Abstract
Sonography currently provides a variety of tools to support the diagnosis and treatment of osteosarcoma and, with the development of therapeutic ultrasound practices, could play an increasingly important role in the treatment of future patients with osteosarcoma. Currently, computed tomography, magnetic resonance imaging, and radiography are the preferred imaging modalities for bone lesions; treatment options for osteosarcoma primarily include highly toxic chemotherapies and surgeries that often result in limb loss. Unfortunately, osteosarcoma is rarely eliminated, and despite this, patients lose their lives after having a significant reduction in quality of life. Evolving therapies such as high-frequency focused ultrasound, ultrasound-enhanced delivery of photodynamic therapy, and low-intensity pulsed ultrasound may offer these patients an improved quality of life while also increasing efficacy of treatment. This canine case study illustrates the various ways in which sonography might contribute to the treatment plan for patients with osteosarcoma. It may provide a comparable model for the reimagining of treatment for future patients with osteosarcoma using a suite of therapeutic ultrasound applications.
Keywords
Current applications of sonography in the diagnosis and treatment for patients with osteosarcoma are varied, and while they support several aspects of a care plan, plain-film radiography is the primary imaging modality for the evaluation and management of this disease. Standard grayscale imaging of abdominal organs may be used in the staging of newly diagnosed malignancies and can offer a safe way to perform routine monitoring in the presence of metastatic disease. Diagnosis may only be confirmed with cytology; therefore, invasive procedures such as needle aspiration and biopsy are integral to the identification of disease. Sonography again supports this vital task with real-time image guidance, thereby improving the accuracy of tissue sample collection. Finally, echocardiography is used to evaluate the cardiotoxic effects of frequently used chemotherapy drugs, helping to balance the desired effects of the drug with the unintended systemic effects.
Interestingly, many aspects of this disease are seen in both veterinary and human medicine, making it possible to study the outcomes of new treatments in both canine and human populations. Future applications of sonography are of special importance, as they potentially offer safer and less invasive treatment options for osteosarcoma patients of either species. Considering the high frequency of osteosarcoma among canine patient populations, an introduction of emerging therapeutic applications of ultrasound to the veterinary community could offer pet owners access to cutting-edge alternative therapies while also increasing opportunities to study patient outcomes in a clinical setting. This type of partnership could be of great benefit to the development and ultimately the approval of ultrasound therapies for use in human medicine.
Discussion of Disease Process in Osteosarcoma: A Comparative Approach
Animal trials are critical to safety testing and approval of new therapies for use in human medicine.1,2 While canine trials represent a particular animal rights challenge due to domestication, there is some evidence that canine models are more predictive of the human response than rats or mice, are a closer genetic match, and are exposed to environmental and lifestyle factors that mirror those found in the human patient population.1,2 Osteosarcoma is particularly well suited for canine study due to the many similarities in etiology, incidence, differential diagnosis, pathophysiology, treatment, and prognosis.1–3
Background
The etiology of osteosarcoma is unknown in both species; however, there is an association with rapid bone growth, bone injury, and subsequent repair.1 –3 Direct injury to the bone contributes to environmental changes (such as pH or hormone), stimulating increased activity of mesenchymal cells, and osteoblasts proliferate in response.1,3 Osteoclasts carry out normal lytic functions, but malignant osteoblasts haphazardly deposit immature bone matrix, leading to tumor growth.1,3 Several genetic alterations contribute to disease progression and are considered risk factors for the disease.1–3
The incidence of osteosarcoma between canine and human patients is remarkably similar. 1 Among patient populations, osteosarcoma is the most common primary bone lesion, and sites most commonly affected are the femur, tibia, and humerus.1–3 The frequency of canine occurrence is significantly higher, due primarily to selective breeding practices. 1 In canines, giant and large breeds are at the greatest risk. 3
Considerable overlap is also seen in differential diagnoses. Differentials shared between the canine and human populations include osteoblastoma, chondrosarcoma, fibrosarcoma, and fibroblastic dysplasia.1,2 In humans, Ewing sarcoma and lymphoma are also important considerations. 4 Metastatic disease is frequently seen in both canine and human patients.1,2 Regardless of species, disease progression begins with malignant changes to mesenchymal cells of the bone metaphysis, leading to the erratic formation of immature bone or osteoid tissue along the interior or exterior surfaces of normal bone tissue. 5 Tumor progression causes focal pain and swelling, which often responds well to nonsteroidal anti-inflammatory drugs (NSAIDs).1,3 Pet owners generally complain of sudden lameness in canines; humans often complain of increasing pain, swelling, and limited range of motion.1,3 Disease progression beyond initial tumor formation almost always includes metastasis—other bone sites as well as vital organs, primarily lungs, liver, or kidneys, are frequently affected; pulmonary metastasis is commonly the direct cause of death.1,2
Prognostic outcomes for patients with osteosarcoma are generally poor. Pulmonary metastatic disease occurs with high frequency and is often the cause of death in both canine and human patient populations.1,2 Surgical treatment options are aimed primarily at complete excision of malignant cells and in many cases leads to limb loss.3,5 Aggressive chemotherapy, frequently doxorubicin, is chosen as a follow-up to surgical intervention, and therapeutic radiation is sometimes used as a palliative approach to treatment. This pattern of care is relevant in both human and canine medicine. Despite these efforts, many patients lose their lives to osteosarcoma. In canines, the disease is considered fatal; however, the later stage disease progression commonly discovered upon initial examination may contribute heavily to this statistic.6–8 The long-term prognosis for human patients is somewhat less grim, although 5-year survival rates are only about 68%.4,9
Case Study
A neutered male Rottweiler, age 7 years and 3 months and weighing 49.6 kg, was referred for symptoms suspicious for appendicular osteosarcoma. The owner reported sudden lameness of the right rear leg. Plain-film radiologic evaluation revealed a lytic lesion in the neck of the right femoral head (Figure 1). Upon discovery of the lesion, additional views were ordered to evaluate the patient. Right lateral, left lateral, and ventral-dorsal views were obtained. At the time of the initial exam, metastasis to the lungs and other bone sites was ruled out. Sonographic examination was offered to the owner to check for solid organ metastasis but was declined. A fine-needle aspiration of the affected area was recommended to confirm diagnosis and was successfully performed under sonographic guidance (Figure 2). Cytology results (Figure 3) confirmed the diagnosis of osteosarcoma, and a treatment plan consisting of amputation of the affected limb and an 18-week chemotherapy regimen of carboplatin was agreed upon. Limb-sparing surgery was not an option for this patient due to the location of the lesion.
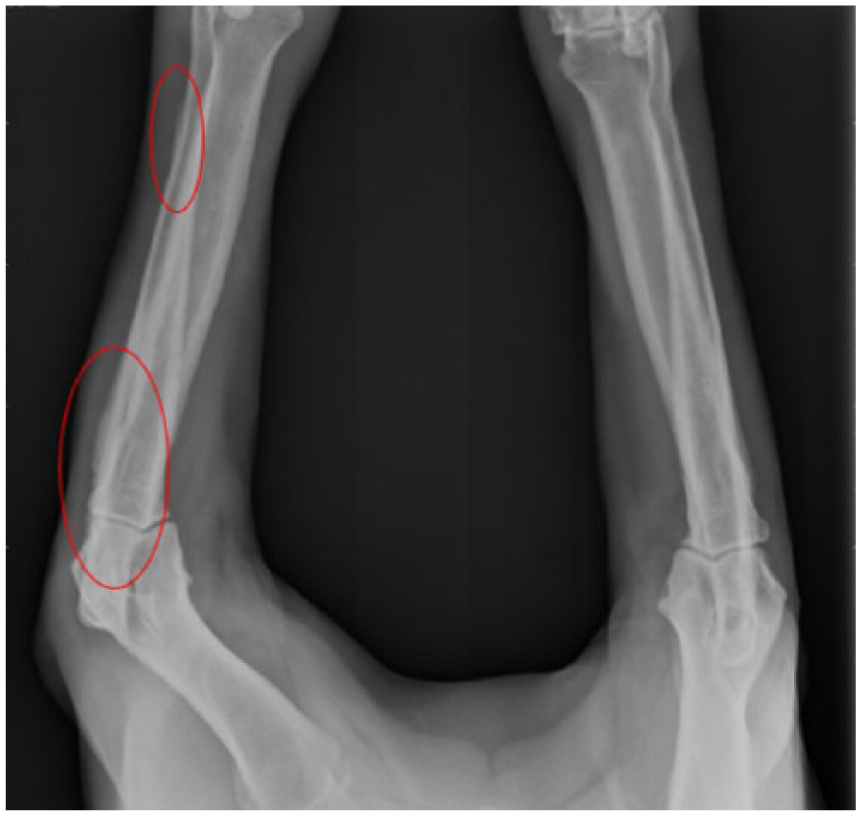
Initial plain-film radiography; collected upon initial consultation. Area of interest (circled in red) reveals a lytic bone lesion along the lateral border of the right femur. Lower extremity anteroposterior view.
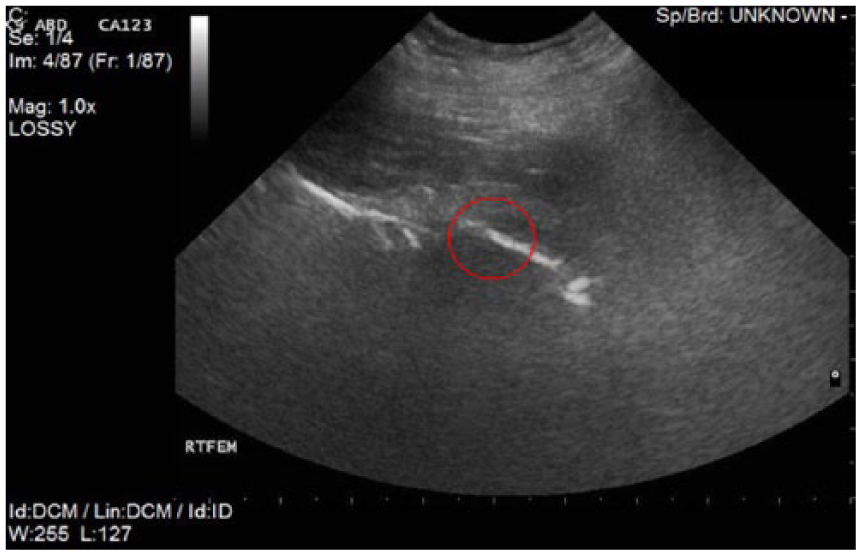
Sonographic image obtained during ultrasound-guided biopsy procedure performed at the right rear lateral hip.
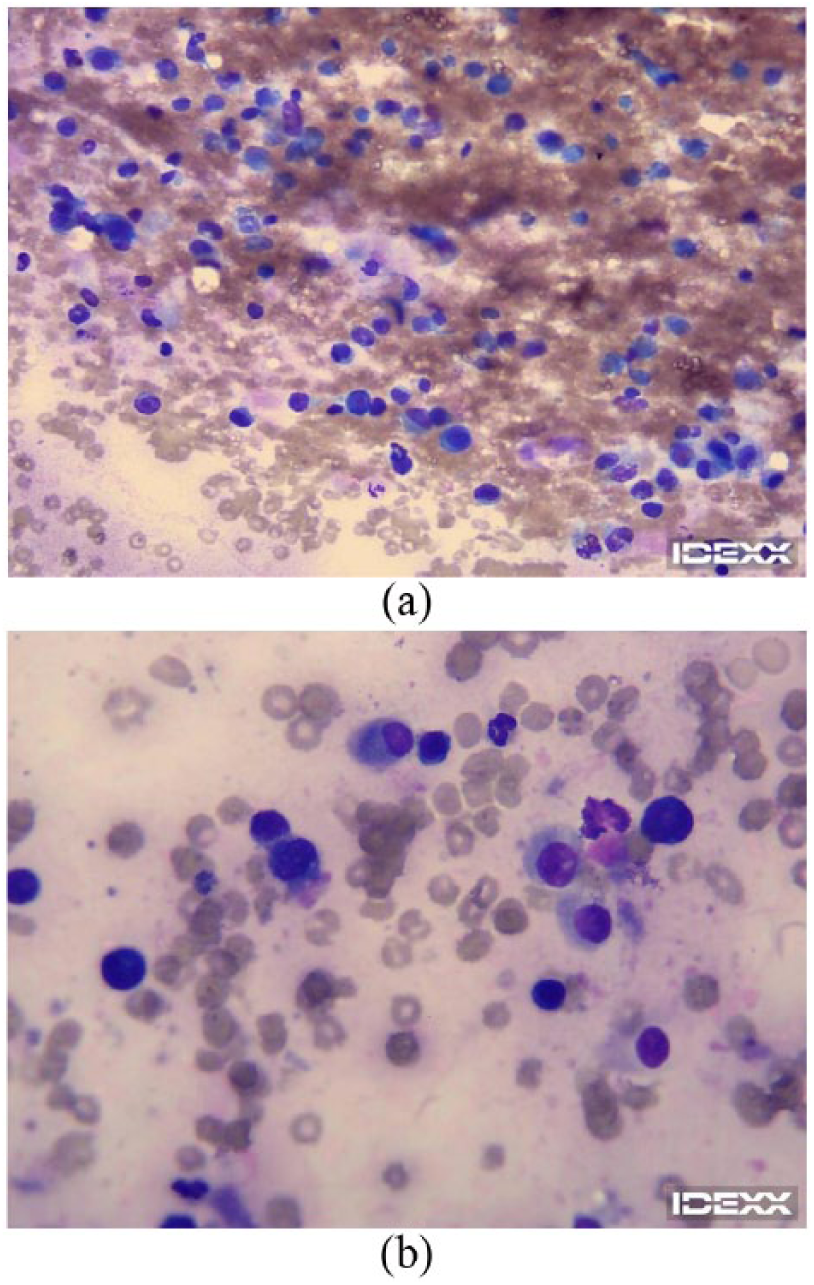
Cytology images from the specimen collected during biopsy of bone lesion. Large round cells stained purple represent malignant osteoblasts.
Thirty days following a successful amputation, carboplatin therapy began; however, after just one round of chemotherapy, the patient’s platelet count dropped below acceptable levels, and the treatment was delayed. Once healthy platelet levels were achieved, treatment was resumed. Although treatments were delayed occasionally due to side effects of chemotherapy, the patient was able to complete the 18-week cycle and begin recovery. Symptoms returned after several months, and metastasis to other bone sites as well as the lungs was confirmed via plain-film radiograph (Figure 4). With the onset of systemic metastasis and amputation a previously exhausted option, an aggressive chemotherapy regimen with doxorubicin, a key treatment choice in the treatment of osteosarcoma, was initiated. Doxorubicin is highly cardiotoxic and therefore prescribed with careful consideration to the patient’s ability to tolerate treatment. 10 Echocardiography is used to evaluate the health of the heart every fourth week of treatment. A fractional shortening test measures the ability of the left ventricle to eject blood; a fraction of 43% is considered safe for the continuation of treatment. Performed with a 5- to 8-MHz pediatric curved transducer, using a Esaote MyLab (Esaote North America, Fishers, IN) portable sonography instrument, a fractional shortening exam was completed. Unfortunately, test results were unsatisfactory (Figure 5), and the patient was unable to complete this therapy due to nonreversible enlargement of the heart muscle. The patient was undergoing palliative care at the time of this article, including nutritional support, NSAIDs, and fentanyl patches, to manage extreme pain associated with significant bone metastasis (Figure 6).
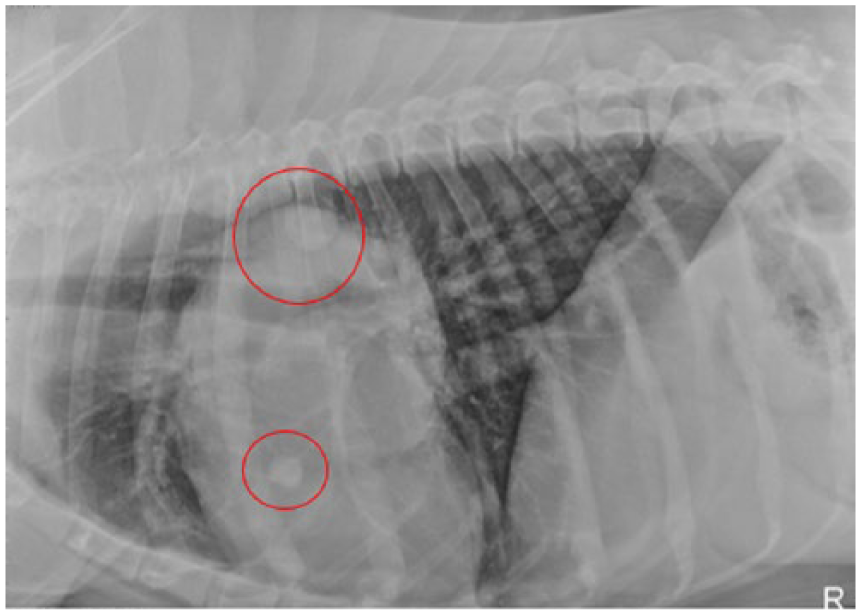
Subsequent plain-film radiography demonstrating metastasis to lung. Right thoracic lateral view.
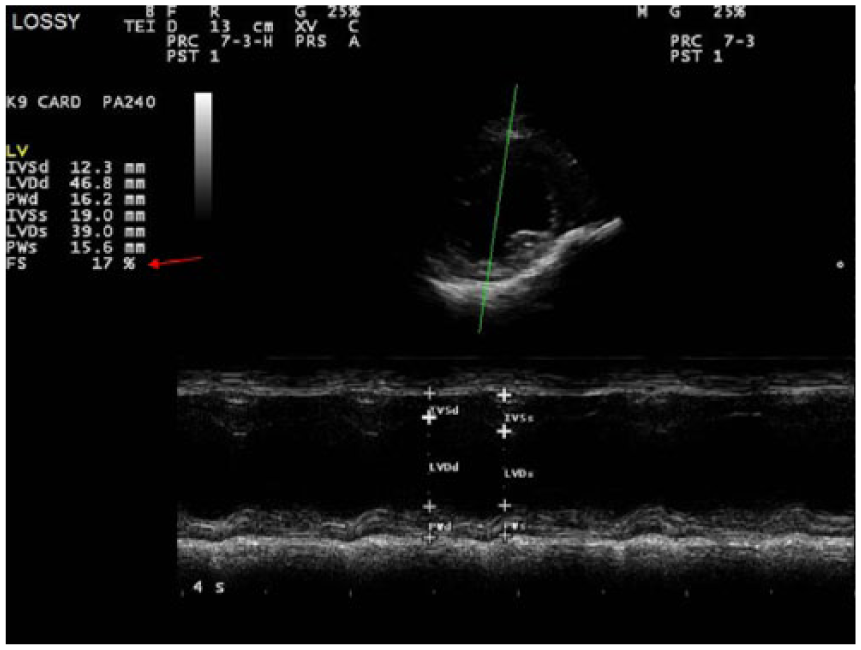
Echocardiogram performed during the fourth week of treatment with doxorubicin. The red arrow indicates the percentage of fractional shortening, measured in this test. Results are 17%, well below the minimum to continue treatment (minimum acceptable fractional shortening percentage is 43%).
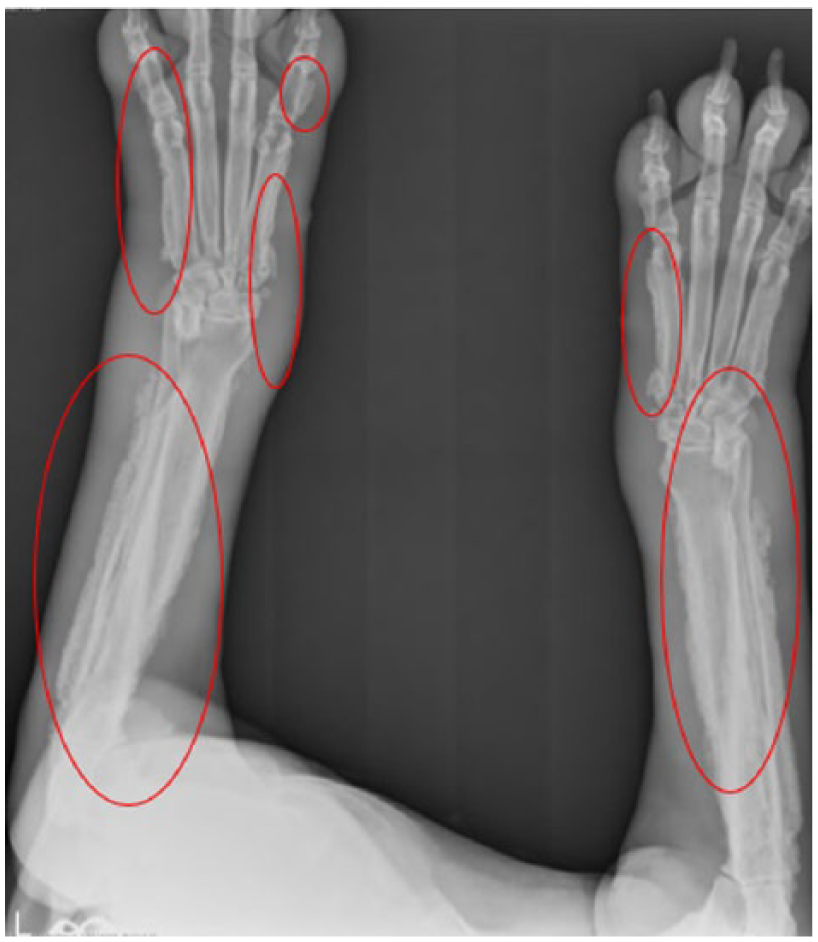
Subsequent plain-film radiography demonstrates significant metastasis to distant bone sites; in this image, medial and lateral surfaces of the right and left humerus, as well as multiple bilateral carpal and digital sites, are affected. Edema is also visible adjacent to metastatic lesions. Upper extremity anteroposterior view.
Discussion
This case study demonstrates two of three ways sonography is currently used to diagnose and support the management of treatment in patients with osteosarcoma. Plain-film radiography is the preferred standard for imaging bone lesions; sonography currently plays a supportive role in the care plan for patients with osteosarcoma.4,9 While not a part of this case, sonographic imaging of the abdomen is often recommended as an avenue to evaluate the solid organs and lymph nodes for metastatic disease, thereby offering support in the staging of disease. 3 Once the lesion was imaged and a lytic bone lesion was identified, sonography was used to improve the accuracy of fine-needle aspiration. The aspirates collected from this procedure confirmed the diagnosis, and an appropriate treatment plan was able to be built based on the cytology findings. Cardiac sonography was used to measure the ejection performance of the left ventricle, offering a warning that chemotherapy was not well tolerated by the patient and should be discontinued.
As is often seen with osteosarcoma, disease progression was rapid and difficult to treat, with the patient experiencing a markedly reduced quality of life; however, exciting advances are being made in the field of therapeutic ultrasound that have the potential to overhaul the approach taken by medical science in the treatment of osteosarcoma. While plain-film radiography is likely to remain the preferred imaging exam to evaluate bone lesions, ultrasound may someday replace difficult surgeries and aggressive chemotherapies. Some of the promising applications currently in research and trial stages include low-intensity pulsed ultrasound (LIPUS), ultrasound-enhanced delivery of photodynamic therapies (PDTs), and high-intensity focused ultrasound (HIFU).
LIPUS already has been established in orthopedics as a safe method to induce fracture healing and bone growth.4,9 Current research in this area suggests that when LIPUS is applied to lytic bone lesions of any tissue origin (primary or metastatic lesions), the natural healing and growth processes are preserved but do not increase the proliferation, migration, or vascularization of malignant cells. 9 Malignant cells in osteosarcoma intermingle with healthy cells, benefiting from the constant source of growth stimulation occurring at the epiphyses and presenting a difficult problem for treatment. 9 Currently, bone lesion removal and repair often result in distant organ metastasis; LIPUS offers a promising solution for both pain and tumor reduction while minimizing unwanted systemic effects.4,9
PDT has had promising results when applied to tumors, successfully deactivating malignant cells through reactive oxygen species (ROS), produced by light activation of a photosensitizer.6,8 One photosensitizer with particular promise is hematoporphyrin monomethyl ether (HMME); however, the poor penetration of light energy through tissue limits the success of this therapy. 6 Recent studies in animal models have demonstrated that the mechanical waves created by ultrasound, known to have predictable tissue penetration properties, greatly improve efficacy of HMME treatments. 6 Following an intravenous injection of HMME, continuous wave ultrasound was applied focally with a 10.5-MHz transducer for measured intervals of time, and tissue samples were evaluated to quantify the percentage of actively proliferating malignant cells. Results from this study indicate that the application of HMME alone had little reductive effect on malignant cell proliferation (64% compared to the untreated control group rate of 66%), but therapeutic ultrasound alone reduced the number of proliferating malignant cells to just 25%; when applied in conjunction with HMME, that percentage was reduced to just 15%.6,8
HIFU is known to cause tissue cavitation and is currently being examined as a means for targeted drug delivery in the treatment of osteosarcoma and other inoperable tumors.10,11 The mechanical waves produced by ultrasound increase cell membrane permeability, allowing the selective treatment of tumors with chemotherapeutic drugs while preserving healthy tissue.11,12 This approach has been limited, however, by molecule size; bubbles needed to be as small as an erythrocyte to pass through to targeted tissues. A recent study using this type of therapy demonstrated that the use of microscopic lipomas (termed bubble liposomes), as opposed to gas bubbles, increased the tumor uptake of chemotherapeutic drugs by 57%. 11 This study is particularly relevant in the treatment of osteosarcoma because subjects exclusively included patients whose osteosarcoma was being treated with doxorubicin, a widely used and well-established choice for chemotherapy treatment.10,11,13
Conclusion
While most of the treatment options discussed are still in experimental stages and obviously cost-prohibitive for the average pet owner, there is evidence that canine trials for medical procedures offer more reliable insight to success in human trials than those conducted in other species. Genetically, canines and humans are a closer match than either species is to mice or rats, and cohabitation ensures strong similarities in environmental and lifestyle factors.1,2,11 Opportunities for pet owners to take advantage of these breakthrough technologies could be a means to preserve the family pet while also strengthening the case for the use of therapeutic ultrasound in human medicine. Osteosarcoma primarily affects young adults in the human population and, similar to canine incidence, is a common primary bone malignancy that carries a dire prognosis.1,2 Disease process and treatment options are parallel between species; so too is the significantly diminished quality of life following treatment.1,2,13
Considering this case study, it is possible to imagine a more positive outcome for the patient as therapeutic applications of ultrasound are approved. Patients with limb loss face a difficult transition; therefore, avoidance of amputation is an important benefit of any effective treatment option. The safe and commonly practiced LIPUS provides a means to stimulate growth and repair of the weakened bone matrix while having no stimulatory effects on tumor cells. Also of high importance in the preservation of patient quality of life is reduction of systemic effects of chemotherapy. PDT with enhanced delivery via ultrasound could offer a less toxic approach to the destruction of malignant cells, thereby avoiding the use of highly toxic chemotherapy drugs. With the similarities between canine and human disease progression of osteosarcoma, increasing the number of preclinical trials could prove beneficial. Opening treatment trials to pet owners interested in alternative therapies offers an interesting avenue to developing therapeutic ultrasound techniques that could entirely change outcomes for patients with osteosarcoma in the future.
Footnotes
Acknowledgements
Contributions to this article were made by the entire team at Veterinary Specialists of North Texas. Special thanks to Dr. Allstadt and Dr. King for providing detailed information regarding patient care and treatment rationale, as well as to Linda Wogeman and Jennifer Conaway for providing research support and knowledge of veterinary sonography. In addition, the authors thank Lynn Schluns, RDMS, RVT, BS, DMS Lynn is the DMS Program Director at El Centro College, and El Centro College Allied School of Health and Nursing, Dallas, TX.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
