Abstract
Malignant melanoma is a cellular cancer that produces pigmentation of the skin. The tendency toward melanoma may be inherited, and risk factors are increased with overexposure to the sun and ultraviolet radiation. Melanomas commonly present as a dark, asymmetrical, mole-like spot that spreads, with an irregular border. It is uncommon to find a melanoma in the auricular regions and even rarer for it to be a primary malignancy of the auricles. Utilizing sonography to evaluate melanoma lesions could serve as a prognostic indicator, regarding Breslow’s depth, an aide in staging, as well as surgical planning. However, utilizing multiple diagnostic imaging modalities is essential in the proper evaluation and staging of a melanoma. Currently there are revolutionary, effective systemic therapies available for patients with a metastatic melanoma. Current therapies are focused on immunotherapy and checkpoint inhibitors. These treatment options provide an opportunity for selected metastatic melanoma patients to achieve healthy long-term success.
A cancerous cell that produces pigment in the basal layer of the skin, a melanocyte, is referred to as cutaneous melanoma (CM) or malignant melanoma. 1 The appearance of a melanoma is commonly found to have an asymmetrical, dark mole-like resemblance with irregular borders. 1 Environmental exposure to ultraviolet radiation (UVR), genetic defects, and a combination of both are known risk factors. Fair-skinned, red- or blond-haired individuals, as well as individuals with one or more first-degree relatives are at higher risk for a melanoma. 1 Exposure to UVR is one of the highest contributing risk factors to the development of a melanoma, as well as one of the most modifiable. 1 Even with greater awareness between the correlation of UVR overexposure and melanoma, predictions of new cases continue. It is believed that more new cases will be recorded, due to Americans developing skin cancer during their lifetime. 2 In the United States, melanoma is ranked as the sixth most common fatal cancer. 2 This also places melanoma as the cause for six of every seven skin cancer–related deaths. 2
Case Report
An 81-year-old Caucasian male presented with a confirmed diagnosis of malignant melanoma of the left auricular helix. He was evaluated as a new patient to the Head & Neck Neuro Interventional Radiology Ultrasound (NIRUS) department. The patient’s history included excessive sun exposure and previously smoking one pack per day, for 15 years. He had quit smoking 28 years ago. The patient also had a history of hypertension, hyperlipidemia, diabetes, congestive heart failure, chronic obstructive pulmonary disease, and a squamous cell carcinoma, of the left chest. He was able to confirm having no known family history of melanoma or skin cancers. Melanoma of the left ear was confirmed via a punch biopsy at the referring facility, which had occurred 30 days prior to arrival. Reviewing his initial clinical presentation, prior to the punch biopsy, it was noted that the patient had an ulcerated, scabbed over, and crusty lesion of the left superior helix, as well as focal auricular thickening (Figure 1A). The patient stated that 5 months earlier, this lesion started out as a small red bump. No associated pruritus, discomfort, or pain was appreciated. Clinical evaluation was performed by the melanoma clinic at the hospital prior to sonographic evaluation at NIRUS. In that examination, they found an approximately 2-mm crusted, dark, asymmetrical lesion of the left superior helix (Figure 1B). Extensive auricular thickness and roughness to the skin, which extended toward the helical tail, was observed.
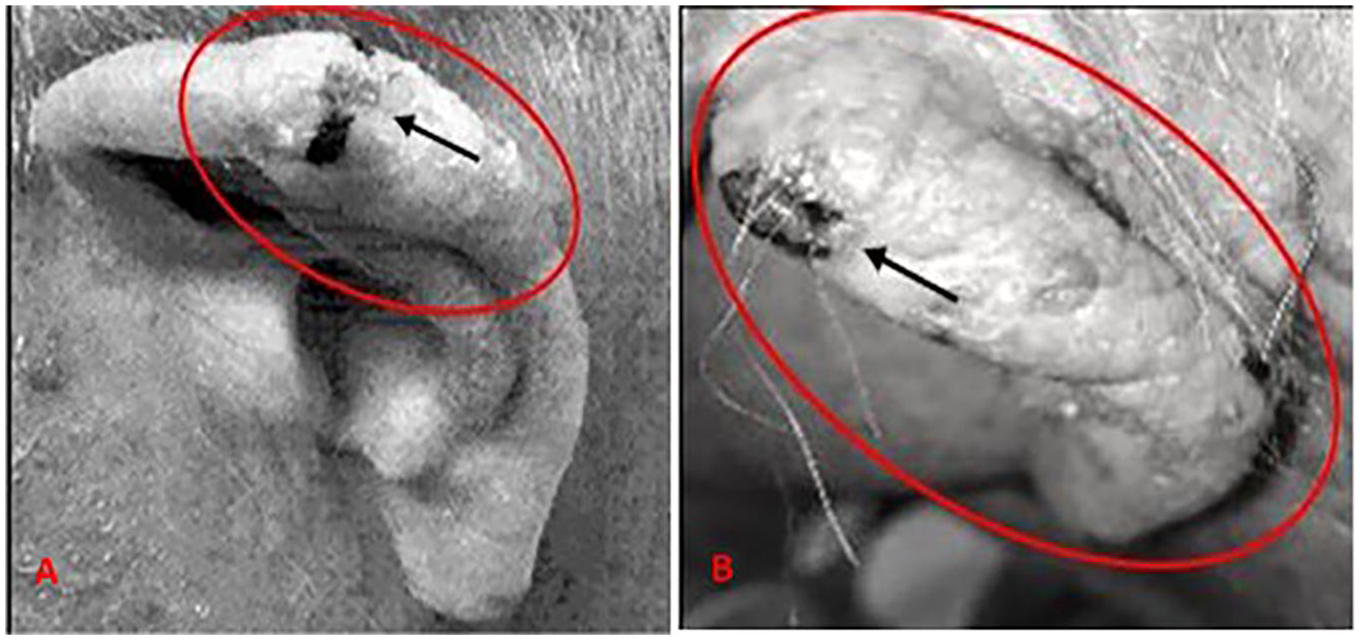
(A) Initial presentation: focal area of thickened skin, with ulcerated lesion (black arrow). (B) In-house presentation (30-day difference): compare extent of diffuse thickened skin of the entire auricle with (A). The black arrow demonstrates the ulcerated lesion used as a focal point for ultrasound imaging.
Multiple diagnostic evaluations were ordered to evaluate the extent of the patient’s disease, as well as to gauge possible metastases. A positron emission tomography (PET) examination was the first diagnostic test completed, postdiagnosis. As expected, there was increased 18F-fluorodeoxyglucose activation, at the level of the left helix. Additional diagnostic imaging included a nuclear medicine lymphoscintigraphy (lymphatic mapping) to identify the sentinel lymph nodes (SLNs; Figure 2A). This examination demonstrated the left auricular thickening as compared with the right (Figure 2B). A contrast computed tomogram (CT) for the soft tissue of the neck was completed. The CT results demonstrated no evidence of metastases to the cervical lymph nodes.
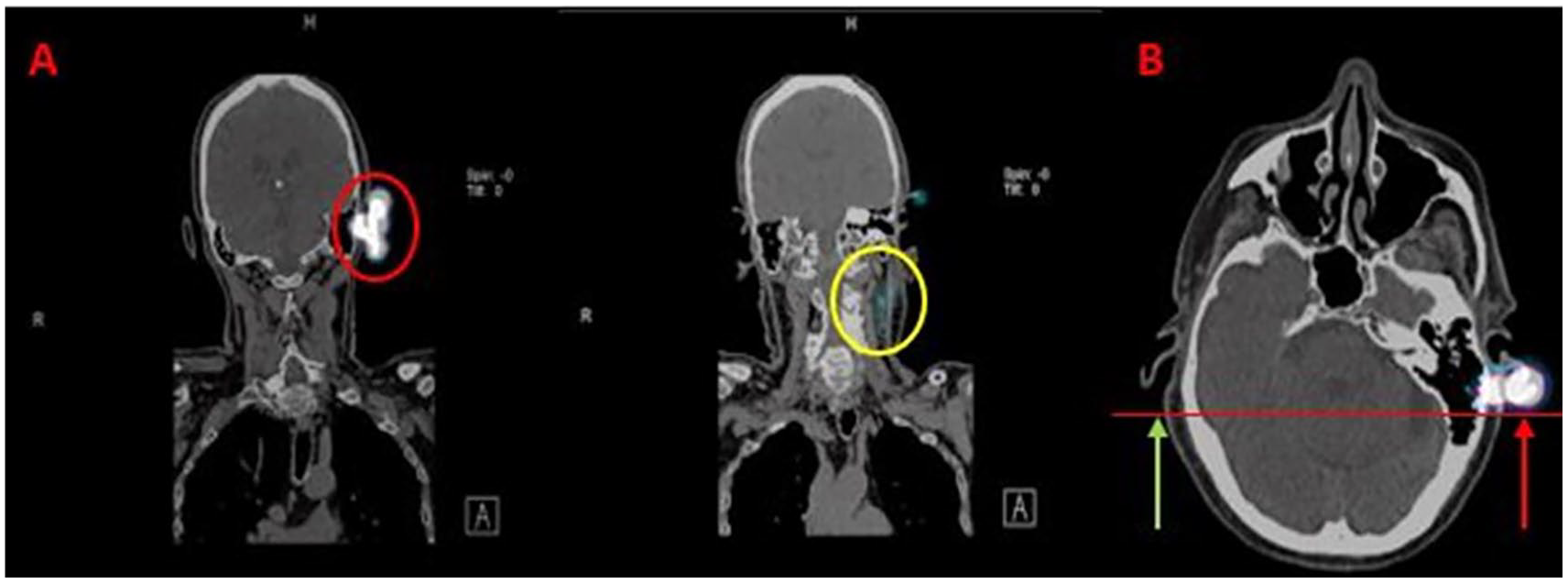
(A) Nuclear medicine lymphoscintigraphy (lymphatic mapping for sentinel lymph node localization). Red circle indicates auricular lesion. Yellow circle indicates sentinel lymph node identification. (B) Transverse view demonstrating thickened helix of the left ear (red arrow) as compared with the normal right ear (green arrow).
A sonogram of the head and neck, to evaluate for any cervical lymphadenopathy, was ordered. The gray-scale sonogram was completed with an Arietta 850 ultrasound unit (Hitachi Aloka) using a 12-2 MHz linear probe transducer. The sonographic protocol included a thorough evaluation of bilateral, lateral, and posterolateral neck, as well as the salivary glands, postauricular, suboccipital, central neck, and suprasternal areas. The sonographic evaluation started from the right supraclavicular inferior neck and included static images that documented, in both planes, up to the right inferior, mid, and superior neck. The examination was conducted with a focus on evaluating each visualized lymph node, including adjacent tissue. The right posterolateral neck was then evaluated in the same manner, from superior posterior neck documenting down to the posterior inferior neck. Evaluation of the right submandibular gland, right parotid gland (including superficial and deep parotid tissue, starting with transducer placement adjacent to the tragus), postauricular, and suboccipital areas followed. The submental, prelaryngeal, pretracheal, midline neck, and suprasternal regions were also evaluated prior to proceeding with repeating the scanning protocol on the left neck.
The patient history was reviewed and evaluated by the ultrasonographer prior to the examination. Being that the surface of the auricular lesion was raised and easily visualized, after following the standard sonographic protocol for head and neck melanoma, the left ear was evaluated. The intention was to evaluate if the lesion could adequately be visualized with sonography. To make better contact with the superficial auricular surface, a generous amount of acoustic gel was applied to the transducer, as an acoustic standoff, and light pressure was applied. This was done in an effort to not impede the integrity of the auricular tissue, lesion, or vascular flow within the lesion. A superoanterior approach of the auricular helix, at the level of the triangular fossa and antihelix crura, was completed in both transverse and longitudinal planes (see Figure 3). Power Doppler was utilized, as it is more sensitive in its parameters to detect slow vascular flow. It was an effective diagnostic tool used to detect possible signs of malignancy within this lesion (see Figure 4). The histological findings were a lesion thickness/depth of 6.0 mm. This finding correlated with the exact anterior/posterior depth measurement of 0.6 cm obtained during the sonographic evaluation. This finding supported the use of sonography as a prognostic indicator of Breslow’s depth in melanoma lesions. The final sonographic report indicated no demonstrated parotid, occipital, superficial, or deep cervical lymphadenopathy detected.
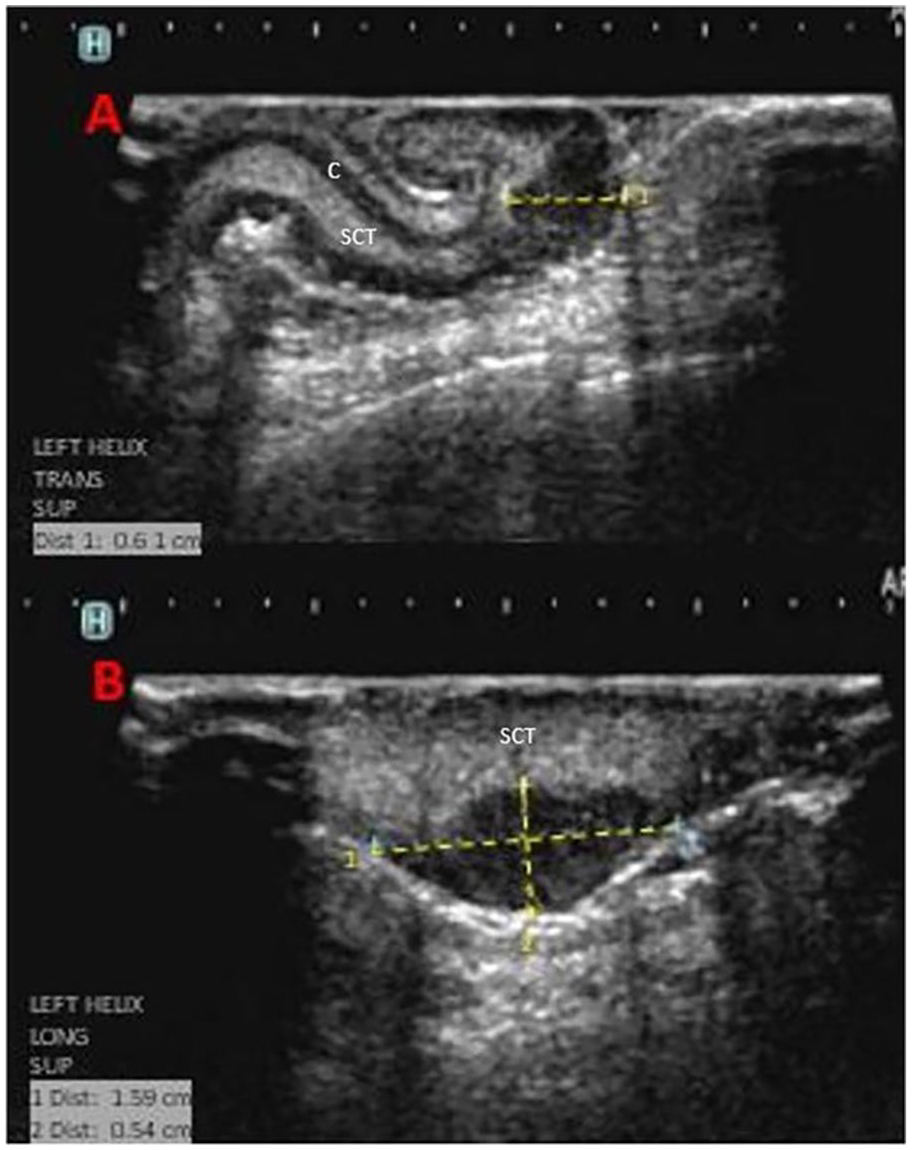
B-mode ultrasound images. (A) Transverse view of Clark level V (6-mm Breslow thickness) invasive melanoma of the left superior helix = 0.6 cm. (B) Sagittal view of the same lesion = 1.6 × 0.5 cm. C, cartilage; SCT, subcutaneous tissue
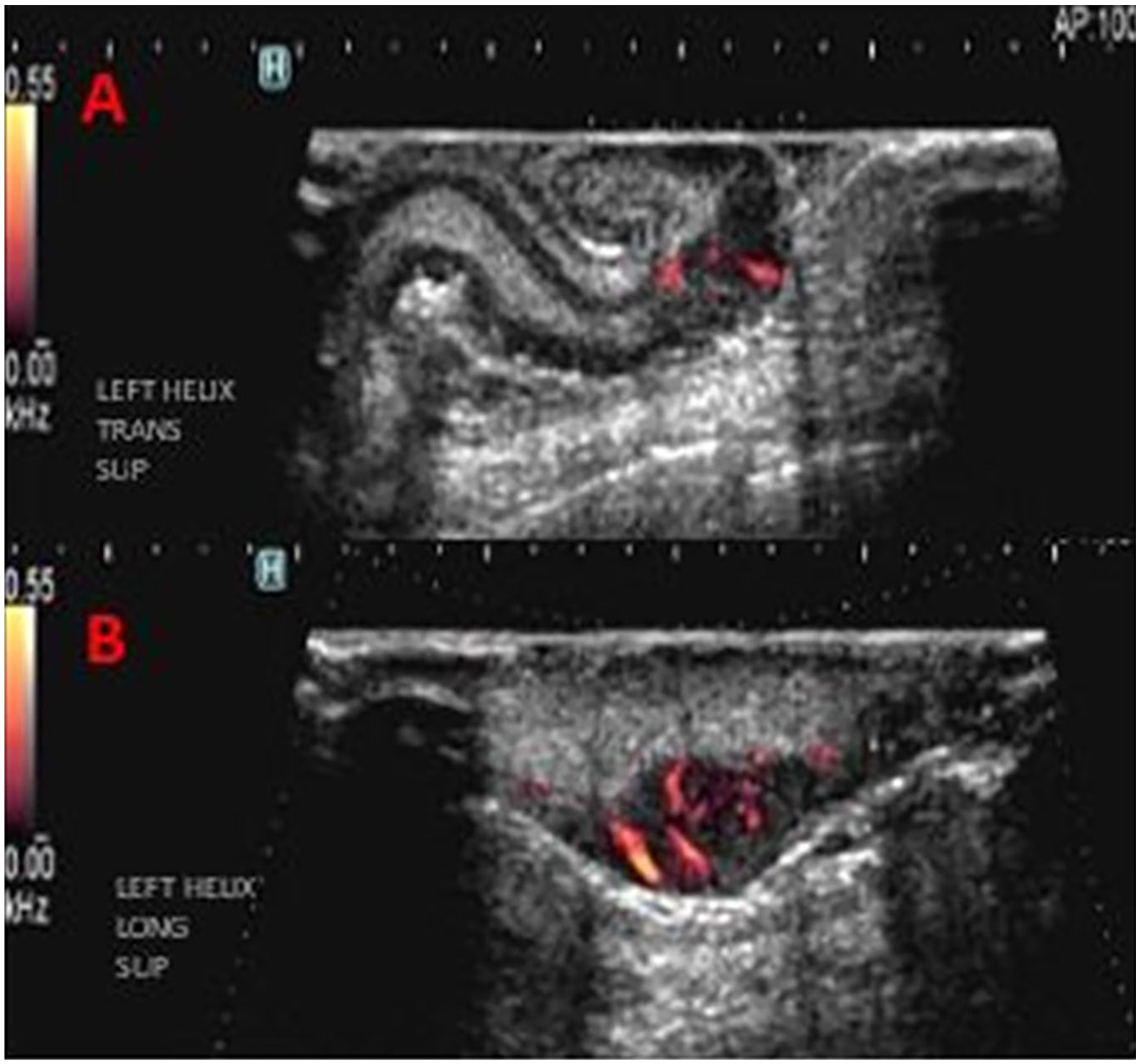
B-mode ultrasound images with power Doppler. (A) Transverse view and (B) sagittal view of the Clark level V (6-mm Breslow thickness) invasive melanoma of the left superior helix. Note the increased vascularity, utilizing the power Doppler function. Hypervascularity within a lesion is a characteristic indicative of malignancy. Light pressure should be applied on superficial lesions as to not disrupt the integrity of the structure or vascular flow.
In the best interest of the patient, surgical intervention was deemed necessary. A left partial auriculectomy, and a lymphadenectomy of the left sentinel node 1 and left level 2A node were performed. The auricular tissue proved invasive malignant melanoma with a Breslow’s thickness of 6.0 mm (Clark level V) and clear margins. The histologic slides were composed of nodular proliferation of atypical spindled and epithelioid melanocytes throughout the sample (Figure 5). Multiple areas of satellitosis (abnormal clustering) were present. Melanoma was not identified in the SLN 1, nor the level 2A lymph node. After postsurgical healing, the area was treated with radiation. Finally, a radiograph of the chest and CT of the chest, abdomen, and pelvis were completed to evaluate for melanoma restaging. A nonspecific subcentimeter nodule was found in the right lung and warranted continued surveillance.
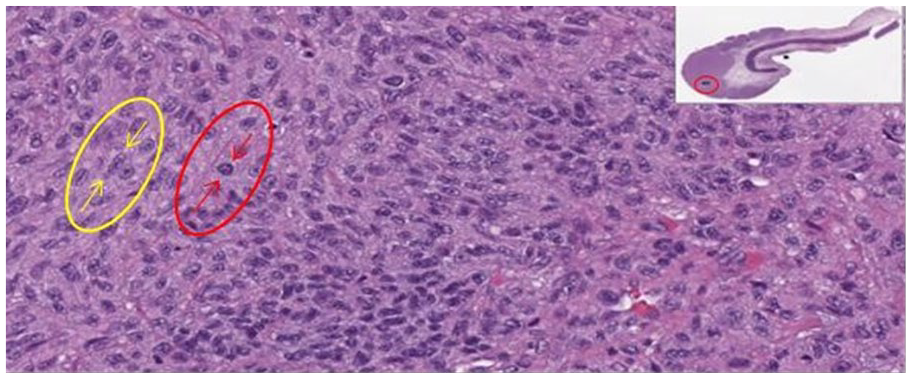
Left auricle-invasive melanoma. Histologic slide demonstrating a cross-section of the most posterior aspect of the surgically excised left auricular specimen, which extends into the anterior aspect of the ear. Highly magnified view of the above sample as indicated by the red circle. This sample demonstrates the nodular proliferation of atypical spindle cell (yellow arrows) and epithelioid cell (red arrows) melanocytes throughout.
Discussion
Reports of melanoma date back as early as the 17th and 18th century. 3 One of the many European reports including that of Highmore in 1651, referenced a “fatal black tumor” and “black fluid in the body.” 3 There are also documented cases of melanoma in 1858, by an established surgeon from England, Oliver Pemberton, that conducted a research study with a group of patients with metastatic melanoma treated over three decades, interpreting their clinical features and metastasis sites. 3 Awareness of metastases and the SLN concept was brought about in 1863 by a German pathologist, Rudolf Ludwig Carl Virchow. 3 Virchow proposed that lymph drained from one site through the lymphatic system to specific lymph nodes, and then onwards to other lymph nodes. 3 This concept formed a revoltionary gold standard of care that is still used today in melanoma staging with evaluation of SLNs. 3
Along with the SLN concept, other methods of prognosis involve characterizing and categorizing attributes of melanoma to determine the extent of invasion and therefore treatment. The scale of invasion based on the layer of dermis infiltration, referred to as the Clark level, is directly correlated to the prognosis for melanoma patients. 3 A melanoma lesion confined solely to the epidermis is considered a level I on the Clark scale, while a lesion penetrating past the deepest layers of dermis (papillary and reticular) reaching the subcutaneous fat is considered a level V on the Clark scale. 4 It was not until the publication in 1970 of Alexander Breslow, that the prognostic significance of vertical tumor thickness was established as a very strong prognostic factor. 3 The anterior/posterior depth (vertical thickness) from the top layer to the deepest invasive cell is referred to as Breslow’s depth. 3 Both the Clark level and Breslow’s thickness parameters are currently used as descriptive classifications of malignant invasiveness.
UVR is considered the highest risk factor for melanoma as it is known to encourage both cell death and malignant mutations of skin cells. 5 Compared with most cancers, melanoma has a greater variance rate amongst different ethnic groups. 6 Fair-skinned Caucasian populations are disproportionately at greater risk due to their lack of melanin, which serves as a natural barrier to decrease UVR through the skin. 6 Melanoma skin cancer is categorized into four main types: superficial spreading, nodular, lentigo maligna, and acral lentiginous. 6 Of the four, the most common is superficial spreading melanoma, accounting for the majority of skin melanomas. 6 Commonly, the locations most susceptible to melanoma are the face, scalp, neck, and ear, respectively. 7
Areas of the head and neck that receive the most exposure to UVR are typically the ears, resulting in being an area of high risk for skin cancers, such as melanoma. 8 The normal anatomy of the auricle consists of a thin layer of skin over minimal subcutaneous tissue encasing the cartilage, excluding the lobule which comprises fatty tissue and skin. 8 In particular, the outermost portion of the auricle is the helix, consisting of a smooth rim, distinguishable from the antihelix crura, antihelix, antitragus, and the lobule (see Figure 6). 9 Normal sonographic features of the auricle portray a distinguishable zone between the upper two thirds and lower third of the ear, where cartilage is present and absent, respectively. 10 The skin is represented as thin echoic layers, while the cartilage is depicted as a thin hypoechoic stripe that tracks the curvatures of the ear. 10 Interruption of the cartilage, or disruption of the layers of the dermis and subcutaneous tissue would be indicative of an abnormality, as seen in this case study (see Figure 3).
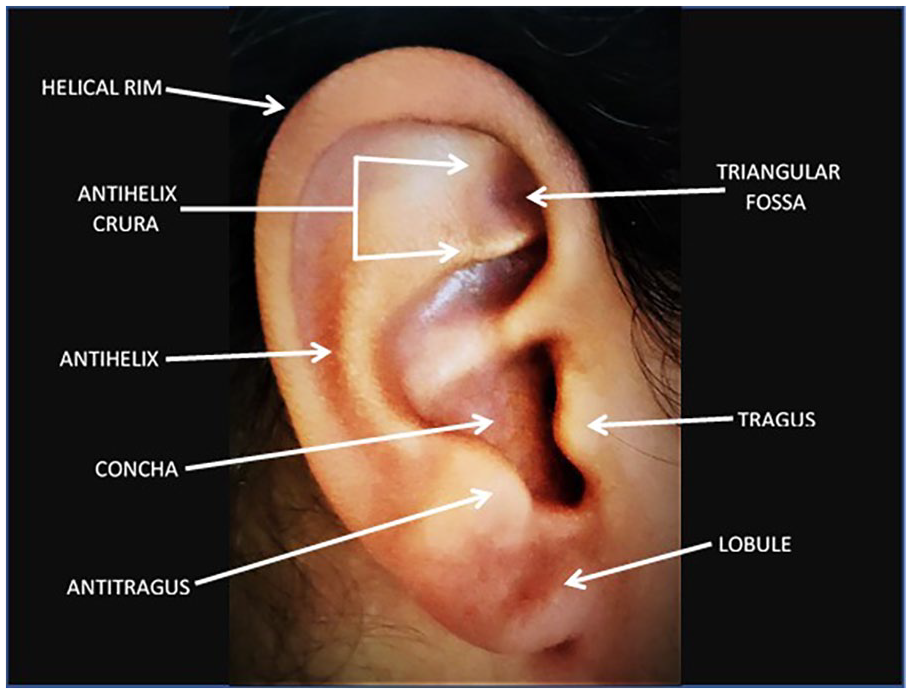
Auricular anatomy.
The most effective melanoma screening method used today is the whole-body skin examination using the “ABCDE” mnemonic. 11 Since melanoma lesions are typically incidental findings discovered during routine skin examinations, the basis for this simple guide sets a standard reference: 11
Asymmetry = one half unlike the other
Border = an irregular, scalloped, or poorly defined border
Color = varied from one area to another (shades of tan, brown or black, sometimes white, red, or blue)
Diameter = usually greater than 6.0 mm (the size of a pencil eraser) when diagnosed, but can be smaller
Evolving = a mole or skin lesion that looks different from the rest or is changing in size, shape, or color. 11
The use of the “ABCDE” reference assists in differentiation between common benign growths and those that should be considered worthy of further investigation. 11
The combination of whole-body skin examinations and diagnostic imaging modalities (PET/CT/sonography) effectively aids in thoroughly evaluating and staging patients who have confirmed cases of melanoma.11,12 The utilization of sonography, where applicable, can provide detrimental information regarding Breslow’s depth of melanoma lesions, as well as identify suspicious lymph nodes accessible to fine needle aspiration. This allows for histopathological review prior to surgical intervention. 13 Carefully evaluating sentinel and cervical lymph nodes in patients, who have melanoma of the head and neck, assists in staging the severity of the disease and contributes to presurgical assessment. 13 In addition, magnetic resonance imaging should be considered for patients with more advanced disease. 12
Utilizing sonography to distinguish disruption between the dermis and subcutaneous layers correlates with Breslow’s depth of preoperatively measured melanomas and the histologically measured thickness of CM. This has been shown to have the highest correlation in the head and neck, compared with other anatomical areas.13,14 Furthermore, the correlation was more significant when the CM had a depth of greater than 2 mm. 13 Sonography provides significant depth information, contributing toward surgical planning, where the incision size, free margins, and SLN can be considered presurgically. 10 Moreover, sonography provides preoperative guided access for fine needle aspiration cytology, identifies SLN metastases, and reduces the need for further surgical procedures. 10 Incorporating sonographic staging, complete with other investigative diagnostic imaging, could become like the Tumor, Node, Metastasis classification system and could cross-over into other forms of cutaneous cancer staging. 15
There are revolutionary, effective systemic therapies available for patients with metastatic melanoma. 16 Treatment approaches currently implemented include surgical resection, chemotherapy, photodynamic therapy, immunotherapy, biochemotherapy, and targeted therapy. 16 Some new therapeutic targets have derived from focusing on melanocytic genetic profiling and identifying molecular factors. These have been responsible for malignant transformation, proving to be a hopeful treatment option, for patients with advanced disease. 16 Given these points, the main form of treatment for melanoma is harvesting of the SLNs at the time of surgery, for staging the primary lesion.3,14
Conclusion
Utilizing diagnostic imaging modalities as part of the evaluation and staging process of melanoma lesions should be considered a standard of care. Specifically, utilizing sonography as a prognostic indicator of Breslow’s depth in qualifying melanoma lesions, as well as guidance for fine needle aspiration cytology, contributes vital information for surgical planning and staging. Furthermore, sonography is an integral tool utilized for evaluation of cervical adenopathy beyond initial assessment, assisting with metastatic recurrence detection. Continued surveillance should include diagnostic imaging, as well as whole-body screenings and plays a significant role in the continued aftercare of melanoma patients.
Footnotes
Acknowledgements
Special thanks to Dr. Dawid Schellingerhout, professor, diagnostic neuroradiology department at the University of Texas MD Anderson Cancer Center. Special thanks and in loving memory of Dr. Beth S. Edeiken-Monroe, professor, diagnostic neuroradiology department at the University of Texas MD Anderson Cancer Center.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Copyright and Permission Statement
To the best of our knowledge, the materials included in this case study do not violate copyright laws. All original sources have been appropriately acknowledged and/or referenced.
