that was not the case with the PV. In patients with VVs, duplication of the PV trunk at the level above the knee was found in only one patient and was associated with a high bifurcation of the PA (Figure 6). In one patient, there was a local division of the main trunk of the PV into two trunks at the level of the knee. In this case, the vessels were located along the lateral and medial surfaces of the PA. Immediately below the knee, they again formed a single PV trunk. Duplication of the PV below the knee joint was found in 87.5% of cases, and the medial trunk was always larger than the lateral one (Figure 7).
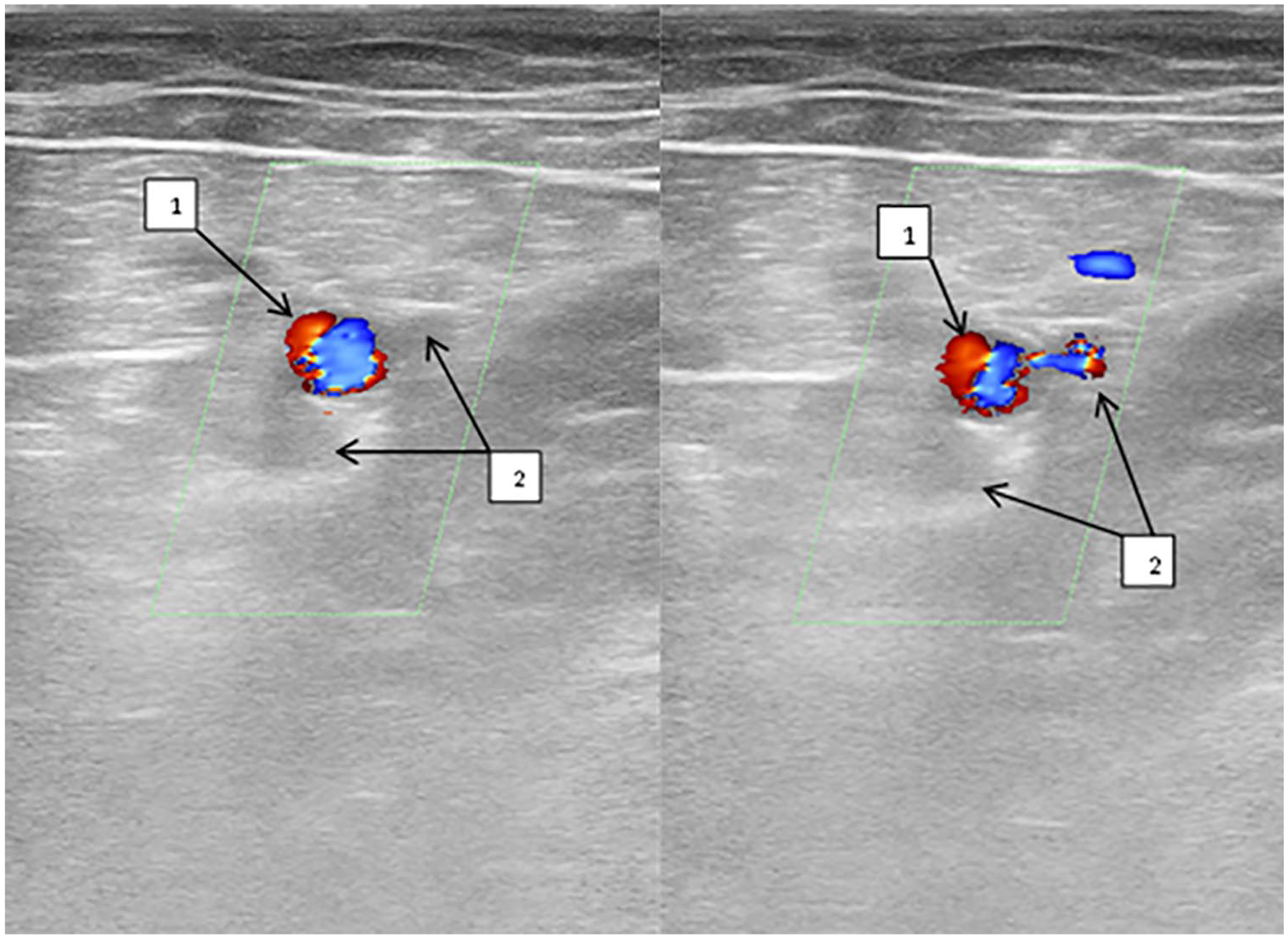
A sample sonogram that demonstrates a high division of the PA, in the proximal part of popliteal fossa. The gray-scale sonogram is denoted with A, B, and C. The color Doppler image is denoted with D. Note that 1 is the PA and 2 is the PV. PA, popliteal artery; PV, popliteal vein.
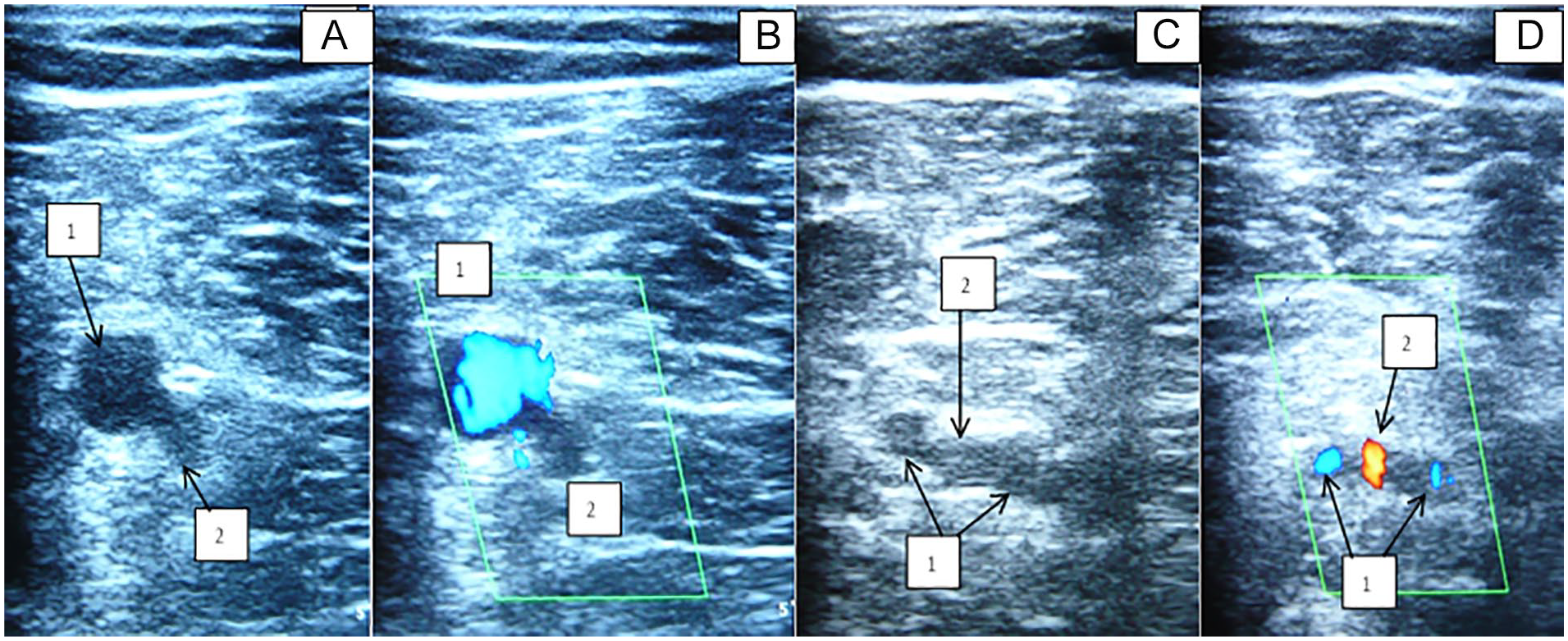
A sample sonogram that provides a topography of the PV, above the level of the knee (A and B). A duplication of the PV is demonstrated, below the level of the knee (C and D). Note that 1 is the PV and 2 is the PA. PA, popliteal artery; PV, popliteal vein.
In patients with PTS, the incidence of PV duplication above the knee level was initially 5%. However, upon detailed study, it was found that the dilated origin of one of the sural veins was mistaken for an additional trunk. In 90.1% of cases, there were two PV trunks below the knee level. In the remaining 9.9% cases, the PV was represented by a single trunk.
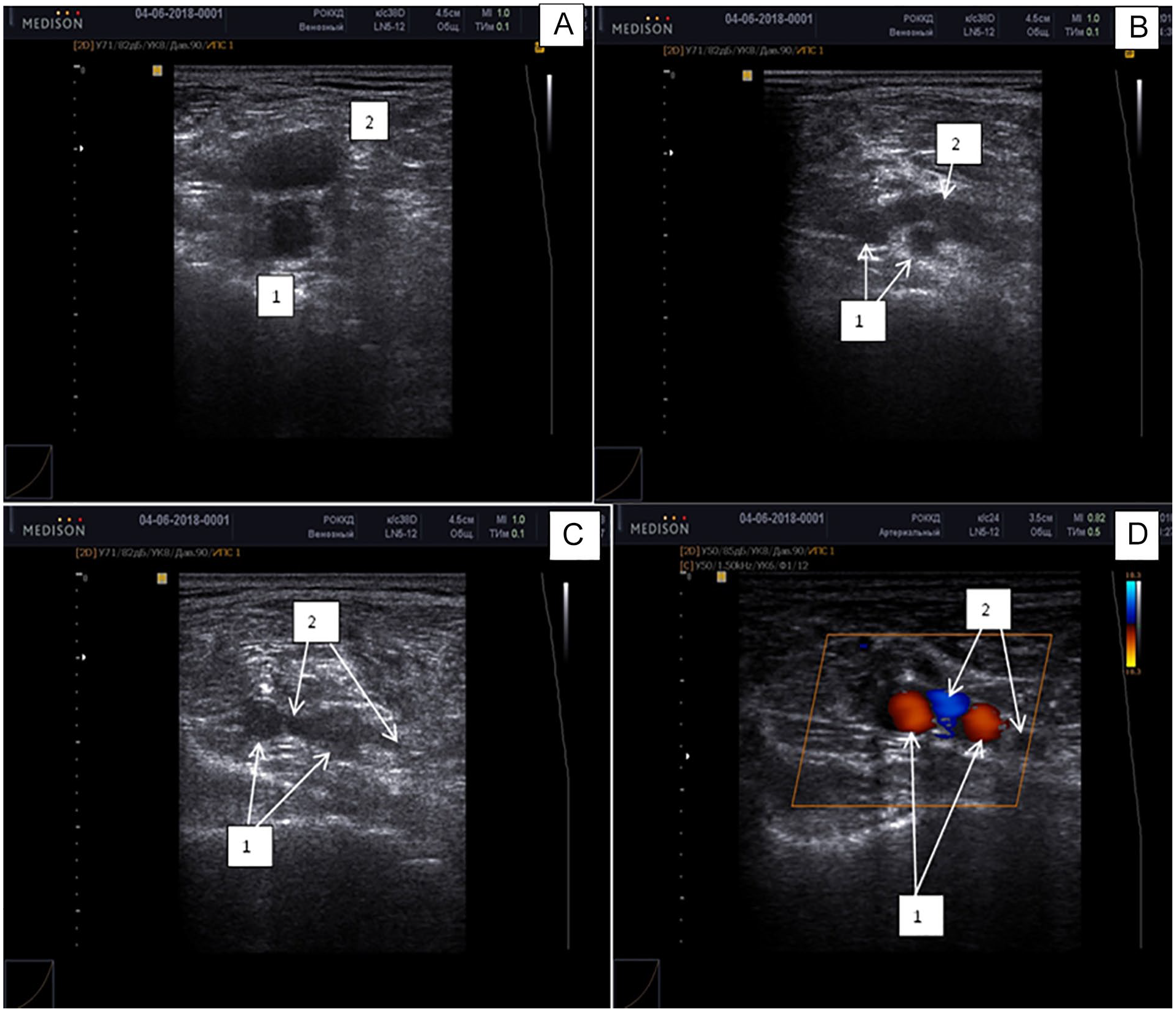
Popliteal vascular topography data were supplemented during anatomical dissection. According to the data obtained, the PV above the knee level was always represented by one trunk; in 83.3% of cases, two trunks of PV were identified below the knee level and were located along the PA (Figures 8 and 9). In 80% of cases, the PV trunks were interconnected in the distal part of the popliteal fossa (Figure 10). In most cases (98.2%), PA was divided into posterior and anterior tibial arteries at the level of the P3 segment, of which, in one patient, it was above the level of the knee (P2 segment). In 1.8% of cases, the PA was divided into anterior tibial and peroneal arteries.
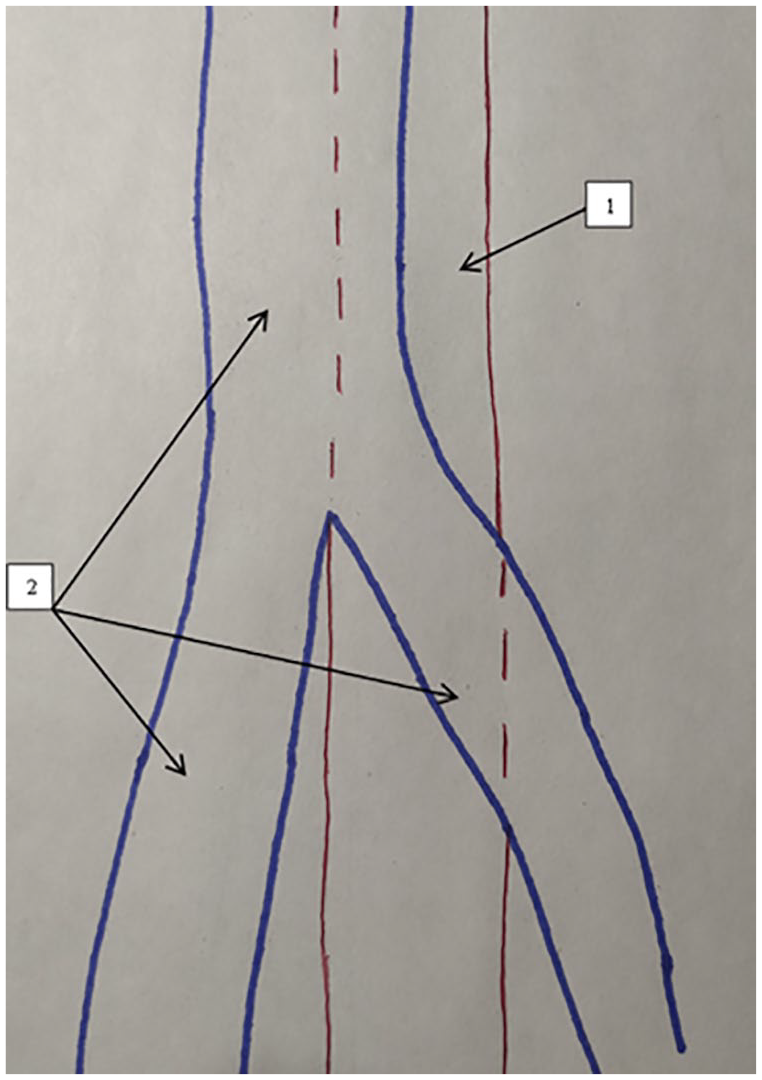
A topography of the popliteal vessels is provided. Note that 1 is the PA and 2 is the PV (one trunk above the level of the knee; two trunks below the level of the knee). PA, popliteal artery; PV, popliteal vein.
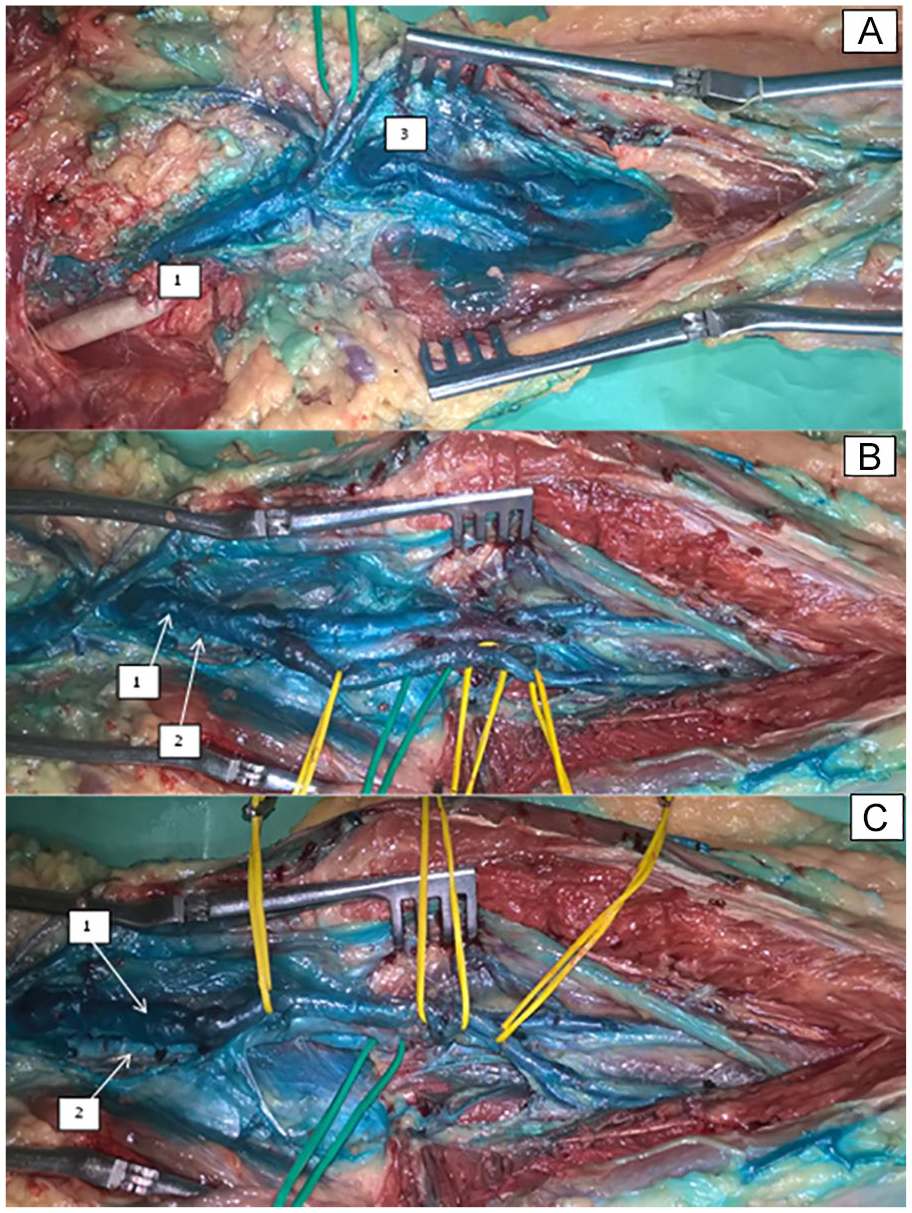
An anatomical dissection of the popliteal fossa. Note that 1 is the PV, 2 is the PA, and 3 is the sural veins. On the yellow vascular loops, the medial trunk of the PV and the origin of the anterior tibial veins (A, B, C) are noted; on the green loops, the origin of the anterior tibial artery is noted (B, C). PA, popliteal artery; PV, popliteal vein.
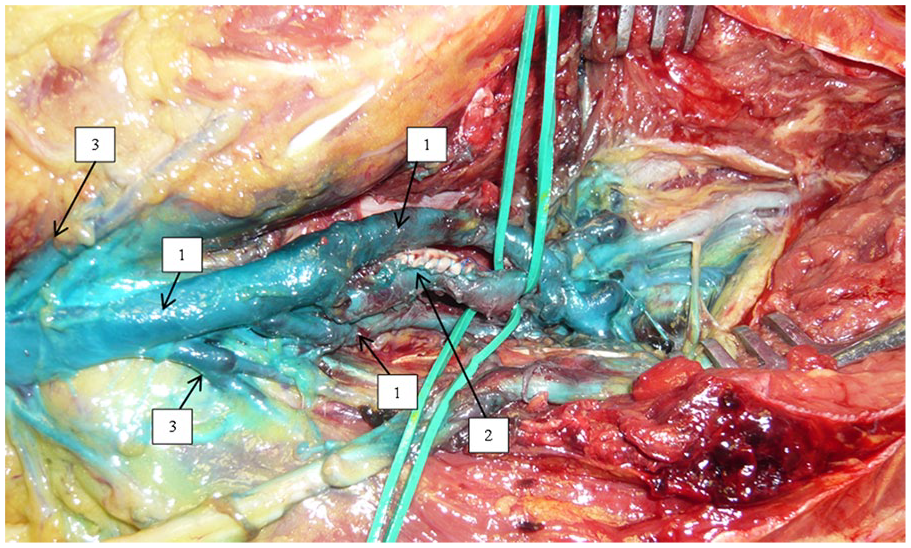
An anatomical dissection of the popliteal fossa. Note that 1 is the PV, 2 is the PA, and 3 is the sural veins. On the green vascular loops, the origin of the anterior tibial vessels is noted. PA, popliteal artery; PV, popliteal vein.
Discussion
Correct anatomical assessment using DUS is not always simple, due to the variant anatomy of the vessels, mainly veins. In addition, there is a concept of developmental anomalies. An anomaly, as an anatomical fact, is the result of an impaired developmental process while maintaining the function of organs and tissues. 11 Therefore, the objective of the clinician is to first differentiate them and second describe the pathological process, to arrive at the correct diagnosis.
Vakhitov et al. 3 reported that during anatomical dissection of the extremities, without signs of CVD, the typical structure of the FV was identified only in 30.2% of cases. Duplication, in one area or another, was identified in 22.6% of cases. Accessory veins with a diameter of up to 2 mm were detected in 47.2% of cases. In 79.2% of patients, a direct connection was identified between the FV and the DFV. At the same time, phlebography revealed a duplication of the FV only in 6.9% of cases. The accessory veins, near the main trunk of the FV, were noted in 7.1% of cases and an anastomosis between the FV and the DFV was found in 8.9% of cases. The authors concluded that the typical clinical anatomy of the deep veins was a risk factor predisposing to the development of VVs. 3 The researchers believed that this was due to a mismatch between the quantitative need for venous outflow and the actual capacity of the deep veins characteristic of normal anatomy.
Although DUS is highly informative, anatomical dissection is much more accurate, 7 as it allows one to isolate and trace absolutely all vessels. Until recently, contrast phlebography was the standard of care for assessing the morphological and functional state of the venous system, in clinical practice. Phlebography, including direct phlebography, is currently only used in difficult diagnostic cases, due to ionizing radiation exposure, invasiveness of the procedure, and the possibility of complications such as thrombosis, allergic reactions to contrast media, hematomas. 12 Retrograde phlebology is most often used, but the best visualization of the venous system is obtained with the intraosseous injection of contrast medium. 5 Quinlan et al., 4 based on the phlebographic findings, reported a duplication of FV in 31% of cases. In that study, 42% of cases demonstrated two deep veins in the popliteal fossa, as well as a complete duplication of PV, detected in 5% of patients. 4
Evidence has been provided that the occurrence of a duplicated FV can occur in up to 5% in CVD, mainly in the middle and lower third of the thigh. 5 There is also a concept of the venae comitantes, or companion veins, or satellite veins to the FV. The difference between the satellite veins and the duplicated trunk of the FV is that there are no valves in the orifice of the latter. Veins connecting FV and DFV are an important source of collateral blood flow. These are small veins, which can be up to 2 mm in diameter. However, if the main trunks are occluded, the companion veins expand two to three times their normal diameter, and they can be mistaken for the main trunk of the FV. Such companion veins are observed in about 35% of patient cases. Most often, they originate in the lower third of the thigh, extending directly from the FV and having connections with the vertical venous plexus at the level of the heads of the gastrocnemius muscles, which in turn are connected with the posterior tibial veins.
Modern diagnostic procedures in patients with CVD are based on the use of minimally invasive techniques.1,12,13 Sonography is the leading diagnostic procedure for patients with CVD.1,2,14 The main advantages are noninvasiveness and the ability to obtain information about the venous system in a dynamic manner. 15 Gray-scale sonography gives a great opportunity to visualize the structure of the venous walls, lumen of blood vessels, and surrounding tissues.
Casella et al. 7 examined patients with VVs and revealed that there was a duplication of the FV in up to 55.1% of cases, of which in 9.6% at its full length; duplication of the PV was identified only in 2.2% of cases. Dona et al. 16 observed a duplication of the FV and PV in 15.7% of cases according to DUS; an isolated duplication of PV was observed in 5% of cases.
In the present study, it was possible to identify a duplication of FV in 14% of cases, in patients with VVs and this is closer to the data published by Dona et al. 16 However, it differs from the study published by Casella et al. 7 At the same time, an increased number of FV trunks was identified in 42.1% of subjects with PTS. Such a sharp increase from 14% to 42.1% in the occurrence of multiple FV trunks can only be explained by the fact that venous occlusion leads to the redistribution of the blood flow through the companion veins. But even if the main trunk of the FV is smaller in size than the additional trunks, it should not change its classical location. Typically found in the upper third of the thigh (at the apex of the femoral triangle) it will be medial and posterior to the FA, in the middle third of the thigh and posterior to the FA, and in the lower third of the thigh and lateral to the artery. All these raise the question of the pathological topographic anatomy of the venous system of the lower extremities.
However, DUS is an operator-dependent method of investigation. The main factor for obtaining objective data is therefore a profound knowledge of the anatomy of the venous system. Subsequently, to clarify this discrepancy, the intention is to continue this study by comparing DUS findings with the results of computed tomographic (CT) angiography and anatomical dissection.
The variant anatomy with the hypoplastic FV and dilatation of the DFV, encountered in one patient with VVs, can be attributed to developmental anomalies. Uhl et al. 17 reported a 2% detection rate for this type of developmental disorder. Another developmental variant identified in the present study was an anastomosis between the DFV and the FV in the lower third of the thigh (Figure 7).
The topography of PV in the present study differed somewhat from the classical anatomy. 1 Radiologists often report two PV trunks. Park et al. 18 reported a high confluence of the tibial veins into the PV in 83% cases, according to the CT angiography. DUS should document the anatomy of both tibial vein and PV and should be carefully assessed, especially when suspecting deep venous thrombosis. 19
Although variant anatomy may sometimes cause difficulties when performing either diagnostic or therapeutic procedures, the duplication of FV or PV has potential benefits in open vascular surgery. The duplicate could be a possible source for bypass conduits or permanent vascular access, since it corresponds to the triad that determines the actions of surgeons. These actions could be based on the anatomical accessibility (a greater length, superficial localization), technical capability,10,20 or physiological sequalae (removal of one of the additional deep venous trunks will not cause significant impairment to the venous flow, thereby reducing the risk of developing venous insufficiency). Variant anatomy of the PA is also of practical importance in open vascular surgery because high bifurcation of the PA requires an access along the entire length of the PA rather than a minor access in the upper third of the medial surface of the tibia.
Therefore, performance of DUS requires profound knowledge in anatomy when assessing the femoral and popliteal vessels. This was done to avoid diagnostic errors and provide a clear anatomical description to the surgeons.
Limitations
The main limitations of the study was the research design as this was a convenient sample that formed a study cohort. In addition, there were difficulties in differentiating pathological conditions from variant anatomy in the absence of a confirmed clinical diagnosis. This may partially explain the different incidence of FV duplication described in the literature. Therefore, the intention is to carry out another study focused on the detection of valve insufficiency in deep veins in patients with VVs and PTS.
Conclusion
In this patient cohort, up to 14% had VVs and 42% with PTS had an increased number of FV trunks. Duplication of the PV below the level of the knee occurred in 87.5% to 90.1% of this cohort. Changes in the topographic anatomy of the FV and PV in PTS occur due to the dilatation of the satellite veins, mimicking the typical veins of the thigh. In this patient cohort, 1.8% had an atypical PA, with the artery splitting into anterior tibial and fibular arteries, rather than the typical anterior and posterior tibial arteries.
Footnotes
Ethics Approval
The study has been approved by the Institutional review board (Local ethical committee) of the Ryazan State Medical University.
Informed Consent
Informed consent has been obtained from the participants of the study.
Animal Welfare
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration
The trial has been registered at the Institutional review board (Local ethical committee) of the Ryazan State Medical University.
