Abstract
Loeffler endocarditis is rare and can initially be a clinically silent cardiac disorder. Given its natural history with evolving stages and its vague initial cardiac presentation, it is important to keep this as a differential diagnosis. Without treatment, this disorder is progressive and may lead to a possible restrictive cardiomyopathy-related heart failure. Here, a rare case who presented with nonspecific chest pain is reported, which was recognized early by a routine noninvasive transthoracic echocardiography (TTE) imaging study. This was followed by the addition of contrast enhancement techniques, which made it possible to solidify the diagnosis of the mass to a right ventricular thrombus (possibly secondary to the hypereosinophilia syndrome), with isolated right ventricular involvement. Magnetic resonance imaging (MRI) of the heart, without contrast, was performed to further analyze the anatomic location of this entity. A high index of suspicion for Loeffler endocarditis helped identify this condition.
Keywords
Case Report
A 67-year-old woman with a medical history of hypertension, hyperlipidemia, coronary artery disease, mitral valve repair, and chronic kidney disease stage III presented for evaluation. She was experiencing sharp, intermittent chest pain over the past 2 days. She was hemodynamically stable at presentation. Serial cardiac enzymes were performed, which were unremarkable. The patient’s lab results were abnormal, including a markedly elevated eosinophil count of 5280/µL and an elevated baseline creatinine of 2.82 mg/dL with a calculated estimated glomerular filtration rate (eGFR) of 24 mL/min/1.73 m2. Her troponin I value was normal at <0.01 ng/mL. Electrocardiogram (ECG) showed a sinus rhythm with nonspecific right ventricular conduction delay, with nonspecific T-wave abnormality (see Figure 1). Transthoracic echocardiography (TTE) was additionally performed on the patient. The study findings revealed her left ventricular ejection fraction to be mildly reduced at 45%–50% (unchanged from her prior TTE), but the right ventricular systolic function to be severely reduced. Right ventricular S-wave velocity was 3.49 cm/s, and tricuspid annular plane systolic excursion was 0.92 cm, corresponding to an estimated right ventricular ejection fraction of 20%–30% (see Figures 2 and 3). In addition, an echo density suggestive of a large intramural thrombus was noted from apex to the mid-ventricular level of the right ventricle (RV), in the parasternal long-axis view (see Figure 4 and Supplemental Video 1) and apical four-chamber view (see Figure 5 and Supplemental Video 2). An ultrasound contrast media agent was administered to improve the visualization of the mass and to rule out normal variants, such as a right ventricular trabecula or a moderator band. The study showed an echo density in the RV that was suggestive of a mural thrombus (see Figures 6 and 7). Cardiac magnetic resonance imaging (MRI), without contrast, was performed to confirm the diagnosis. Steady-state free-precession (SSFP) four-chamber noncontrast MRI, of the heart, demonstrated a right ventricular mass consistent with a thrombus in the RV, separated from the myocardium (see Figure 8). Due to the suspicion of Loeffler endocarditis (LE), diagnostic tests were conducted, including a stool culture for ova and parasites, which was unremarkable. In addition, a hematology consultation was obtained. To rule out malignancy and investigate the underlying causes for hypereosinophilia (HE), the patient underwent a bone marrow biopsy, which was unremarkable. The patient was started on pulse dose steroids, which showed immediate improvement in the patient’s nonspecific chest pain, and her eosinophilic count decreased from 5280/µL at admission to 20/µL. The patient remained stable, was discharged to be followed up by the Hematology and Cardiology staff, and had no further complications.
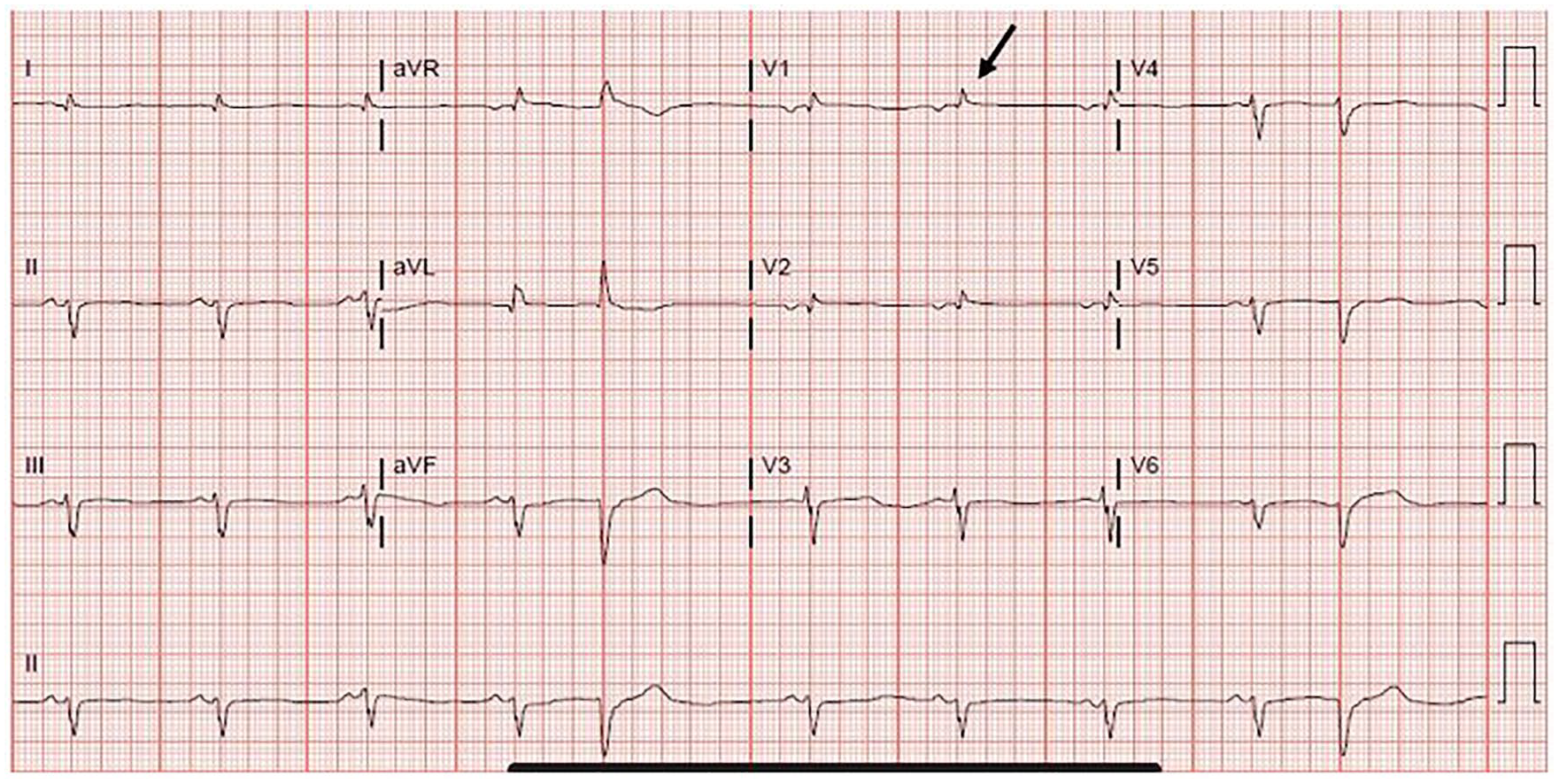
Electrocardiogram showed sinus rhythm with nonspecific right ventricular conduction delay (arrow) and a nonspecific T-wave abnormality.
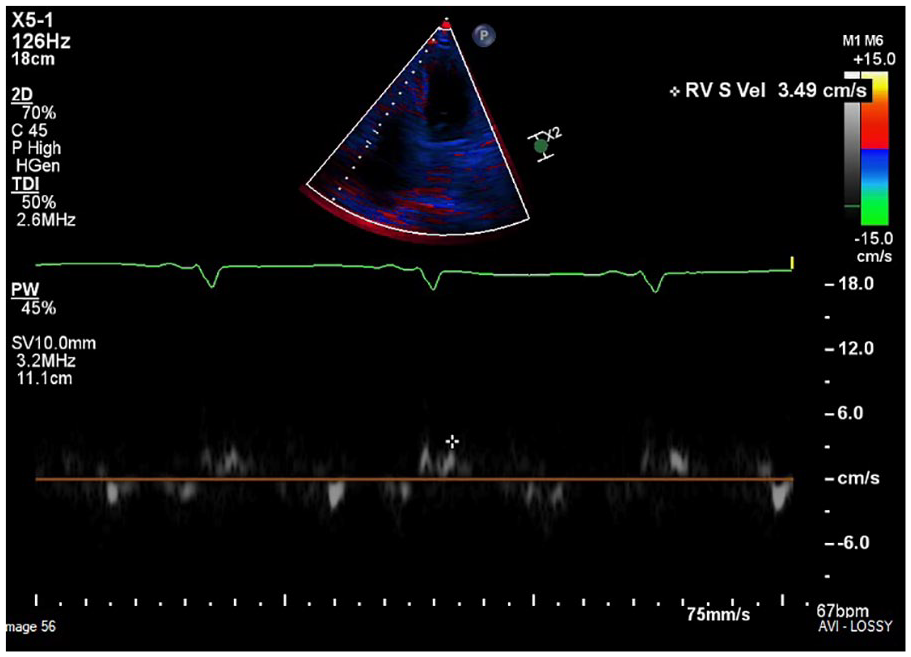
Apical four-chamber view demonstrating the myocardial systolic excursion velocity (s′) wave obtained by tissue Doppler imaging at the lateral tricuspid annulus.
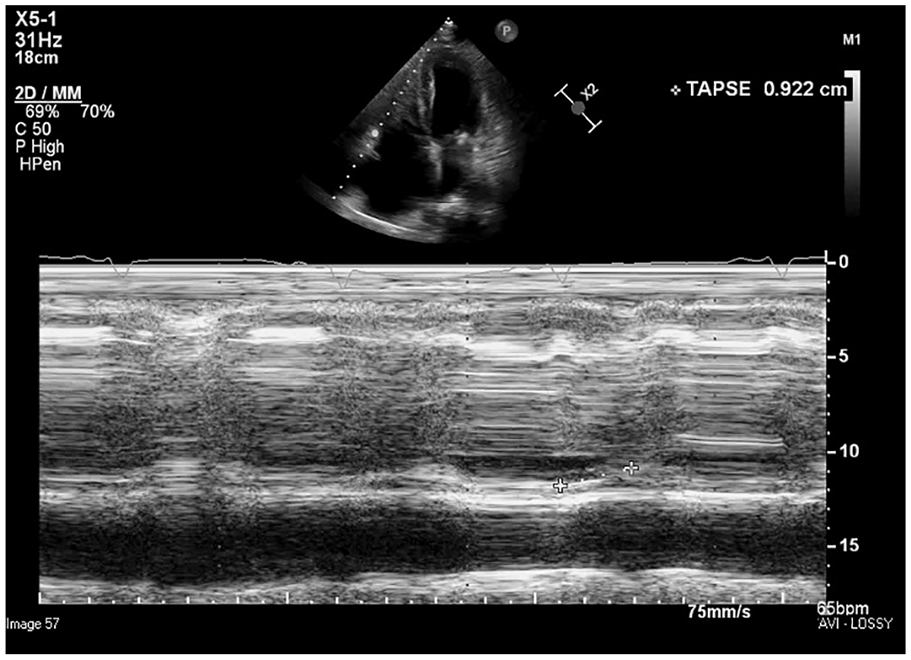
Tricuspid annular plane systolic excursion M-mode measurement at the lateral tricuspid annulus.
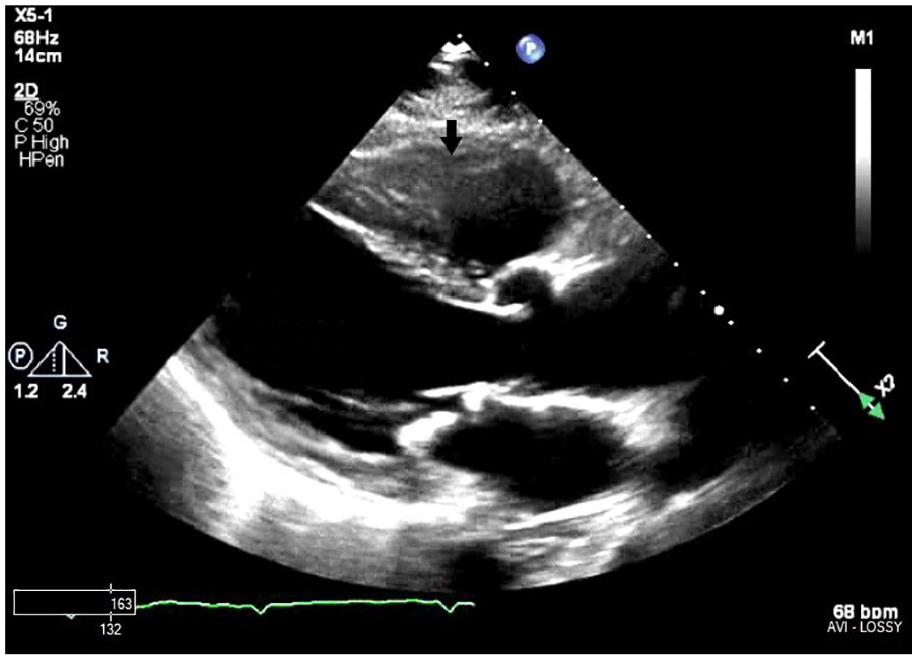
Transthoracic parasternal long-axis view (arrow) depicting a possible echo density in the right ventricle.
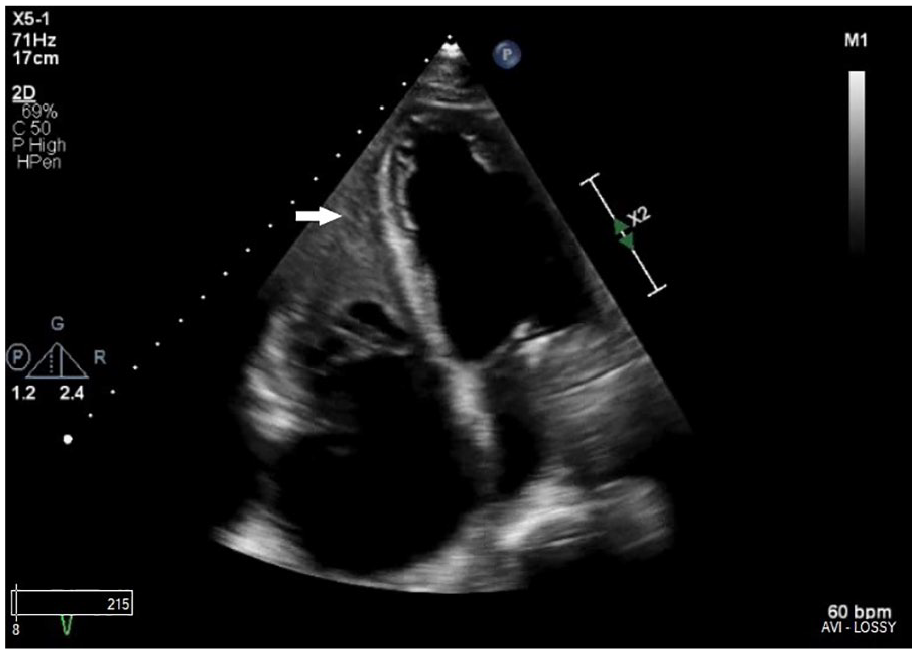
Transthoracic apical four-chamber view depicting a possible echo density (arrow) in the right ventricle.
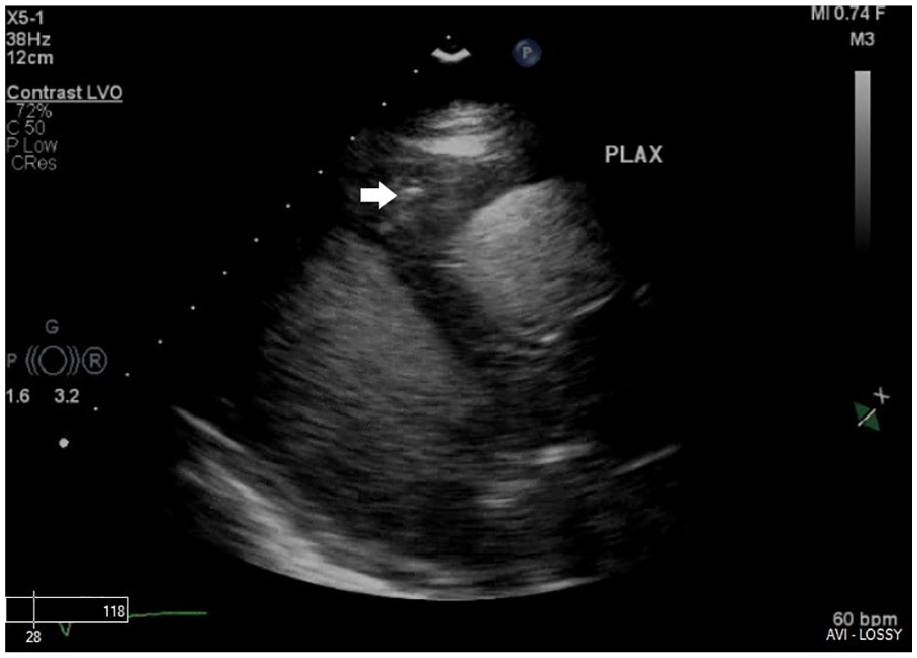
Transthoracic low parasternal long-axis view, with contrast agent administration, demonstrating a filling defect (arrow) in the right ventricle.
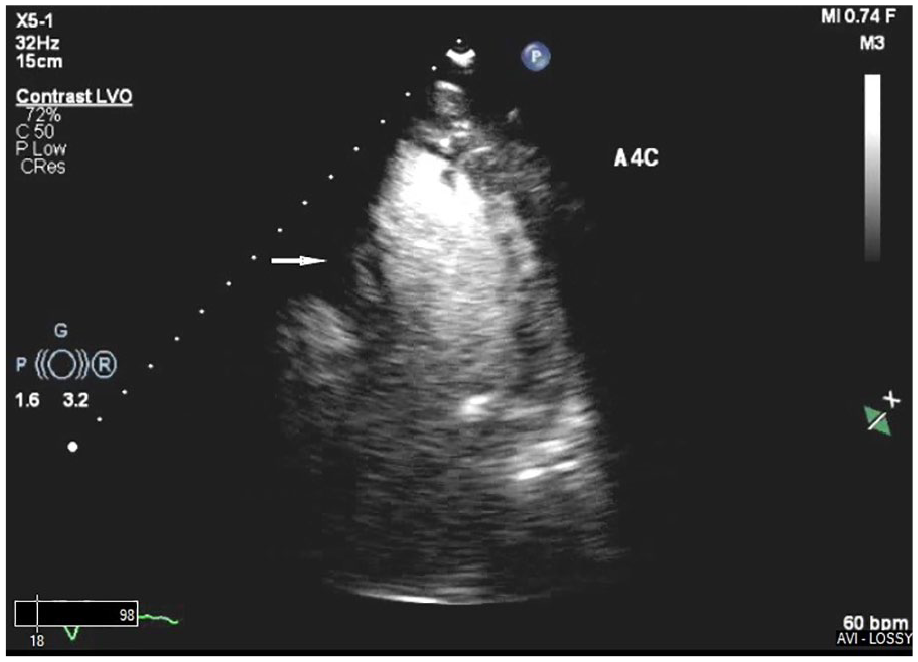
Transthoracic apical four-chamber view, with contrast, demonstrating a filling defect (arrow) in mid to apex of the right ventricle.
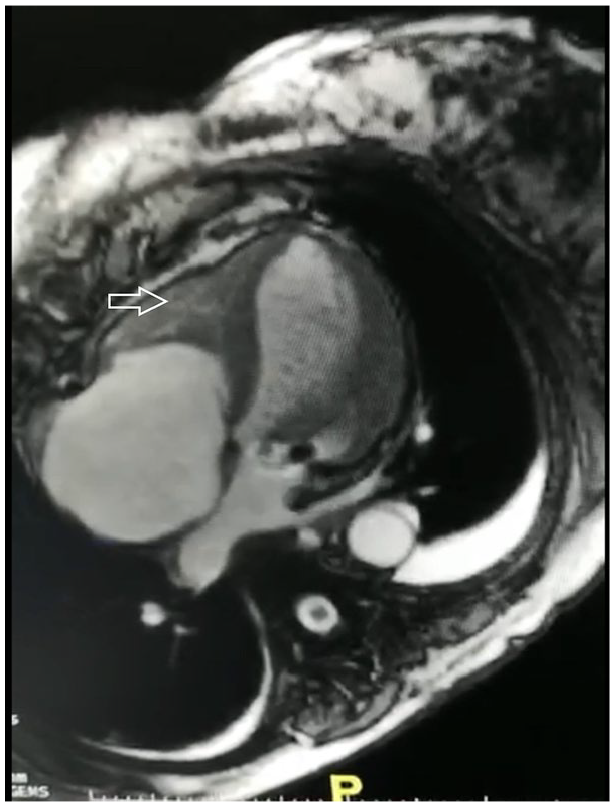
Steady-state free-precession four-chamber noncontrast magnetic resonance imaging, of the heart, demonstrating dark thrombus in the right ventricle (arrow), separated from the myocardium.
Discussion
Eosinophils, one of the three granulocytes in the peripheral blood, account for less than 7% of the circulating white blood cells (leukocytes). Eosinophilia is a condition in which the eosinophilic count is greater than 0.5 × 109/L (500/μL). 1 Peripheral blood eosinophilia has been categorized into mild (0.5–1.5 × 109/L), marked (>1.5–5 × 109/L), and massive (>5.0 × 109/L) eosinophilia. 1 This condition is also classified previously as transient, episodic, or persistent.1,2 Valent et al also proposed the term “hypereosinophilia,” which should be used when marked and persistent eosinophilia has been documented or marked tissue eosinophilia is observed. 1 The underlying cause of HE can be a reactive process, an underlying condition, or a neoplastic process. Persistent eosinophilia is when peripheral blood eosinophilia is recorded on at least two occasions with a minimum time interval of 4 weeks, unless treatment is administered for an emergency. 1 Hypereosinophilia syndrome (HES) is a clinical syndrome in which blood HE can be documented and is directly linked to organ damage, irrespective of the underlying cause of HE. 2 Tissue manifestations, such as endomyocardial fibrosis, thrombosis, or thromboembolism, resembling those described in “historically defined HES,” can occur in patients with HE. When end-organ damage is present, specifically due to eosinophilia, the diagnosis can be changed from HE to HES. This entity is a rare occurrence with an approximate reported incidence of about 0.035–6.5 per 100 000. 3
Infiltration of the myocardium by eosinophilic cells following endomyocardial fibrosis is known as hypereosinophilic endocarditis, also addressed as “Loeffler endocarditis.”4,5 Cardiac involvement can be present in as high as 40%–50% of this entity. 6 Three pathological stages of LE are described: acute phase, thrombotic phase, and fibrotic stage. When eosinophilic granulocytes infiltrate the heart, they secrete granules that damage the endcardium/myocardium and result in platelet activation. The activated platelets later combine to form intracavitary and intravascular thrombi. 4 The eosinophil granule proteins are involved in cardiac injury, producing muscle damage and vascular injury. This injury can further result in the development of endomyocardial fibrosis, which can potentially lead to the development of restrictive cardiomyopathy. 4
Diagnostic studies including laboratory tests with complete blood count with differential can search for secondary causes of eosinophilia, which include parasitic infestations, Churg-Strauss syndrome, medication-induced eosinophilia, malignancies, and so on. ECG may reveal abnormalities specific to the underlying cardiac chamber involved, but not any particular abnormalities that are specific to HES. 6 These findings may include nonspecific T-wave findings with or without atrial enlargement, right ventricular conduction delay abnormalities if the RV is involved, and left ventricular hypertrophy if the left ventricle is involved. 6 Two-dimensional echocardiography (TTE) with color and spectral Doppler analysis is a great initial diagnostic, noninvasive, quick, safe tool to assess cardiac abnormalities due to eosinophilic endocarditis. Findings may show left ventricular hypertrophy, endomyocardial thickening, atrial dilatation, ventricular apical thrombus, and/or posterior mitral valve cusp involvement (enmeshment, chordae tendineae causing valvular regurgitation) or entrapment of the chordae tendineae with valvular regurgitation. Findings consistent with restrictive cardiomyopathy may be evident in the third (fibrotic) stage of the disease process. 5 In this case, the LE was detected while performing a routine TTE with an isolated right ventricular thrombus, as a diagnostic finding. This patient had associated chronic kidney disease as comorbidity. A contrast agent was administered, and through enhanced imaging, the filling defect was demonstrated conclusively and helped to solidify the diagnosis. A noncontrast MRI study was performed to further help confirm the diagnosis through visualization of the thrombus, later in the course of the patient’s hospitalization.
Cardiac involvement in HES is characterized by endomyocardial fibrosis and intracardiac thrombus. Since contrast-enhanced MRI can detect myocardial fibrosis, inflammation, as well as being sensitive and specific, for the detection of thrombus, it is an excellent diagnostic modality to assess for cardiac manifestation in HES. 7 However, in this case, gadolinium contrast could not be administered, due to associated chronic kidney disease. The patient had an eGFR of less than 30 mL/min/1.73 m2 and was contraindicated, per the US Food and Drug Administration’s (FDA) drug safety package insert.
A high index of suspicion can help with identification of this condition. While myocardial biopsy is the gold standard to diagnose this condition, it is an invasive technique. Therefore, a multimodality imaging approach was modified, to the patient’s condition, and was used to diagnose this condition, at an early stage.
Conclusion
Loeffler syndrome is a rare cardiac disorder. Given its natural history with evolving stages and its vague initial cardiac presentation, it is essential to keep it as a differential in a patient presenting with HE and cardiac symptoms. Without treatment, the disorder is progressive with possible restrictive cardiomyopathy-related heart failure. Multimodality imaging tools can help diagnose at an earlier stage and can be useful for subsequent monitoring of the treatment response. Contrast-enhanced imaging is becoming an increasingly useful technique to further enhance the diagnostic capability, in the noninvasive imaging sector. This technique may offer promise in increasing the sensitivity of conventional echocardiography imaging, especially in the setting of limited access or when an MRI with or without gadolinium contrast is otherwise contraindicated due to a patient’s associated comorbidities (chronic kidney disease, pacemaker dependence, clips for the cerebral aneurysm, etc.).
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
