Abstract
Premature closure of the ductus arteriosus is a rare phenomenon. It is thought to be caused by one of three things: abnormal levels of produced and circulating prostaglandin, maternal ingestion of prostaglandin synthase inhibitors, or it can be idiopathic. Idiopathic premature closure of the ductus arteriosus, not caused by maternal exposure to nonsteroidal anti-inflammatory drugs, is an uncommon event that can cause increased pressure on the right ventricle, resulting in pulmonary hypertension. If left untreated, it can result in fetal death.
A possible, but rare, cause of cardiomegaly in utero is premature closure of the ductus arteriosus. The ductus arteriosus is a critical component of normal fetal circulation. Patency of the ductus arteriosus mainly depends on high levels of circulating prostaglandin E2 and prostaglandin E1 that are produced by the ductus arteriosus itself. 1 Maternal ingestion of prostaglandin synthase inhibitors, such as nonsteroidal anti-inflammatory drugs and corticosteroids, could be associated with increased risk of premature closure of the ductus arteriosus. Along with maternal ingestion of prostaglandin synthase inhibitors, it has been found that increased maternal intake of polyphenol-rich foods such as green tea, dark chocolate, and grape juice could increase the risk of ductal closure. 2 Premature closure of the ductus arteriosus causes volume overload on fetal pulmonary circulation, which can result in persistent pulmonary hypertension in the newborn. 1 Premature closure of the ductus arteriosus can also be idiopathic and can lead to cardiovascular dysfunction such as tricuspid and pulmonary regurgitation, right heart dilatation, functional pulmonary atresia, persistent neonatal pulmonary hypertension, and fetal death. 2
Case Report
A 33-year-old woman was referred to the ultrasound department for a second-trimester morphology examination at 19 weeks, 6 days’ gestation. The patient reported current tobacco use. An echogenic focus in the left ventricle and a possible pericardial effusion were noted in the findings. The interpreting radiologist recommended a follow-up ultrasound. The follow-up ultrasound at 24 weeks, 4 days confirmed the pericardial effusion measuring 0.166 cm, but the echogenic focus was no longer visible (Figure 1). Another follow-up sonogram was performed at 30 weeks where the pericardial effusion measured 0.356 cm (Figure 2). At 34 weeks, 5 days, the patient was admitted to the hospital for fetal monitoring after reporting decreased fetal motion to her attending physician. A nonstress test was also performed and where a category 3 strip was recorded. Category 3 fetal heart rate tracings are indicative of hypoxic risk, including one or more of the following: no baseline variability, the presence of recurrent late decelerations, variable decelerations, bradycardia, or a sinusoidal pattern. These tracings have been associated with adverse neurologic sequela, and if these abnormalities are not resolved, the fetus should be delivered immediately. 3 A sonogram was then performed to evaluate amniotic fluid levels and fetal growth. During the sonogram, it was found that there was adequate fluid, but multiple cardiac decelerations were observed. One deceleration was documented on a clip measuring 73 beats per minute (Figure 3) but rebounded back to 146 beats per minute. The heart to chest ratio was 0.63 and the pulmonary artery measured 0.914 cm (Figures 4 and 5). The previously noted pericardial effusion was no longer visible. The fetus’s biparietal diameter, occipital frontal diameter, and femur length were within normal limits. The abdominal circumference was slightly small, measuring 27.99 cm. For a fetus at 34 weeks, the 50th percentile is 28.84 cm and the 5th percentile is 25.98 cm. 4 The fetal stomach was difficult to visualize, with only a small glimpse of the lumen visualized throughout the entire examination. The fetal bladder was enlarged throughout the entire examination, with a maximum volume of 32.3 mL (Figure 6). It was also discovered that the fetus made only a single gross body movement throughout the entire examination of an estimated 45 minutes. The case was presented to the radiologist, who compared the heart and pulmonary artery size to the previous examination, in which they were both within normal limits. The radiologist diagnosed the fetus with cardiomegaly and recommended immediate delivery due to fetal distress. After delivery, a chest x-ray was obtained secondary to fetal distress. The chest x-ray confirmed that the cardiothymic contour was mildly prominent, and perihilar interstitial prominence with fluid was seen in the right fissure, which is consistent with transient tachypnea (Figure 7). The infant was stabilized and airlifted to a children’s hospital. Despite vigorous effort to obtain follow-up information about the baby, no information was available except that, to our knowledge, a postnatal cardiac workup was never completed. The patient is known to have survived without complications but is lost to detailed follow-up. After discussing the clinical findings and outcomes of this case with a cardiologist, a potential diagnosis was achieved. It was determined that the likely cause of the lagging fetal growth, cardiomegaly, enlarged pulmonary artery, and fetal distress is premature closure of the ductus arteriosus.
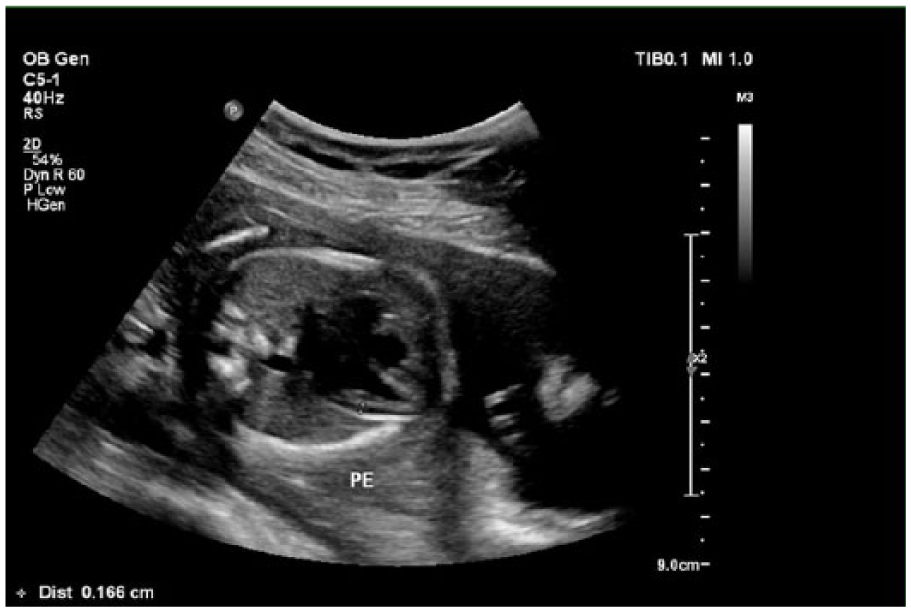
Gray-scale sonogram showing pericardial effusion at a gestational age of 24 weeks, 4 days, measuring 0.166 cm.
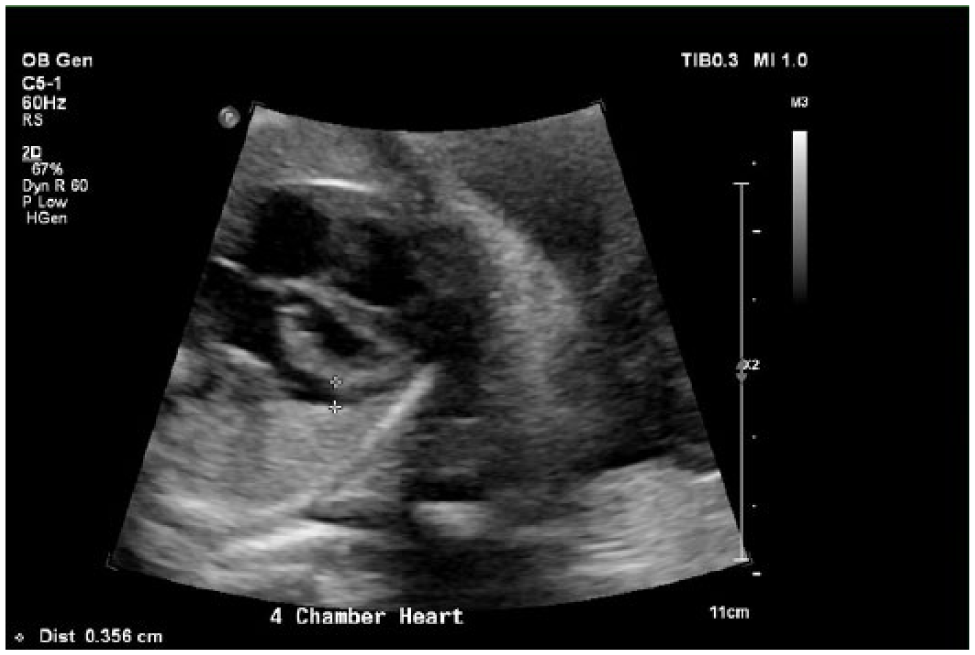
Gray-scale image of pericardial effusion at a gestational age of 30 weeks, 0 days, measuring 0.356 cm.
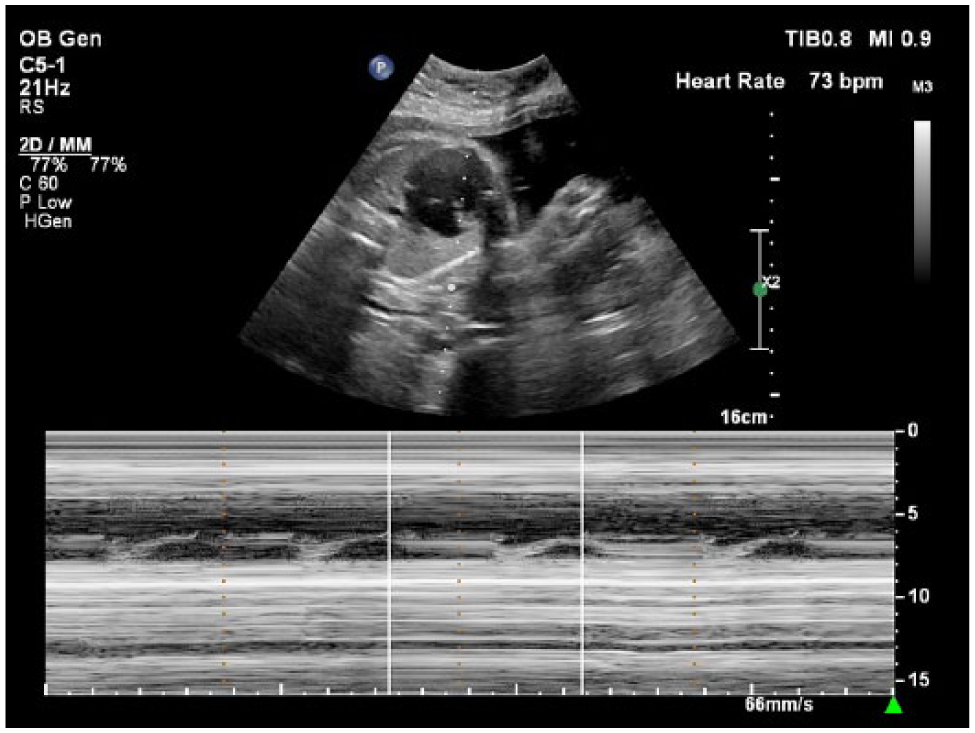
M-mode image depicting fetal cardiac deceleration at a gestational age of 34 weeks, 5 days.
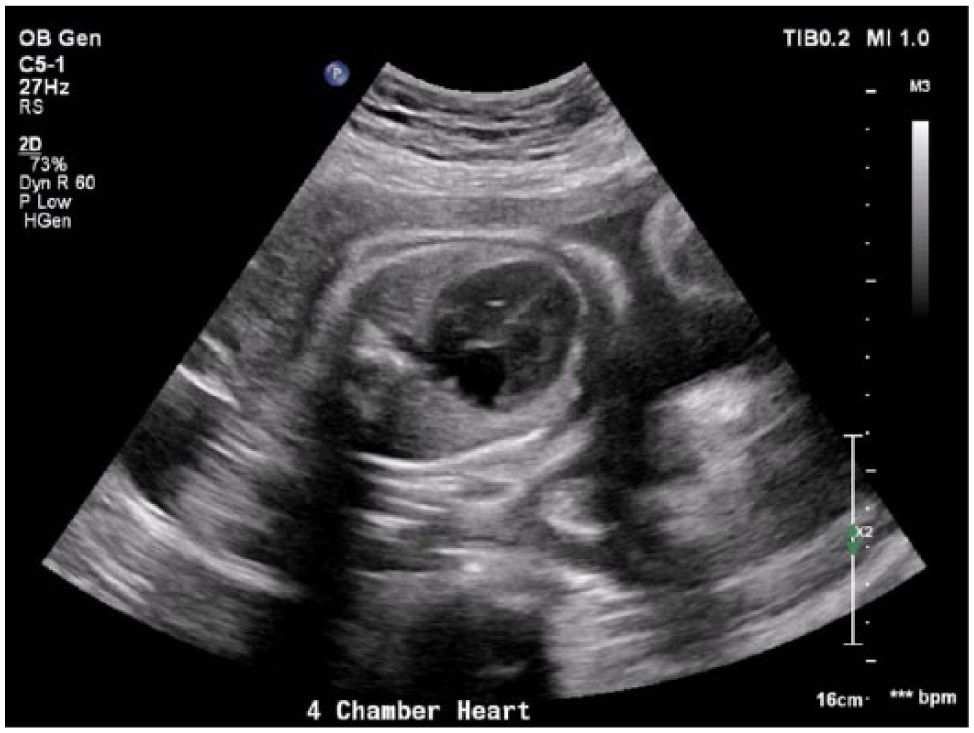
Gray-scale sonogram showing a four-chamber view of the enlarged fetal heart at a gestational age of 34 weeks, 5 days.
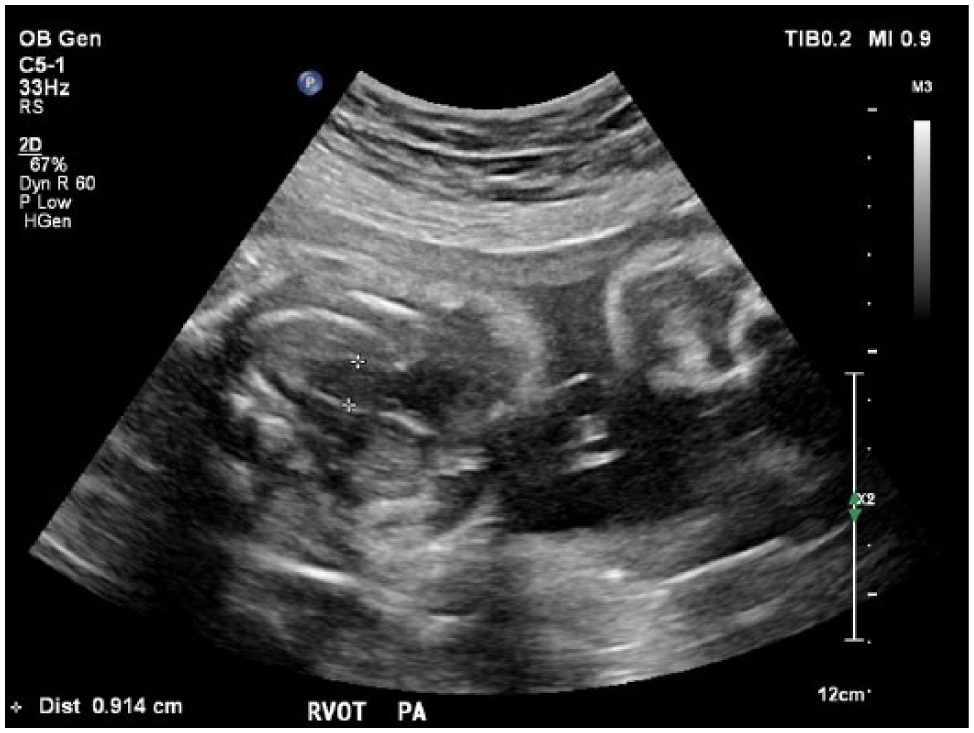
Gray-scale sonogram of the dilated fetal pulmonary artery at a gestational age of 34 weeks, 5 days, measuring 0.914 cm. The 50th percentile for a fetus at 34 weeks’ gestation is 0.743 cm, and the 90th percentile is 0.942 cm. 13
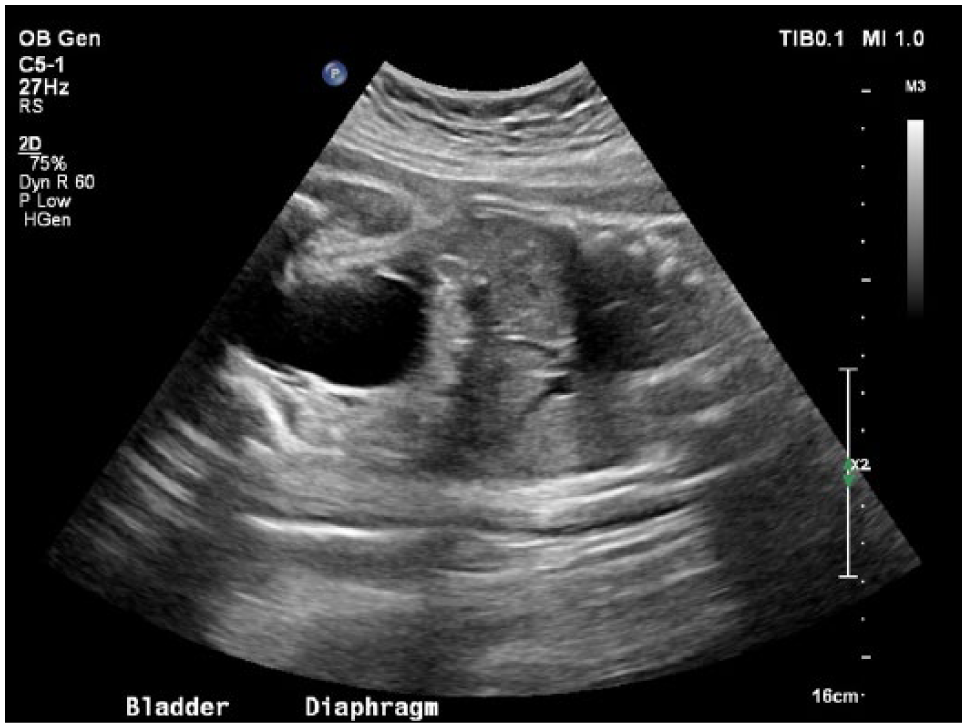
The diaphragm and bladder were easily visualized at a gestational age of 24 weeks, 5 days. The stomach is not clearly visualized.
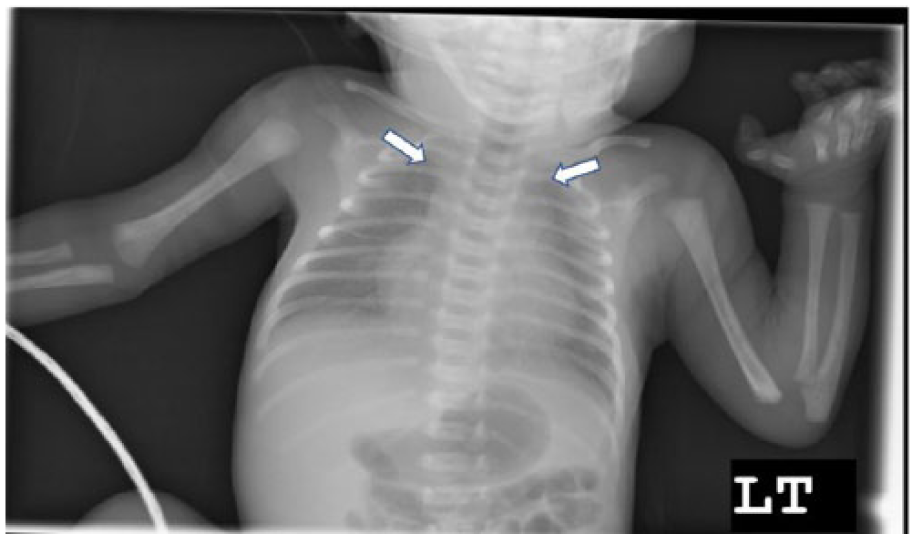
Postnatal chest radiograph performed immediately after birth showing prominent cardiothymic contour on the left and fluid in the right fissure.
Discussion
The ductus arteriosus is formed by the trifurcation of the main pulmonary artery, which arises from the right ventricle, also forming the right and left pulmonary arteries during the fifth week of gestation. The ductus arteriosus carries blood from the pulmonary artery to the descending aorta. 5 The patency of the ductus arteriosus mainly depends on high levels of circulating prostaglandin E2 and prostaglandin E1 produced by the ductus. 1 After birth, the ductus arteriosus generally closes in full-term neonates one to two days after birth. 6
The etiology of idiopathic closure of the ductus arteriosus in the fetus remains unknown. Recently, maternal ingestion of polyphenols or flavonoids present in foods and beverages such as berries, herbal teas, and grape and orange juices has been reported to influence fetal ductal blood flow, indicating a risk for constriction or closure of the ductus. 2 Idiopathic closure of the ductus arteriosus can have a wide variety of clinical presentations depending on the length and severity of the flow obstruction. This diagnosis may be underestimated due to the cases with negative maternal medications or mild cases where symptoms are not present. 2
Clinical symptoms vary with the severity of the flow obstruction. Findings include severe oligohydramnios; cardiac chamber disproportion caused by enlargement of the right atria and ventricle, along with increased diameter of the main right and left pulmonary arteries; and lack of visualization of the pulmonary end of the ductus arteriosus. 7 If one of these abnormalities is detected on a routine sonogram, an immediate delivery is not advised; however, a fetal echocardiogram may be done to confirm the diagnosis. If there is a constriction of the ductus arteriosus, blood circulation through the ductus will be turbulent, as well as have an increased systolic flow and a reduced pulsatility index. If there is complete closure of the ductus arteriosus, no ductal flow will be present. Tricuspid and pulmonary valve regurgitation may also be present. 8
If premature closure of the ductus arteriosus is found on a sonogram, premature delivery should be considered because it provides a favorable outcome. Once the fetus is delivered, there is an opportunity to reverse issues caused by premature closure. The immediate decrease in pulmonary vascular resistance with lung expansion and exposure to oxygen relieves the severe right ventricular afterload, caused by the closed ductus arteriosus, resulting in a positive outcome. A worse outcome has been associated with idiopathic closure of the ductus arteriosus in comparison to those caused by maternal exposure to nonsteroidal anti-inflammatory drugs, including fetal hydrops, persistent pulmonary hypertension, and death. Since this condition can to progress to fetal death, premature delivery is the best option providing the most favorable outcome. 9 When the fetus is delivered prematurely, there are usually no future issues related to premature closure of the ductus arteriosus.
According to the American Institution of Ultrasound in Medicine (AIUM) guidelines, when examining the fetal heart, a sonographer should examine the four-chamber view, the heart size and position, left and right ventricular outflow tracts, and the three-vessel view. 10 The sonographer should start with a cross-sectional view of the fetal chest. The heart should occupy approximately one-third of the fetal chest with the apex pointing to the left. 11 The normal angle of the heart should be 45 degrees to the left of midline, plus or minus 20 degrees. The left atrium should be closest to the fetal spine and the right ventricular wall closest to the anterior chest wall; both atria and both ventricles should be approximately equal in size. The right ventricle should appear slightly larger than the left. Although it is not in the AIUM practice guidelines, it is the authors’ personal experience that both the aortic and ductal arch are imaged. The aortic arch view is a longitudinal view of the aorta arising from the fetal heart, arching over the left atrium, and descending posteriorly along the fetal spine. 12 This view is characterized by visualization of the brachiocephalic, left common carotid, and left subclavian arteries arising from the top of the aortic arch. Angling the transducer slightly results in the ductal arch view, which includes the pulmonary artery, ductus arteriosus, and descending aorta. The ductus arch is not as tightly curved as the aortic arch and the vessels feeding the head and upper limbs are not visualized in this view. The heart should fill approximately one-third of the chest cavity.
Fetal premature closure of the ductus arteriosus is a rare occurrence that is difficult to diagnose. Furthermore, the incidence and prognosis are not well defined. Before the use of sonography, premature closure of the ductus arteriosus was impossible to diagnose until after fetal death. Today, it is easier to diagnose when a fetal echocardiogram is done in the third trimester. Without a fetal echocardiogram, a definitive diagnosis cannot be made. When a fetal echocardiogram is done, the most common findings are cardiomegaly and a dilated pulmonary artery with a lack of flow through the ductus arteriosus and pulmonary valve regurgitation. 10
While the cause of the cardiomegaly in this case was not determined, a likely cause was premature closure of the ductus arteriosus. Other possible causes that were ruled out were coarctation of the aorta and significant placental dysfunction. With significant placental dysfunction, the cardiac outflow tracts would be within normal limits; in this case, the right outflow tract was enlarged. 7
Conclusion
Idiopathic premature closure of the ductus arteriosus is a rare but serious event. Premature closure of the ductus arteriosus leads to volume overload on the fetal pulmonary circulation, eventually leading to persistent pulmonary hypertension and, in some cases, fetal death. The present case demonstrates how sonography plays a critical role in the diagnosis of premature closure of the ductus arteriosus. Prompt detection and diagnosis along with immediate action will result in the best possible outcome.
Footnotes
Acknowledgements
The authors thank Kelly Schwent, RT(R), RDMS, RVT, RDCS, and Matthew Bosner MD, FACC, FACP, for their assistance, leadership, and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
