Abstract
Laminectomy, a common neurosurgical procedure that decompresses the spinal cord, is the definitive surgical treatment for spinal epidural abscesses. Although complications after laminectomy occur infrequently, they can cause significant morbidity and health care resource usage. These complications include soft tissue collections like a persistent and/or new abscess or a hematoma. The preferred method of diagnosis for spinal soft tissue collections is magnetic resonance imaging. However, traditional neuroimaging poses significant challenges in patients with spinal hardware due to metallic artifact precluding appropriate visualization of anatomy and pathology. This was a case of a patient with extensive spinal hardware where visualization of the spinal cord by postoperative sonography was made feasible by a lack of bone after a laminectomy. Point-of-care ultrasound, a convenient bedside tool with the ability to detect soft tissue collections easily, was used to monitor for postoperative recurrence of a spinal epidural abscess in this patient. Patients with an intact spine do not have adequate acoustic windows due to posterior vertebral anatomy. In postlaminectomy patients without posterior vertebral structures, sonography may offer a diagnostic modality for postoperative monitoring. Ultrasonographers can employ spinal cord sonography, particularly when the indication for decompressive laminectomy was a localized fluid collection or abscess.
Laminectomy is a common neurosurgical procedure that decompresses the spinal cord. Decompressive laminectomy and drainage is the definitive surgical treatment for spinal epidural abscesses (SEAs), which are a rare but devastating cause of back pain.1,2 Overall, in-patient laminectomy surgery has a 30-day complication rate of 7%. 3 Postoperative complications include soft tissue collections like a hematoma, a persistent and/or new abscess, or a spinal fluid collection caused by a dural tear.4,5 The most sensitive modality for diagnosis of postoperative spinal complications is gadolinium-enhanced magnetic resonance imaging (MRI); for example, paraspinal or epidural inflammation on MRI with contrast is 97% sensitive for an SEA. 6
However, MRI findings in the postoperative setting are not always diagnostic because increased signal intensity and edema can be a normal postoperative finding as well. 7 Furthermore, the hardware implanted during spinal surgery can obscure the view of the spinal cord due to metallic artifact. Given the persistence of drill bit fragments postoperatively, MRI may even demonstrate metallic artifact when no hardware is present. 8 Despite the challenges of imaging, early diagnosis is critical to avoid irreversible neurologic deficits secondary to postoperative complications such as SEAs. Treatment of such complications often requires emergent repeat surgery for decompression. 9
Sonography, as an imaging modality, is particularly useful for the evaluation of soft tissue collections like abscesses. In neonates, due to the incomplete ossification of the posterior vertebral elements, sonography can be used to visualize the spine. 10 In adults, the posterior vertebral arch, including the spinous process, laminae, and articular processes, obscures the view of the spinal cord. In a laminectomy, the lamina is removed and provides a window through which sonography can produce high-quality images of the underlying spinal cord.
This was a case of an 84-year-old man with prior thoracolumbar spinal fusion. He had an extensive medical history who presented to the emergency department (ED) with 1 week of worsening back pain and subjective weakness in both legs. Point-of-care spinal sonography offered a novel application in monitoring this patient status after emergent decompressive thoracic laminectomy.
Case Report
The patient was an 84-year-old man with a T10-S1 posterior spinal fusion 2 years before, coronary artery disease with stents on dual antiplatelet therapy, heart failure, hypertension, chronic kidney disease, hypothyroidism, polymyalgia rheumatica on chronic steroids, and bipolar disorder. He presented to the ED with 1 week of worsening back pain and symptoms suggestive of neurogenic claudication. He endorsed subjective weakness in bilateral lower extremities but denied radicular symptoms or bowel/bladder involvement.
Further laboratory evaluation on presentation revealed an elevated white blood cell count to 17 × 10*3/µL and elevated C-reactive protein to 257.3 mg/L. Otherwise, laboratories were unremarkable. A computed tomogram (CT) of the spine, on hospital day 1 showed T9 and T10 compression fractures that had been visualized on prior imaging. Of note, CT is best used for visualizing bony structures rather than soft tissue.
Neurosurgery consultation on hospital day 1 recommended thoracic and lumbar MRI with plans for elective T8 vertebroplasty and T9 kyphoplasty to treat his compression fractures. In vertebroplasty, a cement mixture is injected into the fractured bone; in kyphoplasty, a balloon is inserted into the fractured bone to create a space that can be filled with cement. Cardiology and rheumatology were consulted for perioperative recommendations regarding management of dual antiplatelet therapy and chronic steroids use, respectively. On hospital day 4, the patient’s course was complicated by blood cultures that returned positive for Staphylococcus aureus, requiring infectious disease consultation. The patient was started on antibiotics for his bacteremia, and the planned elective T8 vertebroplasty and T9 kyphoplasty procedure were canceled.
Meanwhile, MRI of the thoracic spine obtained on hospital day 4 revealed possible discitis or osteomyelitis at T9-T10 with paraspinous and epidural abscesses, pathologic fractures of T9 and T10, advanced canal stenosis, and cord compression resulting in cord edema. Additional T2 hyperintense fluid collections along the dorsal margin of the thecal sac were reported at L3-L5 of undeterminable origin. Of note, the radiologic imaging evaluation of the canal was limited by susceptibility artifact from hardware (Figure 1).
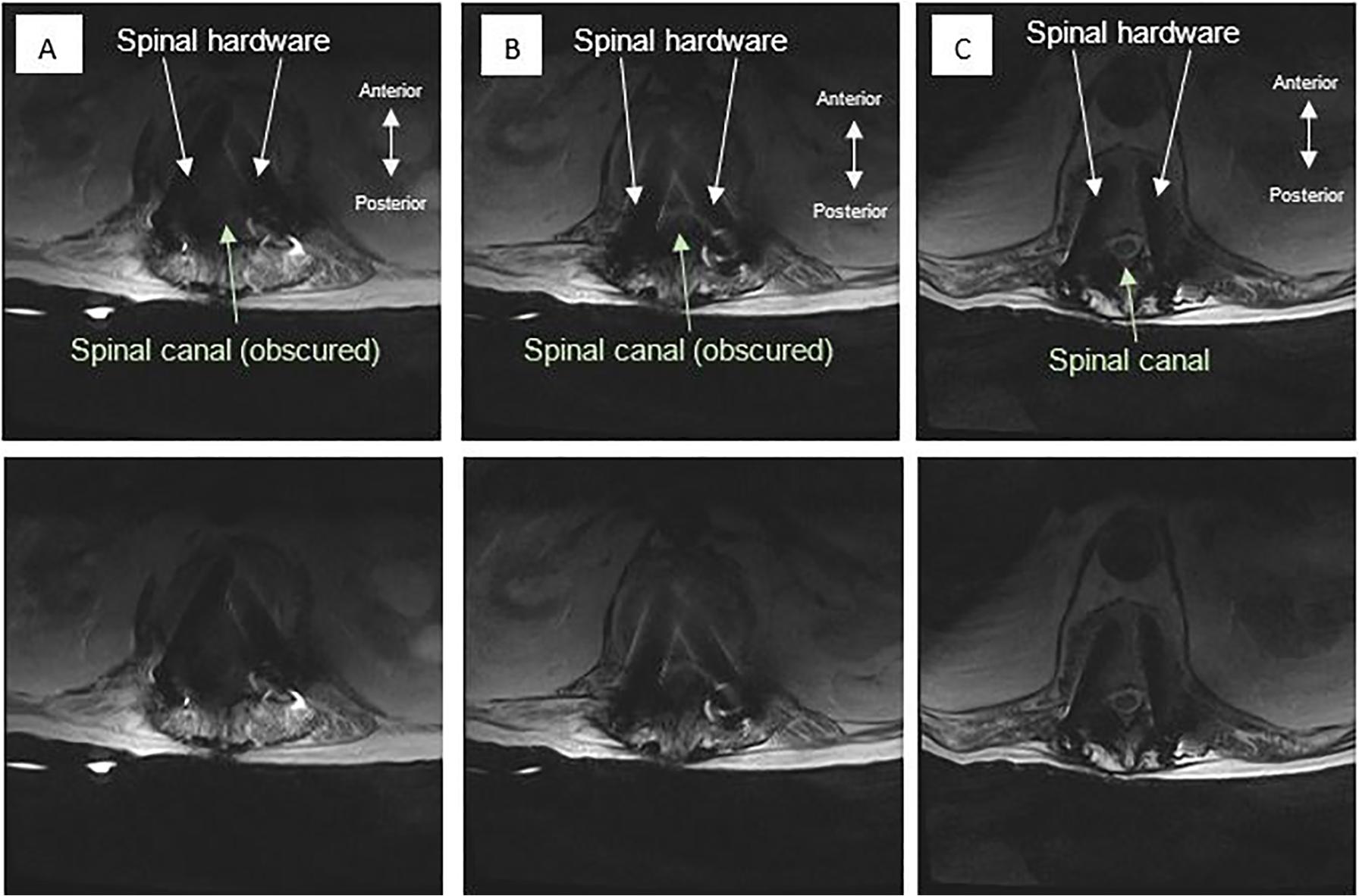
Patient’s magnetic resonance imaging of the thoracic and lumbar spine, axial images at the level of (A) L2, (B) L1, and (C) T12. The patient’s spinal hardware from a prior spinal fusion surgery obscured visualization of the spinal canal. All images were used with patient consent under Wake Forest Institutional Review Board regulations.
With worsening symptoms of paraparesis, on hospital day 5, the concern for a possible epidural abscess causing severe thoracic myelopathy at T9-T10 resulted in an emergent surgery for decompression. Spinal decompression was done with T9-T10 laminectomy and extension of posterior spinal fusion up to T5 from T12 using bilateral segmental instrumentation with complete laminectomy and bilateral facetectomy. Intraoperatively, purulent drainage was noted adjacent to the screws at T10 from his prior thoracolumbar fusion, and an epidural abscess at T9-10 was evacuated.
Postoperatively, the patient continued to recover function but required evaluation for a persistent infective source at the previous epidural collection site. Given the patient’s extensive spinal hardware and uncontrolled pain while supine, bedside sonography with a Sonosite Xporte(c) machine was attempted to assess for possibility of acoustic windows through the removed posterior vertebral structures (Figure 2). Gray-scale sonography was successfully performed in the longitudinal and axial orientation (Figure 3). The images revealed a distinct and unobstructed view of the spinal cord due to the removal of the vertebral lamina. The images confirmed lack of evidence of paraspinal fluid collection suspicious for hematoma or persistent abscess in the visualized portion.
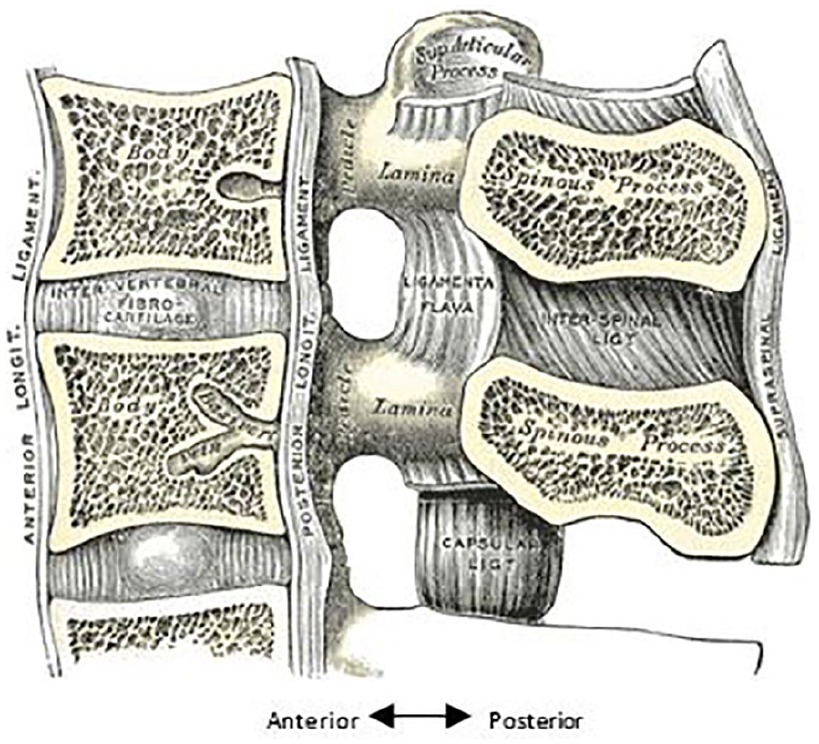
Medical illustration of the median sagittal section of two lumbar vertebrae and their ligaments. 11 Permission was obtained by creative commons licensure.
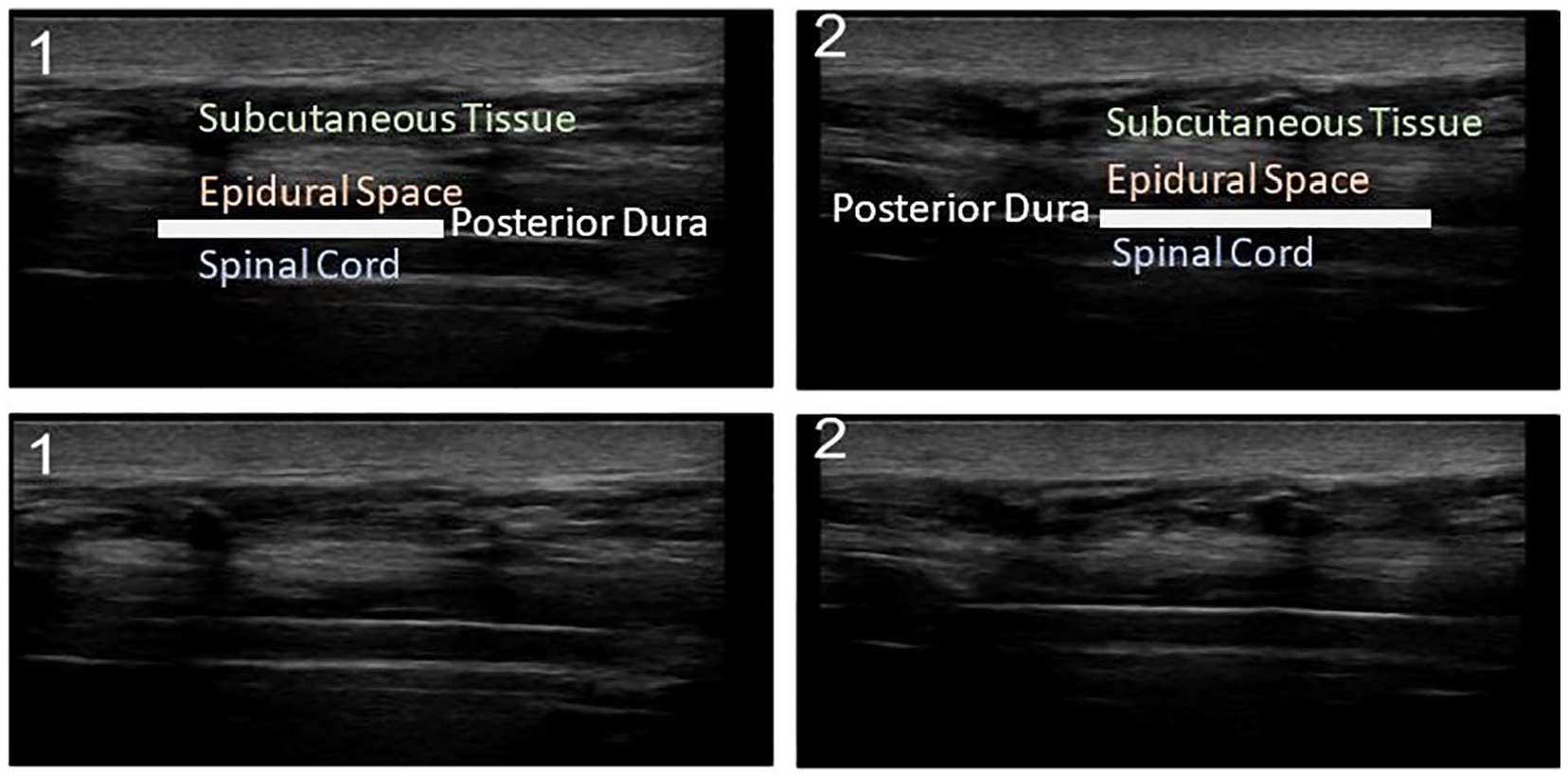
Gray-scale sonograms of the patient’s thoracic spine in the sagittal plane with a linear probe 6–13 MHz. The ultrasonographer insonates the skin and subcutaneous tissue first. From a posterior approach, the epidural space is encountered before the posterior dura. The posterior dura encloses the spinal cord. After the removal of posterior vertebral bony elements including the lamina, the epidural space and spinal cord are clearly visualized. All images were used with patient consent under Wake Forest Institutional Review Board regulations.
The patient was deemed medically stable and discharged to subacute rehabilitation facility for recovery on postoperative day 16 on colchicine, anticoagulation, and dual antiplatelet therapy.
Discussion
Laminectomy is a common neurosurgical procedure that decompresses the spinal cord. Given the 7% complication rate after in-patient laminectomy, postoperative imaging to assess for persistence of pathology and/or new complications is not uncommon. 3 MRI is the modality of choice, given that specific imaging features are diagnostic of specific complications. 12
SEAs are an indication for decompressive surgery as well as a possible serious complication of surgery. In fact, 16% of SEAs are postoperative in etiology. 1 SEAs are classically accompanied by back pain as well as fever and a focal neurologic deficit; however, patients can be normothermic and present with back pain alone. 9 Given the variability in clinical presentation, MRI is the standard for diagnosis. 6 If treated quickly and effectively with antibiotics and surgical decompression as well as drainage, the duration of neurologic deficit predicts the degree of neurologic recovery. 9
MRI, however, can be thwarted by metallic artifact when spinal hardware is in place, which is common in patients after spine surgery. 8 Difficulty with visualization by MRI poses a critical problem. Without early diagnosis and subsequent intervention upon an SEA, neurologic symptoms, such as paralysis, may quickly become irreversible. In fact, one third of SEA survivors may have persistent neurologic deficits due to delayed diagnosis. 9
Due to the potentially catastrophic prognosis of SEAs and importance of an early diagnosis, without adequate visualization of the spine, patients may be unnecessarily taken to the operating room under abundance of caution when an abscess cannot be ruled out. 13 Unlike MRI, gray-scale spinal sonography in a postlaminectomy permits noninvasive imaging unaffected by spinal hardware at the bedside.
Patients with intact spines will have acoustic shadows caused by the skeletal spine (Figure 4). However, in a patient after a laminectomy, the lamina has been removed to decompress and provide access to the spinal cord; in complete laminectomy, the spinous process has been removed as well (Figure 5). This provides a sonographic imaging window of the epidural space and the spinal cord. In this case, sonography was particularly useful because the patient’s spinal hardware preoperatively limited the visibility of the spinal cord with MRI (Figure 1). With extension of his prior fusion from T12 up to T5, the additional hardware would further hinder the use of traditional imaging techniques to assess for recurrence of the abscess postoperatively. Given this patient’s laminectomy, however, the spinal cord was clearly visualized without obstruction of the skeletal spine (Figure 3).
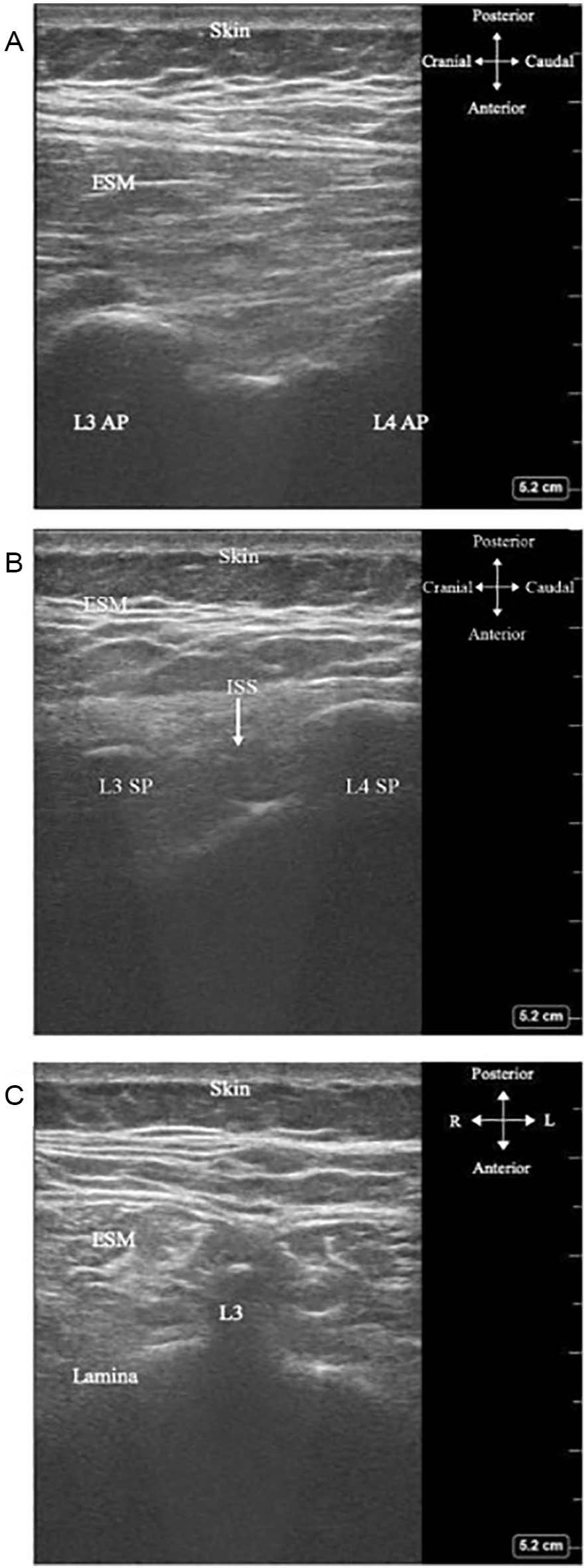
Gray-scale sonography of the spine in a healthy adult female in (A) parasagittal, (B) sagittal, and (C) transverse planes. 14 Insonating the spine from the posterior aspect, first, the skin and subcutaneous tissue are visualized. Then, the erector spinae muscles (ESMs) are encountered. Next, the posterior vertebral arch is made up of seven bony parts, including one spinous process (SP) in the midline, two laminae on either side, and four articular processes (APs) per vertebral level (two superior and two inferior). The placement of the probe determines which of these bony features are visualized. Regardless, as skeletal structures, the SP, lamina, and AP all create acoustic shadows that obscure a clear and continuous view of the underlying spinal cord. The interspinous space (ISS), between two SPs, does not provide an adequate acoustic window. Permission was obtained for use from the original author.
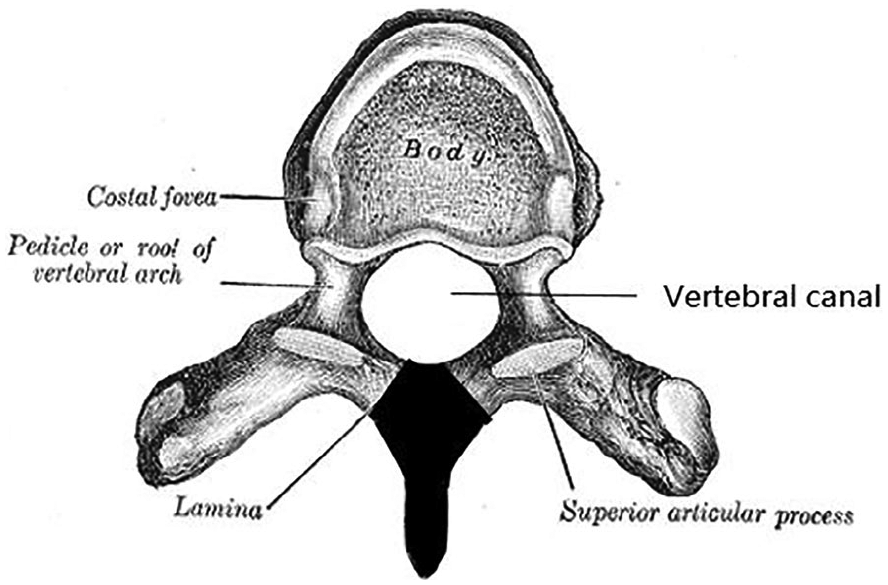
Medical illustration of a typical thoracic vertebra, viewed from above. 15 Black shading (added) identifies the vertebral elements that are resected in a complete laminectomy: the bilateral laminae and the spinous process. The vertebral canal is subsequently decompressed. Permission was obtained by creative commons licensure.
On sonography, an abscess cavity is usually round with an anechoic or hypoechoic center composed of purulent material and blood. The cavity creates the artifact of posterior acoustic enhancement due to insonation through the liquid center (Figure 6). Sometimes, an abscess can appear isoechoic or hyperechoic if loculations or dense fluid is present. When the ultrasonographer applies gentle pressure, the abscess contents can be visualized swirling, and this technique has been called “ultrasound fluctuance.” In addition, many abscesses are surrounded by soft tissue edema or cellulitis, which is noted by “cobblestoning” on a sonogram. The reticular pattern caused by hypoechoic fluid separating the subcutaneous tissue and fat is described as “cobblestoning” because the pathologic fluid is akin to the spaces that separate cobblestones on a street. 16
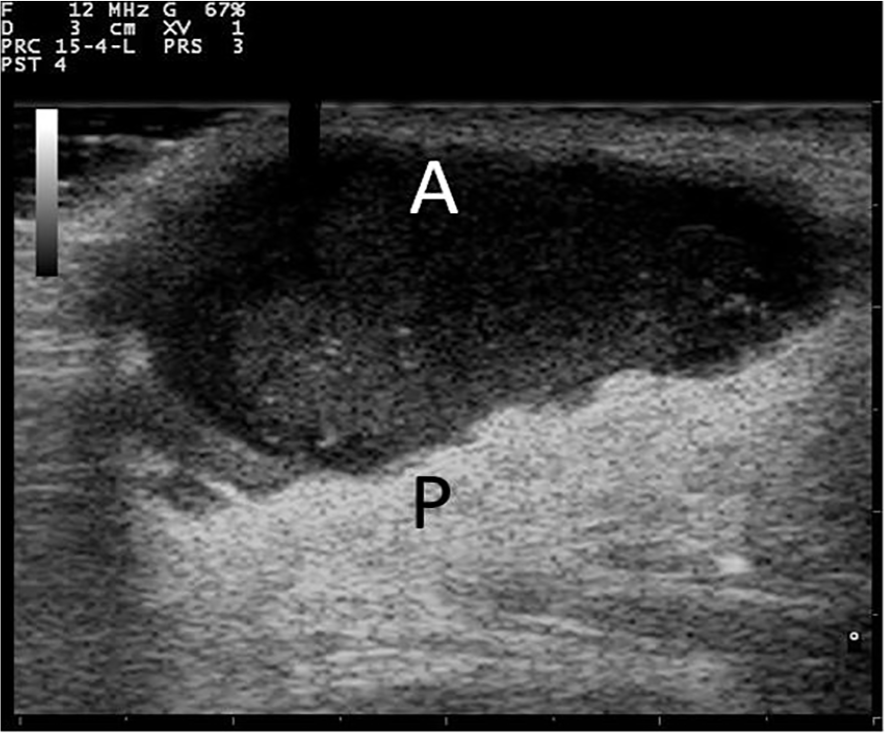
A sonographic example of an anechoic abscess (A) with hyperechoic posterior acoustic enhancement (P). 17 Permission was obtained by creative commons licensure.
In this patient, the epidural space was normal, and he had no focal collection with acoustic shadowing or ultrasound fluctuance, which would have been suggestive of an abscess or fluid collection. Color Doppler was not employed; future studies can investigate the role of color Doppler in delineating spinal pathology in postoperative complications. As a result of this novel application of point-of-care spinal sonography, the patient was able to be adequately monitored and avoid unnecessary interventions, including repeat surgery. The ease of use at the bedside proved supremely beneficial for this patient. Ultrasonographers should be prepared to use sonography for postoperative monitoring, particularly when the indication for decompressive laminectomy is a localized fluid collection or abscess.
Conclusion
Gray-scale sonography of the spinal cord in this postlaminectomy patient suggests the utility of this imaging modality as a tool to examine the epidural space and spinal cord. This is especially important for postoperative complications when traditional imaging is limited or challenging due to spinal hardware. Postoperative sonography permits noninvasive monitoring for resolution of the initial lesion. With bedside visualization of the epidural space and spinal cord in patients with a surgically created acoustic window, sonography may prove vital in avoiding unnecessary returns to the operating room. Ultrasonographers could rely on this tool, particularly when the indication for decompressive laminectomy is a localized fluid collection or abscess. It is important to remember that point-of-care spinal sonography is only helpful to visualize the spinal level where the bony elements have been surgically removed. Future investigations are warranted to determine the reliability and validity of spinal cord ultrasonography in this population.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
