Abstract
Alcoholic liver disease (ALD), a precursor to alcohol-induced cirrhosis, is a disease caused by excessive ingestion of alcoholic substances that directly affects liver functions. Abnormal liver function can cause the liver to damage other organs within the abdomen. This case study examines the physiological nature of alcohol-induced cirrhosis and its pathogenesis, external and internal clinical presentations, and treatment options. Treatments for alcohol-induced cirrhosis include liver transplant for permanent correction as well as varied options to manage symptoms. This case study analyzes alcoholic liver disease within one male patient whose condition highlights the importance of sonography in routinely monitoring patients with ALD.
Alcoholic liver disease (ALD), a precursor toalcohol-induced cirrhosis, is an acquired, degenerative tissue disease that causes fat to accumulate and infiltrate the liver, as well as disrupts the liver’s oxidation mechanisms. Approximately 2 million people in the United States have ALD and the mortality for cirrhotic disease with superimposed ALD is worse than many commonly diagnosed cancers. 1 In 2007, 29,925 deaths were attributed to cirrhosis in the United States, and 48% of those deaths were alcohol related. 2 According to research cited by Barve et al., 1 ALD ranks second, only to hepatitis C cirrhosis, as the reason for a liver transplant. With patients having end-stage alcoholic-induced cirrhosis, the focus is long-term symptom management.
Alcohol-induced cirrhosis is part of a differential diagnosis when a patient presents with an acute onset of symptoms, such as but not limited to hematemesis, abdominal distention, abnormal liver enzyme levels, and a history of excessive alcohol ingestion. Sonography can be used to assess the liver echotexture, tissue echogenicity, and blood flow patterns within the portal veins, hepatic veins, and hepatic artery to aid in diagnosis and staging of the disease.
Case Study
A 40-year-old man presented to the emergency department complaining of abdominal distention, nausea, vomiting, weakness and fatigue, severe abdominal pain, and hematemesis. The patient had a history of excessive smoking and consuming one gallon of hard liquor per week. A series of diagnostic laboratory tests were ordered, which found the patient’s alanine aminotransferase (ALT) and aspartate aminotransferase (AST) to be within normal limits; however, the alkaline phosphatase (Alk Phos) and serum creatinine were considerably elevated.
Due to the elevation of Alk Phos and the patient’s symptoms, a right upper quadrant (RUQ) sonogram with Doppler was ordered to assess the major organs and vascularity of the abdominal structures. Findings of the sonogram demonstrated slight nodularity of the liver with a coarse and hyperechoic echotexture (Figure 1). Doppler interrogation of the portal vein (PV), hepatic artery (HA), and hepatic veins (HVs) was performed. Portal vein thrombosis was suspected due to limited color and spectral Doppler flow observed within the PV (Figure 2). The HA velocities measured 65.7 cm/s peak systolic and 18.5 cm/s end diastolic with a resistive index (RI) of 0.72 (Figure 3). The HVs were difficult to visualize but exhibited little phasicity of flow with respiration (Figure 4). All findings were consistent with a differential diagnosis of portal vein thrombosis (PVT) secondary to alcohol-induced cirrhosis of the liver.
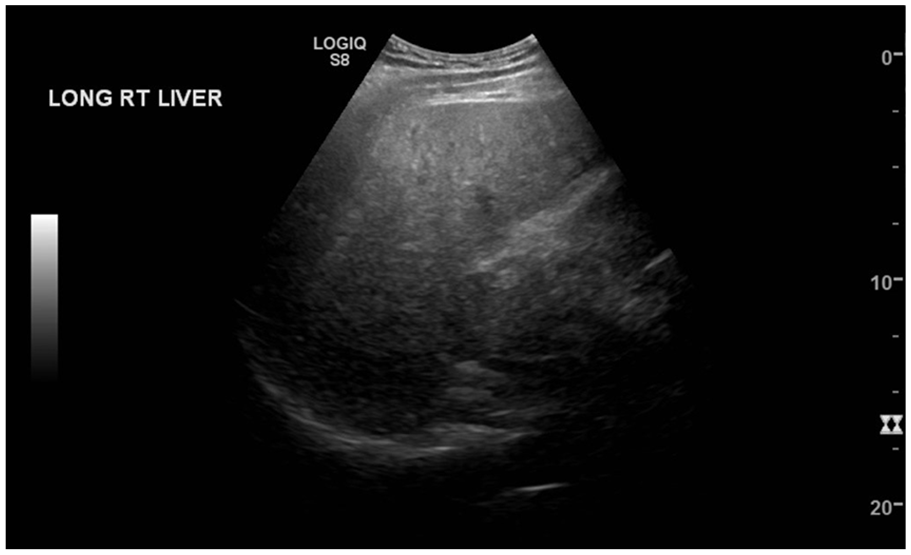
Longitudinal view of the right lobe of the liver demonstrating slight nodularity and a coarse, hyperechoic echotexture.
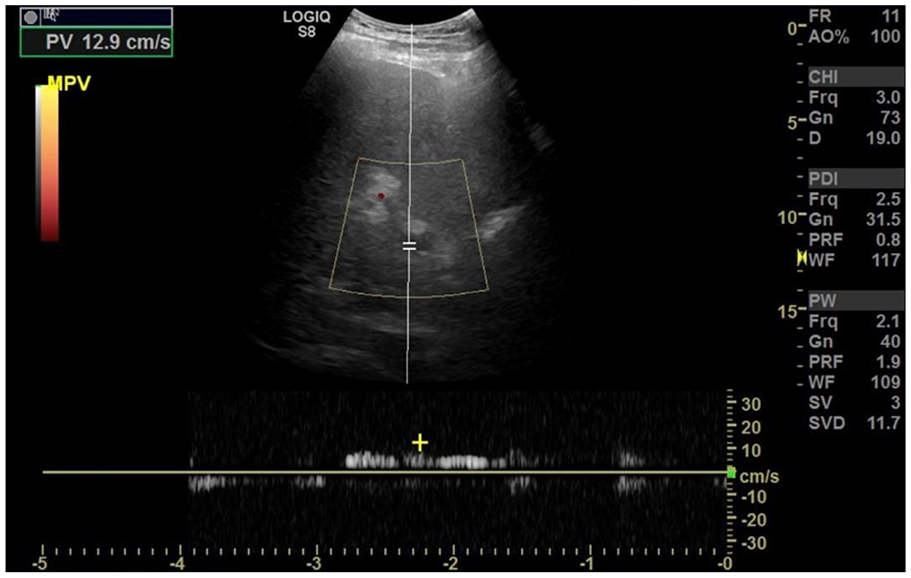
Transverse view of the main portal vein demonstrating limited color and spectral Doppler flow.
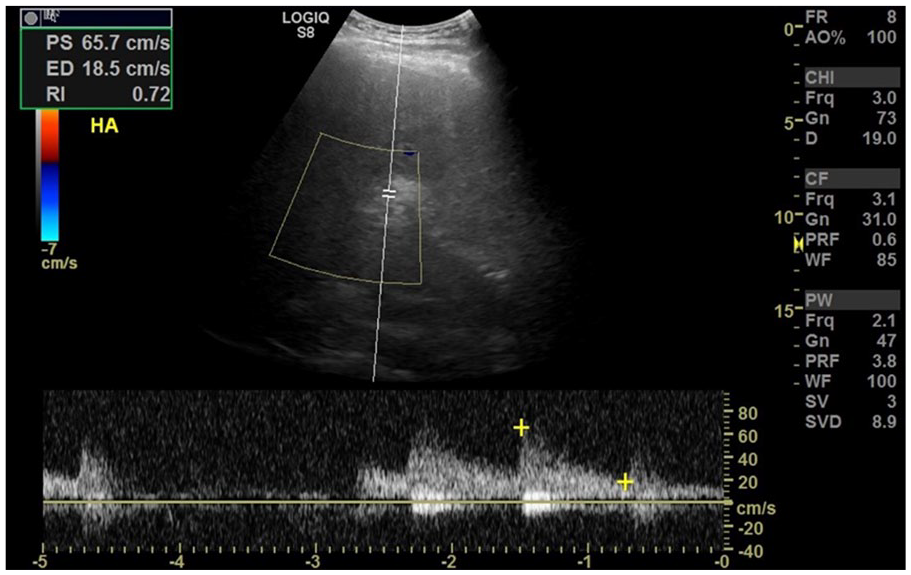
Transverse view of the hepatic artery with a velocity of 65.7 cm/s and a resistive index of 0.72.
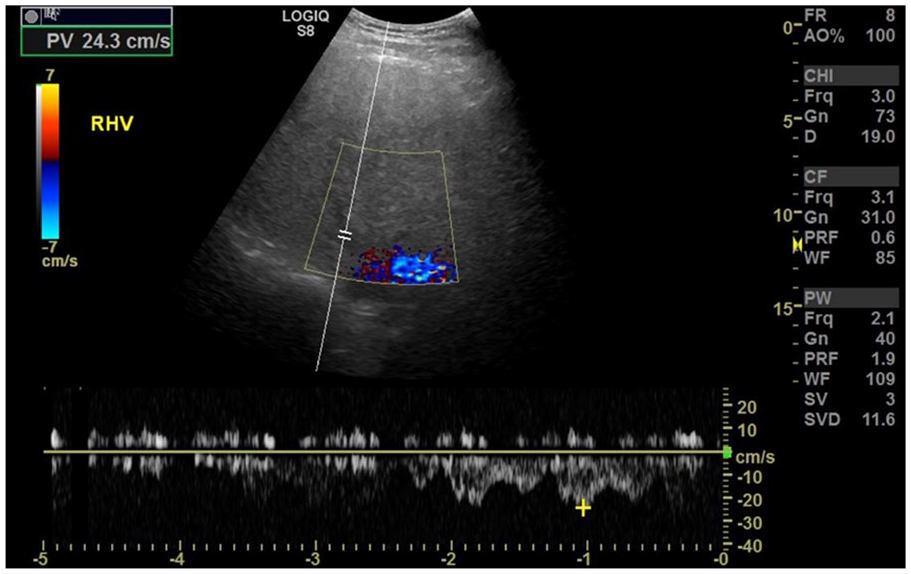
Transverse view of the right hepatic vein demonstrating minimum phasicity of flow with respiration.
After the initial sonographic findings and the clinical assessment of the patient’s symptoms, an esophagogastroduodenoscopy was completed the same day to assess for the possibility of bleeding varices contributing to the hematemesis, secondary to possible portal hypertension, due to alcohol-induced cirrhosis. The findings of the endoscopy revealed large fundal varices that were present along the stomach, as well as multiple grade III varices located in the distal portion of the esophagus.
Immediately following the esophagogastroduodenoscopy, an endoscopic sonogram with fine-needle aspiration (EUS-FNA) was performed. During the procedure, an endoscope was passed through the mouth and advanced to the second portion of the duodenum. There were several 30 × 30-mm varices in the stomach. The varices were successfully embolized using embolization coils, along with Dermabond injections. The varices were firm upon completion of the procedure and a possible residual varix was present, without stigmata. No complications were reported, and the patient was scheduled for a follow-up abdominal sonogram with Doppler and a computed tomogram (CT) to be completed within the following days.
The diagnostic characteristics of the spleen seen on CT (completed 8 days after the EUS-FNA procedure) were consistent with mild splenomegaly measuring 5.9 × 16.5 × 16.6 cm. Embolization coils were visualized on the CT of the abdomen (Figure 5). Ascites and liver nodularity were noted on the CT. Despite small focal areas of cystic lesions seen on the kidneys and 0.2-cm nonobstructing renal calculi, other major organs had a normal appearance.
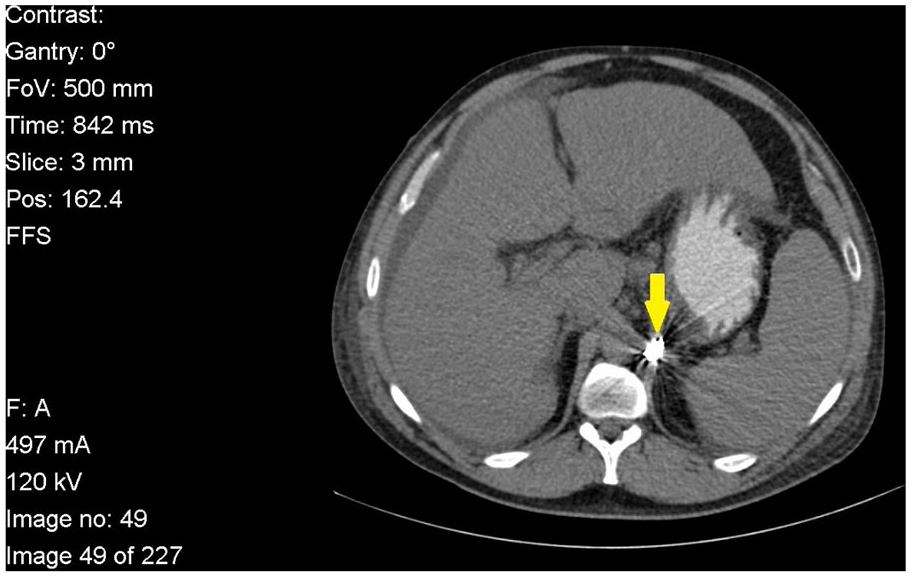
Cross-sectional computed tomography image of the liver, spleen, and gastric embolization coils (yellow arrow). This demonstrates liver nodularity, mild splenomegaly, and ascites.
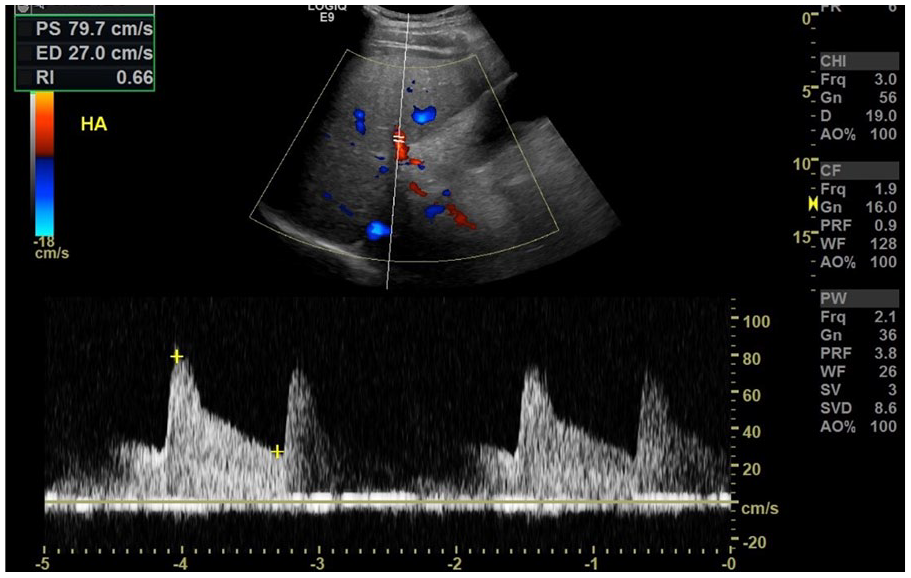
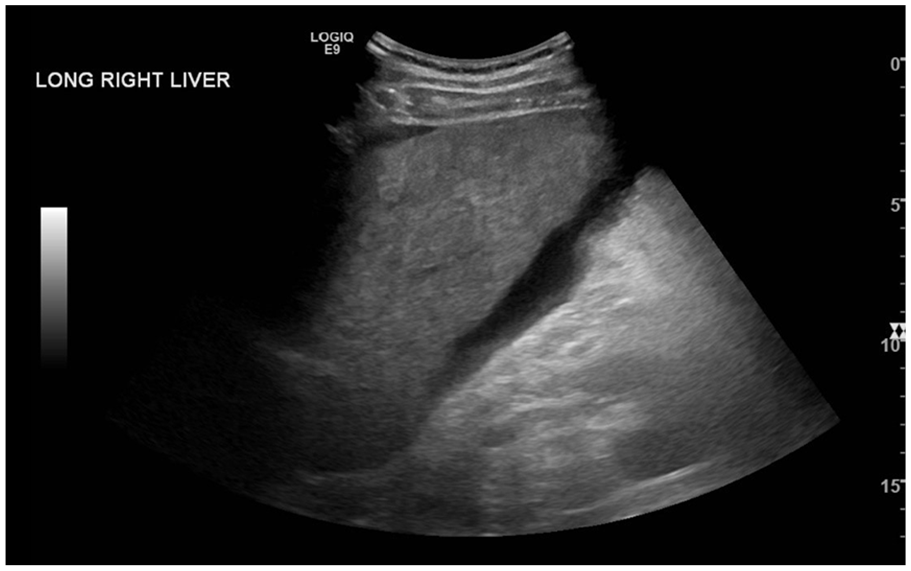
The follow-up sonographic examination (completed 9 days post EUS-FNA procedure) demonstrated slight nodularity of the liver with increased parenchymalechogenicity; no intrahepatic ductal dilatation was noted. However, mild to moderate ascites was present within the peritoneal cavity, as well as a thickened gallbladder wall measuring 0.46 cm. Within the gallbladder, hyperechoic sludge was also visualized (Figure 6). The sonographic characteristics of the spleen were consistent with mild splenomegaly, measuring 14.25 cm. Doppler interrogation revealed that HA resistance had decreased to an RI of 0.66, indicating an improvement. This was compared to a lower resistant Doppler flow profile demonstrated on the previous examination (Figure 7). The HVs were better visualized showing normal Doppler waveforms (Figure 8). The PVs demonstrated improved antegrade flow (Figure 9). Despite the new sonographic findings related to the gallbladder and ascites, color and spectral Doppler assessment showed an improvement in the portal venous, hepatic artery, and venous flow compared to the initial study.
Longitudinal view of the right lobe of the liver. Liver nodularity with a hyperechoic echotexture and ascites is observed.
Spectral Doppler interrogation of the hepatic artery demonstrating velocities at 79.7 cm/s and a resistive index of 0.66.
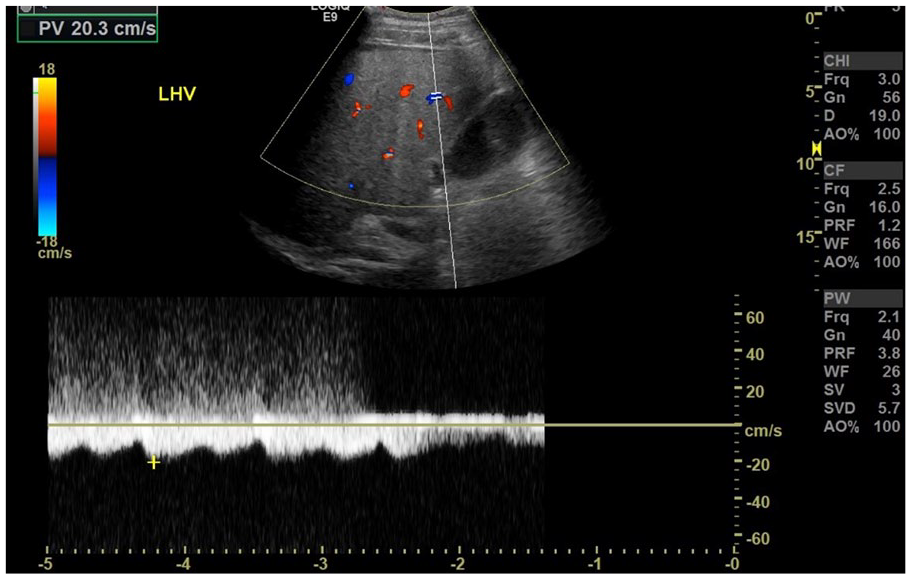
Spectral Doppler interrogation of the left hepatic vein demonstrating improved hepatic vein flow.
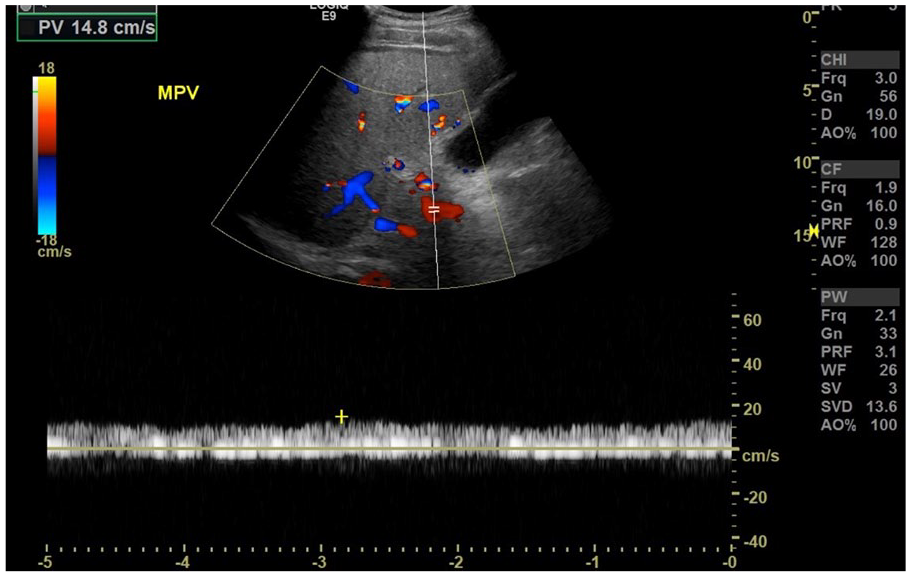
Spectral Doppler interrogation of the main portal vein demonstrating improved, continuous flow.
Twenty-three days after the first EUS-FNA, the patient had another successful EUS-FNA ligation and adhesion procedure performed to treat a residual varix. Recurring outpatient office visits with the gastrointestinal (GI) physician, a complete abdominal Doppler interrogation, and endoscopic examination were scheduled every six months to monitor the stability/progression of disease. These follow-up physician visits allowed for a determination of the presence and prognosis of esophageal varices. It also documented any sonographic changes that were associated with hepatocellular carcinoma, secondary to alcohol-induced cirrhosis.
Three years after the initial diagnosis, the patient remained completely abstinent from alcohol with a recent sonogram confirming that the disease process was stable. Sonographic findings exhibited a mildly nodular and somewhat hyperechoic liver with no ascites present (Figure 10). Spectral Doppler interrogation of the main, right, and left PVs revealed normal antegrade flow (Figure 11). The common hepatic artery (CHA) showed a normal arterial upstroke and normal flow resistance (Figure 12). The right, left, and middle HVs were visualized with normal triphasic waveforms varying with respiration (Figure 13). Mild splenomegaly remained present on the sonogram with the spleen measuring 13.30 cm. The gallbladder, biliary system, and all other major organs appeared had a normal sonographic appearance. Due to the patient’s lifestyle modifications and abstinence from alcohol, a referral for invasive treatment options, such as a transjugular intrahepatic portosystemic shunt (TIPS) and liver transplant evaluation, was not needed.
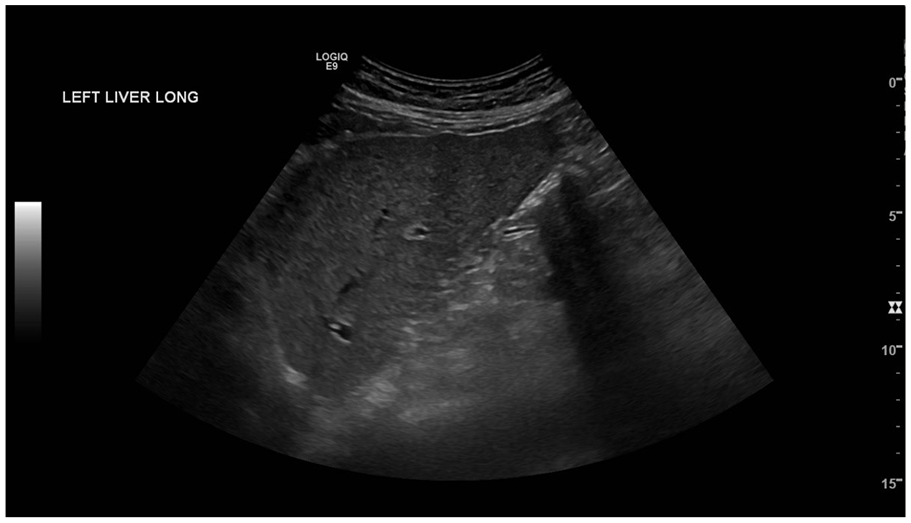
Longitudinal view of the left lobe of the liver exhibiting a mildly nodular and echogenic echotexture with no evidence of ascites.
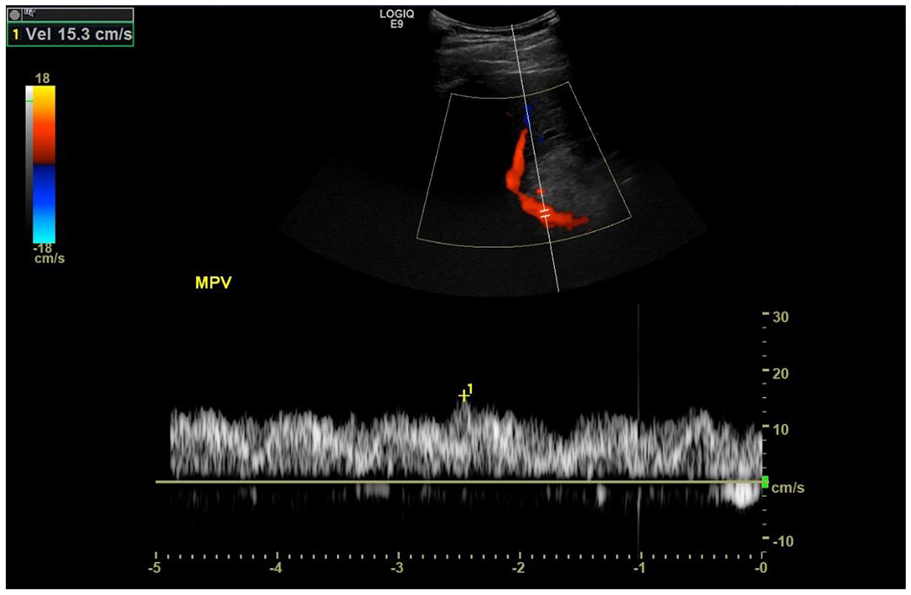
Spectral Doppler interrogation of the main portal vein demonstrating normal continuous, monophasic flow.
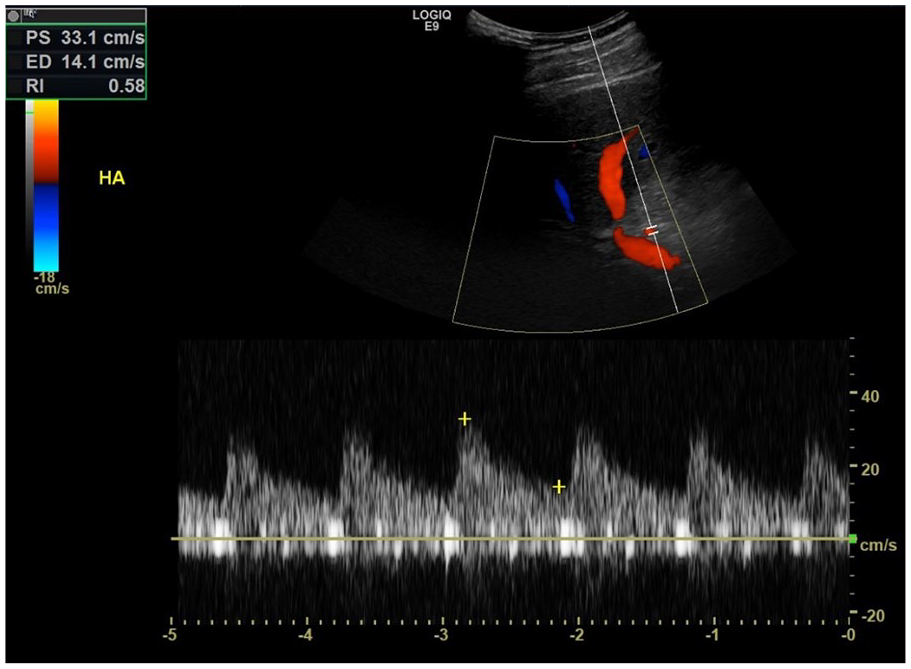
Spectral Doppler interrogation of the hepatic artery demonstrating normal low-resistance flow at a velocity of 33.1 cm/s and a resistive index of 0.58.
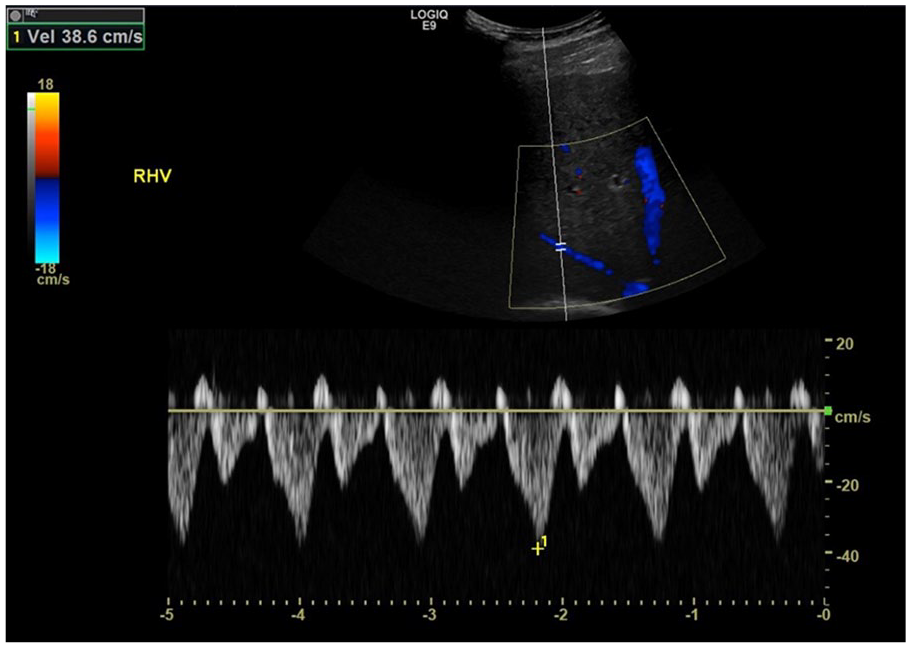
Spectral Doppler interrogation of the right hepatic vein demonstrating normal triphasic waveforms, varying with respiration.
Discussion
The development and progression of ALD relate directly to excessive ingestion of alcoholic beverages. Excessive ingestion is defined as more than 14 alcoholic drinks per week for men and 7 alcoholic drinks per week for women, for generally 10 to 12 years, with one alcoholic drink containing 12 to 14 g of alcohol.2,3 Research into the pathogenesis of ALD has found that excessive alcohol ingestion will first lead to steatosis, the accumulation of fat in hepatocytes. 2 Research has shown fatty acid mitochondrial β-oxidation is inhibited in the hepatocytes secondary to alcohol consumption. As the disease progresses, the fatty infiltration and disruption of the liver’s oxidation mechanisms cause scarring (cirrhotic changes) in the liver. 2 Ethanol has also been proven to inhibit the activation of enzymes (AMP-activated protein kinase, most notably), directly related to fat metabolism. 2 As fat accumulates in the hepatocytes and is not metabolized, liver tissues stiffen and cirrhotic changes intensify. Portal venous blood flow then becomes disrupted, leading to portal hypertension and increased risk of varix formation, to relieve the portal hypertension. It is important to note that patients can have a fatty liver without evolution of cirrhosis. If the condition is diagnosed early, lifestyle modifications can be made in an effort to preserve liver function and prevent scarring. Unfortunately, determining a patient’s predisposition for developing ALD is challenging. According to Younossi and Henry, 3 no modern diagnostic tool exists that directly assesses an individual’s susceptibility of developing the disease. Sonography is a vital diagnostic tool in the monitoring of ALD patients, as hepatocellular carcinoma (HCC) evolves in approximately 1% to 2% of those with ALD and alcohol-induced cirrhosis. 4 Although determining an individual’s susceptibility for developing ALD is not well understood, effectively evaluating and following patients with the disease using noninvasive approaches (such as gray-scale sonography and Doppler) has great diagnostic promise.
Understanding the basic pathogenesis of ALD and the sonographic markers allows for accurate staging of the disease and provides prognostic information for therapy planning. 5 Sonographic characteristics of ALD include nodularity of the liver surface, increased echogenicity, and a coarse echotexture. 5 The sonographic appearance of liver surface nodularity alone has a high diagnostic correlation with cirrhosis. Research conducted by Colli et al. 6 into asymptomatic patients with cirrhosis reported that sonography of the liver surface demonstrating nodularity has a 95% specificity. As the disease progresses, an enlargement of the left and caudate lobes will occur. 7 In the latter stages of the disease, ascites will be present, which has clinical significance, as ascites is a sign of the body decompensating. Decompensation is another term for the functional deterioration of body systems. The sonographic presence of splenomegaly (a spleen measuring greater than 13 cm) suggests clinically significant portal hypertension in patients compensating for the disease. Splenomegaly acts as a predictor for the manifestation of an esophageal varix. Research suggests splenomegaly has a high sensitivity and variable specificity, ranging from 50% to 80%, in cases of cirrhosis where clinically significant portal hypertension is present. 8
Color and spectral Doppler interrogation of the hepatic vasculature (portal veins, hepatic veins, and hepatic artery) complements gray-scale sonography in patients with suspected cirrhosis related to ALD. Due to the fibrotic changes in the liver parenchyma related to ALD, it can be difficult for ultrasound to penetrate the liver tissue. For example, the lack of penetration when performing Doppler interrogation studies can cause vessels to demonstrate an apparent lack of flow. The sonographer should evaluate the Doppler angle to ensure the angle of insonation is within the range of 45 to 60 degrees. If an ideal angle of insonation cannot be achieved, proper documentation is needed. With sonography being an operator-dependent imaging modality, it is imperative the sonographer use all maneuvers (patient breathing exercises, alternative patient positions, knowledge of instrumentation and equipment capabilities) to provide accurate and diagnostic images and data.
Gaiani et al. 9 determined liver surface nodularity, combined with a spectral Doppler portal vein mean velocity below 12 cm/s, holds 80% accuracy in differentiating between chronic hepatitis with severe fibrosis and cirrhosis. Furthermore, sonographic evidence of porto-collateral circulation (such as paraumbilical vein collateral flow) and inversion of flow in the portal system is 100% specific in representing clinically significant portal hypertension related to the cirrhosis. 8 While the present case study did not demonstrate paraumbilical vein collateral flow, sonographically, this would appear as flow in the anterior ridge of the falciform ligament. Sonographic presence of hepatofugal portal venous flow would also suggest porto-collateral venous flow in an effort to relieve portal hypertension. Spectral Doppler flow profiles in the various hepatic vessels exhibit classic characteristics of portal hypertension, secondary to cirrhosis caused by ALD. Thorough evaluation of the portal venous system is the main role of sonography in the evaluation and follow-up of patients with alcohol-induced cirrhosis. While assessing main portal vein velocity can serve as a marker for portal hypertension, research suggests assessment of velocity alone provides highly variable information. 10 A study of 118 patients revealed that main portal vein velocities can range from 7 to 83 cm/s, underscoring that velocity measurement alone are not sufficient to stage portal hypertension. 11 Evaluating portal venous flow patterns serves as a valuable diagnostic tool, in conjunction with velocity assessment. Normal portal venous flow appears hepatopetal, constant, and monophasic. Patients with cirrhosis and portal hypertension may exhibit heptaofugal flow, and the flow is not constant. 9 A complete absence of portal venous flow, with Doppler, may indicate portal vein thrombosis and should be included as a differential diagnosis until effectively eliminated.
Hepatic vein waveforms appearing biphasic or monophasic can predict cirrhosis in patients, but they are not as reliable as portal venous flow characteristics. 9 Hepatic vein waveforms with decreased phasicity and those that present as biphasic or monophasic warrant documentation. Interrogation of the HA should be included in the abdominal Doppler assessment, with special attention focused on the RI. Patients with alcohol-induced cirrhosis commonly demonstrate an elevated HA resistive index, as demonstrated in the case presented. Colli et al. 12 conducted a study of 116 patients and discovered the HA resistive index, measured with Doppler sonography, had an 84% sensitivity when it came to correlating an elevated RI (>0.70) with liver disease.
Transient elastography (TE) is an additional tool to assess liver stiffness, which correlates with degree of liver fibrosis. Since liver tissue becomes fibrotic as part of the pathogenic process of ALD, measuring liver stiffness aids in diagnosing cirrhosis, using a noninvasive approach. TE works by generating a low-frequency elastic wave (50 Hz) and a high-frequency ultrasound wave (1500 m/s), which allows for measurement of the transmission into the liver parenchyma. 13 TE does have limitations in discriminating lower stages of fibrosis, but overall, TE does differentiate cirrhotic from noncirrhotic livers with 77% to 100% sensitivity and 78% to 98% specificity. 13 In a study of 291 patients, TE had a sensitivity of 90.4% for detecting cirrhosis secondary to various etiologies (ALD included) and a sensitivity of greater than 10% compared to a routine sonogram. 13 Obtaining a liver stiffness measurement with TE, in conjunction with a sonographic study, detected cirrhosis in 96.1% of patients evaluated. 13
Once a definitive diagnosis of ALD occurs, treatment planning will commence. A number of factors contribute to each patient’s care plan. Disease management options focus largely on repairing liver damage as much as possible and treatment of symptoms. It is important to note the only cure for alcoholic cirrhosis, secondary to ALD, is liver transplantation. Many researchers discuss the controversy of transplanting a healthy liver into a patient with alcoholic cirrhosis secondary to ALD, given that some consider this a “self-inflicted ailment.” 1 Outcome data on liver transplantation for patients with known alcoholic cirrhosis are equivalent to others receiving liver transplants, but only 5% of those with alcoholic cirrhosis receive a liver transplant. 14 There is risk for relapse after liver transplant, and research collected between 1988 and 2009 indicates that relapse rates range from 10% to 50%. 15 Overall, the one-year survival rate posttransplant for those with alcoholic cirrhosis is 86%, with rates lower at 59% ten years posttransplant. When 10-year survival rates are broken down into relapse versus nonrelapse cases, the difference in survival rate is 45.1% and 85.5%, respectively, and many deaths in the relapse category are attributed to malignant tumors and adverse cardiovascular events. 15 While a liver transplant does cure alcoholic cirrhosis, it does not cure the underlying alcoholism, which may explain relapse rates that can approach 50%.
For patients with ALD, the focus will be on management of symptoms rather than achieving a cure. Lifestyle changes, varix treatment options, and/or placement of a transjugular intrahepatic portosystemic shunt (TIPS) are commonly used to manage symptoms. Of the disease management options, lifestyle modifications can include complete abstinence from alcohol or a reduction in consumption. Abstinence from alcohol, for a minimum of 6 months, is a common requirement at many centers for liver transplant candidates. 16 Abstinence from alcohol leads to resolution of benign steatosis, allowing liver oxidation mechanisms to effectively resume, even in patients who exhibit signs of decompensation due to alcohol-induced cirrhosis. 16 Routine monitoring of ALD patients who are abstaining from alcohol and making lifestyle changes is accomplished with regular sonograms. Patients who commit to alcohol abstinence can show sonographic signs (hepatofugal to hepatopetal portal venous flow, decrease or resolution of ascites) of disease improvement over time.
Patients decompensating and needing immediate relief of hematemesis due to bleeding varices may undergo endoscopic ultrasound procedures to control varices and stop bleeding. The deployment of coils and injected glue are used to obliterate varices and lower renewed bleeding rates compared to band ligation procedures. 17 In a study by Anastasiou and Berzin, 18 the renewed bleeding rate with the combination therapy of coil obliteration and glue injections was 16.6% after a six-month follow-up. Placement of a TIPS is a secondary procedure used to control variceal bleeding for patients with severe liver disease and subsequent portal hypertension, given that pharmacologic and endoscopic treatments have failed. 19 A study of over 900 patients, across 13 randomized clinical trials, concluded that recurrent variceal bleeding rates, after TIPS, were 9% to 40% compared to 20% to 60% with endoscopic ligation treatment. 19 Longitudinal data on the prognosis for patients who undergo a TIPS procedure can vary. A study of 495 patients (91 patients received a TIPS due to alcoholic cirrhosis) found that the one- to three-year survival rates post-TIPS placement were 93.4% and 77.2%, respectively. 20 The TIPS procedure has a higher risk of hepatic encephalopathy formation postprocedure compared to endoscopic ligation procedures, but there is a lower risk for renewed variceal bleeding.
Conclusion
ALD, a precursor to alcohol-induced cirrhosis, continues to be an epidemic within modern society. This degenerative liver disease does not present until later in life and typically during the later stages of disease, which gives patients less time to manage or reverse tissue damage. The external presentation may differ, but internally, the liver will become echogenic, coarsened, and possibly be surrounded by ascites. Without obvious clinical indications, cirrhosis may go unnoticed for years.
The diagnostic and therapeutic aspects of ALD coincide due to the lack of available treatments. Diagnosis and serial evaluation of cirrhosis are highly dependent on sonography due to the easy access of various points of interest, including the right, left, and caudate lobes of the liver; portal and hepatic venous systems; and the spleen. Sonography can also guide interventional procedures to aid in correcting the functions of affected areas indicated by laboratory values, as well as monitor patients after liver transplant.
Footnotes
Acknowledgements
The authors would like to thank the University of Arkansas for Medical Sciences Department of Radiology for providing the images and case information for this paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
