Abstract
Encephaloceles are considered neural tube defects, but their exact cause is unknown. The outcome is dismal, and essential management and counseling are needed for patients. Two-dimensional and three-dimensional sonography can be used to detect encephaloceles as early as 11 weeks, assist in treatment planning, and improve patient care. This case report presents an occipital encephalocele diagnosed by sonography and followed until delivery.
Encephaloceles are rare birth defects where a cranial bone opening results in herniation of neuronal tissue outside of the cranial vault. Encephaloceles are thought to be a form of neural tube defect, but the exact cause is unknown. Encephaloceles are typically congenital but can be secondary due to trauma or injury and are classified according to their location. 1 The size of encephaloceles vary greatly, and most are covered by skin or a thick membrane making them closed defects that are unlikely to be detected by prenatal serum screening. 2
Detection of encephaloceles can be made with the help of two-dimensional (2D) and three-dimensional (3D) sonography as early as 11 weeks, but most cases are diagnosed in the second trimester. 3 The 3D sonography was first used to diagnose encephaloceles in 2006 and since has been shown to contribute to early detection, assist with prenatal diagnosis of associated abnormalities, and help guide genetic consultation.3,4 Prognosis, treatment, and management of encephalocele malformations depend on the size of the sac, contents, and other associated findings. 5 Ultimately, the outlook is dismal. This case report will describe the presentation of an isolated occipital encephalocele diagnosed using 2D and 3D sonography.
Case Study
A woman in her early 30s, gravida 5, para 3 (G5, P3,0,1,3), presented for a routine anatomical sonogram with no prior sonography examinations, for this pregnancy. Formulated by her last menstrual period, the fetus was estimated to be 23 weeks 3 days gestation. The patient was not taking prenatal vitamins or supplementary folic acid and was positive for syphilis, which she was subsequently treated. A comprehensive metabolic panel from a year prior showed a glucose level of 230 mg/dL (normal range is 70–99) and a current 1-hour glucose screen value of 303 (normal range is 70–140). An A1C level was drawn with the results being 8 therefore, underlying, undiagnosed diabetes mellitus was suspected (normal level is below 5.7).
A detailed sonographic evaluation was performed with 2D and 3D sonography using a GE Voluson E8 (GE Medical Systems, Milwaukee WI) with a 2 to 8 MHz curvilinear-array transducer and Rab6-D 3D probe. The sonogram demonstrated a viable fetus measuring 19 weeks 3 days and <1% based on estimated fetal weight (EFW) for the gestation age that was expected by Last Menstrual Period (LMP) at 405 g. A large, high occipital posterior bony defect was visualized with a large, protruding mass consistent with an encephalocele (see Figure 1) measuring 4.5 cm in the largest dimension. The cerebellum was hypoplastic, measuring 1.55 cm, and appeared to be contained within the defect (see Figure 2) along with the lateral ventricles and choroid plexus. The head was measuring small at <1% based on EFW for gestational age, consistent with microcephaly (see Figure 3). A pericardial effusion measuring 3.2 mm was seen. All other anatomy measured within normal limits and had normal sonographic appearances.
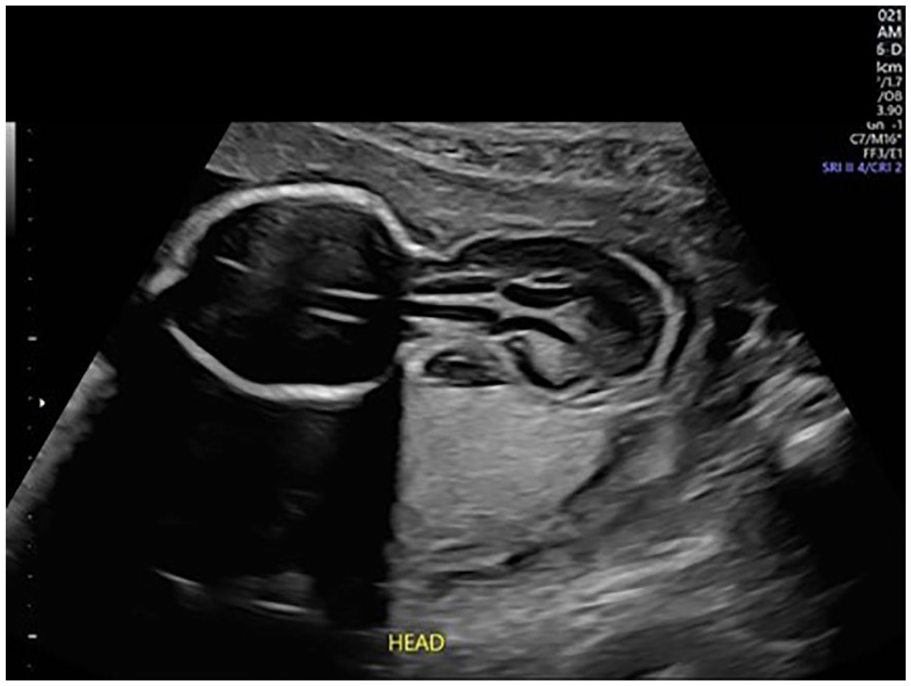
A transverse sonographic image through the fetal head demonstrating a large posterior defect with herniated brain contents.
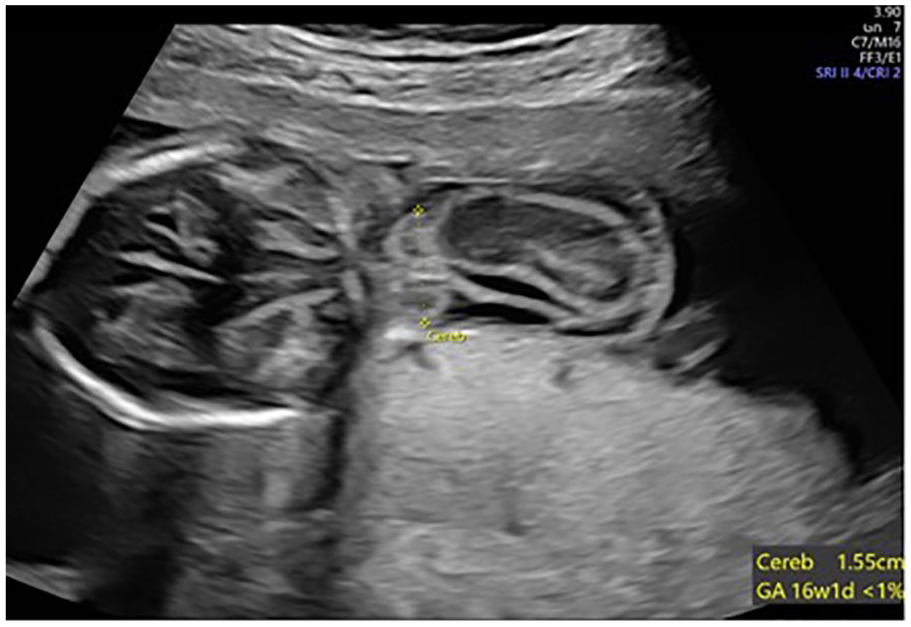
A transverse sonographic image through the fetal head demonstrating the cerebellum measuring <1% contained within the posterior encephalocele.
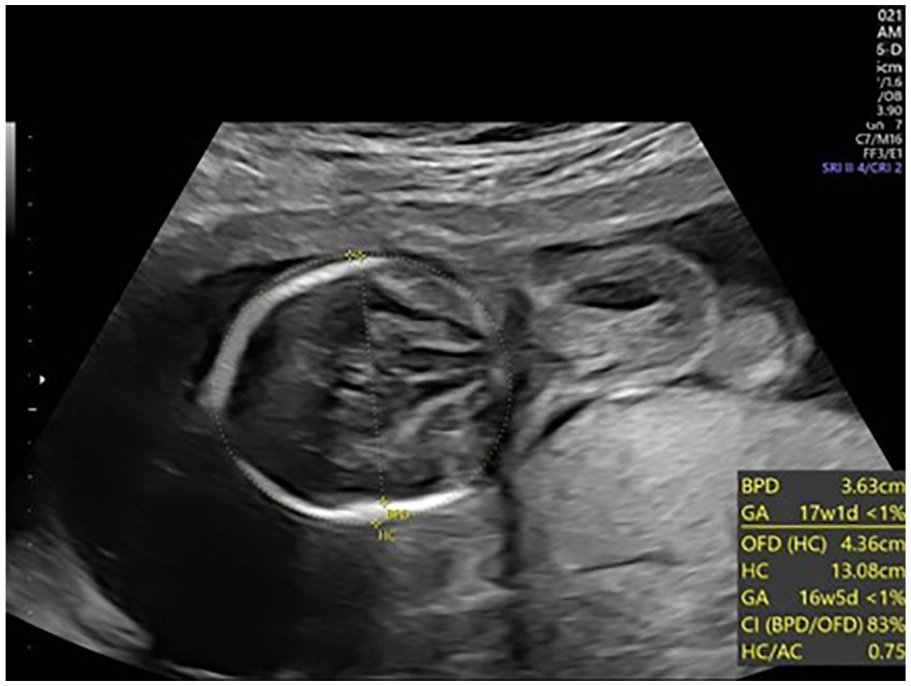
A transverse sonographic image through the fetal head demonstrating the Biparietal Diameter (BPD) and Head Circumference (HC) measuring <1%, consistent with microcephaly.
The patient elected to pursue an amniocentesis which was performed 1 week later at 24 weeks 3 days gestation. Prenatal interphase fluorescence in situ hybridization (FISH) analysis was completed on the amniotic fluid, and the results showed a female fetus (46XX) with no evidence of aneuploidy. Genetic testing revealed a normal chromosomal microarray. Alpha-fetoprotein (AFP) within the amniotic fluid was 3412 ng/mL, a negative result for an open neural tube defect. Acetylocholinesterase and fetal hemoglobin were tested for within the amniotic fluid, but neither were detected.
A growth evaluation, biophysical profile (BPP), and 3D sonogram were performed at 28 weeks 3 days gestation, which demonstrated similar findings to the original anatomy examination. The BPP was scored as an 8/8. An EFW of <2% for gestation age or 932 g was obtained. The encephalocele containing brain tissue (see Figures 4 and 5) and the pericardial effusion were again documented.
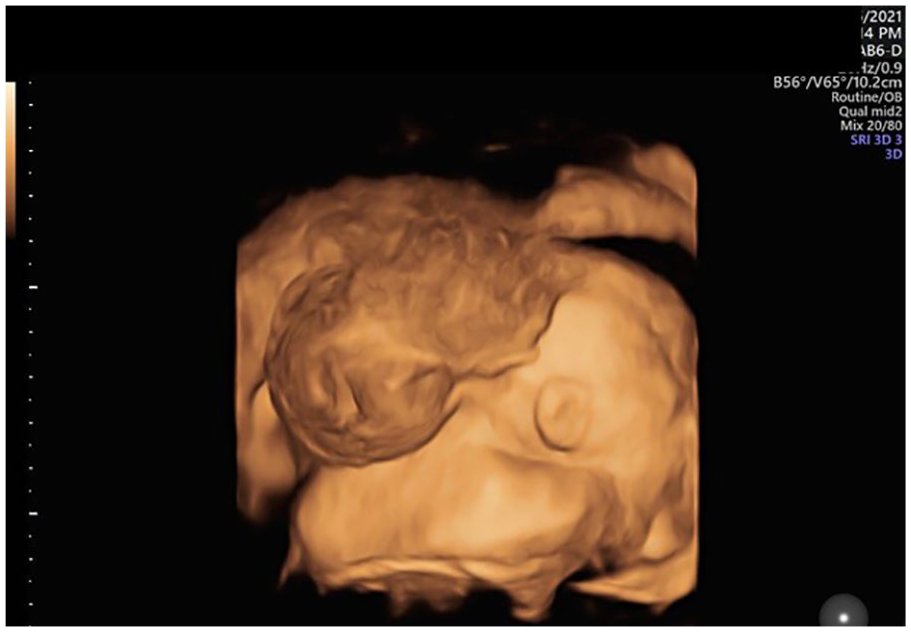
A 3D sonographic image of the posterior head demonstrating a large encephalocele.
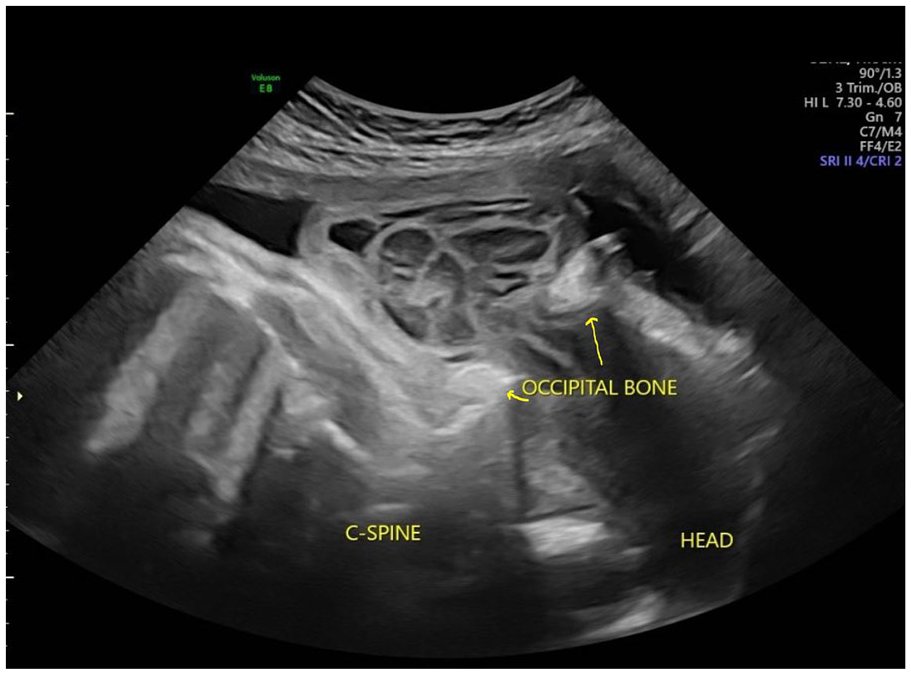
A sagittal sonographic image of the posterior head demonstrating a large encephalocele through the occipital bone defect (yellow arrows).
A fetal magnetic resonance imaging (MRI) was performed at 32 weeks gestation which demonstrated a singleton fetus in the cephalic position with the fetal back toward the right side of the uterus. An encephalocele in the occipital region measuring 7 × 3.8 × 6.4 cm3 was visualized, as identified on prior sonograms. The encephalocele contained cystic structures believed to be the trigones of the lateral ventricles—T2 hypointensities along the margins of the cystic structures, likely representing subependymal gray-matter heterotopia (normal neurons in an abnormal location). The superior sagittal sinus was also noted extending into the encephalocele. The basal ganglia, thalamus, and brainstem were not well delineated on the examination, but the cerebellum was visualized within the encephalocele and measured small for gestational age. The residual intracranial brain was dysmorphic without well-defined ventricles and showed significant volume loss due to the herniation/protrusion of intracranial contents (the meninges, brain, and part of the ventricles) through the defect into the encephalocele. Within the residual cranium, sulci and fissures were seen coursing in the anterior-posterior (AP) direction along with an elongated fluid-filled structure on the right, possibly representing a rudimentary ventricle.
The patient continued with twice-weekly surveillance including non-stress tests with her primary obstetric provider BPPs with a maternal fetal medicine provider. Polyhydramnios was later diagnosed during the BPP sonograms, as well as a depressed frontal bone (see Figure 6). The patient consulted with pediatric neurosurgery and neurology. A vaginal delivery with palliative care for the neonate was planned. A viable female infant was born at 37 weeks 2 days gestation, weighing 7 pounds 1.6 ounces with a length of 18.5 inches. APGAR scores were not recorded. The neonate was intermittently dusky with extensive periods of breath holding and apnea. Morphine and Ativan (Lorazepam) were administered every 3 hours for the breathing issues, restlessness, and discomfort. The patient was discharged after 2 days to hospice for home palliative care for the infant. The primary terminal diagnosis for hospice was a giant encephalocele with brain exposure with excessive periods of apnea. Due to the terminal diagnoses, the parents elected to sign a do-not-resuscitate (DNR) order. The prognosis was dependent on whether the infant would pass from lack of nutrition or breathing issues. If death was due to breathing issues, prognosis was 24 to 48 hours. If death was due to lack of nutrition, since the infant was not able to suck or feed, prognosis could extend up to 3 weeks. The infant later died at her home on the morning of day 3.
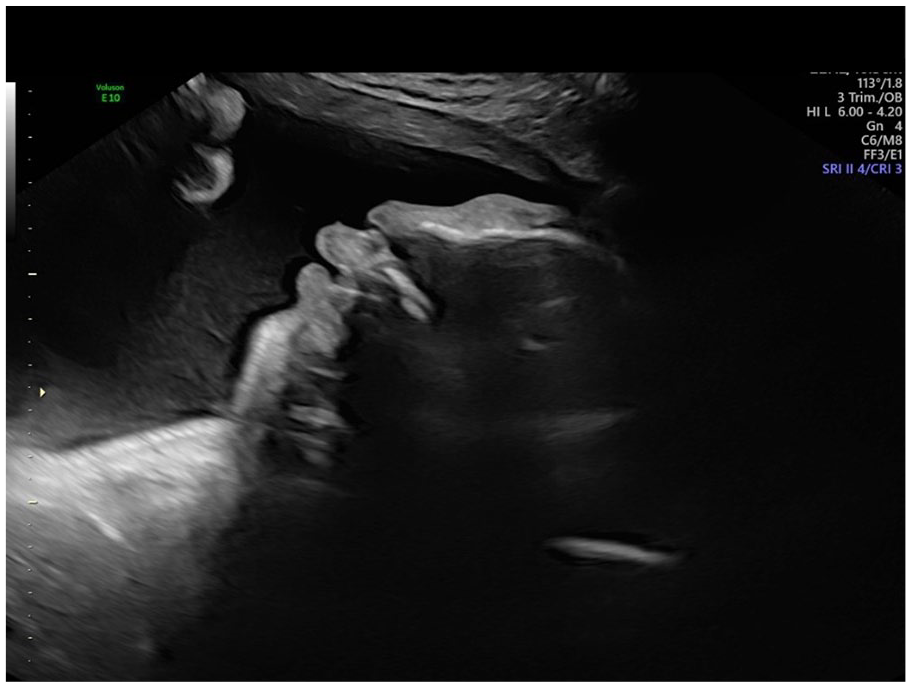
A sagittal profile sonographic image obtained in the late third trimester, demonstrating a depressed frontal bone.
Discussion
Encephaloceles are thought to be a neural tube defect and are characterized by a pedunculated lesion containing brain tissue that protrudes through a defect in the dura mater and cranium. 2 Cephaloceles is the broad term used to describe any protrusion of intracranial contents through a defect in the skull. 2 Encephaloceles are the third most common neural tube defect after anencephaly and spina bifida and are found in approximately 1 in every 10 500 babies born in the United States. 6 However, the true incidence rate is higher because many cases result in pregnancy termination. Most encephaloceles are congenital or primary, but some can be acquired secondary to injury or trauma. 1 Congenital encephaloceles are classified according to their location in either the occipital, frontal, temporal, or parietal regions of the head. The most common anatomical site depends on patients’ geographical location; occipital encephaloceles are most common in the Western hemisphere while frontal locations are more common in Southern Asia, particularly Thailand.3,7
Neural tube defects are commonly grouped together such as anencephaly, spina bifida, and encephaloceles. 8 Neuralization, the process in which the neural tube forms, occurs by days 22 to 23 with fusion being complete within 3 to 4 days. The neural tube begins to fuse in the cervical area and then proceeds more caudally. The rostral neuropore closes on day 25 with the caudal neuropore closing 2 days later. The failure of the rostral neuropore to fuse during the fourth week, between days 25 and 29, causes neural tube defects. 9 Although a defective closure of the neural tube is thought to cause encephaloceles, the exact mechanism is uncertain with some arguing that encephaloceles occur post neural tube closure.8,10 Rolo et al 10 found that non-syndromic cases of encephaloceles occur later in pathogenesis, after the neural tube has fully closed. A disruption of the surface ectoderm occurs and allows brain matter to herniate through a defect. The researchers concluded pathogenesis occurs later with encephaloceles than with open neural tube defects and also established that both can have interchangeable genetic causation. 10
Genetic and environmental factors have been linked to encephaloceles.3,11,12 TORCH infections (toxoplasma, rubella, cytomegalovirus, herpes simplex virus), consanguineous marriages, and family history have all been documented contributing factors. 13 Similar to other neural tube defects, risk factors may include diabetes, obesity, hyperthermia, and folic acid insufficiency. 14 Pregnant women exposed to Zika virus also are at an increased risk for abnormal fetal brain development including encephaloceles. 15 Cases may be isolated, but 60% of encephaloceles are associated with other malformations such as a cystic hygroma, a single umbilical artery, amniotic band, Dandy-Walker defect, Arnold-Chiari defect, and hydrocephalus. 3 More than 30 syndromes have been associated with encephaloceles which include aberrant tissue band syndrome, Knobloch syndrome, Warfarin syndrome, and other chromosomal anomalies. 11 The most recognized syndrome associated with an encephalocele is Meckel-Gruber syndrome, which is characterized by large polycystic kidneys, postaxial polydactyly, and an occipital encephalocele.11,16 Chiari III malformation is a congenital hypoplastic disease of the hindbrain associated with a low occipital/high cervical encephalocele and myelomeningocele. 2 The location of the encephalocele and myelomeningocele should aid in the diagnosis of Chiari III versus an isolated encephalocele.
The pregnancy in this case study was complicated by maternal diabetes and syphilis. The risks of congenital anomalies of pregnancies complicated with diabetes is two to five times the risk of nondiabetic pregnancies, with half of the anomalies being cardiac. 17 Unplanned pregnancies with elevated hemoglobin A1c (HbA1c) levels within the first trimester have higher rates of congenital anomalies. 17 In this case, the patient’s HbA1c level was 8.0%, suggesting pre-existing, undiagnosed type II diabetes mellitus. Current research has shown that Pax3, a gene that is required for normal neural tube closure, is greatly reduced when maternal diabetes is present. 18 A co-existing factor to consider in this case study was the diagnosis of syphilis at the patient’s routine anatomical ultrasound, the first prenatal care of this pregnancy. Maternal infections, including syphilis, have been shown to be capable of causing congenital malformation of the central nervous system structures.8,13
The sonographic diagnosis was straightforward. An encephalocele can be suspected when a paracranial mass with a gyral pattern continuous with the brain and a bony defect of the cranial bones are identified on sonography.2,19 Encephaloceles impair the normal circulation of cerebrospinal fluid (CSF) and therefore are commonly associated with hydrocephalus. Large encephaloceles may be associated with microcephaly. 2 Sac size, suboptimal fetal head position, or location of the defect can limit sonographic visualization.2,19 However, sonography has been estimated to detect approximately 80% of all encephaloceles. 20 Most sonographic diagnoses occur in the second trimester, with some cases being diagnosed as early as 11 weeks. 3
The sonographic evaluation of encephaloceles should include the location of the calvarium defect, identification of internal contents, and associated findings such as hydrocephalus. Demonstration of an intact fetal cranium during the late first trimester and early second trimester is important and can help exclude encephaloceles. The bony defect of an encephalocele can best be seen in an axial head image where there is a loss of normal, hyperechoic cranial bones (see Figure 1). Sagittal imaging planes of the fetal head may also assist in the diagnosis. 21 A 3D sonography can also aid in the diagnosis and provide additional information to patients, thus improving overall patient care.3,22
The value of fetal MRI is widely accepted and considered superior for central nervous system anomalies detected by ultrasound. Fetal MRI has higher contrast resolution than ultrasound and can provide high-resolution images in cases where sonography may be limited, such as cases with low amniotic fluid or artifacts from ossified bones. 23 Fetal MRI is limited by fetal motion and small fetal size; therefore, it is not typically performed before 22 gestational weeks. 23 Fetal MRI provides superior detail of encephalocele contents and location, which contributes to safe delivery planning.23,24
Most encephaloceles are covered with skin or a thin membrane, resulting in a normal second trimester prenatal screen.2,25 A maternal serum alpha-fetoprotein (MSAFP) screen is the earliest non-invasive biochemical test to provide information about the developing fetal neural axis. Produced by the yolk sac, gastrointestinal tract, and liver, MSAFP levels rise until 30 weeks and thereafter start to decrease. In the case of an open neural tube defect, AFP from the fetal circulation leaks into the amniotic fluid, thus increasing the MSAFP levels.26,27 An elevation of MSAFP 2.0 to 2.5 times higher than the multiples of the median (MoM) indicates a possible exposure of spinal or brain tissues to amniotic fluid. 2 Normal MSAFP levels are seen in 97% of encephalocele cases.25,27 With a normal prenatal serum screen, sonography may be the first detection method for finding such severe defects.
An amniocentesis is often performed to determine the cause responsible for any elevation of MSAFP levels. Levels of AFP, acetylcholinesterase (AChE), and a genetic microarray test may be performed on the fluid as well. AChE is found in tissues such as red blood cells, muscles, and neural tissue, but is not normally found in amniotic fluid. Levels of AFP and AChE are elevated with open neural tube defects but can be normal with closed defects like the encephalocele in this case study. 28
The prognosis of a fetus with an encephalocele varies depending on the amount of brain tissue present within the sac, defect location, and accompanying findings. 29 The presence of intracranial abnormalities in addition to an encephalocele is a significant predictor for developmental delay. 30 In addition, microcephaly and brain tissue within the sac are associated with poorer outcomes. In a study performed by the Children’s Hospital of Los Angeles over the course of 18 years, the presence of additional cranial anomalies, hydrocephalus, microcephaly, and a larger-sized encephalocele were found to be the most common factors associated with significant neurologic deficits. 31 Only 50% of fetuses with an encephalocele will survive until birth, and most deaths occur in fetuses with an occipital encephalocele. 32 Half of those that survive delivery will experience neurodevelopmental impairment or drug-resistant seizures. 33 Morbidity and mortality rates for occipital encephaloceles vary according to geographical location but are improving given the advanced surgical techniques and postoperative care. 33
The only treatment for occipital encephaloceles is surgery to remove the sac and put the brain contents back within the skull. Most surgical techniques involve surgery shortly after birth or within the first 3 months. New techniques are being trialed with fetal surgery. 34 Surgical repair of occipital encephaloceles is a difficult task for both neurosurgeons and anesthesiologists. The neurosurgeons may face problems with fetal intraoperative blood loss, intraoperative hypothermia, and prolonged anesthesia, while the anesthesiologist may face maternal problems with tracheal intubation, excess blood loss, and cardiorespiratory complications. 35 The postnatal surgical technique involves excision and repair of the herniated brain contents with the removal of all the nonviable brain tissue that is within the sac. Ventriculoperitoneal (VP) shunts are placed when hydrocephalus is present. 33 If microcephaly is present, the cranial cavity must be expanded prior to placing viable brain contents back within the skull. 33 Complications following postnatal surgery of an occipital encephalocele may include postoperative meningitis, CSF leakage, hydrocephalus, wound infection, wound dehiscence, or possibly death. 35 Cavalheiro et al 34 studied occipital encephalocele patients who underwent fetal correction. The authors found that neurocognitive development was improved, and microcephaly was reversed in fetuses that underwent fetal repair. Compared with those undergoing surgery after birth, fetal surgery was also associated with a better cognitive outcome. 34
Conclusion
Encephaloceles are named according to their cranial location and associated with multiple syndromes. Occipital encephaloceles have the worst prognosis and are the most common type found in the Western hemisphere. Care should be taken to determine the sac contents and other associated findings. Surgical correction is the only treatment available, with fetal encephalocele surgery showing promise to improve patient outcomes. Both 2D and 3D sonography play an important role in the diagnosis, treatment planning, and patient counseling of encephaloceles. Given the high incidence of encephalocele cases with normal AFP levels, sonography is often the first tool used to find and diagnose this pathology. This case study described a high occipital encephalocele containing a large amount of brain contents that was diagnosed with sonography and confirmed with fetal MRI. The case was further complicated by undiagnosed maternal diabetes mellitus and syphilis, both conditions that are known to cause neural tube defects.
Footnotes
Informed Consent
Written informed consent was obtained from the patient for the publication of the case report and the accompanying images.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
