Abstract
Congenital diaphragmatic hernia (CDH) is a condition that occurs during fetal development where the abdominal viscera herniates through a defect in the diaphragm. CDH manifests on either the right or left side of the diaphragm, but more often on the left side. With advancement in sonographic imaging, this condition can be diagnosed as early as the first trimester. Knowledge of the sonographic appearance and any differential diagnosis of CDH will allow sonographers to document this condition. This permits for diagnosis and early intervention for the fetus. A case report is reviewed of a right-sided CDH with the gallbladder, liver, and bowel present in the thoracic cavity.
Congenital diaphragmatic hernias (CDHs) have an incidence of 1 in 2200 to 5000 live births.1,2 The diaphragm is composed of muscle and other fibrous tissues that separates the abdominal organs from those in the chest.3,4 The diaphragm typically forms during the 4th and 12th weeks of pregnancy.5,6 A hole in the diaphragm can occur when the pleuroperitoneal membrane fails to close at the end of organogenesis. 4 When the fetal midgut migrates back into the abdominal cavity (usually by week 10–12) the pressure forces the abdominal organs to herniate through the defect in the diaphragm. 2
Complications to take into account in neonates with a CDH include pulmonary hypoplasia and primary pulmonary hypertension. 7 The mass effect of the organs herniating into the chest does not allow the lungs to expand as the fetus develops.7,8 Neonates that are born with this condition are usually placed on extracorporeal membrane oxygenation (ECMO) to replace oxygen if the neonate’s lungs are hypoplastic. 9 To further complicate this condition, 15% to 45% of fetuses also present with other abnormalities and anomalies. 9 This includes cardiac abnormalities and chromosomal defects such as trisomy 18.9,10 Genetic counseling is usually advised for a fetus with CDH to assess the risk for additional chromosomal defects. 6
Case Report
A healthy woman in her late twenties, G3, T2, P0, A0, L2, presented to an outpatient obstetric imaging center for a second trimester anatomy survey. At that time, the gestational age of her fetus was 19 weeks and 0 days by the patient’s last menstrual period (LMP).
The second trimester anatomy survey was performed on a GE Logic 5 (GE Healthcare, Milwaukee, WI) with a 5.0-MHz curvilinear transducer. Sonographic examination revealed a single intrauterine pregnancy with a normal heart rate. The fetal heart was displaced to the left and anterior in the thoracic cavity. A cystic structure was visualized to the right of the displaced heart in the thoracic cavity (Figure 1). The cystic structure measured 0.8 × 0.7 cm (Figure 2). An image of the cystic structure was obtained in the sagittal view (Figure 3). The fetal growth was appropriate for gestational age and there were no other abnormalities noted at the time.
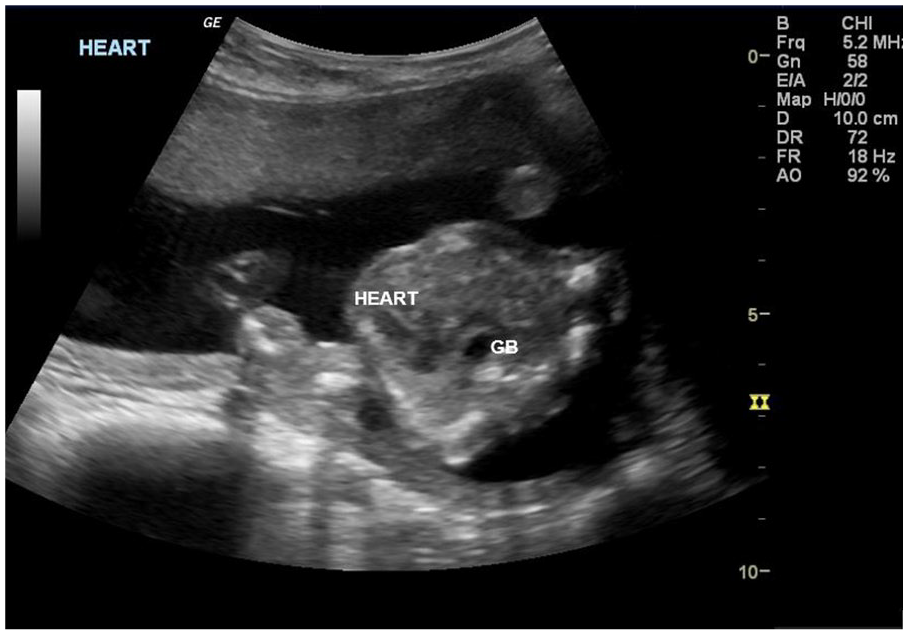
A transverse view of the fetal chest at 19 weeks and 0 days with a four-chamber heart view that is shifted to the left. An avascular cystic structure was noted anterior to the spine displacing the heart.
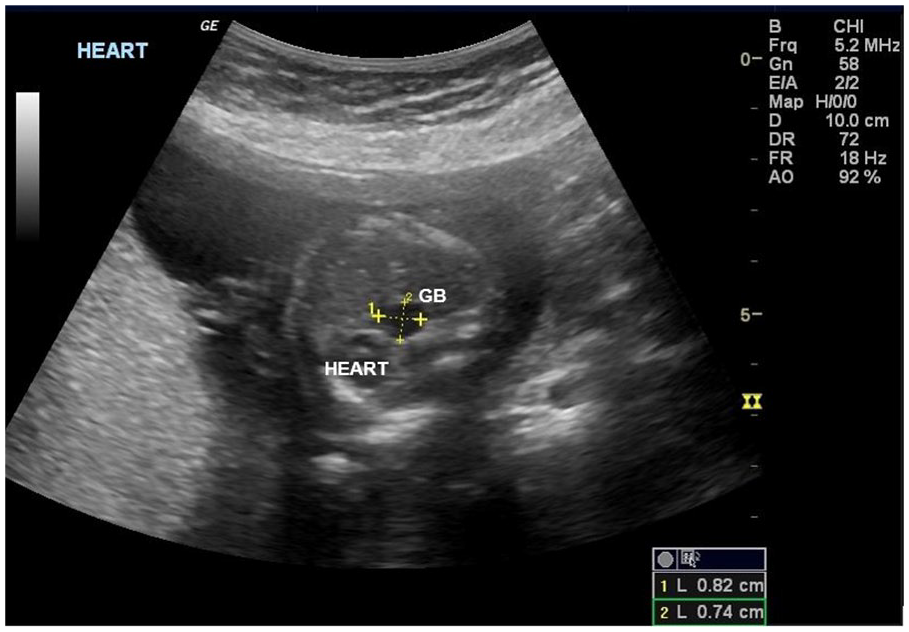
The cystic structure that displaced the heart measured 8 × 7 mm.
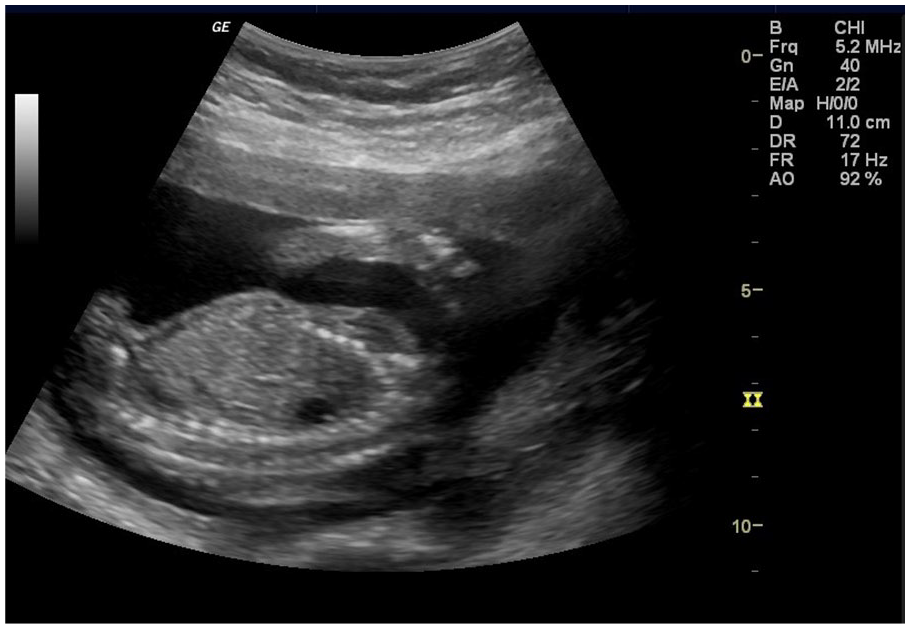
A sagittal view of the chest cavity was obtained that shows the anechoic area.
The patient was scheduled for a targeted sonogram for additional assessment of the cystic structure. The targeted sonogram was performed on an Acuson S2000 Siemens (Siemens Medical, Los Angeles, CA) machine with a 6C2 curvilinear transducer. The gestational age by LMP was 20 weeks and 4 days. No cardiac anomalies were visualized. The mediastinal shift to the left was once again documented during the exam. The anechoic structure was visualized as well and appeared to have no vasculature. With additional interrogation, the anechoic structure appeared to be the gallbladder that had herniated into the chest (Figure 4). The stomach was visualized in the correct position on the left side of the abdomen.
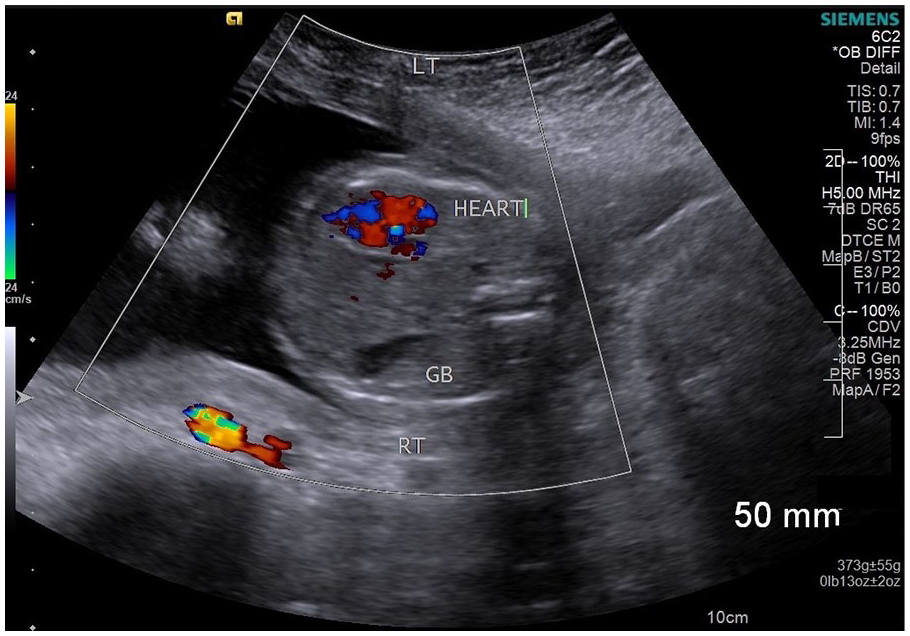
This image of the transverse fetal thorax was taken during the targeted exam. The image shows the fetal heart displaced to the left side of the chest wall and a right-sided cystic mass thought to be the gallbladder.
The patient was scheduled for another targeted sonogram at 28 weeks and 4 days by LMP. The fetus measured 28 weeks and 1 day. The fetal heart once again presented with a mediastinal shift anteriorly and to the left side of the thorax (Figure 5). The gallbladder was once again visualized in the same plane as the fetal heart (Figure 5). Another transverse section of the fetal chest demonstrated liver and bowel adjacent to the fetus’s lung (Figure 6). The amniotic fluid index (AFI) was appropriate for the gestational age.
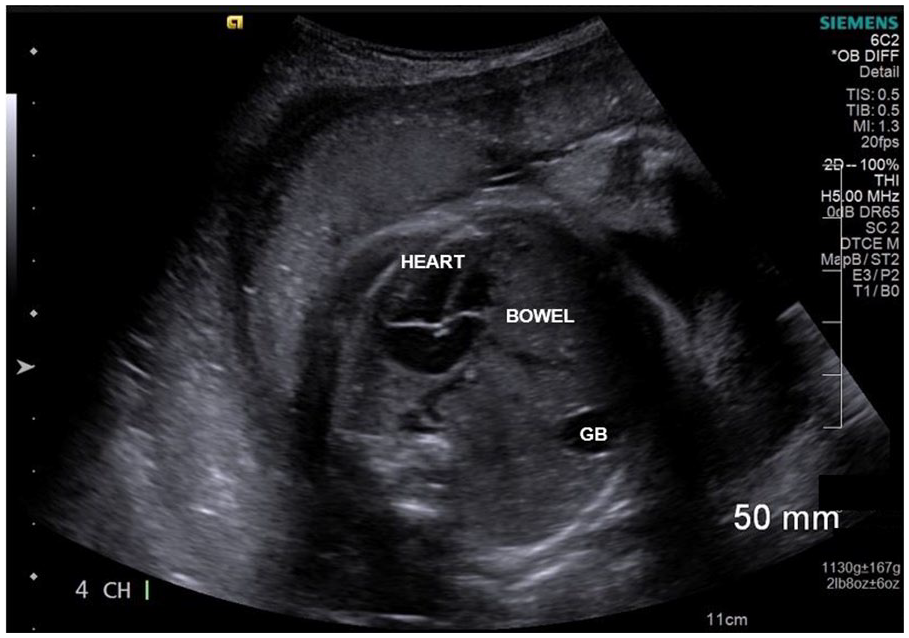
At 28 weeks and 4 days, this view confirms the location of the liver and gallbladder adjacent to the fetal heart.
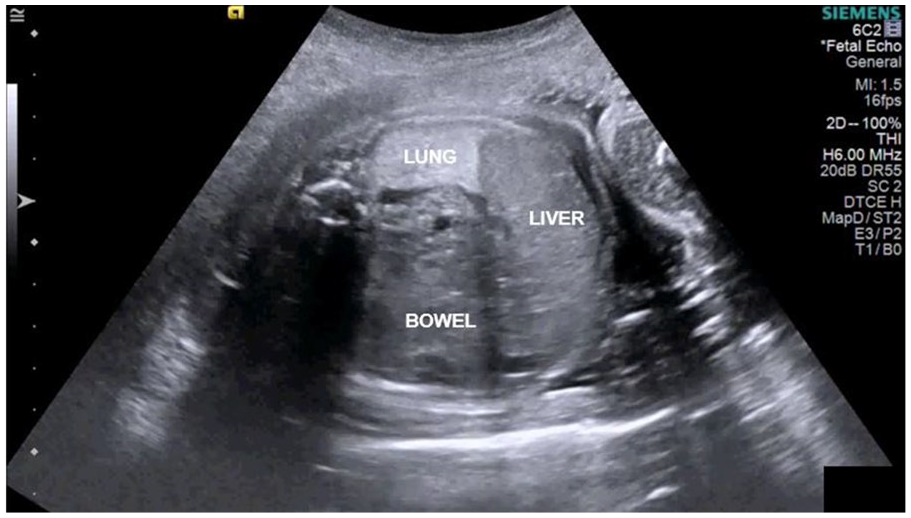
A transverse view of the fetal chest cavity. Liver and bowel appear to be displaced next to the fetal lung.
The patient was transferred to a different hospital for the remainder of her pregnancy and delivery. No further information is available for this case.
Discussion
Even with advances in sonography, a CDH can be difficult to detect before 16 weeks of gestation. 7 Up to 11% of CDH cases are missed prenatally and therefore diagnosed in the postnatal period. 11 A congenital diaphragmatic hernia is often found during a routine anatomy scan when obtaining the fetal heart views. This is why it is vital that sonographers carefully inspect the fetal heart’s orientation and location in the chest cavity. Determining whether an anechoic structure in the thoracic cavity is stomach or gallbladder will contribute to the diagnosis of a right- or left-sided CDH. Right-sided CDH cases will have the heart displaced to the left and possibly gallbladder located in the thoracic cavity. 8 For left-sided CDH, the heart will be displaced to the right and the stomach will be located on the left side of the thoracic cavity.8,9 Both right- and left-sided hernias can also present with bowel and/or liver herniating into the chest cavity. 9 Left-sided diaphragmatic hernias are noted in 85% of cases, while 15% of cases are right-sided. 10
Right-sided CDH cases are often missed during a prenatal sonogram. 9 The liver has the same echogenicity as lung tissue, which can be challenging to differentiate when inspecting the chest cavity. 12 Thus, it is important to carefully examine the thorax for both lung tissue and liver tissue. 12 Sonographically, a right-sided CDH includes a mediastinal shift of the heart to the left of the chest cavity. 13 This can be so severe that the heart appears to be severely pushed into the chest wall. The stomach may appear in the normal position in the abdomen cavity. 12 However, in 2012, Conturso et al. reported that a horizontal orientation of the stomach in the abdominal cavity may indicate an indirect sonographic sign of a right-sided CDH (Figure 7). 12 Experts suggest that this is due to the medial stretching of the hepatogastric ligaments caused by the rising of the viscera into the diaphragm, which then creates a shift of any connected structures. 12 Bowel, liver, and gallbladder may be visualized sonographically in the thoracic cavity. When a CDH is suspected during a prenatal ultrasound, the sonographer should also attempt to obtain a sagittal view of the diaphragm to confirm that there is a separation of the chest and abdominal cavity. 11 Coronal and sagittal views may help visualize any defects in the diaphragm. Polyhydramnios has also been observed in fetuses with a diaphragmatic hernia, which is thought to be caused by an accompanying upper gastrointestinal tract obstruction from the herniation of any gastrointestinal organs. 13
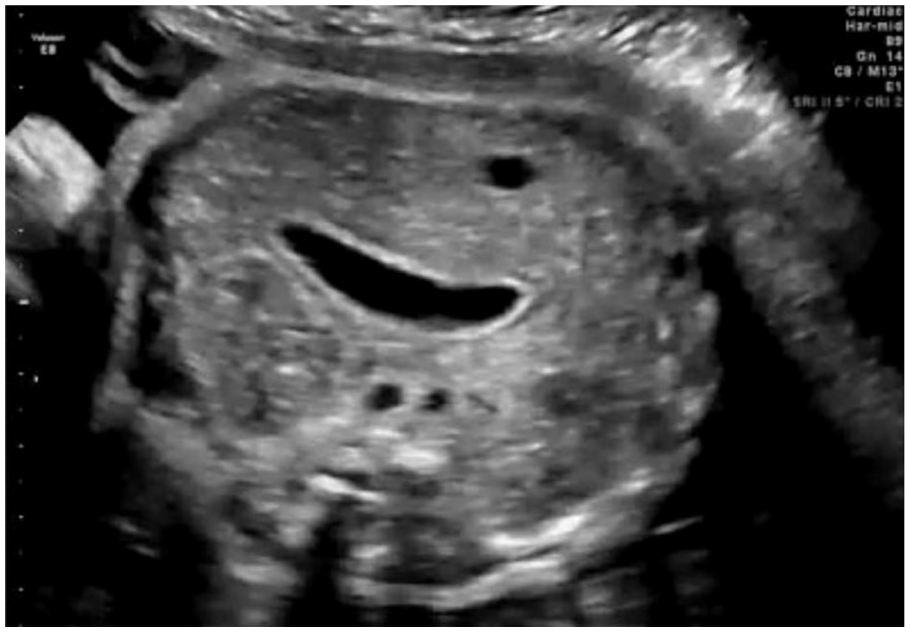
A horizontal orientation of the fetal stomach that has been linked to being a possible early indicator of a right-sided diaphragmatic hernia. 11
Multiple factors must be considered in order to determine the prognosis for a fetus with a right-sided CDH. One major factor used universally to determine the prognosis is the lung-to-head ratio (LHR). LHR is an estimate of the size of the lung in relation to the fetus’s head. 14 An LHR should be obtained at the level of the four-chamber heart, with the diaphragmatic defect closest to the transducer if possible. 14 The image should be magnified to only visualize the fetal thorax. 14 Each facility will have a different protocol to measure the chest cavity, but studies are currently being conducted to determine a universal protocol to aid in standardization of the technique. 14 A high LHR usually results in a better prognosis for the fetus. 15 The survival rate is related to the size of the defect in the diaphragm, with larger defects causing severe complications for the fetus. 15 Overall, a large defect in the diaphragm, a low LHR (lower than 0.9), and a liver that has herniated into the thoracic cavity will result in a poor prognosis for right-sided CDH cases.8,14,15
Due to evolving methods in the healthcare field, patients with a severe case of CDH could be candidates for fetal intervention. If the patient presents with “liver up” (intrathoracic liver) and a low LHR, a tracheal occlusion surgery may be an option. 8 These are cases where there is a high probability the neonate would not survive the postnatal surgical repair due to the severity of pulmonary hypoplasia. 8 Experts have employed a different approach called a fetoscopic endoluminal tracheal occlusion (FETO) surgery, which allows the fetal lungs to expand, lessening the severity of pulmonary hypoplasia. 8 A FETO is usually performed on a fetus that is anywhere from 27 to 30 weeks of gestational age and presents with no other abnormalities. 16 For this surgery, the trachea is temporarily blocked with a balloon, which allows fluid to build up in the thoracic cavity and expand the lungs. 16 This stimulates the growth and development of the lungs to hopefully increase the odds of survival and decrease the severity of pulmonary hypoplasia. 16 The balloon is usually removed prenatally or upon delivery. 16 Once the neonate is stable, a surgical patch surgery is performed to repair the defect in the diaphragm. 16 If a FETO procedure is performed at the optimal gestational age, the results have proven to improve the chances of survival for severe cases. 16
Due to the potential for early intervention when CDH is suspected, it is vital that sonographers obtain optimal images to rule out any differential diagnosis. This will aid in the treatment plan before and after the fetus is born. Multiple congenital lung abnormalities may present with polyhydramnios, a mediastinal shift, and a anechoic mass in the thoracic cavity. 17 This is why it is imperative that sonographers interrogate the thoracic cavity carefully to assist the physician in diagnosing the fetus. Pulmonary sequestration is a condition that should be considered when visualizing a mass (cystic or solid) in the thoracic cavity. 18 A feeding artery usually branches off the thoracic or abdominal aorta in cases of pulmonary sequestration. 19 The best way to discriminate pulmonary sequestration from a diaphragmatic hernia is to locate a feeding vessel to any foreign structure in the chest cavity. 19 Another differential associated with CDH is congenital pulmonary airway malformation (CPAM), which is a developmental abnormality of the lung that results in cell death to the tissue. 20 Solid or cystic masses can form in the lungs, which can cause a mediastinum shift. 17 The main way to distinguish this from a diaphragmatic hernia would be to investigate the chest for liver or bowel which could herniate with a CDH. 18 Fetal magnetic resonance imaging (MRI) would further aid in differentiating congenital pulmonary abnormalities from a diaphragmatic hernia. 18 An MRI is also useful in determining the size of the defect and confirming which organs have herniated into the chest. 18
Conclusion
Knowing the sonographic appearance of a CDH will better assist physicians in developing a treatment plan for severe cases. As right-sided CDH cases are more often missed during a prenatal sonogram than left-sided ones, it is important that sonographers become familiar with their appearance. By carefully examining the fetal chest for a cystic structure, a left mediastinal shift of the heart and liver above the diaphragm will better aid in the diagnosis of right-sided CDH.
Footnotes
Acknowledgements
I would like to thank Lynn Schluns, RDMS, RVT, for her guidance and revisions to this article. I would also like to thank Wendy Brooks, RDMS, RVT, for her advice and feedback to help me throughout this process.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
