Abstract
The purpose of this research was to compare the sonographic features of papillary thyroid microcarcinomas (micro-PTCs) to those of macro-PTCs. Patients with known PTC were retrospectively reviewed and divided into micro- or macro-PTC groups, according to the largest nodule diameter, ≤10 mm or >10 mm, respectively. Eighty-nine nodules (62 macro-PTCs and 27 micro-PTCs) were evaluated. Contour irregularity was more common in micro-PTCs than macro-PTCs (
Keywords
The incidence of thyroid cancer has been reported to have increased considerably, and papillary thyroid carcinoma (PTC) has been the most common occurring type.1,2 The reason for this has been the increased detection of small PTCs, measuring less than 10 mm in size, by the more common use of high-frequency ultrasound equipment,1–5 although most of these are nonpalpable and clinically unapparent. However, the prognostic significance and appropriate management of micronodules (diameter <1 cm) with PTCs are controversial, given that neck and distant metastases of microcarcinomas have been reported.3,4,6 The malignancy rate of subcentimeter nodules is reported to be no less than that of macronodules (diameter >1 cm).7,8
The presence of hypoechogenicity, microcalcifications, irregular borders, absence of peripheral halo, and internal vascularity are generally considered suspicious sonographic characteristics for malignancy of a thyroid nodule and therefore are important in determining the need for fine-needle aspiration biopsy (FNAB).3,9–13 Although the indications for biopsy of micronodules under certain clinical scenarios depend on suspicious sonographic features, it is not well documented whether microcarcinomas differ sonographically from macrocarcinomas.9,10,12,13 Not knowing the possible differences in suspicious sonographic findings may lead to misdiagnosis of microcarcinomas because biopsy is not routine. A search of the literature revealed only one report similar to this work, which compared sonographic features of micro-PTCs to those of larger carcinomas. 14
In the present study, the purpose was to document the sonographic characteristics of micro-PTCs and compare them to those of macro-PTCs. This information was hypothesized to improve the efficacy of sonographic diagnosis.
Materials and Methods
The study protocol was approved by the institutional review board, and informed consent was waived. Patients who had undergone ultrasound-guided FNAB of a thyroid nodule between 2011 and 2015 were retrospectively reviewed. Before FNAB, all patients had undergone sonographic imaging using an Aplio scanner (Toshiba Medical Systems, Tokyo, Japan) and a 7.5-MHz linear array probe. All FNABs were performed by an experienced single attending interventional radiologist.
The nodules with a biopsy result positive for PTC were included in the study and divided into two groups as micro- or macro-PTCs, according to the largest diameter of the nodule, ≤10 mm or >10 mm, respectively. Sonography reports and images before biopsy of these nodules were retrieved from the hospital records and the nodule characteristics were reviewed in terms of hypoechogenicity, heterogeneity, contour irregularity, absence of peripheral halo, presence of microcalcifications, cystic component, and internal vascularity by color Doppler imaging.
Chi-square test was used for statistical analysis to compare the sonographic features between micro- and macrocarcinoma groups using SPSS Statistics for Windows, Version 19.0 (SPSS, Inc, an IBM Company, Chicago, Illinois).
Results
Eighty-nine nodules in 89 patients (31 males, 58 females) were found to have a positive biopsy result for a PTC. The patients’ ages ranged between 20 and 67 years (mean, 39 years). Sixty-two nodules (70%) were macro-PTCs, while 27 (30%) nodules were micro-PTCs.
With sonography, the macrocarcinoma group showed hypoechogenicity (n = 47), heterogeneity (n = 32), contour irregularity (n = 21), microcalcifications (n = 13), internal vascularity (n = 8), absence of peripheral halo (n = 54), and cystic component (n = 16) (Figure 1). Microcarcinomas demonstrated hypoechogenicity (n = 20), heterogeneity (n = 9), contour irregularity (n = 17), microcalcifications (n = 11), absence of peripheral halo (n = 25), and cystic component (n = 3) (Figure 2). No microcarcinomas demonstrated internal vascularity by color Doppler imaging. Sonographic characteristics of the nodules are summarized in Table 1.
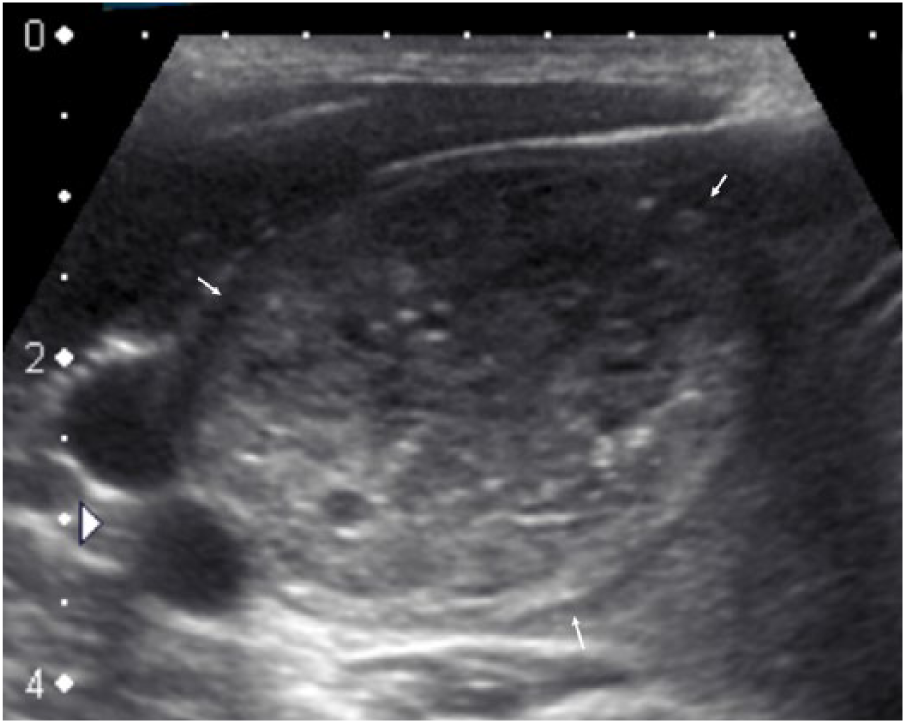
Sonogram demonstrating a heterogeneous thyroid nodule (arrows) measuring 31 × 37 mm with cystic components, microcalcifications, and peripheral hypoechoic halo in a 36-year-old male patient. Fine-needle aspiration biopsy revealed papillary thyroid carcinoma.
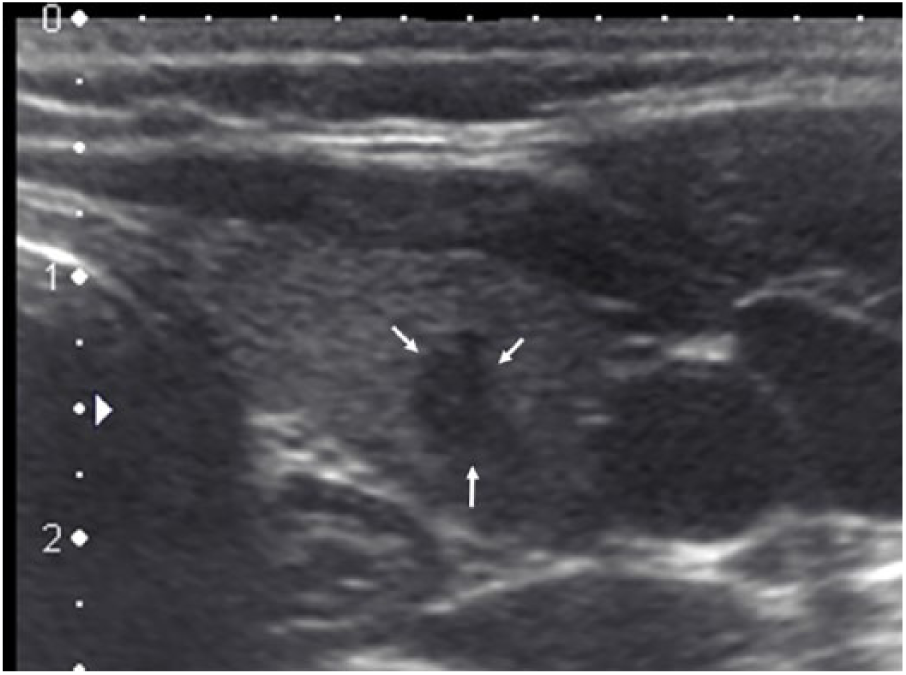
Sonogram showing a hypoechoic thyroid nodule with irregular contours (arrows) and taller-than-wide shape measuring 6 × 4 mm in a 39-year-old female patient. Fine-needle aspiration biopsy revealed papillary thyroid microcarcinoma.
Distribution of Sonographic Features of Macro- and Micro-PTCs.
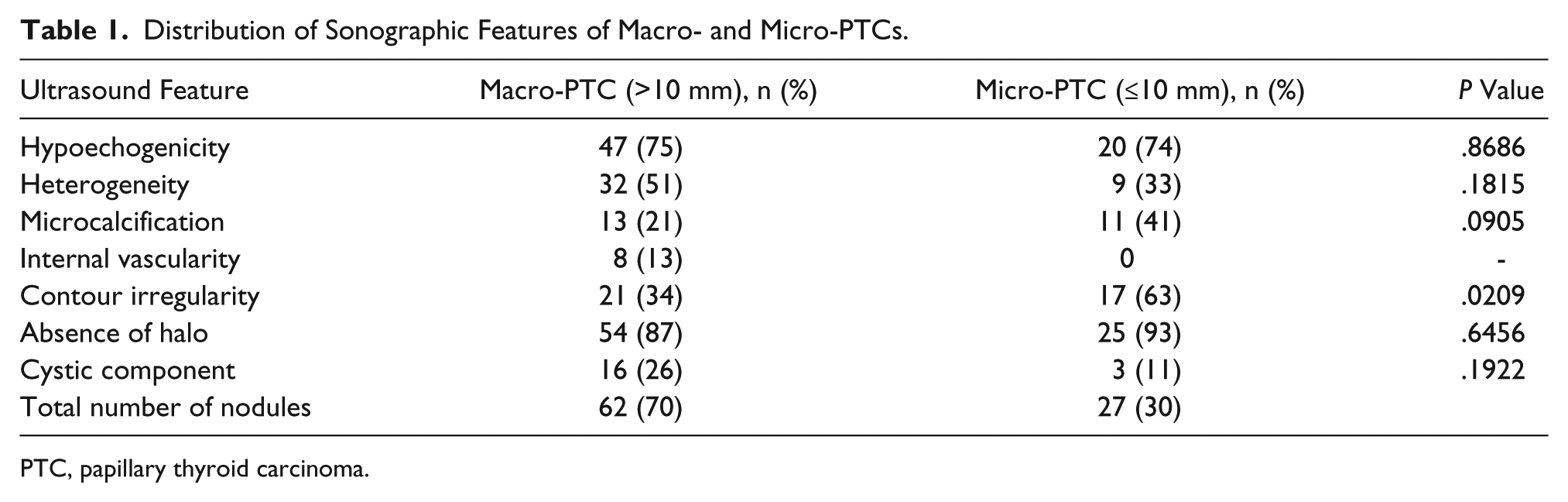
PTC, papillary thyroid carcinoma.
The presence of contour irregularity was found to be significantly higher in the microcarcinoma group (63%) compared to that of macrocarcinoma group (34%) (
Discussion
Sonographic findings of PTCs were reported to include hypoechogenicity, solid composition, presence of microcalcifications, and a taller than wide shape.1,5–7,15–20 Different reports emphasized the importance of certain features such as the presence of microcalcifications, hypoechogenicity, or taller than wide shape. However, no single sonographic feature was found to be capable of predicting malignancy in isolation, while the presence of more than one suspicious sonographic finding was reported as more accurate for predicting malignancy.1,5,6,12,13,18,20,21 The Thyroid Imaging, Reporting and Data System (TI-RADS) provided by the American College of Radiology has proposed a sonography-based risk stratification system by giving points for each suspicious feature. This would include composition, echogenicity, shape, margin, and echogenic foci of the nodule, where the total point value equates to the TI-RADS level as well as the malignancy risk. 12 In addition, the documentation of central vascularity and the absence of elasticity of a nodule, by color Doppler and sonoelastography, respectively, have proven to be useful to determine nodules. These imaging factors have increased the risk of malignancy when combined with other suspicious sonographic features. 1 The suspicious sonographic findings are considered a more valuable indication for biopsy than nodule size; however, significant overlaps were also reported between benign and malignant nodules.5,10,11,22
According to American Thyroid Association (ATA) and American Association of Clinical Endocrinologists (AACE) guidelines, when a subcentimeter nodule is detected, FNAB is not routinely recommended. It may be considered depending on specific clinical conditions such as patient age, preference, history, or other findings.9,10 According to the TI-RADS classification, which reportedly better classifies nodules compared to ATA guidelines, biopsy of micronodules may be appropriate given a clinician’s preference or submarginal location of the nodule.12,13 Particularly for such clinical scenarios, recognition and reporting of suspicious nodules by sonography is still essential, whether for the purposes of biopsy or a clinical decision to follow up the patient.
The documentation and comparison of sonographic features of micro-PTCs to macrocarcinomas are limited in the literature. Our search of the literature revealed only one comparative study of sonographic findings of micro- and macro-PTCs by Zhang and Qian,
14
who reported that there were statistically significant differences between features of margins, ratio of length/width, cystic component, vascularity, and lymph node metastasis (
Contour irregularity, one of the most common findings of micro-PTCs in the present series, emerged as a significant outcome of the current study. It appears that irregular contours in a micronodule should increase suspicion and consideration for biopsy. Microcalcifications and hypoechogenicity showed no statistically significant difference between micro- and macro-PTC groups; both findings are equally important in both groups. It is reported in the literature that micronodules with a solid internal composition have a high risk of being malignant.17,19 Microcarcinomas in this series displayed a cystic component (11%) less commonly than macrocarcinomas (26%), but the difference was not statistically significant (
The presence of internal nodular vascularity by color or power Doppler is generally considered a suspicious sonographic feature for malignancy. 11 Although this finding has been excluded from the 2015 ATA guidelines, AACE guidelines include the presence of internal vascularity in suspicious sonographic features regardless of nodule size.9,10 Internal vascularity was detected in none of the micro-PTCs in the present study. This may be because of nonvisualization of flow due to smaller nodule size. Similarly, Kim et al. 15 have reported that increased vascularity was not significantly associated with malignant subcentimeter nodules. It would seem that, in evaluation of micronodules, internal vascularity should not be used as a criterion, and the absence of internal vascularity by color Doppler imaging should not defer the need for biopsy in a micronodule with other suspicious features.
This study has some limitations. First, sonographic examinations were not performed by a single radiologist. Second, cytopathological findings could not be confirmed by histopathologic results in all patients because some of the patients had undergone thyroidectomy at different institutions. Therefore, these results are descriptive of only these patients.
Conclusion
The sonographic features of micro- and macro-PTCs appear similar. The presence of irregular contours in a micronodule should increase suspicion and consideration for biopsy when clinically indicated. In micronodules, the absence of internal vascularity should not decrease the suspicion of malignancy in the presence of other suspicious sonographic features.
Footnotes
Acknowledgements
Preliminary results were presented at European Congress of Radiology in 2016 as an electronic poster (not published printed or online).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
