Abstract
High-intensity focused ultrasound (HIFU) is a noninvasive, nonionizing means to therapeutically treat various medical conditions. Although HIFU has proven useful in the treatment of a variety of conditions, in recent years, more research has been conducted on how HIFU treatments can be used to treat conditions related to the female pelvis. Some of the medical conditions being researched are uterine fibroids, adenomyosis, cervicitis, and polycystic ovaries. Clinical studies have demonstrated the effectiveness of HIFU in the treatment of these medical conditions unique to the female pelvis. This literature review will be used to introduce the technology of HIFU, present an overall analysis of HIFU, provide a review on the latest clinical research concerning HIFU clinical applications for pathologic conditions of the female pelvis, and identify the impact of HIFU on patients and the ultrasound community.
Keywords
Since the late 1960s, diagnostic medical sonography has been aiding the field of medicine as a cost-effective, real-time, noninvasive, nonionizing means of imaging the human body. 1 Generally viewed as a safe method for imaging normal anatomy and pathology, little thought is given to the potential biologic effects that can be caused by ultrasound. In fact, with a high enough intensity, ultrasound has the ability to permanently damage cells by creating both thermal and nonthermal bioeffects. For this reason, sonographers use the “as low as reasonably achievable” or ALARA principle to minimize patient exposure to ultrasound in an attempt to avoid any potential biologic effects. 2 Conversely, what if this harmful damage to cells could be used to treat pathology? High-intensity focused ultrasound (HIFU) is a noninvasive, nonionizing means to therapeutically treat various medical conditions. 3 Such medical conditions include the ablation of both benign and malignant tumors throughout the human body, as well as the treatment of Parkinson disease, atrial fibrillation, and a means of drug delivery to the human body.
Although HIFU has proven useful in the treatment of a variety of pathologies, in recent years, more research has been conducted on how HIFU treatments can be used to treat conditions related to the female pelvis. These conditions include but are not limited to uterine fibroids, adenomyosis, cervicitis, and polycystic ovaries. As technology advances and research continues, HIFU is proving useful in the therapeutic treatment of pathologic conditions unique to the female reproductive system. This article will be used to review the technology of HIFU, present an overall analysis of HIFU, and provide an overview of the published evidence concerning HIFU clinical applications for pathologic conditions of the female pelvis.
Background
History
Robert Williams Wood and Alfred Lee Loomis first discussed the damaging effects of high-intensity ultrasound on biologic organisms and living tissues in 1927. 1 The first therapeutic theory was introduced more than a decade later. In 1942, Lynn et al. hypothesized that soft tissue could be destroyed through localized thermal destruction with the use of focused high-intensity ultrasound waves. 4 Subsequently, the first clinical application came about after more research had been conducted. It was not until 1958 when the first therapeutic treatment with HIFU was used in the neurosurgical treatment for movement disorders, neuropathic pain, and hypersensitivity. 5
In the past few decades, HIFU technology has reached several more milestones. The 1980s brought forth the first HIFU device approved by the Food and Drug Administration (FDA) for the treatment of glaucoma to be performed in the United States. 5 Current HIFU applications use real-time monitoring by magnetic resonance imaging (MRI) and sonography. MRI has the ability to monitor the temperature changes of tissue being treated with HIFU. The first FDA-approved use of MRI-guided HIFU was for the treatment of uterine fibroids in 2004. 6 Clearly, HIFU technology has matured since its first clinical use in 1958. The technology will continue to advance based on the outcomes of clinical studies and the continued approval of therapeutic applications by the FDA.
Process/Physics
HIFU procedures harness the bioeffects created by high-intensity ultrasound to therapeutically treat a variety of medical conditions. The bioeffects can be placed into two groups: thermal and nonthermal. The type of procedure being performed determines which type of bioeffect is desirable and used for each treatment.
Thermal biologic effects
As ultrasound propagates through tissue, the energy is absorbed and converted to heat. During a HIFU procedure, thermal biologic effects are created by tightly focusing the high-intensity ultrasound waves onto the targeted area. Transducer frequencies for HIFU treatments vary and depend on the type of procedure being performed and the depth of penetration needed. The typical range of frequencies for HIFU thermal ablation is 600 kHz to 7 MHz. 7 Ablation treatments require the use of continuous ultrasound waves that are focused onto the desired target area. For ablation to occur, the tissue must heat up to 60°C in the focused area. 4 Tissue coagulation and necrosis will begin as the tissue proteins are heated.
Nonthermal biologic effects
Also known as mechanical effects, nonthermal biologic effects are able to damage tissue without the use of heat. One such nonthermal effect used with HIFU is known as cavitation. When HIFU is applied to specific tissue, micro-bubbles of gas within the tissue begin to oscillate rapidly. This rapid oscillation causes the gas bubbles to collapse, creating inertial cavitation. The force from the collapse will in turn destroy or liquefy the cells of the targeted tissue. Unlike the continuous waves of ultrasound used to create thermal effects, nonthermal effects are created using extremely short pulses of ultrasound. To create cavitation within the targeted tissue, these short pulses of sound last approximately 1 to 2 ms with high amplitudes up to 100 MPa. 4 A procedure used to destroy areas of tissue by creating multiple cavitation collapses through inertial cavitation is known as histrotripsy.
Result of bioeffects
Once bioeffects have been produced in the targeted tissue, tiny elliptical shape lesions, known as focal spots or sonications, are left behind. 5 Sonications correspond with the focal size of the HIFU system. Current focal spot size ranges from 4 to 60 mm in length and 2 to 16 mm in diameter. 5 To cover a larger volume of a targeted area, multiple sonications are placed over the desired area without gaps. HIFU procedures can be lengthy depending on the size of the target being treated. A pause is needed between each sonication treatment to prevent overheating and boiling of the surrounding tissues. 4 Exposing tissue to high-intensity ultrasound has proven to be an effective means to causing intentional death to targeted tissue. In fact, exposing tissue to a temperature of 56°C (130°F) for a single second is enough to cause thermal damage to cells from protein destruction. 5
Equipment
The equipment used to administer therapeutic ultrasound treatments consists of two main components. The first is the piezoelectric ultrasound transducer. One type of transducer used is self-focusing and spherical shaped with a fixed aperture and focal length, which is the simplest, most economical, and most popular design used for transducer construction. 4 Another type of transducer used in HIFU treatments has a phased array. Phased-array transducers comprise multiple elements and are constructed with a concave design for a natural focus.4,8 Mechanical movement of the transducer is used for the overall position of the focal spot, whereas the electronic steering of the ultrasound beam is used for the fine control of focal spot location. 8 An advantage to the phased-array design is the ability for fast electronic beam-steering and beam-forming capacity through the adaptive selection of elements. 4
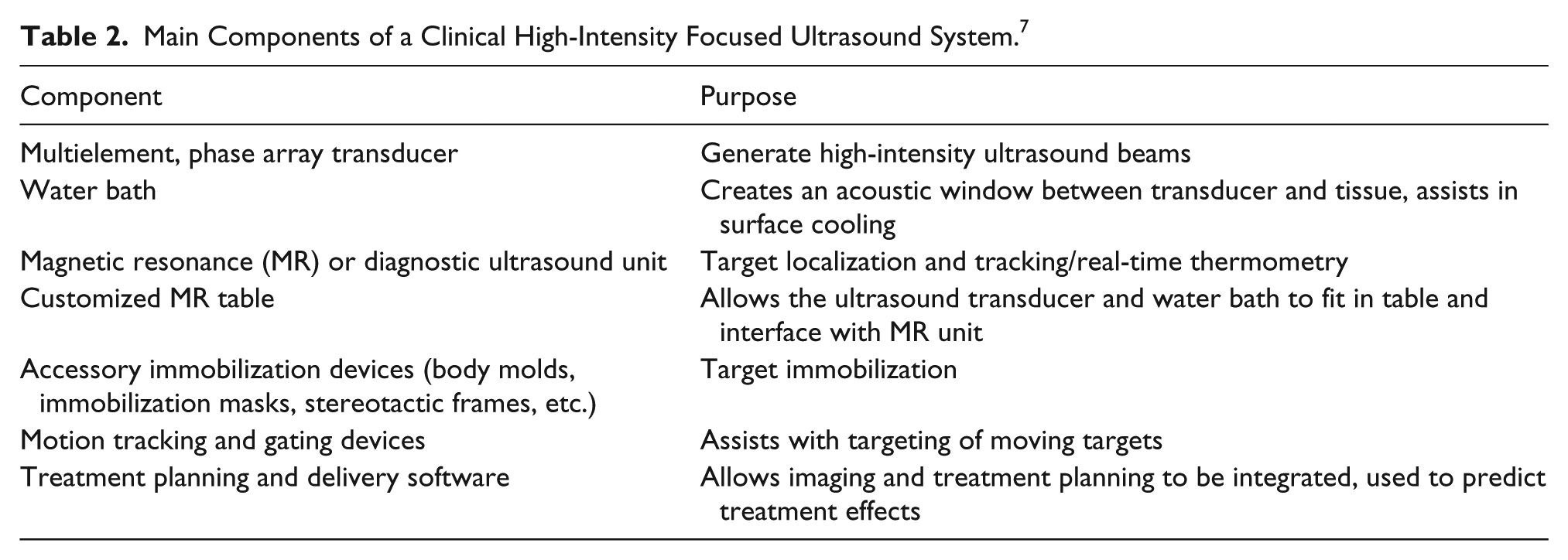
The imaging modality used to monitor the ablation process is the second component of HIFU. Real-time imaging is essential to the monitoring of treatments. Two medical imaging modalities are used for monitoring. These two modalities are sonography and MRI. Imaging is vital during all stages of the therapeutic procedures to ensure the safety and efficiency of the treatment (Table 1). 5 The systems used for both sonography and MRI-guided HIFU treatments are made by several manufactures (Figures 1 and 2),9,10 but the systems all have similar elements. Table 2 lists the main components of both ultrasound and MRI-guided HIFU systems, as well as the purpose of the listed components. 4
Stages of High-Intensity Focused Ultrasound (HIFU) Therapeutic Procedures. 4

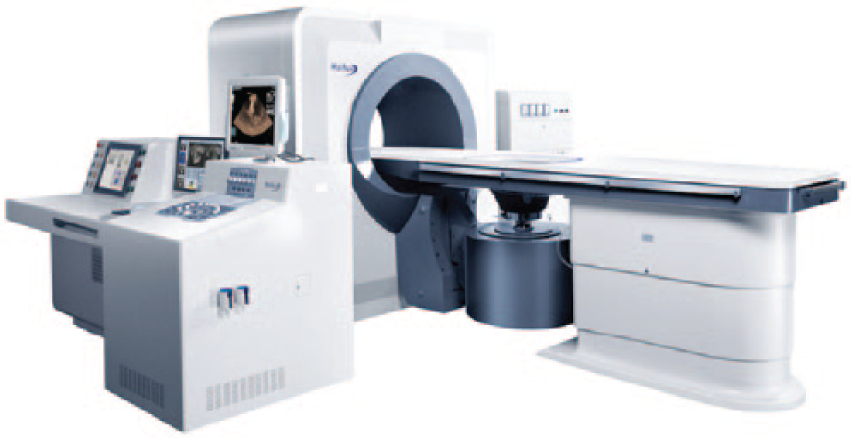
One example of an ultrasound guided high-intensity focused ultrasound system produced by Chongqing Haifu Technology Co., Ltd. (Chongqing, China). 8
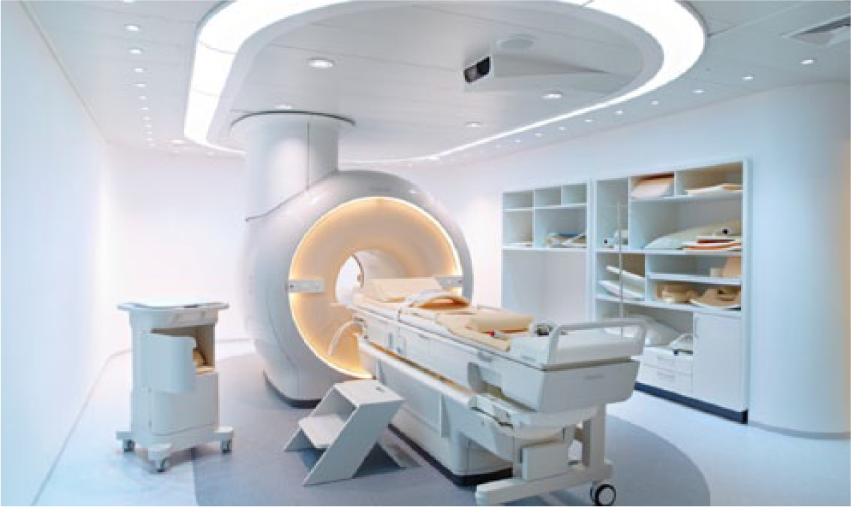
One example of a magnetic resonance–guided high-intensity focused ultrasound system produced by Philips Healthcare. (Philips Healthcare, Bothel, WA). 9
Main Components of a Clinical High-Intensity Focused Ultrasound System. 7
Sonography
Sonographic imaging is used to monitor the HIFU treatments. Real-time sonographic imaging is used to document patient anatomy and procedure information. During the HIFU treatments, feedback is displayed in real-time frame rates by identifying areas of increased echogenicity of treated tissue. 8 Another advantage to using sonography for HIFU guidance is the cost-effective nature of the modality. Planning and monitoring with sonography is an inexpensive imaging option compared with MRI.
One pitfall associated with the use of sonography-guided HIFU is the inability to accurately document temperature changes in the treated tissues. 8 Some success has been seen with the detection of temperatures using several ultrasound parameters. These parameters include the speed of sound, attenuation, or reflection coefficient. 4
MRI
Guidance imaging by MRI allows for preprocedure, target localization and contrast-enhanced postprocedure imaging to verify tissue ablation. 8 Unlike sonographic guidance, MRI has the ability to monitor temperature changes of the tissue. This real-time monitoring of temperature changes in tissue is known as magnetic resonance (MR) thermometry.
A disadvantage for the use of MRI is the costliness of the procedure. MRI equipment is expensive and costly to operate. In addition, a strong magnetic field is used for MR image production. Special equipment is needed to work in the magnetic field; therefore, any equipment used during an MR-guided HIFU procedure must be nonmagnetic.
Analysis of HIFU
Benefits
HIFU is a noninvasive, nonionizing method used to therapeutically treat various medical conditions. Whether sonography or MRI is guiding the HIFU treatment, patients are not exposed to any ionizing radiation.
The recovery time for patients who have had a HIFU treatment is considerably less than that of patients who underwent any traditional surgical treatments. The decreased risk of complications allows for a short recovery time, which, in turn, allows patients to return to work and other normal life activities, sometimes as early as the day after the procedure. 9
Another point to consider is the cost-effectiveness of HIFU treatments compared with traditional medical treatments. Cost comparison for HIFU procedures is unknown at this time. Sonography in general is a more cost-effective means of medical imaging compared with MRI or computed tomography (CT); however, little is known about the cost-effectiveness of HIFU treatments compared with other conventional treatment options. Research is currently being conducted to compare the cost benefits of HIFU procedures and traditional treatments.
Limitations
As with any medical imaging procedure, limitations also exist with HIFU. Some of the limitations associated with HIFU are similar to the ones associated with sonography. Much like sonographic imaging, if lesions are obscured by bowel gas or bone, HIFU treatments are not recommended due to the inability of the therapeutic ultrasound to pass through these barriers effectively. Overlying bone presents a problem by absorbing the ultrasound energy 90 times greater than that of soft tissue, causing a much greater energy needed to ablate tissue lying in the shadow of the overlying bone. 8 In addition, bowel gas poses a problem by reflecting the sound waves back toward the transducer, which in turn has the potential to create tissue damage in the near field. 8
Treatment times are another limitation to the use of HIFU. The actual amount of time the ultrasound energy is applied to a single sonication area is small; however, many sonication areas are required to treat abnormal tissue. Furthermore, normal tissue around the treated area also absorbs some of the energy during each sonication. To avoid damaging normal tissue from the heat absorption during treatments, a break period between each sonication is used to allow the normal tissue to cool. The combination between the times needed for each sonication and break period results in an overall long treatment session. As an example, the time needed to treat a medium-sized (50–100 cm3) fibroid tumor frequently takes several hours to complete treatment. 8 During this time, the patient must remain completely still and the treatment staff members are unavailable for any other procedures.
Accessibility
HIFU is an advanced therapeutic specialty that may not be available in all areas. Procedures are usually performed in larger medical facilities where HIFU treatment suites are located. Travel to a larger city may be required for some patients who are not located near a facility that offers HIFU treatments. In addition, not all treatments are currently performed in the United States. The FDA has only approved the use of HIFU for the treatment of uterine fibroids and bone metastasis. The limited approval from the FDA limits the accessibility of HIFU to patients desiring to use the technology as a treatment option.
The Focused Ultrasound Foundation is an organization committed to the advancement of the focused ultrasound field. The Focused Ultrasound Foundation was created to aid in the growth and adoption of focused ultrasound, as well as to improve the lives of patients with serious medical conditions. 10 Located on the organization’s website, www.fusfoundation.org, resources are available for patients and medical professionals interesting in HIFU information and locating facilities where commercial treatments and clinical trials are conducted.
Current Clinical Status
Although used routinely worldwide, the use of HIFU in the United States is limited. Clinical studies are being conducted on a variety of medical conditions; however, these trial treatments cannot be conducted routinely on patients until the FDA has approved. The only procedures that have been given clearance in the United States are uterine fibroids, prostate tumors, and, most recently, palliative treatment of bone metastases. 8
Clinical Applications for the Female Pelvis
Literature Review
Numerous research studies have been conducted over the years to determine the usefulness of HIFU as an effective, noninvasive, nonionizing treatment option for a number of medical conditions. This literature review focuses on the use of HIFU treatments specific to the female pelvis. These treatments include uterine fibroids, adenomyosis, cervicitis, and polycystic ovaries. PubMed and Academic Onefile were the search engines used to locate articles from peer-reviewed journals.
Uterine fibroids
Uterine fibroids are classified as benign tumors that arise from the smooth muscle cells of the uterine myometrium. 11 Identified as the most common tumors of the female reproductive system, uterine leiomyomata or fibroids occur in at least 20% to 25% of all reproductive-age women.12,13 Symptoms can vary from patient to patient depending on the location of the fibroid tumor. In general, symptoms can include pelvic pain, pressure, menorrhagia, and infertility. Treatments for uterine fibroids range from minimally invasive to surgical. Minimally invasive treatments include hormonal therapy, endoscopic surgery, uterine artery embolization (UAE), and HIFU treatments. 14 Surgical options for patients include hysterectomy or myomectomy.
HIFU treatments create coagulation necrosis within a specific area of uterine fibroids without damaging the overlying tissue. 13 Research studies have been conducted to evaluate the effectiveness of uterine fibroid HIFU treatments. Conducted worldwide, the results of these studies have proven that HIFU treatments of uterine fibroids are safe, feasible, and cost-effective. 15 Recently, more studies have been conducted to evaluate the long-term effectiveness of HIFU treatments on uterine fibroids. One such retrospective study was conducted to evaluate the long-term effects of uterine fibroid HIFU treatments. The study was designed to evaluate symptom improvement and volume change ratio of treated uterine fibroids over a 24-month period. 15 The treated fibroids were divided into three categories based on the signal intensity on pretreatment T2-weighted images: type 1, very low-intensity image comparable to that of skeletal muscle; type 2, an image intensity lower than that of myometrium but higher than that of skeletal muscle; and type 3, an image intensity equal to or higher than that of the myometrium. 15 The mean volume change ratio for type 1 and 2 fibroids decreased from −36.5% six months posttreatment to −39.5% twenty-four months posttreatment (P < .001). 15 Type 3 myomas did not demonstrate a reduction in size at the six-month follow-up (–9.1% ± 44.8%, n = 9); however, there was a change in size at the 12- and 24-month follow-up. 15 The study concluded that MR-guided HIFU is an appropriate treatment option for uterine fibroids due to the reintervention rate being low at <25% for type 1 and 2 fibroid patients and symptom improvement with moderate shrinkage of treated patients (Table 3). 15
Reintervention Rates at the 6-, 12-, and 24-Month Follow-up, by Myoma Type. 14
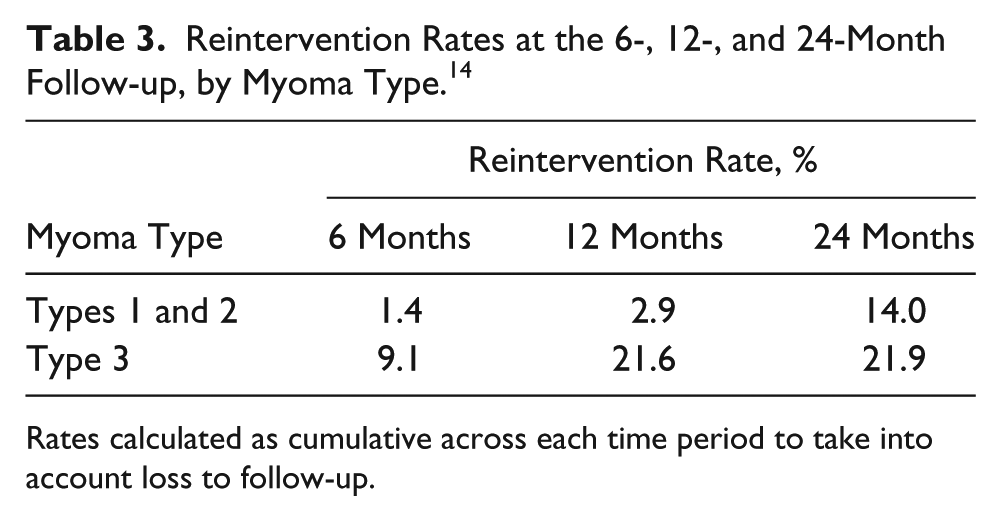
Rates calculated as cumulative across each time period to take into account loss to follow-up.
Adenomyosis
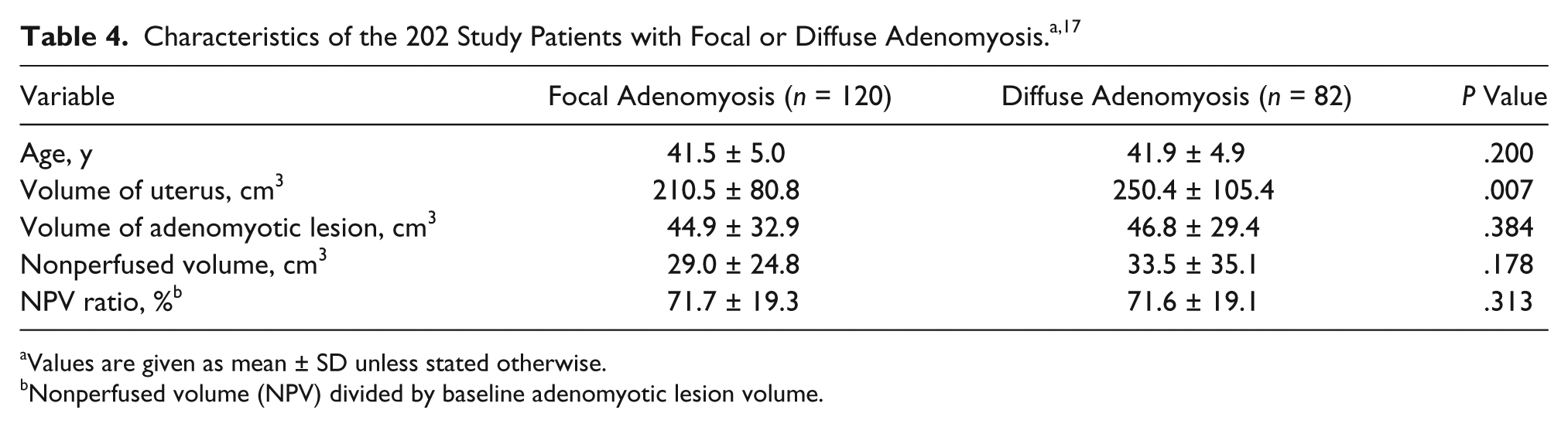
Adenomyosis is common gynecologic pathology characterized as a benign invasion of endometrial tissue into the myometrium of the uterus. 16 Endometrial tissue responds to the monthly changes in female hormones related to the menstrual cycle no matter where the endometrial tissue is located. For this reason, symptoms reported by patients include menorrhagia, dysmenorrhagia with pelvic pressure, and urinary frequency. 16 The rate of incidence for adenomyosis varies between races, resulting in a wide range of occurrence from 1% to 70%. 16 Treatment options for adenomyosis include hysterectomy; however, hysterectomy is only an option for women beyond childbearing years and not opposed to surgery. 16 For patients opposed to a hysterectomy or not considered surgical candidates, other treatment options are available and include myomectomy, gonadotropin-releasing hormone (GnRH) analogues, intrauterine devices (IUDs), and UAE. 16
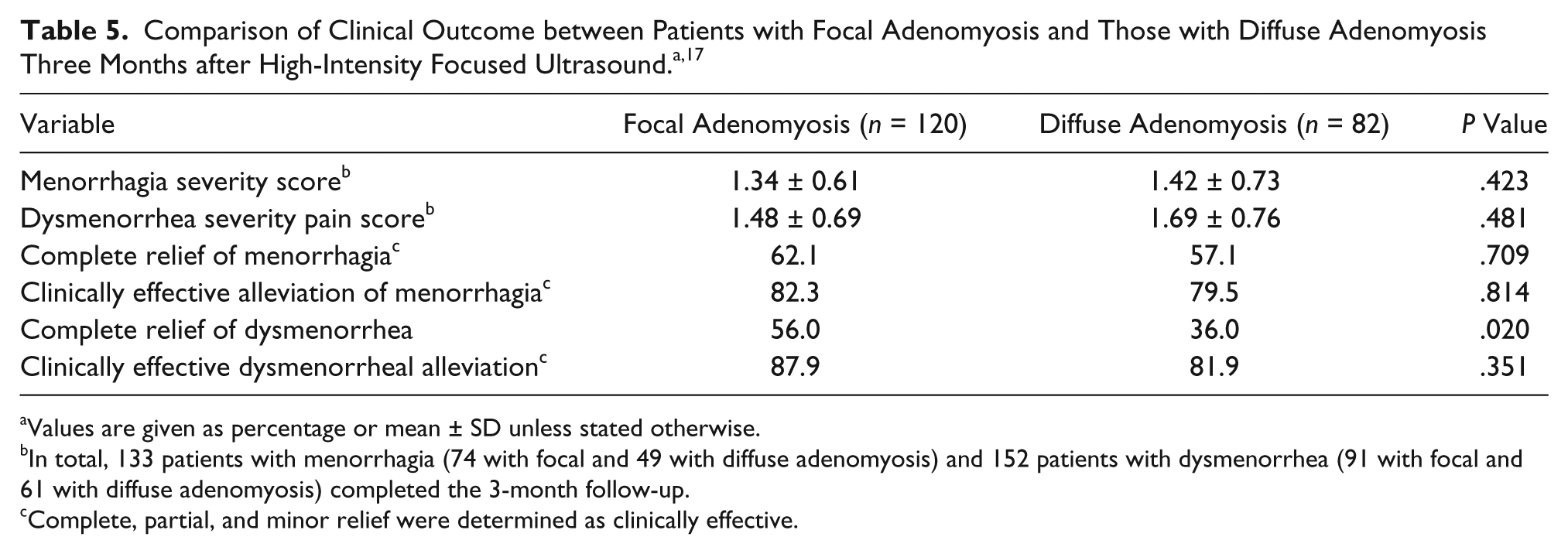
On patients previously diagnosed with adenomyosis, one retrospective study used MRI to image and measure adenomyotic lesions both pre- and postprocedure to obtain initial volumes and nonperfused volumes (NPVs) after the procedure. 17 The NPV measures just the volume of ablated tissue that no longer has blood flow within it. The pre- and postprocedure volumes were compared to determine the successfulness of ablating the adenomyotic tissue. The average postprocedure NPV ratio for focal adenomyosis and diffuse adenomyosis patients was 71.7% ± 19.3% and 71.6% ± 19.1%, respectively (Table 4). 17 Patients also reported a decrease in menorrhagia and dysmenorrheal following HIFU treatment (Table 5). 17
Values are given as mean ± SD unless stated otherwise.
Nonperfused volume (NPV) divided by baseline adenomyotic lesion volume.
Values are given as percentage or mean ± SD unless stated otherwise.
In total, 133 patients with menorrhagia (74 with focal and 49 with diffuse adenomyosis) and 152 patients with dysmenorrhea (91 with focal and 61 with diffuse adenomyosis) completed the 3-month follow-up.
Complete, partial, and minor relief were determined as clinically effective.
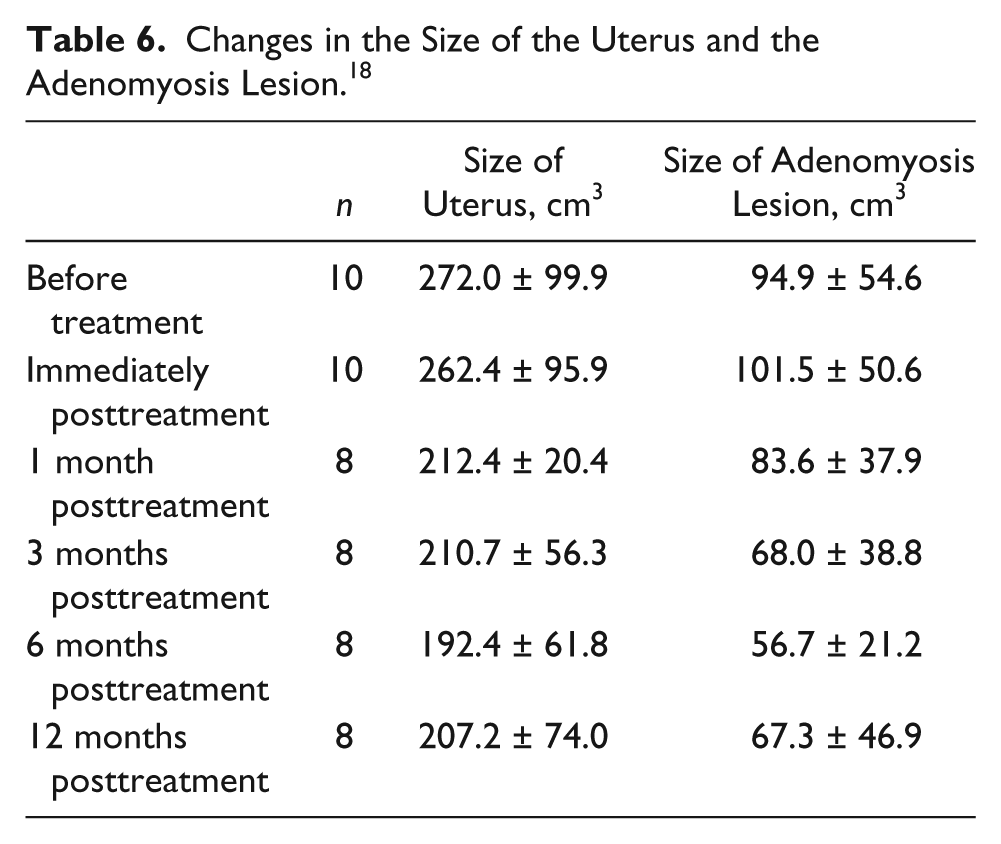
In a similar but smaller study, researchers again found the use of HIFU for ablation of adenomyotic tissue to be successful by measuring the pre- and postablation tissue volumes. 18 Prior to the treatment, the average volumes of the uterus and adenomyotic tissue were 272.0 ± 99.2 cm3 (range, 148.8–440.0 cm3) and 94.9 ± 56.6 cm3 (range, 30.4–208.4 cm3), respectively. 18 During the follow-up periods of the study, the volumes of both the uteri and adenomyotic lesions demonstrated an overall decrease in size. The average NVP was 66.6 ± 49.4 cm3 (range, 27.3–179.1 cm3), and the average percentage of NPV was 62.5% ± 21.7% (range, 35.6%–92.1%) (Table 6). 18 HIFU has demonstrated the potential to decrease the overall size of adenomyotic tissue in the two studies presented; however, research will need to continue to prove the long-term effectiveness and safety.
Changes in the Size of the Uterus and the Adenomyosis Lesion. 18
Cervicitis
Inflammation of the cervix is known as cervicitis and can result from a sexually transmitted infection (STI). Human papillomavirus (HPV) is a very common STI and is estimated to be the most common STI worldwide with an overall prevalence of 26.8% among US females between the ages of 14 and 59 years. 19 The main cause of cervical cancer has been linked to genital HPV infection, making cervical cancer the second most common cancer among women globally. 19 Patients with cervical lesions are generally treated with laser ablation, cryosurgery, and electrocautery.
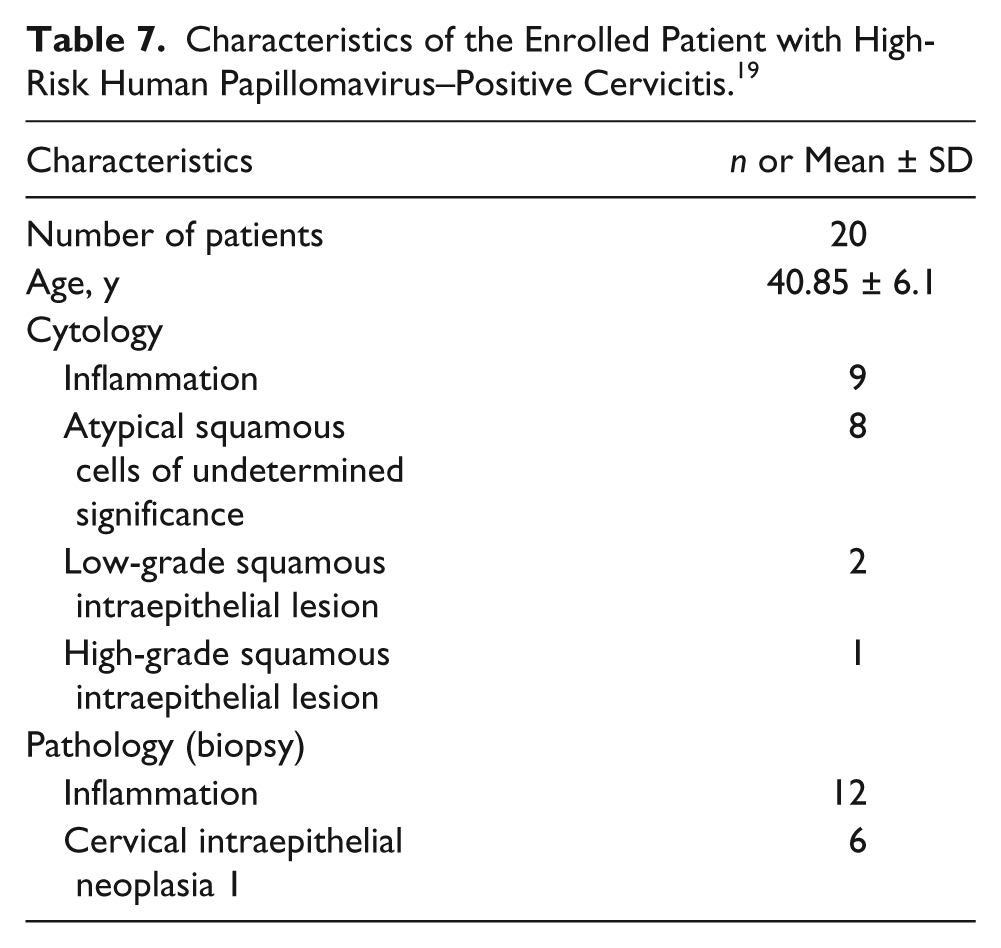
A research study was conducted to determine if HIFU could be used as a possible noninvasive treatment option for patients diagnosed with abnormal cervical cells. Twenty women diagnosed with high-risk HPV infection participated in the study. Characteristics of participating patients can be further examined in Table 7. After the treatments, reevaluation of the participants showed no evidence of cervicitis to be visualized on colposcopic examinations during the 6-month follow-up period in 15 of 20 patients. 19 These results also included 11 patients with known cervicitis and four with confirmed cervical intraepithelial neoplasia (CIN) grade 1 (Table 8). 19
Characteristics of the Enrolled Patient with High-Risk Human Papillomavirus–Positive Cervicitis. 19
Changes in Symptoms and Cytology before and after Ultrasound Therapy in Patients with High-Risk Human Papillomavirus–Positive Cervicitis. 19
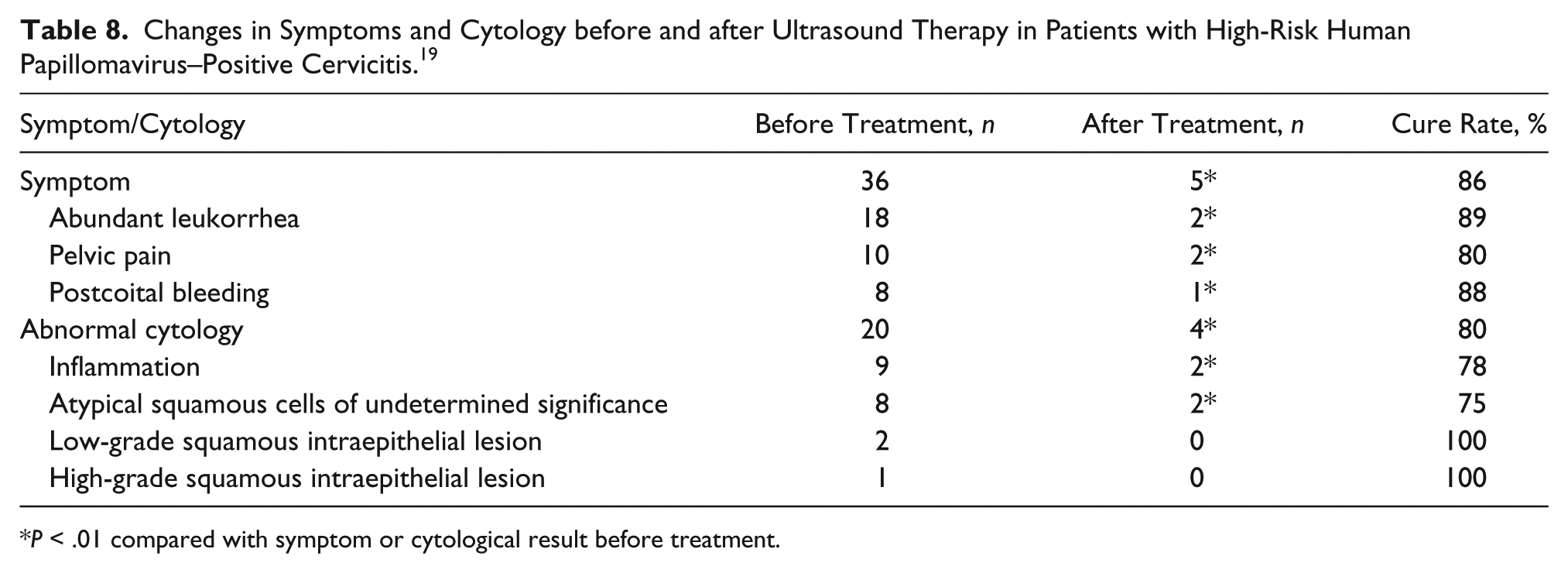
P < .01 compared with symptom or cytological result before treatment.
Polycystic ovaries
Polycystic ovarian syndrome (PCOS) is classified as an endocrine disorder associated with chronic annovulation. 20 Symptoms experienced by patients diagnosed with PCOS include oligomenorrhea, infertility, hirsutism, and obesity. 20 PCOS has been classified as the most common gynecological condition in women of reproductive age and the most common cause of annovulatory infertility, affecting 8% to 10% of women worldwide. 20 Laparoscopic ovarian drilling (LOD) is a surgical treatment option for patients with PCOS struggling with infertility issues. Using electrocautery or a laser, surgeons are able to thermally ablate some of the androgen-producing tissue of the ovary to decrease the hyperandrogenism in patients with PCOS. 21
In a recent pilot research study, Shehata et al. 21 hypothesized that HIFU could be used as a less traumatic alternative treatment to LOD while accomplishing the same therapeutic outcome. Using ex vivo canine and bovine ovaries submerged in molten gelatin, HIFU was applied to the ovaries with imaging guidance from ultrasound. 21 Standard gray-scale imaging was used to identify the ovaries in real time. Researchers used a dual-mode ultrasound array (DMUA) advanced transducers that are able to image and produce HIFU treatment simultaneously. 21 After using different exposure times to adjust the exposure protocols, the HIFU-treated ovaries were histologically examined by an experienced veterinary pathologist. The results of the study demonstrated that it may be possible to create damage inside the ovary using HIFU without causing damage to the surface of the ovary. Again, this study was not conducted on human test subjects. More research needs to be conducted to determine the safety and effectiveness prior to translation for clinical application.
Conclusion
The use of therapeutic ultrasound has grown progressively since it was first used in the 1940s. Since its inception, researchers have conducted studies to determine the usefulness of HIFU in the treatment of various medical conditions. In fact, studies are and have been conducted worldwide for several decades. Through this ongoing research, the use of HIFU in the treatment of uterine fibroids was the first pathology to gain approval for use in the United States from the FDA. Although the FDA has approved HIFU as a noninvasive treatment option for uterine fibroids, more research needs to be conducted before HIFU will be approved as a treatment option for adenomyosis, cervicitis, and polycystic ovaries. Ultimately, the studies presented in this article have shown that HIFU is a promising technique for treating several pathologies unique to the female reproductive system. Only further research will provide the evidence needed to determine whether HIFU could be a standard treatment option for female patients, especially for those patients with pathology related to the reproductive system.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.