Abstract
The goal of this study was to compare the sonographic assessment of endometrial cancer diagnosis and staging using three-dimensional transvaginal sonography with the staging that was ultimately found at surgery. Three-dimensional transvaginal sonography was performed for 20 consecutive cases of presumptive endometrial cancer using power Doppler angiography, virtual organ computer-aided analysis, and volume contrast imaging. These results were compared with the surgical staging found for those identified cases. The sonographic findings predicted the correct minimum endometrial cancer staging in 100% of the 20 cases that were examined and correlated to surgical outcomes. The successful prediction of endometrial cancer staging demonstrated by others was replicated in the organization of this case series. The findings compared favorably with other imaging modalities used for this purpose. This study assists in demonstrating the value of using presurgical three-dimensional sonography to plan for the optimal surgical excision and overall treatment of endometrial cancer.
Atypical endometrial hyperplasia (AEH) is also referred to as carcinoma in situ, or endometrial carcinoma (EC), stage 0, although this is not currently consistent with the International Federation of Gynecology and Obstetrics staging. As the continuum of endometrial neoplasia is recognized to extend from AEH through EC, we may need to predict noninvasively its natural biological behavior, to better prepare the gynecologic oncologist surgeon planning to perform the required surgical treatment(s). 1 It may be particularly useful to identify women experiencing postmenopausal bleeding (PMB) for evaluation, because a significant number has yet been diagnosed with EC. 2
When EC is found, its staging determines the optimal surgical treatment, and the staging itself correlates with the depth of myometrial invasion and lymphatic spread. 3 Recently, determining the cancer invasion in a sentinel node has been used to predict the likelihood of cancer spreading to those pelvic lymph nodes and therefore the possible need for lymphadenectomy.4–6 Specifically, for stage IB or greater, lymphadenectomy should be performed for maximum patient survival. Considering the lymphedema associated with lymphadenectomy, the need for performing the procedure may be ideally predicted prior to the surgical treatment itself, so as to possibly avoid or reduce the associated morbidities. If the need for lymphadenectomy can be determined by presurgical three-dimensional transvaginal sonography (3D TVS), benefit to both the surgeon and patient may be provided. Therefore, this study was designed to investigate the use of 3D TVS techniques and to identify the features of this neoplastic entity preoperatively, compared with those features with postsurgical histology.
Materials and Methods
All patients in this investigation who were clinically diagnosed with AEH or EC were examined sonographically prior to surgery using 3D TVS gray-scale power Doppler angiography (PDA) and virtual organ computer-aided analysis system and volume contrast imaging (VCI) from April 1, 2015, to January 3, 2018. The protocol for the VCI, previously described by Jantarasaengaram et al., 7 was followed for each case. All patients underwent a subsequent surgical procedure within 3 months after TVS, and the resulting histology was compared with the preoperative ultrasound findings. A Voluson E8 system (GE Healthcare Ultrasound, Milwaukee, WI, USA) with transvaginal probe (RIC 5- to 9-MHz transducer) was used for all sonographic analyses in this investigation, which was approved by the Institutional Review Board of the organization where the clinical care was provided.
Sonographic Analysis
For all patients enrolled in the study, 2D and 3D TVS was used to identify the staging of each case of EC. Respective sonographic analysis determined the tumor/uterine anteroposterior (AP) ratio. Deep myometrial infiltration was diagnosed when the tumor was greater than or equal to 50% of the total thickness of the AP diameter of the uterine wall, as first described by Karlsson et al. 8 Using 3D virtual navigation, later described by Alcazar et al., 9 this investigation calculated tumor-free minimal margin tumor distance to serosa (TDS). Deep myometrial infiltration was diagnosed when the minimal margin of the TDS was less than 9 mm. 9 One sonographer (C.M.F.) provided all of the sonogram volume acquisitions and analyses.
The presence of stromal tumor invasion of the uterine cervix was also determined using the approach described by Epstein et al. 10 This methodology included the objective findings of the distance from the external cervical os to the lower margin of tumor (Dist-OCO) and the subjective findings of the dynamic sonographic technique to demonstrate tumor extension into the cervical stroma. A negative “sliding test” (absence of vascularity when moved) enables possible demonstration of vascularity directly to the tumor. A cutoff value of 20 mm was used for the determination of invasion.
Results
The identified sonographic features of the cases enrolled in this study were compared with the intraoperative and postoperative pathologic findings. After analysis of 20 patients, the sonographic findings predicted the minimum EC staging 100% of the time. Deep myometrial invasion (⩾50%) and/or cervical stromal invasion was specifically identified in eight cases and was associated with the surgical staging of EC. Table 1 illustrates this association, noting the sonographically predicted and actual histology for each case.
Case Descriptions.
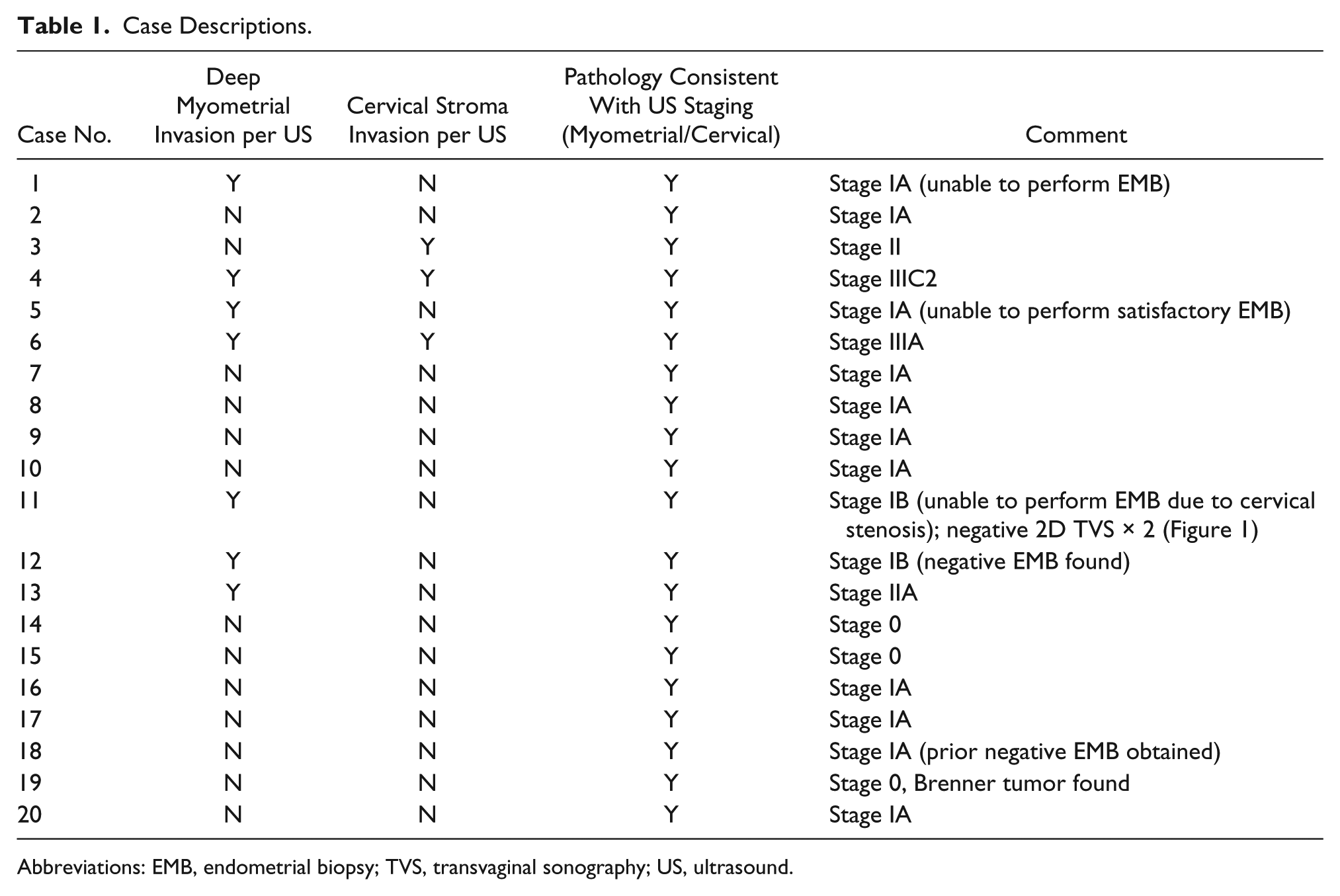
Abbreviations: EMB, endometrial biopsy; TVS, transvaginal sonography; US, ultrasound.
As is noted in Table 1, presurgical histologic sampling of the uterine cavity could not be accomplished in three cases because of cervical stenosis (cases 1, 5, and 11). In two cases, a negative biopsy was obtained prior to the sonogram, although both subsequent 3D TVS and surgical pathology report revealed EC. Figure 1 displays a normal 2D TVS. Figure 2 displays an abnormal vascularization using 2D with PDA. Figure 3 reveals a suspected stage IIB EC using 3D TVS with a 1-mm VCI scan of that same case with two normal previous 2D ultrasound and inability to perform endometrial biopsy in a case of PMB due to cervical stenosis. The deep myometrial invasion toward the right fundus can be seen in this image. Stage IIB EC was confirmed with pathology.
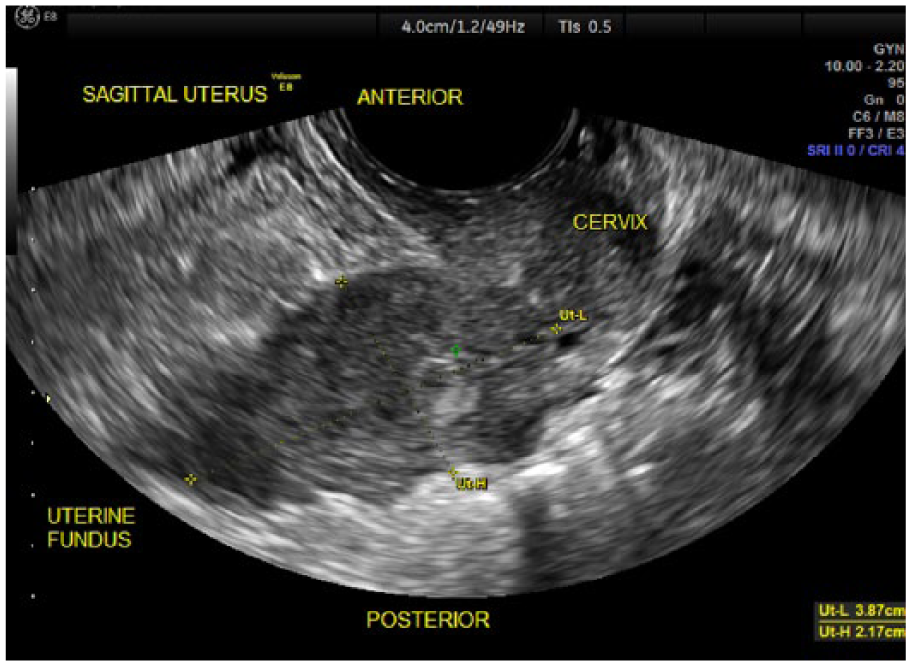
Two-dimensional transvaginal sonography, demonstrating poor visualization of the endometrial echo in a sagittal view of the uterus.
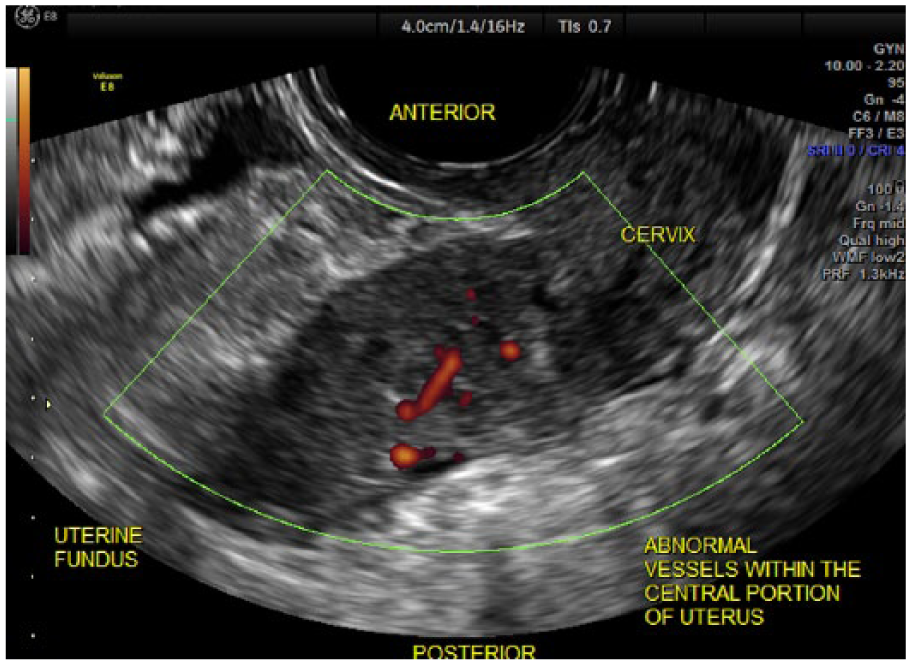
Two-dimensional transvaginal sonography with power Doppler angiography, demonstrating abnormal vessels in the central portion of the uterus.
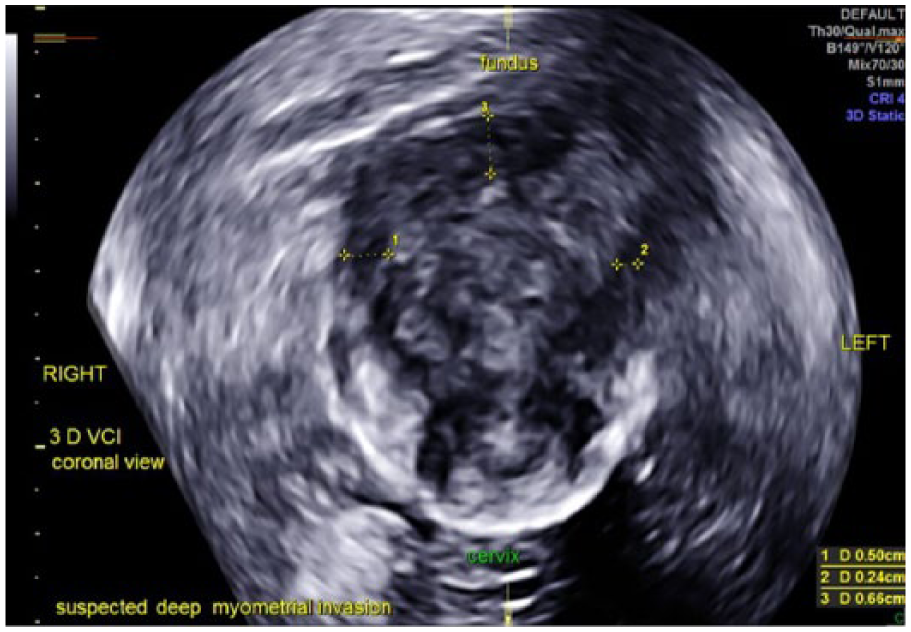
Suspected stage IIB endometrial carcinoma using three-dimensional transvaginal sonography with 1-mm volume contrast imaging scan, demonstrating asymmetric tumor borders, short tumor to serosa distance of 2.4 mm, and deep myometrial invasion of the left fundus of the uterus.
Conclusions
This investigation validates the findings of Alcazar et al. 9 and others9–14 that using sonography to identify the features of EC can properly predict the optimal surgical treatment of this oncologic condition. The methodology employed in this investigation is consistent with that of the International Endometrial Tumor Analysis collaborative group.15,16 The clinical importance of these methodologies should be recognized for the impact they can have on patient care, since there is often difficulty in properly obtaining a diagnosis of EC when it presents as PMB.17,18 An example of this situation is when cervical stenosis is encountered, which can prevent histologic sampling of the endometrium, as was encountered in this case series. Even the histologic sampling of a presumed neoplastic lesion can have limitations of accuracy, especially in women with PMB. 19 In addition, diagnostic uncertainty from a 2D sonographic examination can further contribute to difficulties in the management of this EC condition, as was true in one case in this series (case 11).
Other medical techniques have been used to predict the staging of EC, which can be compared with 3D TVS.20–26 Table 2 compares various imaging modalities that have been used for predicting the staging of EC. It is noted that intraoperative frozen section, which may not be available in all institutions, has its own limitations of accuracy, although its prediction at one institution was excellent. 27 It may be observed that there exists a wide variance in the prediction of EC staging from 3D TVS, which explains why the predictive value this technology may have is very user dependent. Nonetheless, 3D TVS has previously confirmed 100% sensitivity for detecting EC stage IB or greater, as demonstrated in our small series of cases. A recently reported meta-analysis did not find comparable results to this study. However, that may underscore the operator dependence of such an assessment. 28
Prediction of Endometrial Cancer Staging/Lymphatic Spread (Sensitivity).
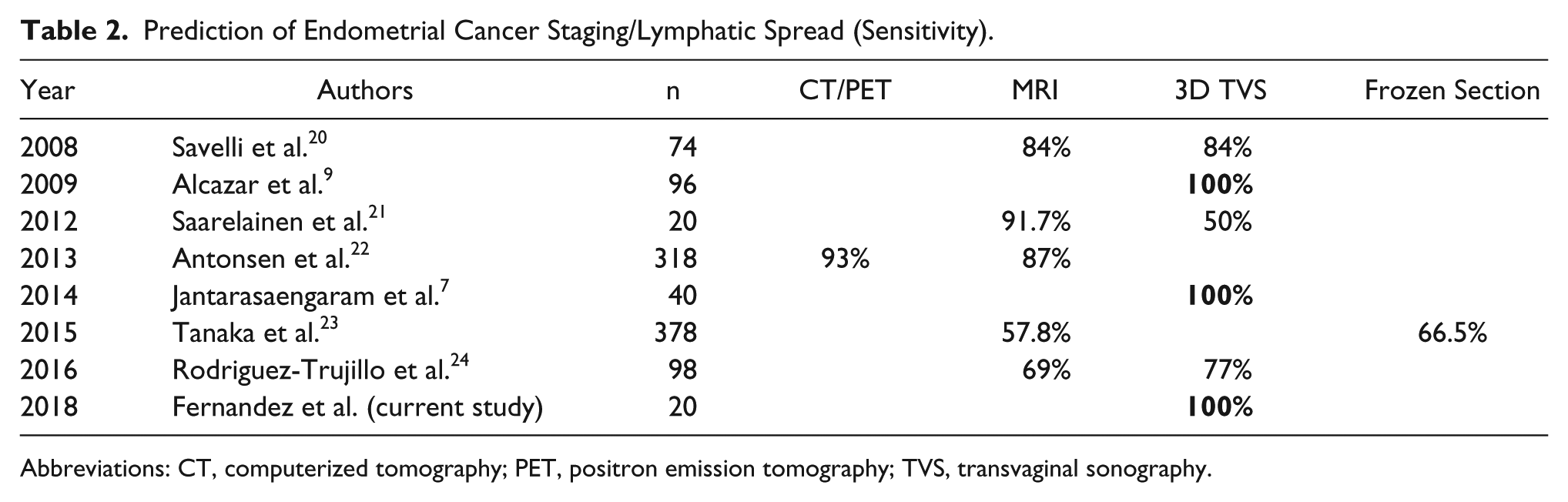
Abbreviations: CT, computerized tomography; PET, positron emission tomography; TVS, transvaginal sonography.
Recently, determination of cancer invasion in a sentinel node has been used to predict the likelihood of cancer spread to the pelvic lymph nodes and therefore the possible need for lymphadenectomy. The extent of the efforts needed to satisfactorily perform sentinel lymph node mapping4–6 may need to be considered, along with the potential predictive value of 3D TVS. Different methodologies have been used for mapping the sentinel node (e.g., colorimetric, radionuclear, and near-infrared methods), and the efforts required for mapping should not be underestimated.
The limitation of this current study is the small convenient sample of patients. An advantage is the illustration of the value and capabilities of current technology for the care of patients with EC. Another strength is the demonstration of the practical use of 3D TVS in a clinical setting. Having this information provided prior to the scheduled surgery may be the greatest benefit provided by presurgical 3D TVS. As the recognition of PMB and the proportion of EC that it represents continue to be reinforced by gynecologists, the routine use of 3D TVS appears to play an important diagnostic role. Given the correlation between the surgical staging of EC and presurgical 3D TVS, this new application should be considered as a routine primary diagnostic method for patients who are likely to have EC.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
