Abstract
Objective:
The objectives were to assess factors associated with a higher likelihood of predicting acute coronary atherothrombosis (ACA) in non-ST-segment elevation myocardial infarction (NSTEMI), evaluate the impact of adding echocardiographic data, and develop an algorithm that would reduce overutilization of emergent angiography.
Methods:
Data were retrospectively analyzed on a patient cohort presenting to an emergency department of an urban community hospital with NSTEMI from October 1, 2015, to July 31, 2018. The inclusion criterion was any adult patient with a first-time, primary diagnosis of NSTEMI without high-risk features. The main outcome variables were the presence of ACA on angiography.
Results:
Seventy-three patients with NSTEMI were included in this study. Logistic regression analysis identified the following independent variables useful for predicting ACA: age, wall motion abnormality on echo, and levels of low-density lipoprotein. The model’s overall fit was highly significant (P = .0012).
Conclusion:
An integrative approach was demonstrated for the management of patients with NSTEMI presenting to the hospital. Although the positive predictive value of echo in predicting ACA was limited, when combined with demographic attributes and risk factors, it proved to be successful in determining the need for angiography in patients with NSTEMI.
Coronary artery disease (CAD) is the most common type of heart disease and the leading cause of death in both men and women in the United States. 1 The clinical manifestations span stable angina and acute coronary syndrome (ACS). The rupture of stable atherosclerotic plaques initiates thrombosis/emboli, causing acute coronary atherothrombosis (ACA); this type of etiology is classified as a type 1 acute myocardial infarction. 2 Depending on the degree of ACA, it is useful to “visualize” acute coronary syndrome on a spectrum ranging in severity. Three distinct diagnostic entities constitute the spectrum: unstable angina (UA), non-ST-segment elevation myocardial infarction (NSTEMI), and ST-segment elevation myocardial infarction (STEMI). 3
While the diagnosis and treatment of STEMI are unambiguous, 4 the treatment of UA/NSTEMI remains a challenge for clinicians in a hospital setting. Similar to the management of STEMI, early detection and intervention in the subgroup of patients with ACA presenting to the emergency department (ED) with NSTEMI have been associated with improved outcomes.5–9 Currently available high-sensitivity troponin assays have increased the frequency of diagnosing NSTEMI. This has led to overutilization of angiography.10–12 It is challenging to accurately predict ACA in patients with NSTEMI. It is estimated that as many as 30% of patients referred for invasive coronary angiography demonstrate less than significant/critical stenosis that may have been better identified using noninvasive imaging (computed tomography angiography, and/or noninvasive fractional flow reserve). 13
Echocardiographic (echo) evidence of regional wall motion abnormality (RWMA) is frequently seen in patients with NSTEMI. The reported sensitivity and specificity of echo in one study were 90.9% and 100%, respectively. 14 However, the implications of echo as a potential discriminator of ACA in patients presenting to the ED are largely unknown.
The purpose of this study was to assess the potential diagnostic capability of echo and its effect on the management of patients with a presumed diagnosis of NSTEMI. The study objective was to assess factors associated with a higher likelihood of predicting ACA in NSTEMI after incorporating data from echocardiography. A secondary objective was to develop an algorithm that could potentially reduce overutilization of angiography.
Methods
Study Design, Setting, and Population
The study received expedited institutional review board (IRB) approval from the review committee at New York Presbyterian–Brooklyn Methodist Hospital (NYP-BMH) in September 2018, due to limited risk to the study population. The only identified potential risk posed to study participants was breech of privacy data. Patients’ protected health information was deidentified to protect privacy, and the coded key to the list was kept on secured venues, such as an encrypted flash drive. IRB exemption was obtained for informed patient consent prior to study enrollment due to nonexistent risks posed to patients by performing a chart review.
This study was a noninterventional, observational, retrospective cohort chart review of patients who were admitted through the ED of NYP-BMH for suspected NSTEMI. The patients reviewed were discharged with a final diagnosis of type 1 NSTEMI from October 1, 2015, to June 31, 2018.
Study Protocol
A search query of all patients with a final discharge diagnosis of type 1 NSTEMI was executed on electronic medical records (EMRs) in the Cerner medical records database at NYP-BMH. The goal was to identify within these patients those who received an echo during their hospital course. Inclusion criteria included any adult patient with a first-time, primary diagnosis of type 1 NSTEMI without signs or symptoms of transient ST-segment elevation, hemodynamic instability, refractory chest pain, or malignant arrhythmia. Patients with documented impaired renal function, prior myocardial infarction, congestive heart failure, or cardiac conditions that interfere with echo RWMA were excluded (Figure 1).
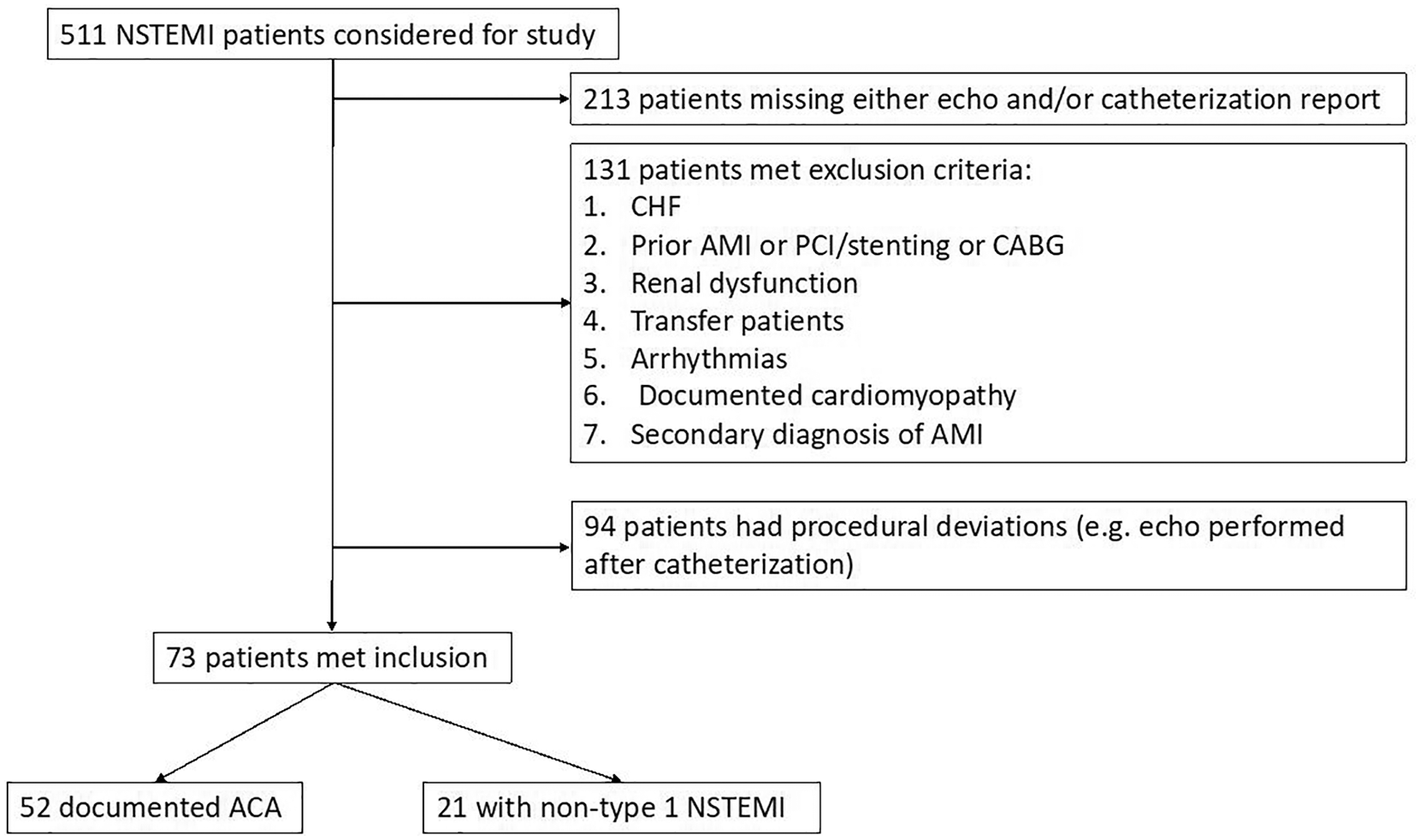
Flow diagram of patient selection.
The following data were extracted from the EMR: age, sex, ethnicity, body mass index (BMI), date/time of presentation, presence of persistent/progressive chest pain (defined as similar chest pain or unstable angina within the past month before the episode inciting the ED visit), medical and social history, serum laboratory data (such as lipid levels, troponin levels), 12-lead electrocardiogram findings, thrombolysis in myocardial infarction (TIMI) risk score, time and magnitude of troponin draws, and report of the transthoracic echo. All data except those from the echo and angiography were obtained in the ED during initial workup of the patient unless otherwise noted. The data extracted from the echo report consisted of the estimated ejection fraction (as calculated using Simpson’s rule) and presence of RWMA (assessed according to the American Heart Association guidelines and graded as normokinesis, akinesis, or dyskinesis).15,16 The echos were performed on the general medical floors upon admission and before left-heart catheterization by the patient bedside. A credentialed and trained sonographer using a protocol in line with the American Society of Echocardiography/Intersocietal Accreditation Commission performed the scans, and it was interpreted by a board-certified cardiologist on staff at the hospital. The data extracted from the echo report consisted of the estimated ejection fraction and presence of RWMA as obtained from the following cardiac views: parasternal long axis, parasternal short axis, apical four and five chambers, apical two chamber, apical three chamber, subcostal, and suprasternal. The data extracted from the catheterization report consisted of the degree of stenosis visualized and type of intervention performed (e.g., balloon angioplasty and/or stent placement). A data abstraction sheet was made for each EMR and the data tabulated on an Excel spreadsheet for subsequent analysis. All patients admitted to the hospital for acute coronary syndrome at our institution are given an echocardiogram, but not all patients are imaged in time before going for angiography. Thus, the decision as to which patients received an echo before left-heart catheterization was not based on clinical decisions that may be prone to bias.
Measures
Main outcomes
The main outcome variable was the presence of ACA on angiography, defined as a significant coronary occlusion greater than 70% that was amenable to stenting or coronary artery bypass grafting. Descriptive statistics are reported as part of this research. A univariate analysis, as well as a multivariate logistic regression, was completed to develop a prediction model.
Data Analysis
Statistical analysis and sample size
A subset of 73 patients was identified as having received an echo prior to left-heart catheterization with a discharge diagnosis of type 1 NSTEMI.
Primary Analysis
Several 2 × 2 table groupings based on diagnostic test results (either a troponin level above or below 5 ng/mL or the presence of RWMA on echo (ECHO/RWMA(+)) were tabulated for the following groups: the entire cohort, those greater than 65 years of age, and those below the age of 65. A test of proportions (two-tailed z test) comparing the percentage of patients with a false-positive ACA based on troponin I testing was carried out.
Single-variable analysis included comparison of means and proportions (one-tailed Mann-Whitney U and two-tailed z test, respectively) for variables between the ACA and non–type 1 acute myocardial infarction (AMI) groups. In conjunction, the Pearson correlation (r) for variables was reported alongside P values.
Multivariate logistic regression analysis was performed with the dependent variable being the detection of RWMA and independent variable being the various risk factors abstracted from the EMR. The variables were analyzed in pairs as a means to identify statistically significant variables predictive of an ACA. Diagnostic test analysis was performed based on the results of the prediction algorithm.
Secondary analysis
Single-variable analysis included comparison of means and proportions (one-tailed Mann-Whitney U and two-tailed z test, respectively) for variables between the ECHO/RWMA(+) and ECHO/RWMA(–) among those with ACA.
Results
Study Participants
A cohort of 510 patients was assessed for eligibility. Based on the inclusion criteria, 73 patients with NSTEMI were included in the study with 60% males (Table 1). The mean ± SD age was 57.5 ± 21.7 and the mean ± SD troponin I was 3.6 ± 27.2. Of all the patients in the study, 35.6% had RWMA on echo and 65% had ACA on angiography.
Baseline Demographic and Clinical Characteristics of Patients Included in the Study (n = 73). a
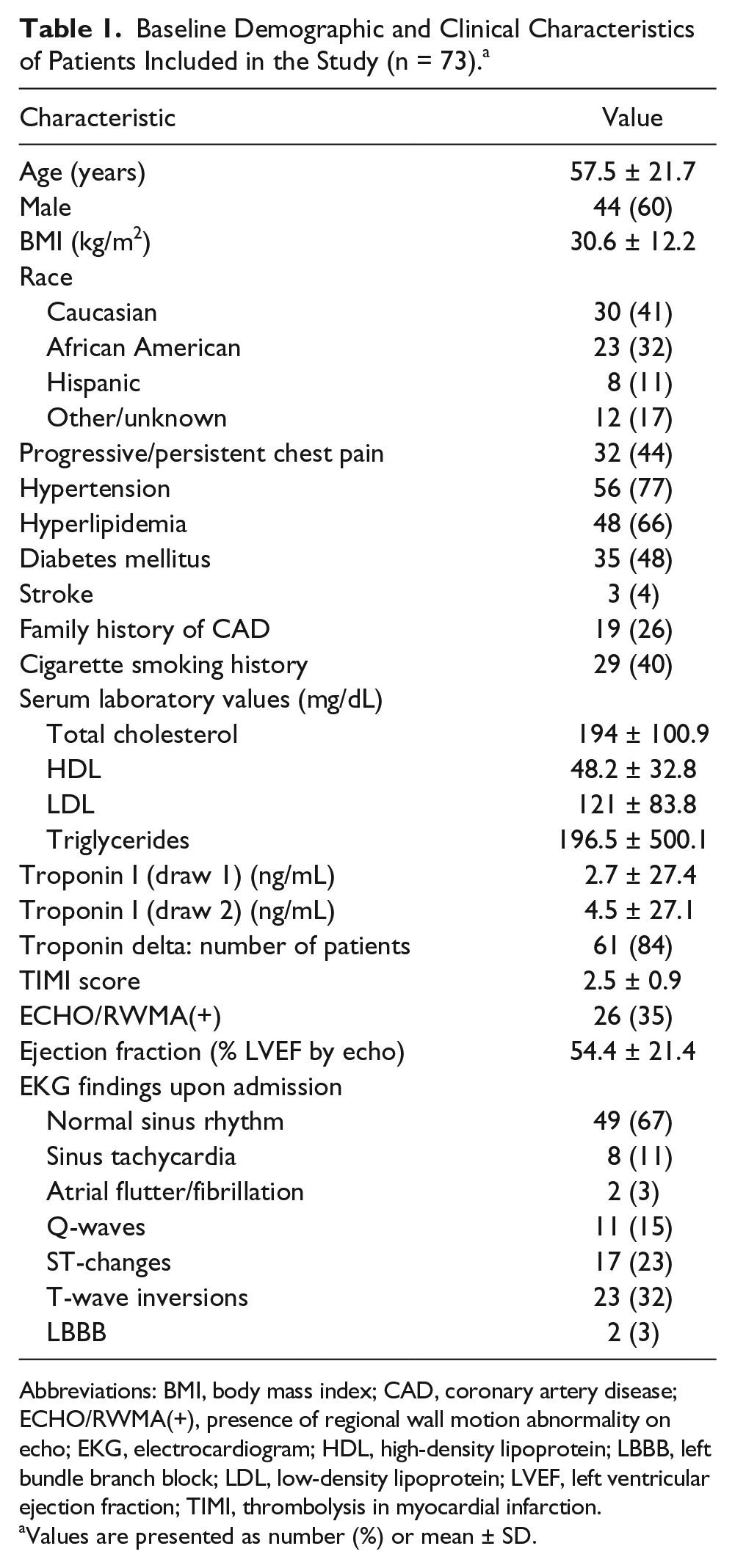
Abbreviations: BMI, body mass index; CAD, coronary artery disease; ECHO/RWMA(+), presence of regional wall motion abnormality on echo; EKG, electrocardiogram; HDL, high-density lipoprotein; LBBB, left bundle branch block; LDL, low-density lipoprotein; LVEF, left ventricular ejection fraction; TIMI, thrombolysis in myocardial infarction.
Values are presented as number (%) or mean ± SD.
Primary Analysis
Troponin I successfully classified 71% of study participants to the ACA group, compared to 30% for ECHO/RWMA(+) (P < .0001) (Table 2). Further analysis revealed a difference in the false-positive ratio of troponin I testing in this study cohort when stratifying patients into two groups, those above and below the age of 65. The difference in false-positive ratio was statistically significant but underpowered (P = .0438) (Table 2). Younger patients were more likely to have a positive troponin than older patients, regardless of having a non–type 1 NSTEMI (32% vs. 18%).
Comparative Analysis of Diagnostic Capability for Troponin Testing vs. Echocardiography.
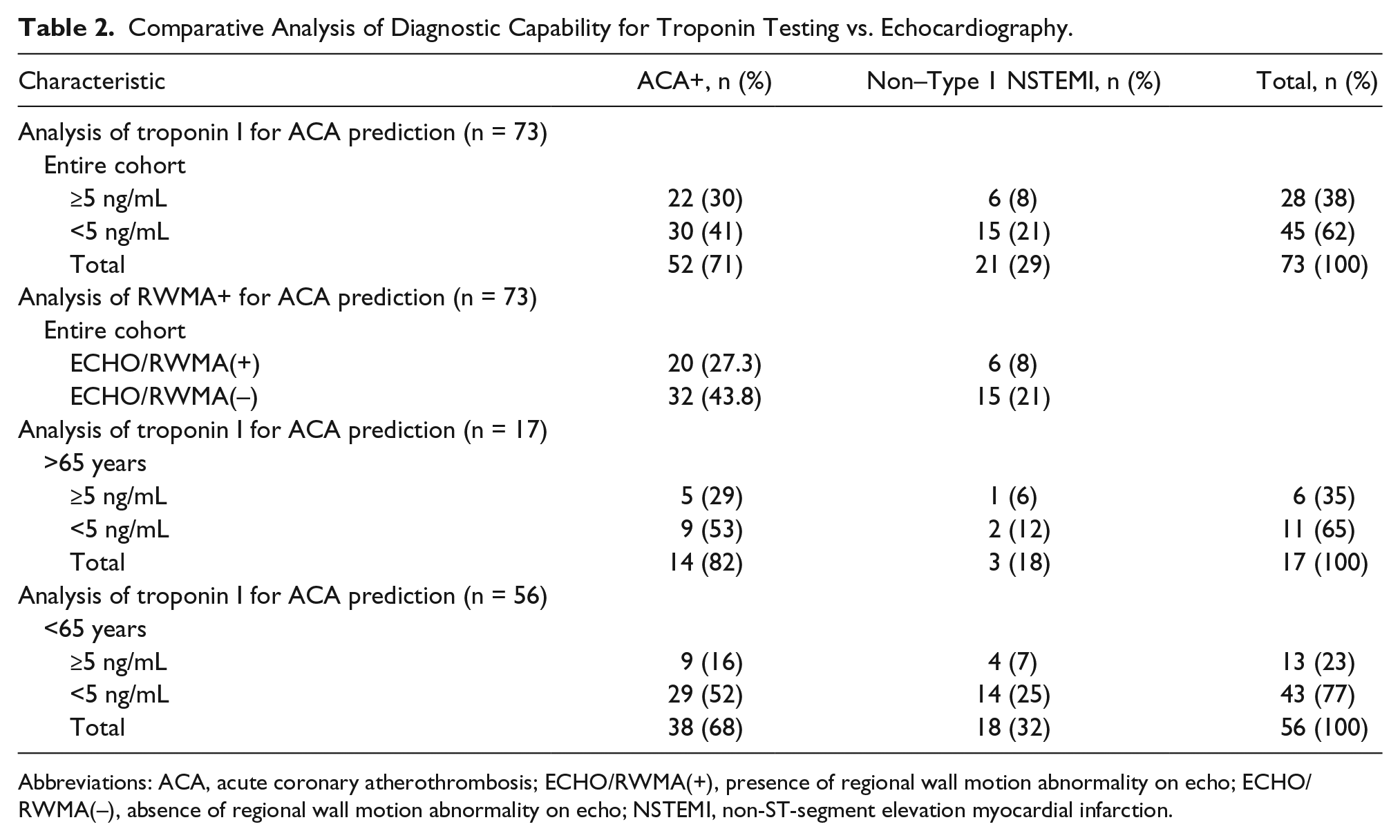
Abbreviations: ACA, acute coronary atherothrombosis; ECHO/RWMA(+), presence of regional wall motion abnormality on echo; ECHO/RWMA(–), absence of regional wall motion abnormality on echo; NSTEMI, non-ST-segment elevation myocardial infarction.
In the single-variable analysis (see Table 3), age (r = 0.14, P = .022), African American descent (P = .015), persistent/progressive chest pain (P = .0071), high-density lipoprotein (HDL; r = 0.15, P = .005), low-density lipoprotein (LDL; r = 0.15, P = .023), and TIMI score (r = 0.20, P = .0032) were statistically significantly associated with ACA. Subsequently, a modeled prediction algorithm was posed for younger patients (n = 56 who were at greater risk for false-positive ACA) and could benefit from early cardiac coronary catheterization (see Table 4). Troponin alone yielded a false-positive rate for ACA of 32% (18/56, Table 2). When the prediction algorithm was applied to the 52 young patients with complete data, a false-positive ACA was seen in only 17% (9/52, Table 5). The number of false positives was reduced after applying the algorithm that also included RWMA. This would presumably reduce unnecessary trips to the catheterization laboratory in younger patients by a little less than 50%.
Patient Characteristics: ACA vs. Non–Type 1 NSTEMI (n = 73). a
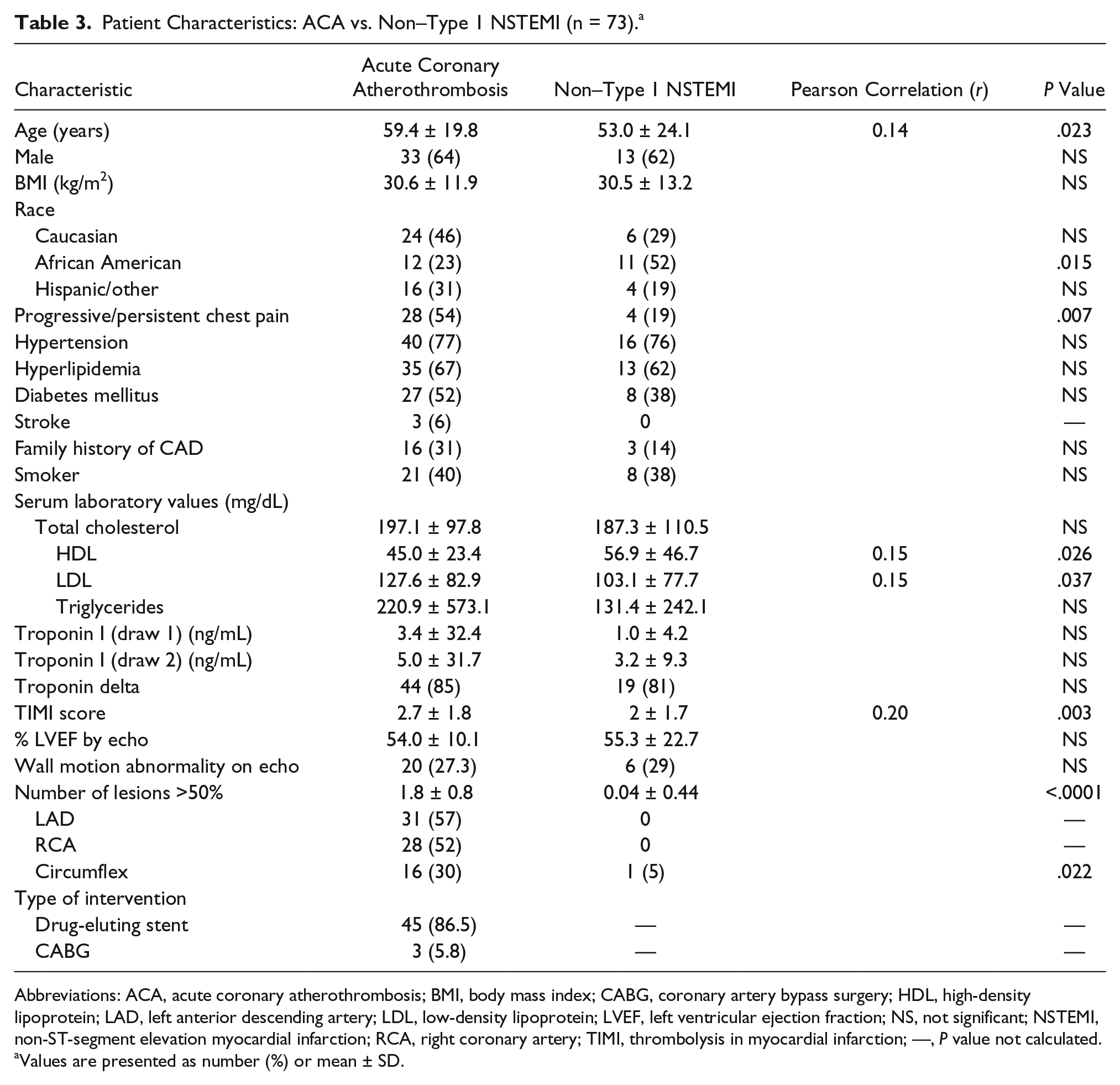
Abbreviations: ACA, acute coronary atherothrombosis; BMI, body mass index; CABG, coronary artery bypass surgery; HDL, high-density lipoprotein; LAD, left anterior descending artery; LDL, low-density lipoprotein; LVEF, left ventricular ejection fraction; NS, not significant; NSTEMI, non-ST-segment elevation myocardial infarction; RCA, right coronary artery; TIMI, thrombolysis in myocardial infarction; —, P value not calculated.
Values are presented as number (%) or mean ± SD.
Summary of the Algorithm for ACA Prediction (n = 52).a
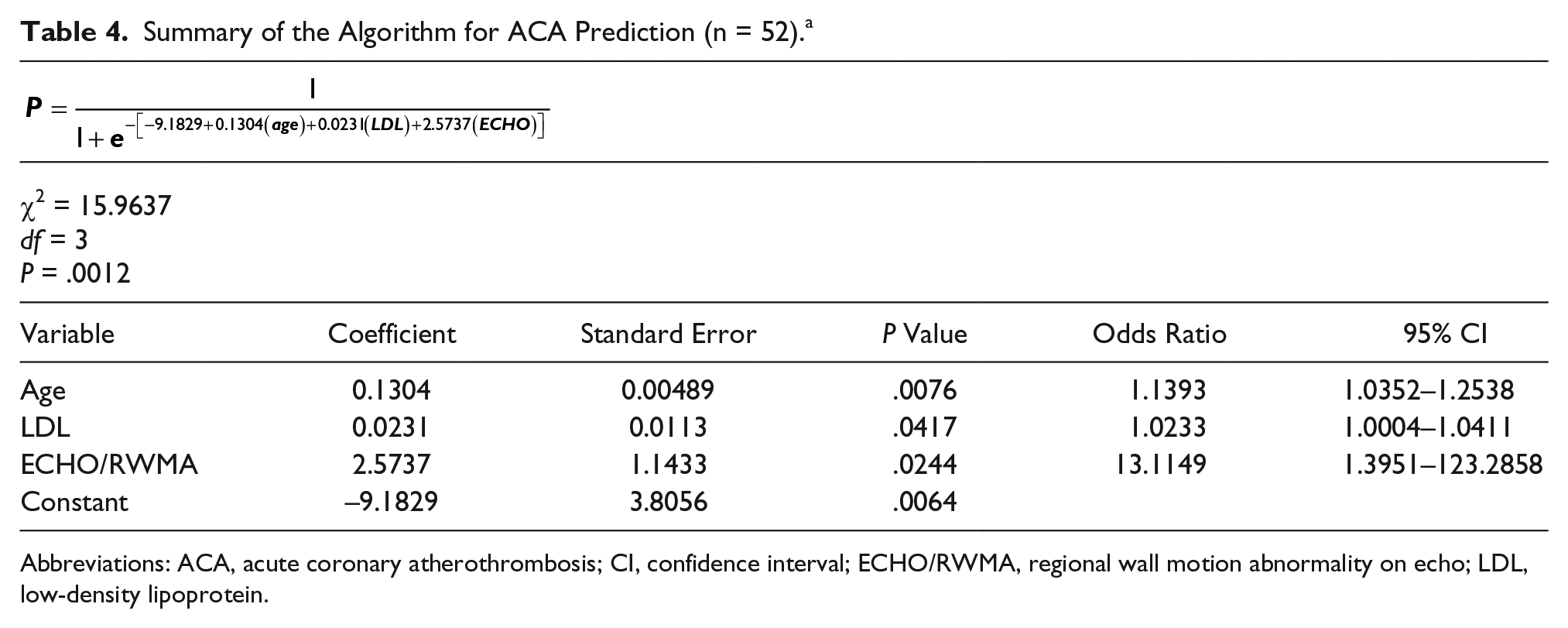
Abbreviations: ACA, acute coronary atherothrombosis; CI, confidence interval; ECHO/RWMA, regional wall motion abnormality on echo; LDL, low-density lipoprotein.
Diagnostic Test Values for ACA Prediction Algorithm (n = 52). a
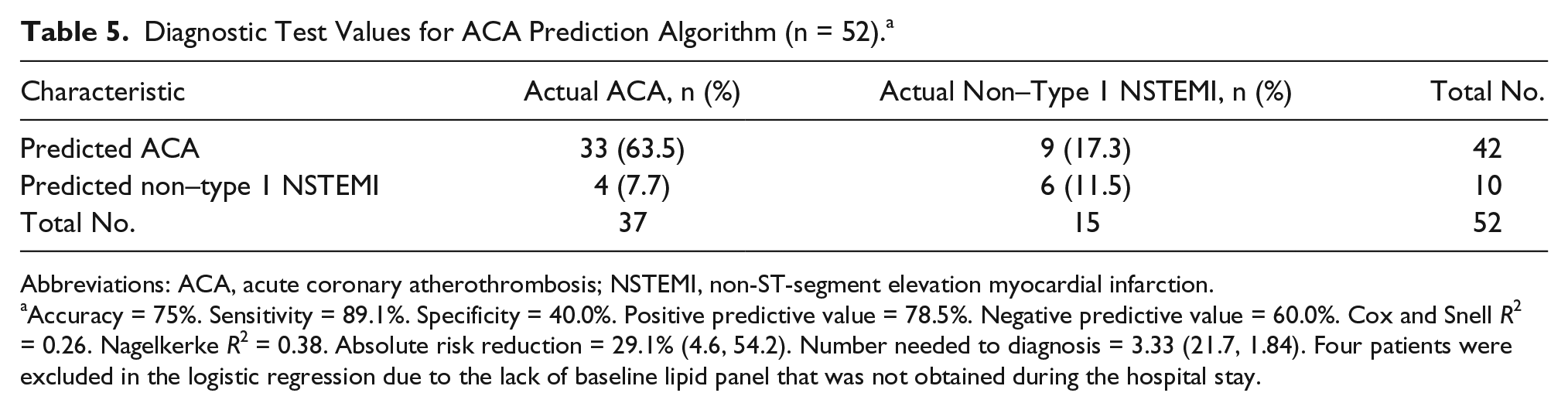
Abbreviations: ACA, acute coronary atherothrombosis; NSTEMI, non-ST-segment elevation myocardial infarction.
Accuracy = 75%. Sensitivity = 89.1%. Specificity = 40.0%. Positive predictive value = 78.5%. Negative predictive value = 60.0%. Cox and Snell R2 = 0.26. Nagelkerke R2 = 0.38. Absolute risk reduction = 29.1% (4.6, 54.2). Number needed to diagnosis = 3.33 (21.7, 1.84). Four patients were excluded in the logistic regression due to the lack of baseline lipid panel that was not obtained during the hospital stay.
Secondary Analysis
To understand the full diagnostic capability and limitations of echo, additional analysis was done to more thoroughly characterize the population within NSTEMI patients with positive ECHO/RWMA. Single-variable analysis revealed BMI (r = 0.20, P = .007), Caucasian race (P = .007), troponin delta (defined as a 20% change in value between blood draws) (P = .002), left ventricular ejection fraction (LVEF) (r = 0.27, P < .0001), and critical stenosis in the left anterior descending artery (LAD) as statistically significant variables between ECHO/RWMA(+) and ECHO/RWMA(–) in those with demonstrated ACA (Table 6).
Patient Characteristics Between ECHO/RWMA(+) vs. ECHO/RWMA(–) Among Those With ACA. a
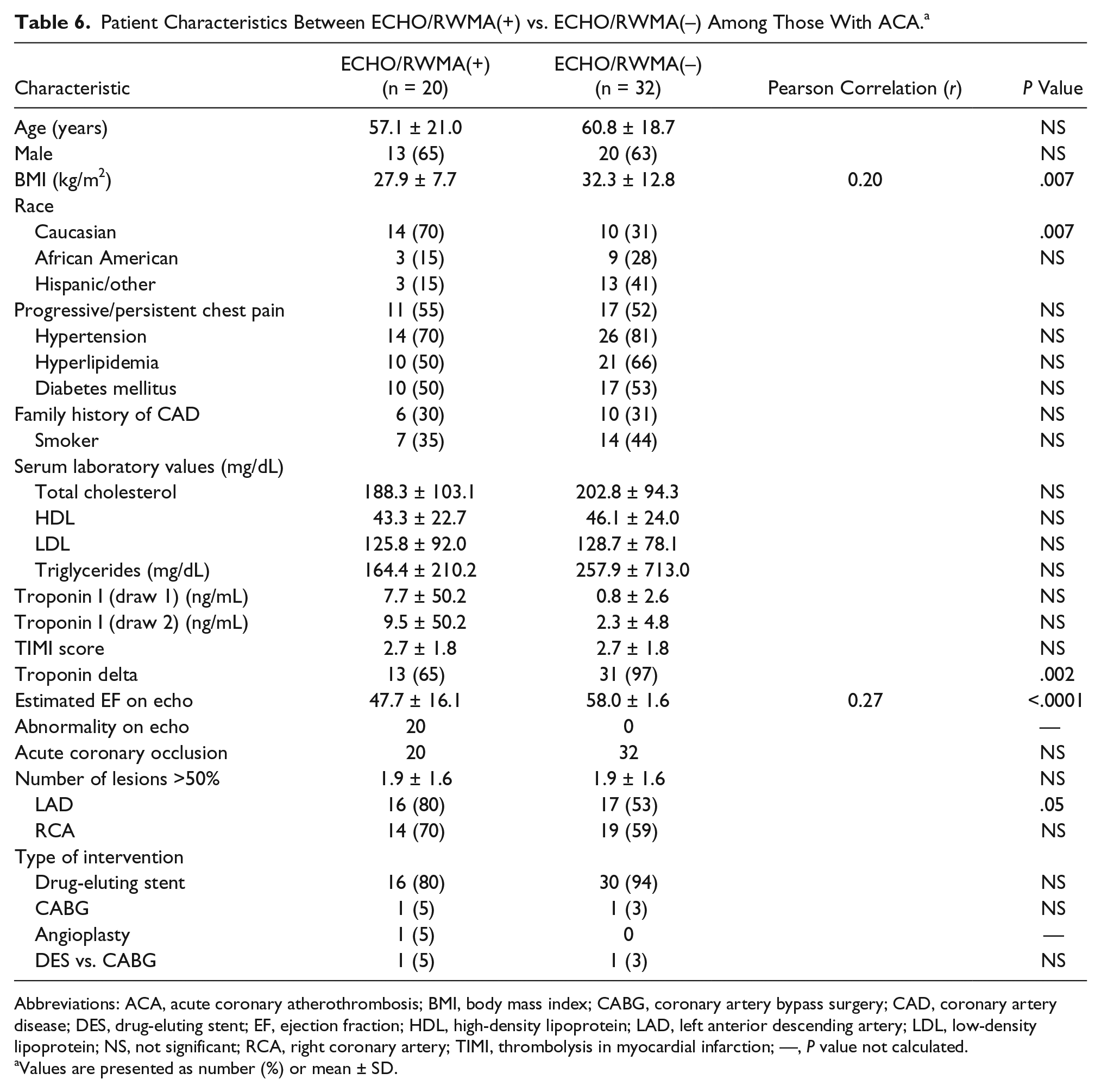
Abbreviations: ACA, acute coronary atherothrombosis; BMI, body mass index; CABG, coronary artery bypass surgery; CAD, coronary artery disease; DES, drug-eluting stent; EF, ejection fraction; HDL, high-density lipoprotein; LAD, left anterior descending artery; LDL, low-density lipoprotein; NS, not significant; RCA, right coronary artery; TIMI, thrombolysis in myocardial infarction; —, P value not calculated.
Values are presented as number (%) or mean ± SD.
Discussion
In this study, the noninvasive diagnostic modality of echo was assessed in a retrospective cohort of patients who were admitted to the ED and discharged with a diagnosis of NSTEMI. One of the strengths of the study was that every patient who met inclusion criteria had an in-hospital echo done prior to receiving an angiography, which allowed for correlation with clinical findings. This study design allowed for a comparison between subsets of the study cohort, based on clinical findings of ACA versus non–type 1 NSTEMI, demonstrated on angiography.
This analysis was informative in several ways. Several key clinical risk factors were found to be statistically different between patients found to have ACA versus non–type 1 NSTEMI on angiography, which may be useful predictors in the acute care setting. Increasing age, recurring anginal symptoms, LDL, TIMI score, and decreased HDL were found to be useful predictors of an ACA. These results confirm the current paradigm of CAD being a disease of age and progressing from silent/stable angina. Interestingly, these results highlight the relatively late effects that LDL cholesterol may play in the progression of CAD to an AMI, which reaffirms previous recommendations of aggressive LDL lowering in this patient population.3,17,18
Next, a study objective was to better characterize the limitations and strengths of echo to understand its role in the management of NSTEMI. Interestingly, BMI, troponin I levels, troponin delta, and LVEF were variables highlighted as statistically significant between ECHO/RWMA(+) and ECHO/RWMA(–) groups. These results highlight several important characteristics of echo. ECHO/RWMA(+) was more prevalent in those with lower BMI. Currently, there is no known study documenting a medical explanation for the increased prevalence of RWMA in those with lower BMI individuals when compared to those with higher BMI. It is likely that RWMA is less frequently detected in those with higher BMI because degradation of image quality is a known limitation of echo. 19 Utilization of ultrasound contrast imaging may improve the sensitivity of echo for this patient population. It is interesting to note that echo positivity among the ACA subcohort was more likely to not exhibit delta changes in troponin values. This is an interesting phenomenon that may be due to nonsalvageable necrotic cardiomyocytes from prior silent AMI. Low LVEF is known to be an independent predictor of increased mortality in patients with NSTEMI.20,21 It is plausible that patients with NSTEMI who present with RWMA may benefit from earlier angiography or tailored post-AMI medical therapy to address impaired ventricular function and/or remodeling and development of subsequent heart failure.
The optimal timing of cardiac revascularization for patients with NSTEMI is not known. A meta-analysis has demonstrated no difference in mortality or risk of a second myocardial infarction for patients with NSTEMI between early (usually less than 24 hours) and delayed (usually after 24 hours) left-heart catheterization with a slight mortality benefit for those considered higher risk patients. This has been defined as having greater than 140 on a risk stratification scoring system known as the GRACE score.13,22 The hypothesis was that one of the reasons that prior studies have not been able to detect statistical significance in outcomes between early and delayed revascularization strategies has been because as many as 30% of patients with NSTEMI do not have a true ACA. Therefore, better identification of patients who may benefit from coronary artery reperfusion, using the proposed prediction model for ACA and echocardiographic RWMA information, is needed. In this way, it may inform future prospective trials that attempt to determine the optimum timing of invasive cardiac catheterization for patients with NSTEMI (Figure 2). 23
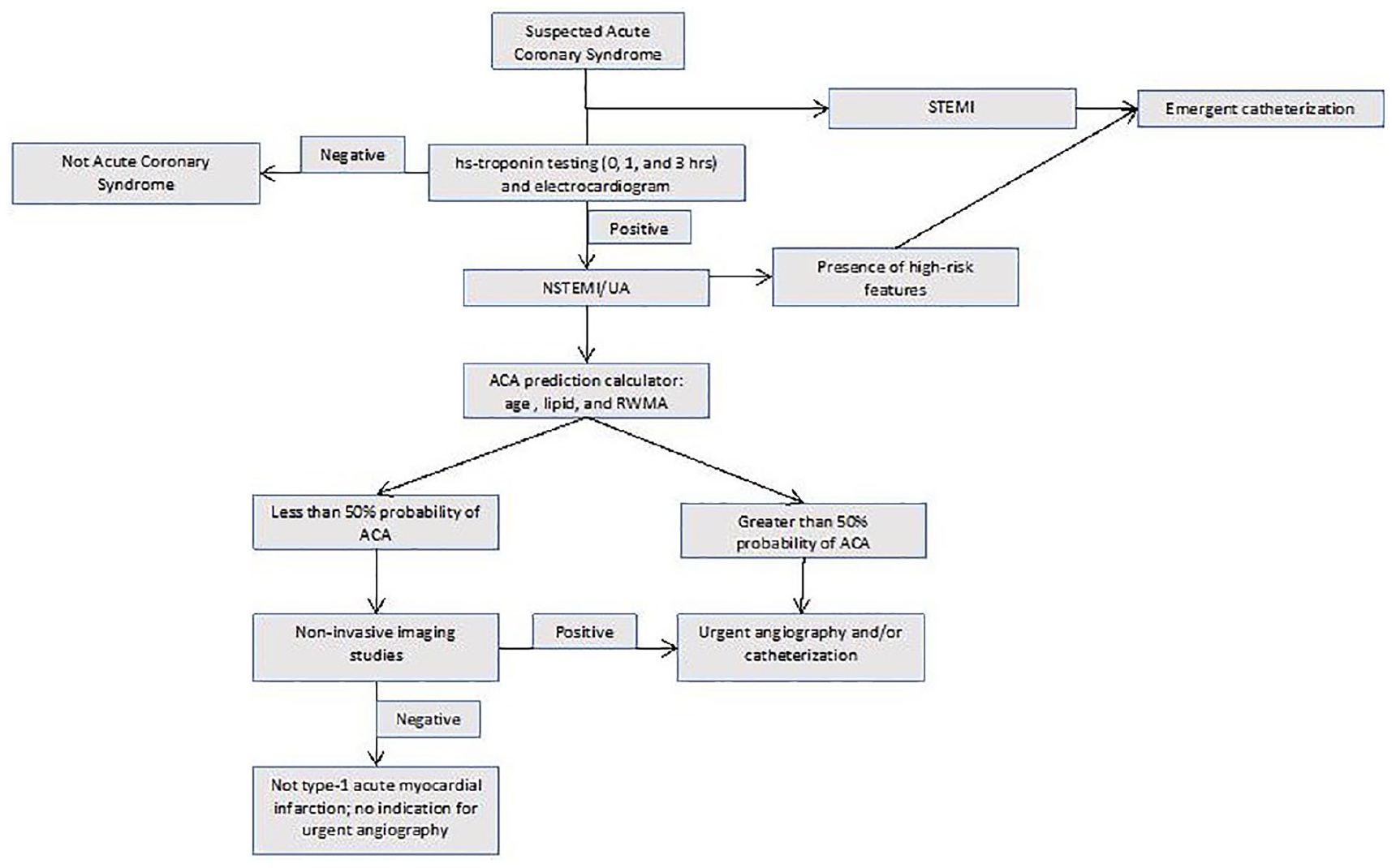
Flow diagram of patient triage in patients with NSTEMI.
More frequent utilization of echo also has the potential to identify those more at risk for increased mortality, evidenced by depressed LVEF and RWMA, and minimize false-positive diagnosis of ACA in the setting of NSTEMI. 24 A lower false-positive rate for ACA would result in fewer patients receiving unnecessary cardiac catheterization. These findings may also have an impact on the timing of dual antiplatelet therapy in patients with NSTEMI prior to diagnostic cardiac catheterization. Finally, there are potential economic implications derived from this study. The cost of overdiagnosis and overutilization of expensive left-heart catheterizations when compared to relatively cost-effective, noninvasive imaging modalities is an important consideration in the management of patients with NSTEMI, all of whom may not have a clear benefit from revascularization.25–27
Limitations
The study participants consisted of those with a first-time diagnosis of AMI, and thus the utility of echo with preexisting RWMA or ventricular dysfunction is unknown. According to Figure 1, the clinical impact for patients with NSTEMI may appear low: 14% among this NSTEMI cohort. However, 60% who otherwise might have met inclusion simply had procedural deviations that prevented them from being included in the present analysis. The percentage that did not meet inclusion criteria based on medical comorbidities was much lower at 26%. Second, the study was a single-center study. Third, this study was a proof-of-concept study, and as a result, it was underpowered, particularly in the nonobstructive CAD group, with a power less than 80% for most of the univariate analysis. Future studies are warranted to more robustly investigate these preliminary findings.
Conclusions
Most current guidelines by the American Heart Association and European Society of Cardiology do not incorporate the routine use of echo for the diagnosis and management of NSTEMI in the ED setting. 9 This study identified a cohort of patients with NSTEMI (those younger than 65 years) in whom a prediction algorithm incorporated RWMA from cardiac echo in addition to traditional predictive variables. This resulted in a lower false-positive rate for identifying ACA on coronary angiography. The results presented in this study support development of larger, prospective cohort studies aimed to further characterize echo and its use as a diagnostic modality for the management for patients with suspected NSTEMI presenting to the acute care hospital setting.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
