Abstract
Objectives:
To differentiate between the classic (A/B) from the nonclassic (C/D) phenotypes of the polycystic ovarian syndrome (PCOS) with sonography.
Materials & Methods:
Retrospective analysis was performed upon 72 female patients with PCOS. All patients underwent transvaginal sonography. The patients were classified into classic and nonclassic phenotypes of PCOS. Image analysis was performed for the ovarian volume, follicle counts, follicle size, and endometrial plate thickness.
Results:
There were significant differences in the ovarian volume (P = .001), follicle counts (P = .001), follicle size (P = .001), and endometrial plate thickness (P = .001) between classic and nonclassic phenotypes of PCOS. The threshold value for ovarian volume, follicle count, follicle size, and endometrial plate thickness used to differentiate classic from nonclassic phenotypes were 12.5, 10.5, 5.25, and 5.75, respectively, with an area under the curve of 0.79, 0.82, 0.83, 0.77 and an accuracy of 75%, 73.6%, 79.2%, and 68.1%, respectively. The serum testosterone level and the body mass index were significantly higher in patients with classic than nonclassic phenotypes of PCOS (P = .001, .04), respectively.
Conclusion:
Sonography findings can differentiate classic from nonclassic phenotypes of PCOS.
Polycystic ovarian syndrome (PCOS) is a highly prevalent complex endocrine disorder in women of reproductive age worldwide. 1 The clinical features of PCOS include menstrual irregularities (oligomenorrhea/anovulation), infertility, hyperandrogenism (HA), either clinical (acne, hirsutism) or biochemical (elevated free testosterone or free androgen index).2,3 The most common finding of PCOS includes elevated serum levels of free testosterone (T), androstenedione, dehydroepiandrosterone sulfate (DHEAS), an excessive amount of luteinizing hormone (LH), elevated LH/FSH ratio. Anti-Müllerian hormone (AMH) has been considered a marker for the ovarian function and reflects the severity of PCOS, which is defined by its two cardinal endocrine elements: anovulation and HA.3–5 Patients with PCOS have a high risk of endometrial hyperplasia, obesity, dyslipidemia, high blood pressure, insulin resistance, diabetes mellitus type II, cardiovascular, and cerebrovascular disorders.4–6
Different studies discuss the role of sonography in the diagnosis of PCOS.7–10 Transvaginal sonography (TVS) depends mainly on the morphological changes,10–14 the ovarian volume,15–17 the number of follicles,18,19 and the endometrial plate thickness, 20 for diagnosis of PCOS. Color Doppler can detect increased vascularity of the ovaries in patients with PCOS21,22 and advanced sonography such as three-dimensional sonography23,24 and sono-elastography 25 is used to diagnose PCOS, but their results are overlapping. Few limited studies discuss the role of magnetic resonance imaging (MRI) and diffusion-weighted MRI for diagnosing PCOS, without definite diagnostic criteria.26,27
In 2012, the National Institute of Health (NIH) recommended the addition of phenotype classification of PCOS. According to the Rotterdam European Society for Human Reproduction and Embryology and the American Society for Reproductive Medicine (ESHRE/ASRM), their definition recognizes four PCOS phenotypes. This is based on the presence of oligomenorrhea/anovulation (O), HA, polycystic ovary morphology (PCOM) with four phenotypes: phenotype A (O + HA + PCOM), phenotype B (O + HA), phenotype C (HA + PCOM), and phenotype D (O + PCOM). These phenotypes were categorized into classic PCOS (phenotype A/B) and nonclassic (phenotype C/D) PCOS.1,28–30 The classic PCOS is associated with more obvious menstrual dysfunction, higher prevalence of obesity, HA, deranged lipid profile, increased insulin levels, higher rates of insulin resistance, high level of AMHs, increased body mass index (BMI), and risk of metabolic and cardiovascular disorders than others. The nonclassic PCOS has the mildest degree of endocrine and metabolic dysfunction and the lowest prevalence of metabolic disorders.1,30–32
Differentiation between phenotypes of PCOS is essential for the prognosis of the severity of the disease, the outcome of fertility, and planning for the lines of treatment. While the classic type of PCOS is associated with a higher risk for metabolic dysfunction compared with the nonclassic type of PCOS. This necessitates a close observation and hormonal management.33,34 It would appear that there is no previous study in the English literature to discuss the role of sonography in phenotyping of PCOS.
The aim of this study was to differentiate between the classic from nonclassic phenotypes of PCOS with sonography.
Materials and Methods
Study Subjects
The institutional review board approved this retrospective study of 76 women of reproductive age, and informed consent was waived because this was a retrospective study. The inclusion criteria were female patients with Rotterdam criteria 33 who presented with irregular menstruation, hirsutism, and/or infertility who underwent TVS. Among 76 patients retrospectively and initially diagnosed with PCOS, with the Rotterdam criteria, 72 patients met the inclusion criteria for the study. Four patients were excluded due to the presence of dominant follicle >10 mm and nondiagnostic quality of the image. The mean age of the remaining 72 patients were 28 years old with a range of age 23 to 43 years. The patients were classified into classic phenotypes including phenotype A (n = 36) and phenotype B (n = 10) and nonclassic form including phenotype C (n = 16) and phenotype D (n = 10) according to Rotterdam (ESHRE/ASRM) criteria.1,28–30 The BMI and the serum testosterone level were calculated by blood sample.
Sonographic Examination
All sonographic examinations of 72 patients were performed using an ultrasound machine with a 10 to 18 MHz transvaginal transducer (AU5; ESAOTE, Genoa) by two radiologists (A.A.K.A.R., H.A.E.), experts in TVS since 25 and 15 years, respectively. The TVS was done with an empty urinary bladder. Participants were evaluated with gray-scale sonography of two ovaries, uterus, and included the endometrial stripe. The ovaries were scanned from the inner to the outer margins in both transverse and longitudinal planes; first with gray-scale sonography, the ovarian volume on both sides was calculated. The ovarian volume was calculated using a simplified formula 0.5 × length × width × thickness. At the conclusion of the examination, the follicular size and count were assessed.
Data analysis
Two radiologists performed the image analysis and disagreement between both radiologists was resolved with consensus. The image analysis was done for the ovarian volume, the follicles number, and the follicle size of both ovaries and the endometrial plate thickness. The ovarian volume was calculated using a simplified formula for a prolate ellipse (0.52 × width × length × thickness). The sonographic images were evaluated for the number of antral follicles in the sectional plane, and the longest diameter of a large follicle was calculated. The endometrial plate thickness was measured in the longitudinal plane of the uterus.
Statistical analysis
Statistical analyses were done using commercially available Software Package for Social Sciences version 22 (SPSS, Chicago, IL). Quantitative data were described as a mean and standard deviation (SD). Independent samples t test was used to compare the normally distributed ovarian volume, the follicle number and the follicle volume, and the endometrial thickness between PCOS phenotypes. Probability (P) values less than .05 were considered a statistically significant at a confidence interval of 95%. The receiver operating characteristic (ROC) curves of sonographic findings of both groups were done with the calculation of the area under the curve. The optimum threshold values of different sonography parameters with the highest accuracy were selected to differentiate the classic from the nonclassic phenotypes of PCOS with the calculation of the sensitivity and the specificity.
Results
Table 1 shows the sonographic findings used to differentiate phenotypes of PCOS. Figure 1 shows the sonographic findings of classic phenotype of PCOS and Figure 2 shows the sonographic findings of nonclassic phenotype of PCOS. Table 2 and Figure 3 demonstrate the ROC results of the threshold value of sonography parameters used to differentiate classic from nonclassic phenotypes of PCOS.
Sonography Parameters Used to Differentiate Classic From Nonclassic Polycystic Ovarian Syndrome.
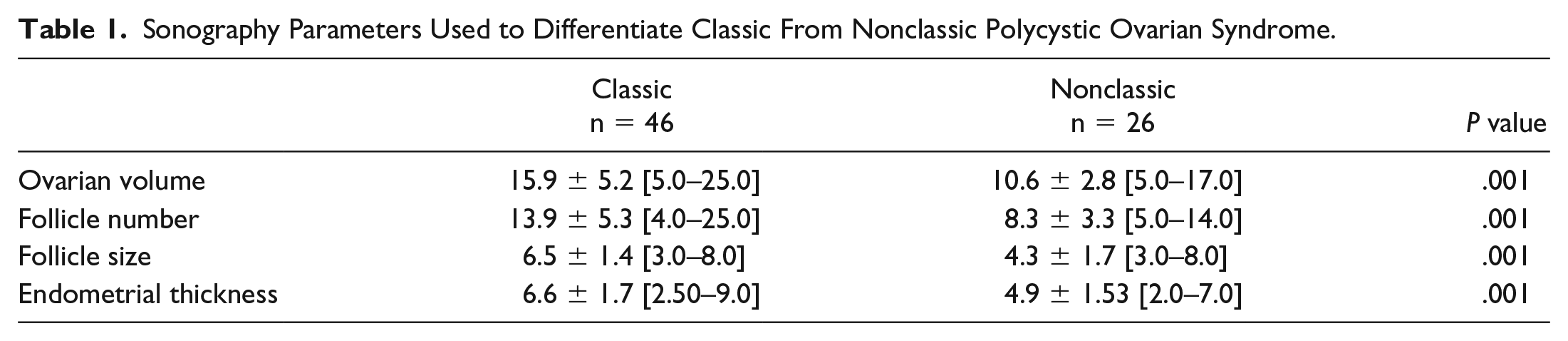
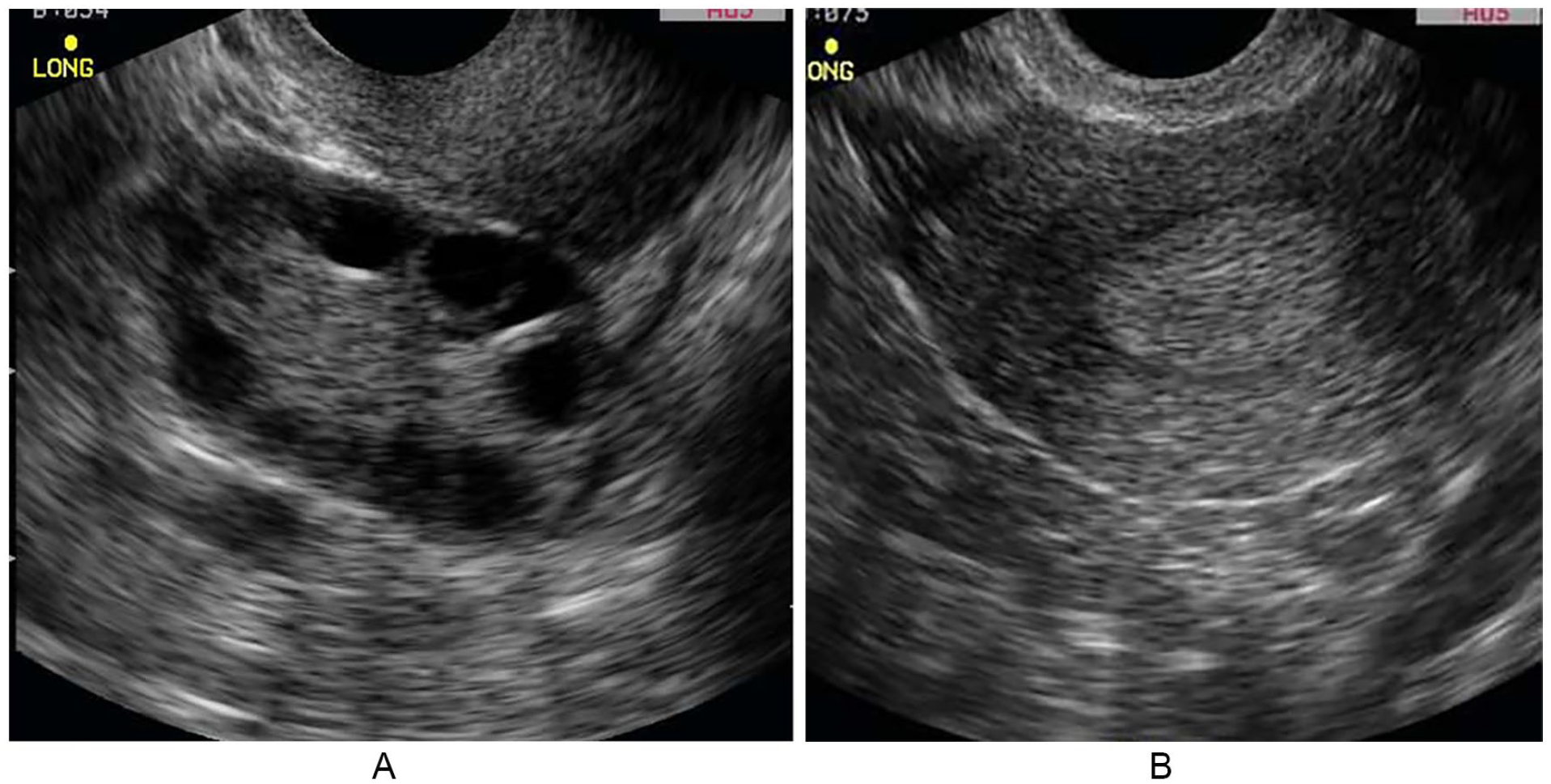
Classic phenotype of PCOS. (A) Transvaginal sonography (TVS) shows few follicles within the ovary with the ovarian volume of 13.5 cm3, the follicles number is 10, and the size of follicle is 4 mm. (B) TVS shows increase endometrial plate thickness in patients with PCOS. PCOS, polycystic ovarian syndrome.
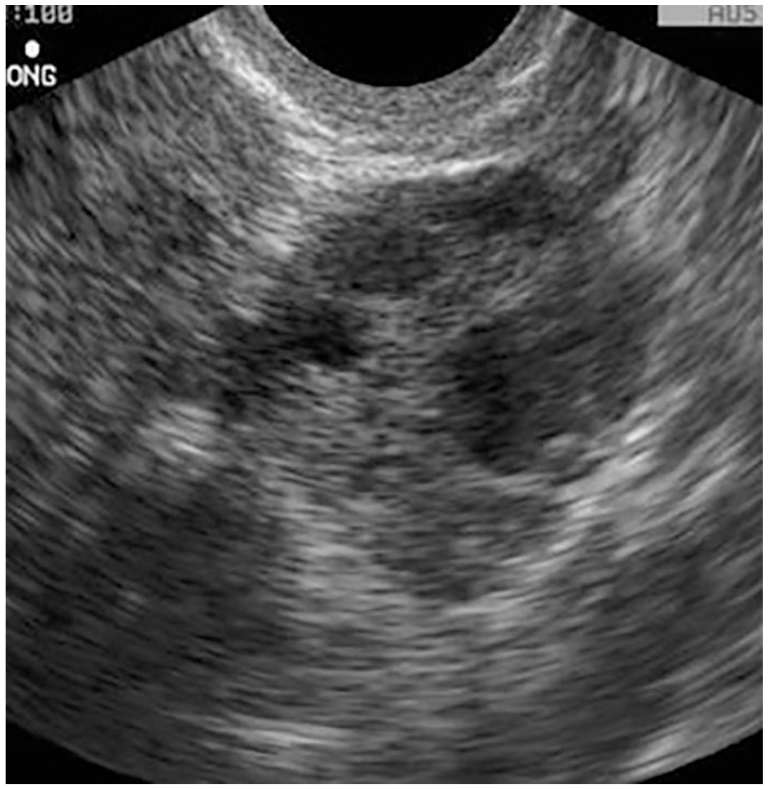
Nonclassic phenotype of polycystic ovarian syndrome. Transvaginal sonography shows multiple follicles within the ovary with the ovarian volume of 8.8 cm3, the follicle number is 5, and the size of the follicle is 5 mm.
Receiver Operating Characteristic Results of Sonography Parameters Used to Differentiate Classic From Nonclassic Polycystic Ovarian Syndrome.
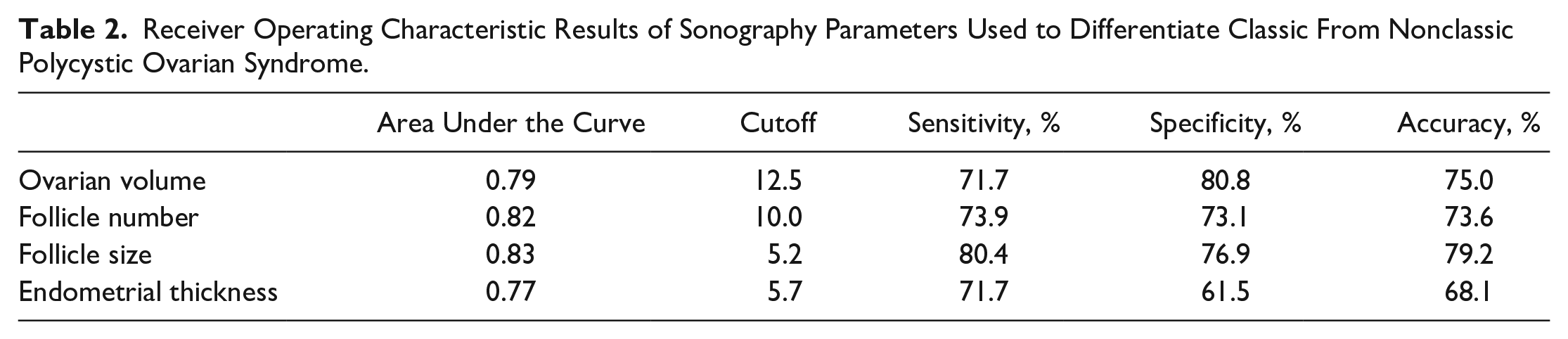
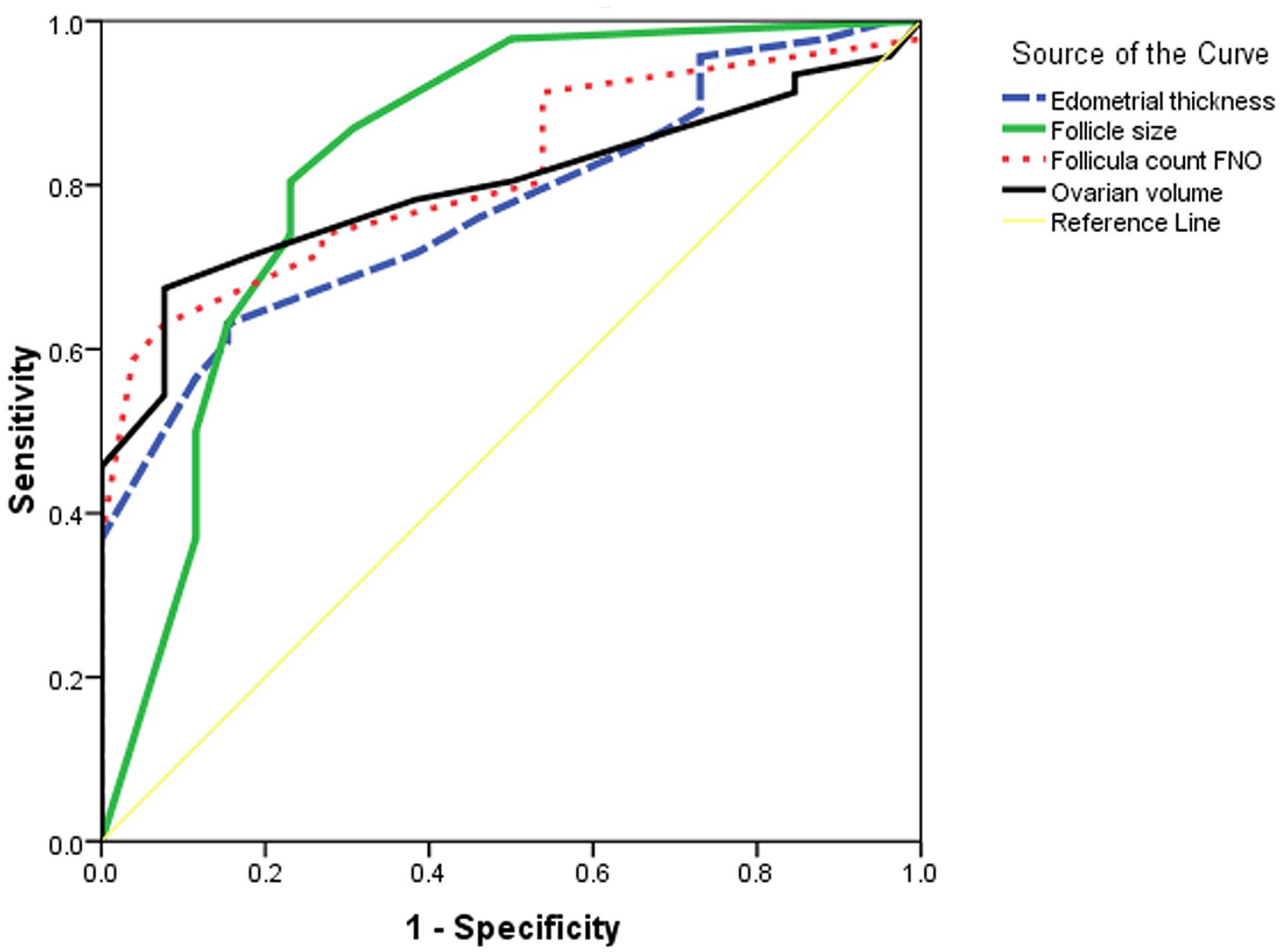
Receiver operating characteristic used to differentiate classic from nonclassic phenotypes of polycystic ovarian syndrome. The threshold value for the ovarian volume, the follicle count, the follicle size, and endometrial thickness used to differentiate the classic from the nonclassic subtypes are 12.5, 10.5, 5.25, and 5.75, respectively, with an area under the curve of 0.79, 0.82, 0.83, 0.77 and an accuracy of 75%, 73.6%,79.2%, 68.1%, respectively.
The mean ovarian volume of the classic phenotype was 15.9 ± 5.2 (5.0–25.0) cm3 and of the nonclassic phenotype was 10.69 ± 2.8 (5.0–17.0) cm3, with a significant difference (P = .001). The selection of (12.5) cm3 as a cutoff point of the ovarian volume used to differentiate classic from nonclassic phenotypes revealed an accuracy of 75%, the sensitivity of 71.7%, and the specificity of 80.8%.
The mean number of the follicles in the classic phenotype was 13.9 ± 5.3 (4.0–25.0) and in the nonclassic phenotypes was 8.35 ± 3.3 (5.0–14.0) with a significant difference (P = .001) between both forms of PCOS. Selection of (10) as a cutoff point of the number of follicles used to differentiate classic from nonclassic phenotypes revealed an accuracy of 73.6%, the sensitivity of 73.9%, and the specificity of 73.1%.
The mean size of the follicles in the classic phenotype was 6.5 ± 1.4 (3.0–8.0) mm and for the nonclassic phenotype was 4.3 ± 1.7 (3.0–8.0) mm with a significant difference (P = .001) between both phenotypes of PCOS. Selection of (5.2) mm as a cutoff point of size of follicles used to differentiate classic from nonclassic phenotypes revealed an accuracy of 79.2%, the sensitivity of 80.4%, and the specificity of 76.9%.
The mean endometrial plate thickness in the classic phenotype was 6.6 ± 1.7 mm (2.5–9.0) and in the nonclassic phenotype was 4.9 ± 1.53 mm (2.0–7.0) with a significant difference (P = .001) between both forms of PCOS. Selection of (5.7) mm as a cutoff point of the number of follicles used to differentiate the classic from the nonclassic phenotypes revealed an accuracy of 68.1%, sensitivity of 71.7%, and specificity of 61.5%.
The serum testosterone was significantly higher (P = .001) in patients with classic (91.80 ± 11.3 ng/dL) than nonclassic (57.6 ± 25.2 ng/dL) phenotypes of PCOS. The serum testosterone was correlated with the ovarian volume (r = .56; P = .91), the follicular size (r = .64; P = .76), the follicular number (r = .57; P = .71), and the endometrial plate thickness (r = .47; P = .50). The BMI was significantly higher (P = .04) in patients with classic (27.7 ± 2.5) than nonclassic (25.66±2.4] phenotypes of PCOS. The BMI was correlated with the ovarian volume (r = .34; P = .001), the follicular size (r = .24; P = .01), the follicular number (r = .33; P = .001) and the endometrial plate thickness (r = .27; P = .007).
Discussion
Polycystic ovarian syndrome has a prevalence of 6.6% in women of the reproductive age and affects four to five million women of the reproductive age in the United States. Although this condition is recognized a common endocrine abnormality in women of the reproductive age, there is still no universal consensus on the diagnostic criteria for PCOS. Moreover, the cause of PCOS remains incompletely understood.3–7 According to the consensus definition, PCOS is present when one or both ovaries demonstrate 12 or more follicles measuring 2 to 9 mm in diameter or the ovarian volume exceeds than 10 cm3. Only one ovary that meets either of these criteria is sufficient to establish the presence of PCOS. 8
The management of PCOS targets the most common symptomatology of the patients as menstrual irregularities, infertility, hirsutism, and acne. The guidelines strongly recommend modification of the lifestyle. Oral contraceptive pills are used as lifestyle first-line treatment for HA and lifestyle menstrual irregularities in patients with PCOS. Clomiphene citrate is the first line of treatment for anovulatory infertility. Exogenous gonadotropins, in vitro fertilization, and ovarian drilling are considered as the second line of management. Inositol stereoisomer is the new drug of choice.1,28–32
Differentiation of classic and nonclassic phenotypes of PCOS has its impact on the treatment planning, prognosis, and different infertility outcomes. Patients with nonclassic phenotype of PCOS have better prognosis, less aggressive form of therapy, and better fertility outcome.1,29–31
In this study, there was a significant difference in the ovarian volume between the classic and the nonclassic phenotypes of PCOS, with an accuracy of 75%. One study reported that nulliparous women with PCOS have a smaller ovarian volume than that ethnic groups and the right ovarian volume is larger than the left ovarian volume. 17 The ovarian volume is one of the diagnostic criteria for PCOS, and the consensus definition of PCOS includes an ovarian volume more than 10 cm.1–4
In this study, there was a significant difference in the number and size of the follicles between the classic and the nonclassic phenotypes of PCOS, with the most predictor is the follicular size with an accuracy of 79%. A previous study reported that PCOS is diagnosed using 12 or more follicles per ovary and is one of the diagnostic criteria of PCOS. However, PCOS is also encountered in normal-ovulatory women. 18 Some metabolic changes of the patients as insulin resistance could be associated with a specific sonographic pattern in patients with PCOS differ from noninsulin dependents. 19
In this study, there was a significant difference in the endometrial plate thickness between the classic and the nonclassic phenotypes of PCOS. A previous study reported that several related endometrial pathologies might be associated with patients with PCOS. These changes vary from increased the endometrial thickness, endometrial hyperplasia, and the rare occurrence of endometrial cancer. 20
In this study, the serum testosterone level and BMI were significantly different between patients with classic and nonclassic phenotypes of PCOS. These parameters are well correlated with sonographic parameters, especially the follicle size. Previous studies reported that serum testosterone and BMI are important parameters that help to differentiate between both phenotypes of PCOS.1–5
The merits of sonography are a noninvasive procedure that can be repeated, safe, low cost, and can be easy to perform at the bedside. Limitations of sonography are operator-dependent and the parameters depend on the ultrasound equipment.35–39
There are a few limitations of this study. First, this study includes a small number of patients. Multicenter studies on a large number of patients improve the results. Second, this study used morphological findings of the sonography only for analysis. Further studies using power Duplex, sonographic elastography, MRI, diffusion tensor imaging, and arterial spin labeling40–52 could improve results. Third, this study looked at the participants as classic versus nonclassic phenotypes of PCOS. Future studies attempt to differentiate between the four phenotypes of PCOS on large number of patients. Fourth, the sonographic examinations were conducted by only two radiologists.
Conclusion
This research concluded that sonography could differentiate between the classic PCOS versus the nonclassic phenotypes of PCOS, with this cohort of patients. These results have future implications for improved patient, diagnostics, prognosis, and management.
Footnotes
Ethics Approval
All procedures followed were following the ethical standards of the responsible committee on human experimentation (Institutional and National) and with the Helsinki Declaration of 1964 and later versions.
Informed Consent
Informed consent was waived because this is a retrospective study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because this study is on humans.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial registration
Trial registration was not applied due to study type.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
