Abstract
Objective:
The aim of this study was to compare the stiffness of different histological types of breast lesions by obtaining shear wave elastography (SWE) and apparent diffusion coefficient (ADC) values, and to determine the contribution of these two methods to the diagnosis.
Materials and Methods:
In total, 70 patients with biopsy-proven breast lesions were included in the study. The mean SWE values of breast lesions were recorded and ADC values of these lesions were calculated. Receiver operating characteristic (ROC) curve analyses and the diagnostic accuracies of SWE-ADC values were determined.
Results:
The mean SWE values were 45.47 ± 25.11 kPa and 3.51 ± 1.04 m/s in benign group, and 161.11 ± 219.34 kPa and 5.96 ± 1.06 m/s in malignant group, respectively. The mean ADC values were 1.38 ± 0.32 (×10–3 mm2/s) in benign group and 0.96 ± 0.22 (×10–3 mm2/s) in malignant group, respectively. When the diagnostic performances of both imaging modalities on mass stiffness are evaluated, statistically significant negative correlations were found between SWE lesion values and ADC lesion values.
Conclusion:
Evaluation of tissue elasticity has recently been used frequently in the diagnosis of breast diseases. SWE-ADC values, which are negatively correlated in the diagnosis of breast masses, may prove to be a powerful alternative diagnostic tool that can be used interchangeably, as appropriate.
Currently, breast screening is performed with periodic breast ultrasonography (US) and mammography for patients 40 years and older. Additional imaging modalities such as magnetic resonance imaging (MRI), tomosynthesis, and diagnostic biopsies are preferred when necessary depending on the tissue content. Knowing the degree of stiffness of the tissue in breast masses can provide information about whether a suspected mass is benign or malignant. The diagnostic accuracy can be increased, in a short time, with methods such as shear wave elastography (SWE) and apparent diffusion coefficient (ADC) values that provide numerical data on tissue stiffness. It may be possible to prevent unnecessary biopsies in lesions defined as Breast Imaging Reporting and Data System (BIRADS) 3-4, and provide additional information in predicting breast cancer prognosis and understanding response to neoadjuvant chemotherapy. With the increased specificity of gray-scale US, elastography can provide a noninvasive assessment of the “stiffness” of a breast lesion.1–3 Briefly, it is possible to obtain the physical parameter, Young’s modulus, with elastography. Young’s modulus provides different values in various biological tissues, creating significant contrast in the characterization of tissues, and allows clinicians to evaluate tissue stiffness with quantitative values, equivalent to palpation. 4 SWE is an up-to-date sonographic method that can transform the stiffness of tissues into quantitative information by following the shear waves passing through the tissue. 5 While high kPa values are observed in malignant tissues, breast lesions containing benign and fibrotic tissues are generally represented with low kPa values. Therefore, the specificity of SWE helps to reduce the number of biopsies to be used in the diagnosis of benign breast lesions.
MRI performed in conjunction with ADC paramaters, can be very sensitive in the diagnosis of breast lesions of different sizes, and is another method that can be used to evaluate the elasticity of breast lesions. Diffusion-weighted imaging (DWI) is based on the random diffusion principle of water molecules. Naturally, diffusivity will decrease in dense and highly cellular-containing tissues. ADC is the apparent diffusion coefficient, which describes the diffusion rate and is defined as the average area occupied by a water molecule per unit time (mm2/s). ADC provides numerical data on diffusivity.6–9 In the comparison of ADC values, it was revealed that malignant tumors showed lower values than benign tumors. 8 Different studies on ADC values of breast lesions have shown that ADC parameter is associated with proliferation markers, cellular accounts, and histopathological features. 9
The benefits of both ADC and SWE techniques in determining large-sized solid breast masses have previously been stated.10–12 The aim of this study was to compare the quantitative values collected from SWE-ADC evaluations, when performed on the same breast lesions with different pathologies, to investigate its diagnostic contribution.
Materials and Methods
Patients seen at the host clinic and diagnosed with breast lesions between April 2018 and November 2020 were invited to this prospective study. The Institutional Review Board approved this research, and it was conducted in accordance with the Declaration of the Helsinki World Medical Association. Each patient provided an informed written consent before entering the study. Within this patient cohort, there were breast complaints prior to enrollment in the study. Of those, there were 70 cases that had breast masses which were detected during routine evaluations (e.g., sonography, mammography) and the diagnosis was confirmed histopathologically, with a tru-cut biopsy procedure.
The inclusion criteria for this patient cohort were those who had a clinically suspected breast lesion, had sudden breast pain, experiencing severe axillary pain, palpable breast masses, a history of breast cancer in their close relatives, follow-up for biopsy-proven breast lesions, and those with radiologic signs of a breast lesion.
The exclusion criteria for this patient cohort were those patients who did not qualify for MRI (presence of MRI-incompatible prosthesis or materials, claustrophobia, etc.), have systemic diseases affecting the chest wall (e.g., systemic lupus erythematosus, dermatomyositis, scleroderma), had a history of breast surgery, and those patients who received chemotherapy before their US and/or MRI examinations.
All of the breast MRI examinations as well as the US and SWE examinations were analyzed and performed by the same radiologists (M.D.A., O.T.). These physicians had more than 14 years of experience with MRI, more than 14 years of performing US, and more than 5 years of elastography. All images were obtained by consensus, adhering to the same conditions. Patients were asked to lie down in a supine position with their hands under their heads in such a way that they felt comfortable. The axillary region and breast US was conducted using an ultrasound equipment system, capable of SWE measurements, using a 14 MHz linear-array transducer (Aplio 500 Platinum; Canon, Japan). Mean SWE values were calculated as in kPa and m/s units. MRI was performed using a 1.5 Tesla scanner (Signa HD, GE Healthcare, Milwaukee, WI, USA) in prone position with bilateral 16-channel phased-array breast coils. After routine sequences were taken, diffusion-weighted sequence (repeat time (TR)/echo time (TE): 6050/95, flip angle: 90°, slice thickness: 3.5 mm, field-of-view (FOV): 250–350, matrix 128 × 192) suppressed fat in the axial plane, with single-shot echo-planar imaging (EPI) sequence, in all three directions (
Statistical Analysis
The data were analyzed with IBM SPSS v23 program. Compliance with normal distribution was examined by the Shapiro-Wilk test. An independent two-sample
Results
There were a total of 70 patients with a mean age of 43.50 (21–74) years. The average age of the benign group was 36.5 ± 11.36 years and the average age of the malignant group was 54.10 ± 9.79 years. Group 1 consisted of 41 patients with benign breast lesions, and Group 2 consisted of 29 patients with known malignant lesions. Histopathological distribution of malignant lesions included invasive ductal carcinoma (
There was a statistically significant difference between the mean age values of the groups (
When the groups were evaluated according to ADC lesion measurements, there was a statistically significant difference between the mean values (
There was a statistically significant difference between the SWE lesion kPa mean values according to the groups (
There was a statistically significant difference between the mean SWE lesion values as m/s according to the groups (
Comparison of Parameters by Groups.
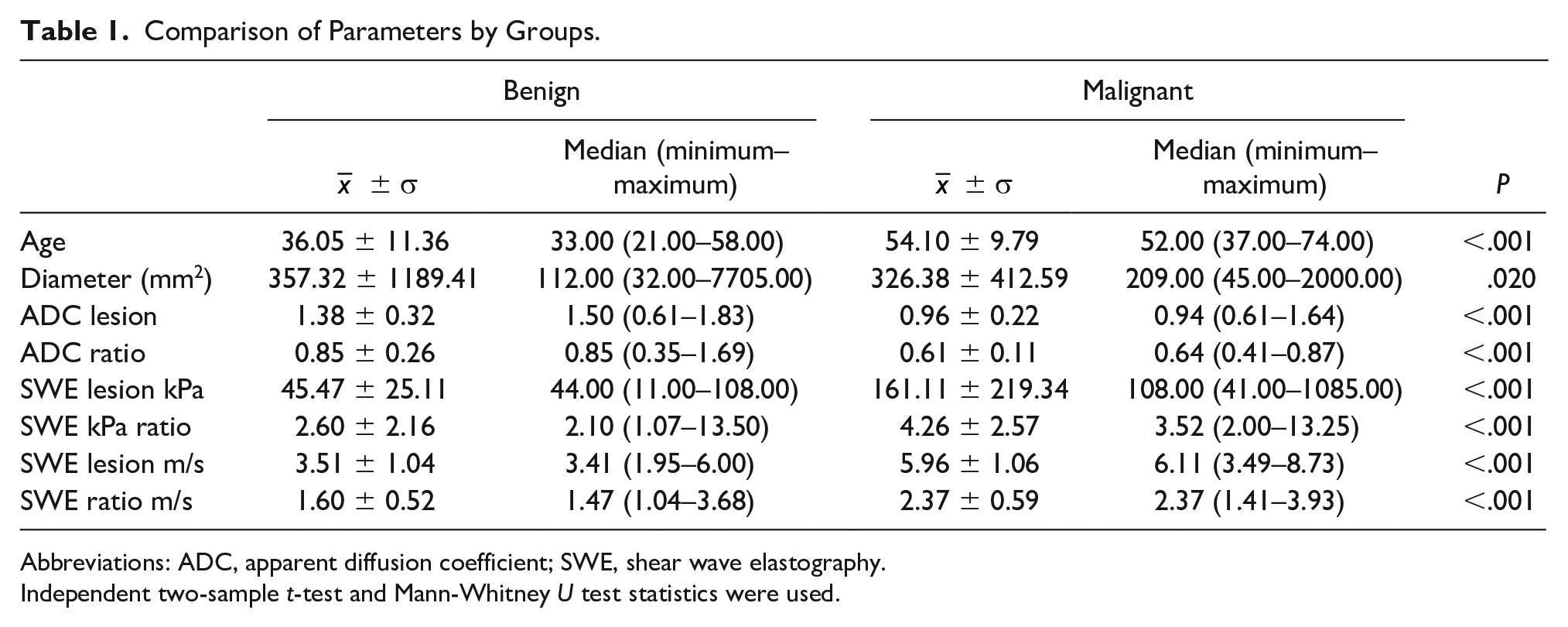
Abbreviations: ADC, apparent diffusion coefficient; SWE, shear wave elastography.
Independent two-sample
When the data were compared according to the age and lesion sizes, a statistically significant weakly negative relationship was noted between ADC lesion measurement values and age (
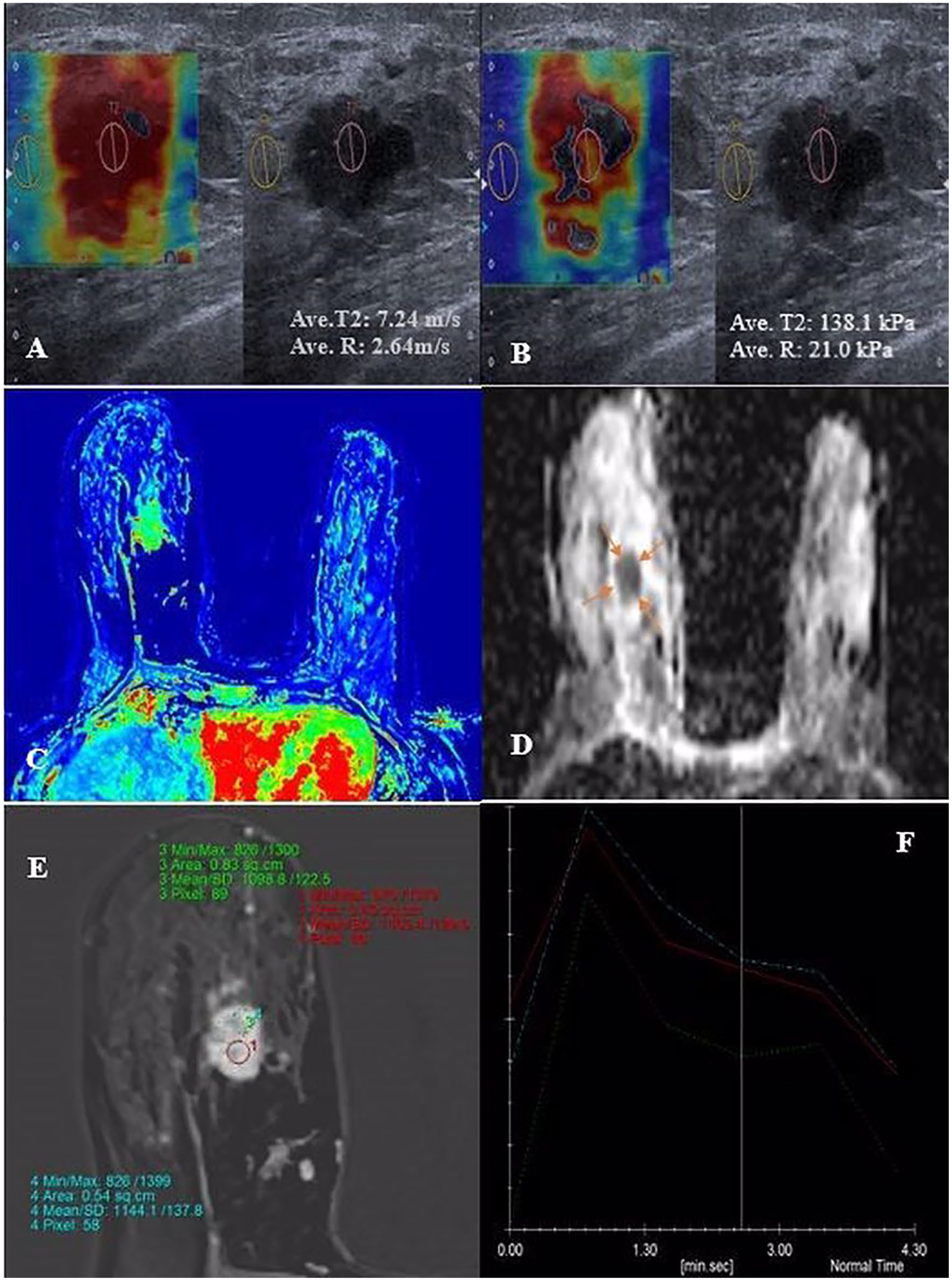
Female patient in her late 50s with right breast invasive ductal carcinoma. Axial plan SWE measurements reflect increased stiffness with high measurements as in kPa (B) and m/s (A). Notice the color change of the mass area from blue to red indicated increased stiffness. ADC color map (C) and neutral (D) images depict diffusion restriction (ADC value: 0.68 × 10−3). Postcontrast dynamic images with different ROIs (E) and type 3 enhancement curves (F) indicate early washout compatible with breast cancer. ADC, apparent diffusion coefficient; SWE, shear wave elastography; ROIs, regions of interest.
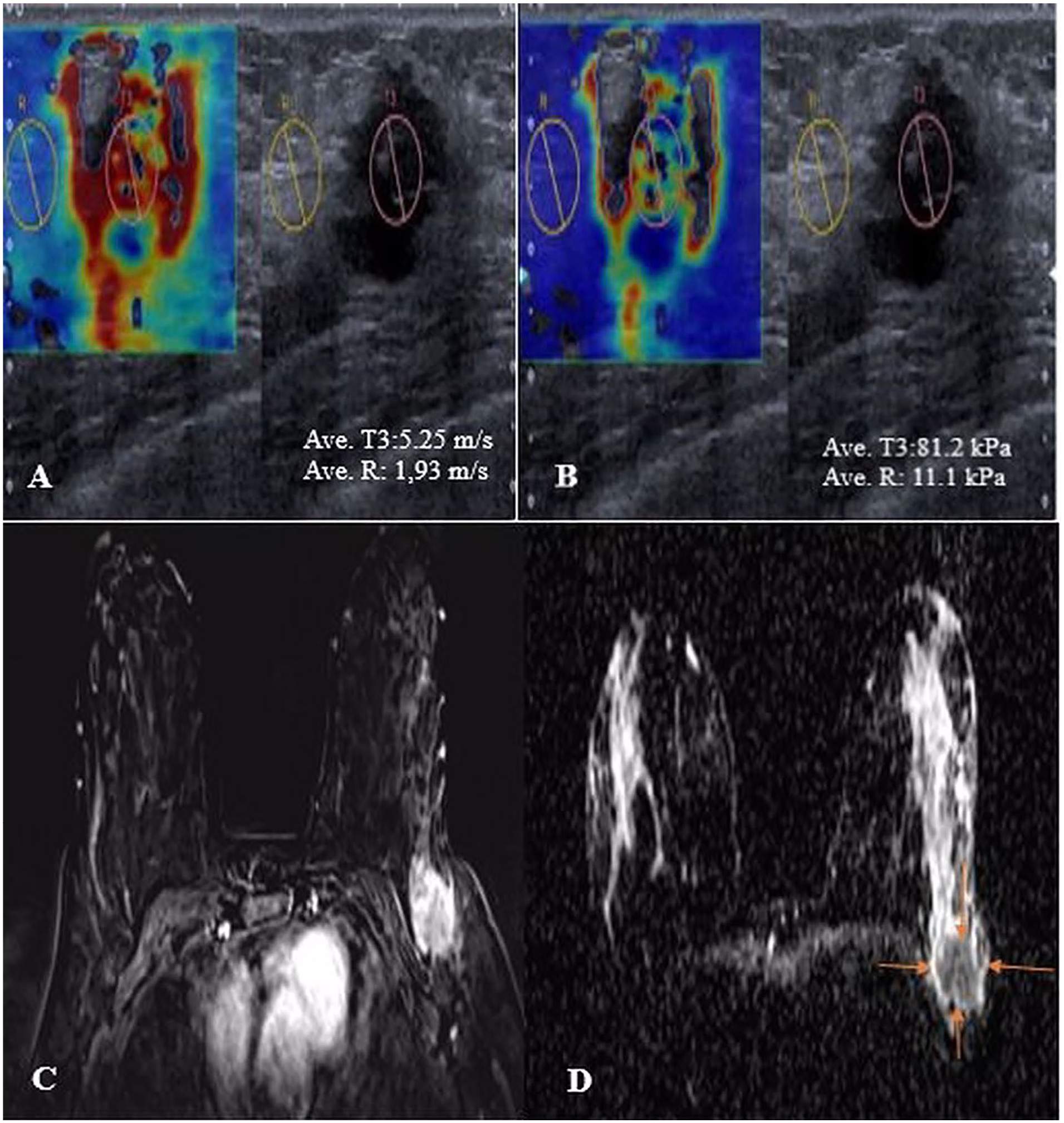
Female patient in her early 50s with left breast fibroadenoma. Axial plan SWE measurements reflect lower stiffness with moderate measurements as in kPa (A) and m/s (B). DWI (C) and ADC (D) images depict diffusion restriction (ADC value: 1.83 × 10−3). ADC, apparent diffusion coefficient; DWI, diffusion-weighted imaging; SWE, shear wave elastography.
When comparisons were made between SWE measurements and ADC values, a statistically significant moderately negative correlation was noted between SWE lesion kPa and ADC lesion values (
Sampling of the Relationship Between ADC and SWE Values.
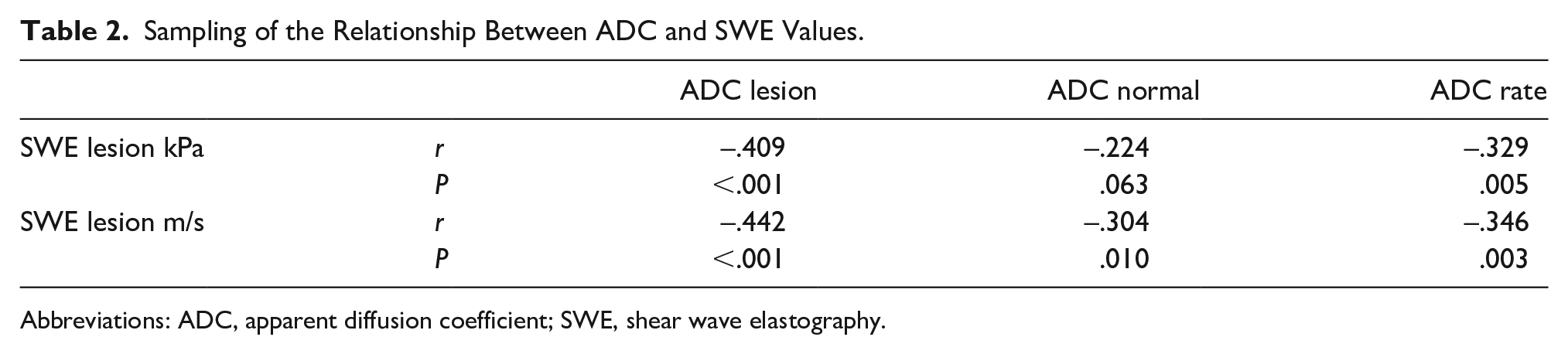
Abbreviations: ADC, apparent diffusion coefficient; SWE, shear wave elastography.
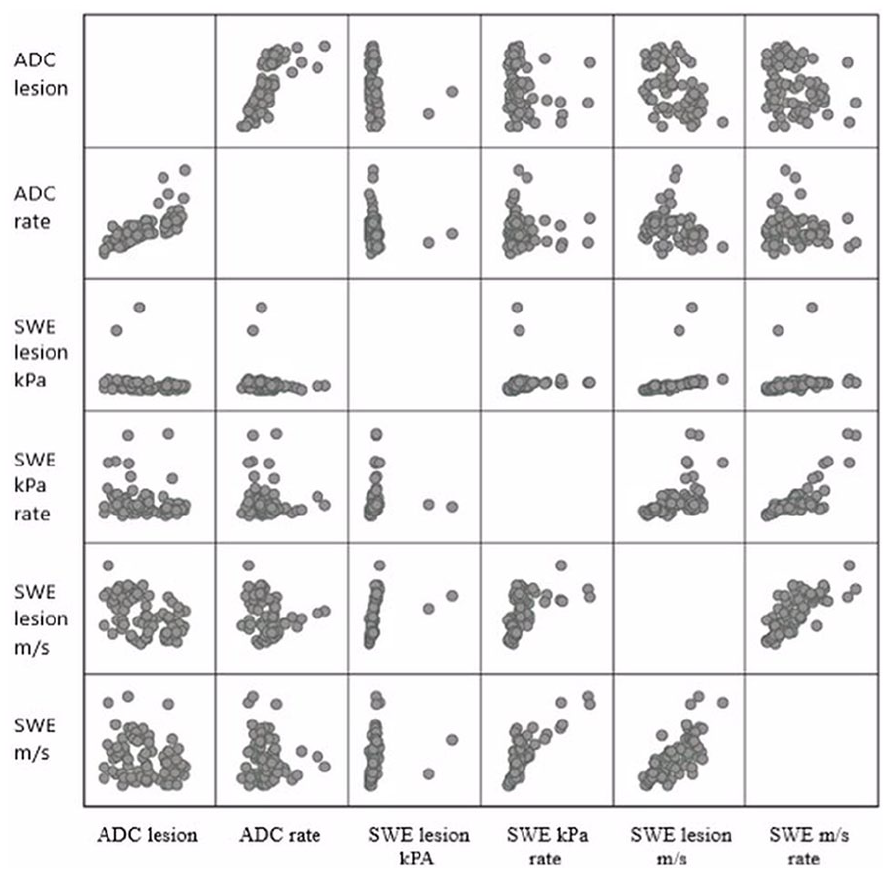
Matrix scatterplot showing the significant negative correlations between ADC and SWE. ADC, apparent diffusion coefficient; SWE, shear wave elastography.
It was important to define a cutoff value, with significant differences, in the total numerical data obtained. When the cutoff value for ADC lesion was taken as 1.115 (×10−3 mm2/s), the area under the curve (AUC) was obtained as 0.843. This obtained value was statistically significant (79.3% sensitivity and 78% specificity;
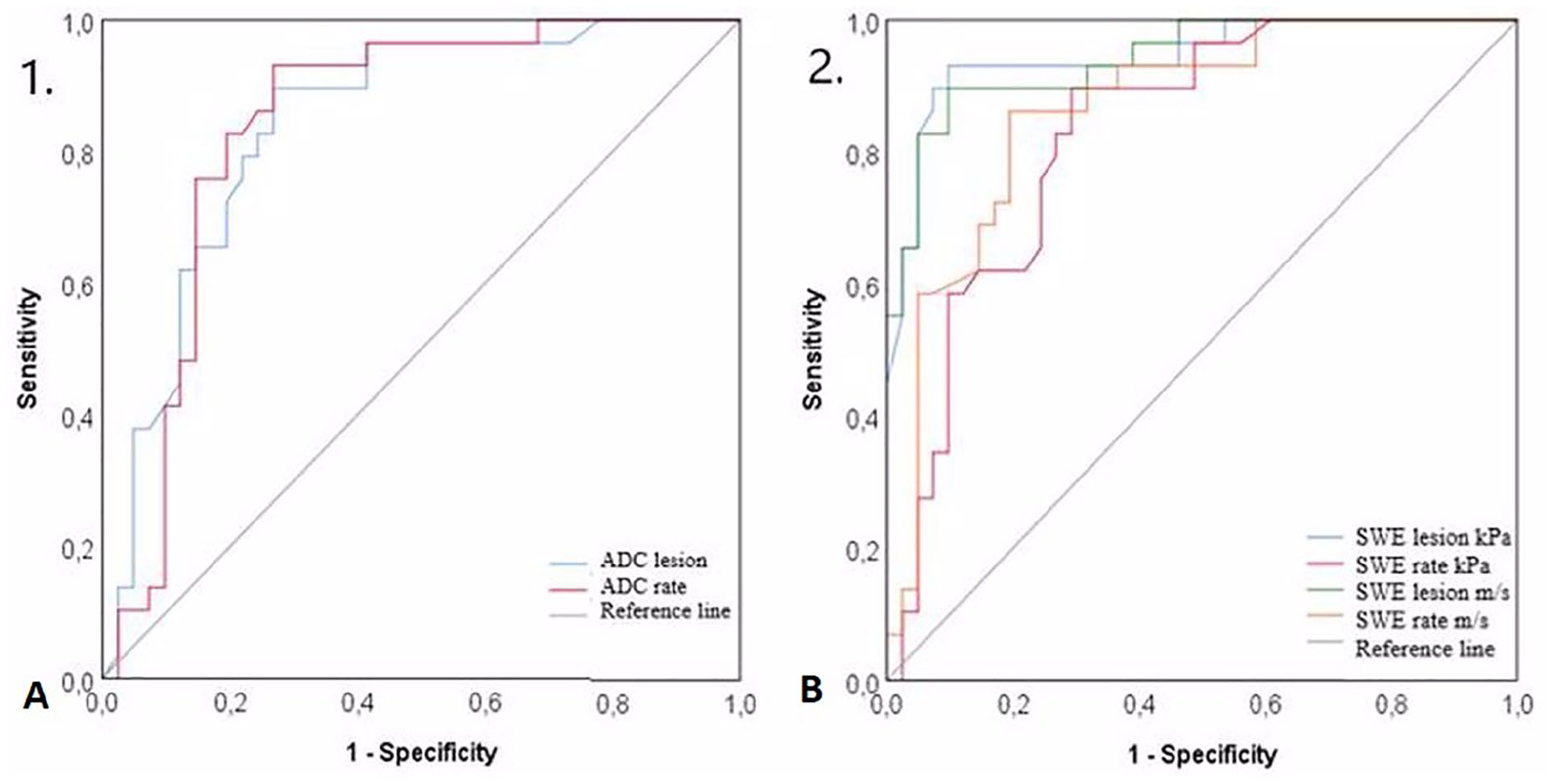
ROC curves of ADC (A) and SWE (B) measurements for the determination of the breast lesions. ADC, apparent diffusion coefficient; SWE, shear wave elastography; ROC, receiver operating characteristic.
Discussion
Quantitative data on the stiffness of tissues are obtained based on the principle of following the shear waves passing through the tissue in SWE examination, which is an increasing useful diagnostic method. 13 The information about tissue stiffness obtained by the SWE method serves as a valuable descriptor for the evaluated tissue components. In general, malignant breast lesions are stiffer than nearest breast tissues, and the majority of such lesions shows fibrous changes. Elastography can be a useful method to examine different types of breast lesions and to differentiate between malignant lesions and benign lesions. Sonoelastography can be used to obtain information about the stiffness of the tissues as well as showing high sensitivity in distinguishing benign and malignant breast lesions as Itoh et al. 14 showed in their study. Elastographic features of lesions of different histopathological types can be found elsewhere. 15 Besides, there are several pathologies that can produce false positive results, such as fat necrosis, mastitis, and low incidence of fibroadenoma. Lymphoma has the only false negative result.16,17
Images obtained by MRI provide multiplan tissue characterization features; fibrous changes appear on T2-weighted or short tau inversion recovery (STIR) images with low signal. However, it is not the only indicator of fibrosis, so T1-weighted series, dynamic phase, and contrast-enhanced examinations are added to the imaging protocols. In recent years, DWI has been used to detect breast cancer in whole body scans. With an overview, DWI is useful in detecting and evaluating extensions of breast carcinomas, as well as aiding in MRI sequences in differentiating malignant and benign lesions.18–21 ADC values reflecting cellular density also can be achieved by DWI. Cellular density is higher in many types of cancer consisting of high-grade malignant lesions than benign lesions. 22
The data obtained in the current study showed that elastographic findings are different in breast lesions. The current study demonstrated a significantly higher mean SWE values in malignant lesions as in kPa and m/s. Conversely, the average ADC values of the malignant group were significantly lower. It was also observed that statistically significant negative correlations existed between SWE lesion values as in kPa and m/s with ADC lesion values. This cohort study is unique in that it provides the opportunity to compare SWE values with ADC values of benign and malignant breast lesions.
According to the study of Au et al.,
23
the most important factor that affecting SWE values is the size of the tumor. The reason of more peripheral desmoplastic reactions in large lesions can be explained with more tumoral cellularity, angiogenesis, and edema than small lesions. Another study by Chang et al.
24
revealed that average elasticity values increase as the size of the lesion increases. In the present study, a statistically significant weak positive correlation was found between SWE lesion kPa, SWE lesion m/s, and lesion size values, given the limited number of patients (
As Itoh et al. 14 explained in their work, as well as in the current study, that SWE values of malignant lesions were significantly higher than those of benign lesions. However, there is still no clear consensus on recommending cutoff values for benignity and malignancy, especially when considered for malignant lesions. Due to histological differences, significant overlaps occur in the assessment of the elasticity of malignant and benign lesions. The vast majority of breast cancers tend to be severe, and SWE values are usually measured above 50 kPa. In some studies, quite different suggestions have been made for breast cancer cutoff values.25–27 Our cutoff value for malignancy was 75 as in kPa.
In some previous studies, it was stated that there is a correlation between the degree of liver fibrosis and ADC values.28,29 Similarly, breast cancers are usually described with a fibrotic stroma; it is thought that there may be a link between the degree of fibrosis and the ADC value. Matsubayashi et al. 30 reported in their previous studies that dynamic contrast-enhanced MRI images of breast lesions were affected by many histological and morphological factors, including fibrosis. In their recent studies, they mentioned that there is a meaningful relationship between SWE and ADC values.30,31 Similar to their data, the current study revealed a statistically significant negative correlations between SWE lesion values as in kPa and ADC lesion values as in m/s.
When reviewing the current data obtained with SWE and ADC, it is possible to summarize that the accumulation and distribution of collagen in the masses reflect the aggressiveness of the tumors. Thus, having information about the histological background of the tumor will gain importance in planning treatment options and evaluating the response to treatment. In addition, it may be possible to accurately define the pathological response of the tumor following preoperative neoadjuvant chemotherapy, to provide information about the postoperative scar-residue distinction, and perhaps to observe metastatic nodes.
Developing real-time stress maps can solve some of the problems with learning curves in SWE. Thus, it can also contribute to the selection of the appropriate ROIs during measurements. While discussing its curative effect on outcomes, Tian et al. argued that US-based diagnostic accuracy will increase in their studies that enable three-dimensional (3D) SWE examination, which will be of great importance in the future. Compared with the standard method, more stable measurements will result given the lower impact of artifacts on 3D SWE. Coronal perspective, being able to examine the elasticity of the complete lesion, and being able to scan the whole breast will be the advantages of 3D examination.32,33 Apart from this, promising future mechanical devices such as vibrating SWE, which generate shear waves that can be detected by gray-scale US, have begun to be used. Hudert et al. in their study that allows for variation in the frequency of possible mechanical stress provided additional diagnostic information. 34 Opto-acoustic (OA) imaging is another promising technique that can be used to identify benign and malignant breast lesions with its success in reducing the postprocedural BIRADS category grade. 35
Limitations
This cohort study contains several limitations mainly due to the research design and a convenient sample of patients. In addition, the ADC calculation was quite difficult for lesions located along the periphery of the breast. In addition, falsely low SWE values can be obtained, as seen in large sizes of infiltrative cancers, because US cannot penetrate highly into scirrhous tissues. The sample size was smaller due to the study being managed in a single center. As not all of the patient cases were surgical in nature, parameters such as lymphatic invasion, vascular invasion, stromal reaction, and lesion character could not be evaluated.
Conclusion
In determining the characteristics of breast lesions, especially in the presence of cancer suspicion, SWE’s contribution to US is a proven diagnostic tool. The degree of stiffness of a carcinoma depends on the rate of fibrosis it contains, so the use of combined imaging methods (such as SWE-ADC) that provides numerical data could be very useful in distinguishing breast cancer from benign breast lesions. The combined use of different techniques can contribute to reducing the frequency and time of scanning, and controlling erroneous measurements. ADC values and SWE measurements have significant reverse correlations in most of breast lesions, and the method of comparing diffusion-weighted MRI with SWE in selected cases has the potential to alternatively evaluate these lesions in more detail. Future studies should consider adding new techniques with a larger number of patients, as this will provide more accurate assessment of elastograms and stronger clinical data.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Local Institutional Review Board approval was obtained before starting the study (#2018.498). The study was performed according to the World Medical Association Declaration of Helsinki.
Informed Consent
Written informed consent was obtained from all subjects before the study.
