Abstract
Atrial fibrillation is the most common cardiac arrhythmia, affecting approximately 1 out of every 250 people. It is associated with thrombus formation in the left atrium, decreased cardiac output, and deterioration of the myocardium. There are multiple treatments available for paroxysmal atrial fibrillation. This literature review will assess the benefits, risks, and complications associated with radiofrequency ablation and cryoablation, which utilize heat and freezing mechanisms, respectively. The review will also assess the role of echocardiography in determining which treatment, if any, is appropriate for atrial fibrillation.
Atrial fibrillation (AF) is the most commonly diagnosed arrhythmia, with a prevalence of approximately 0.5% in the general population. 1 The incidence of AF is projected to double from 2010 to 2030, by an estimated 2.6 million cases, increasing total prevalence to 12.1 million cases in the United States. 2 This conduction abnormality is associated with thromboembolic events and decreased cardiac function and can potentially reduce the quality of life in some patients with symptomatic atrial fibrillation. 3 As the understanding behind the pathophysiology of AF develops and new techniques for treatment are introduced, echocardiography has become an increasingly useful tool for both determining which therapy is most appropriate and ruling out a cardiac thrombus prior to cardioversion or an ablation procedure. Two specific treatments are currently available, radiofrequency and cryoballoon ablation, both of which create transmural lesions to disrupt the arrhythmogenic foci causing AF. 4 Depending on clinical presentation and echocardiographic assessment, one technique may be preferred over the other; therefore, it is crucial for sonographers to become familiar with each method and better understand the role of sonography in evaluating for an ablation procedure.
Atrial Fibrillation
Atrial fibrillation is defined as a supraventricular tachyarrhythmia characterized by irregular activation of the atria. 4 The primary mechanisms in initiating and maintaining atrial fibrillation are local ectopic firing and one or more electrical reentry circuits in the atria or pulmonary veins. 5 The mechanism employed can vary depending on the type of AF, which can generally fall into one of three categories: paroxysmal (self-terminating), persistent, or chronic. 4 Studies have shown that chronic forms of AF are more resistant to remaining in normal sinus rhythm and patients with chronic AF may not be viable candidates for an ablation; therefore, this literature review will primarily focus on paroxysmal atrial fibrillation. 6
Treatment
Currently, two primary mechanisms exist for managing AF: rhythm control and rate control. 4 Rate control is a pharmacological solution, which includes beta blockers and calcium channel blockers, with the ultimate goal of achieving a ventricular response rate of under 80 beats per minute. 5 Rhythm control is the preferred option for patients who are symptomatic in AF and can refer to cardioversion (either electrical or pharmacological), surgical, or catheter ablation. Surgical ablation is known as the Cox-Maze procedure, also called the “cut and sew” method. 7 This surgery works by creating multiple lesions in the atria to isolate the pulmonary veins, forming a “maze” that disrupts the electrical pathways; however, this procedure is highly invasive and complex. 7 The hybrid Maze procedure utilizes both extracardiac surgical ablation as well as intracardiac catheter ablation simultaneously. 7
Catheter ablation is a relatively new technique in managing arrhythmias; this method was first studied and utilized to treat AF in 1998. 8 As it pertains to AF, the purpose of an ablation is to destroy the specific foci that are triggering atrial fibrillation, which can exist in the left atrium (LA), inside the pulmonary veins, or near the coronary ostia. 4 Radiofrequency and cryoballoon are two specific modes of ablation that use different techniques to achieve the same result: destruction of arrhythmogenic foci.
Ablation Techniques
Radiofrequency (RF), which is known as one of the first energy forms utilized for cardiac ablation, cauterizes the target cardiac tissue.9,10 Devices can be unipolar or bipolar, which both utilize alternating currents, anywhere from 100 to 1000 kHz, to create a transmural lesion to disrupt abnormal electrical pathways. 9 However, unipolar RF ablations, which utilize a single electrode, have been shown to be less effective; studies have estimated that only between 7% and 20% of unipolar RF lesions are transmural, increasing the risk of recurrent AF status post ablation. 9 Bipolar RF technology incorporates two electrodes, either between the jaws of a clamp or side by side, to create a therapeutic lesion. 9 This technique is considered quicker and safer than unipolar RF ablation but is not without risks. Major complications have been reported in about 5% to 6% of bipolar RF ablations for AF, including cardiac tamponade, thromboembolism, and perforation leading to an atrial-esophageal fistula. 3
Cryoballoon ablation is performed by pumping nitrous oxide under high pressure into a hollow balloon adjacent to an electrode, where the liquid is converted to a gas; the cryothermal energy produces ice crystals that disrupt the cell membranes and destroy the myocardium.9,11 Unlike RF energy, cryoablation does not affect tissue collagen, making it preferable for arrhythmias that originate near sensitive valvar tissue. 9 However, one side effect of the healing process after cryothermal injury is extensive fibrosis; although this scar tissue has a minimal capacity for conducing AF, currently cryoballoon ablation is reserved for patients who have not undergone an ablation before to prevent the buildup of fibrotic tissue.3,9
Studies comparing these techniques have varied somewhat in their findings. A 2008 study performed on sheep paced into an artificial atrial fibrillation found that while both modalities are equally effective in creating transmural lesions, the RF bipolar clamp completed the procedure faster, was reportedly easier to manipulate than the cryoballoon, and reduced the potential risk of adjacent injury. 12 A larger clinical study, which was performed between 2011 and 2014, compared pulmonary vein isolation via cryoablation versus RF for atrial fibrillation. 13 This study found procedural time for cryoballon ablation to be significantly shorter (by 80 minutes on average), and the long-term success rates were 5% greater than for the RF ablation group; however, the cryoablation group also had a higher occurrence of phrenic nerve palsy post procedure. 13 A third study, performed in 2010, compared the two techniques under varying degrees of sedation and found that less analgesic was required for cryoablation, decreasing the risk of drug overdose. 14 Additional studies have found no significant difference between the two techniques for the treatment of paroxysmal atrial fibrillation.15,16
Transesophageal Echocardiography
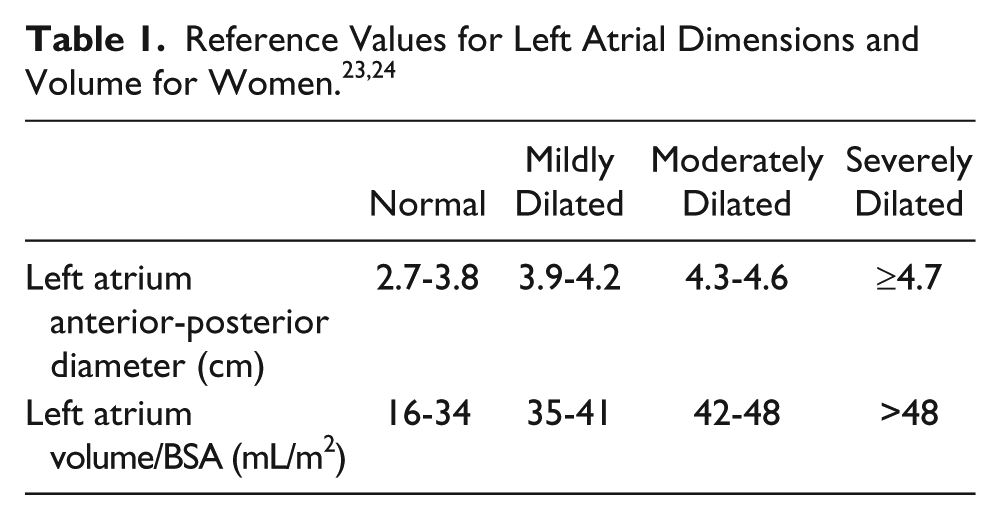
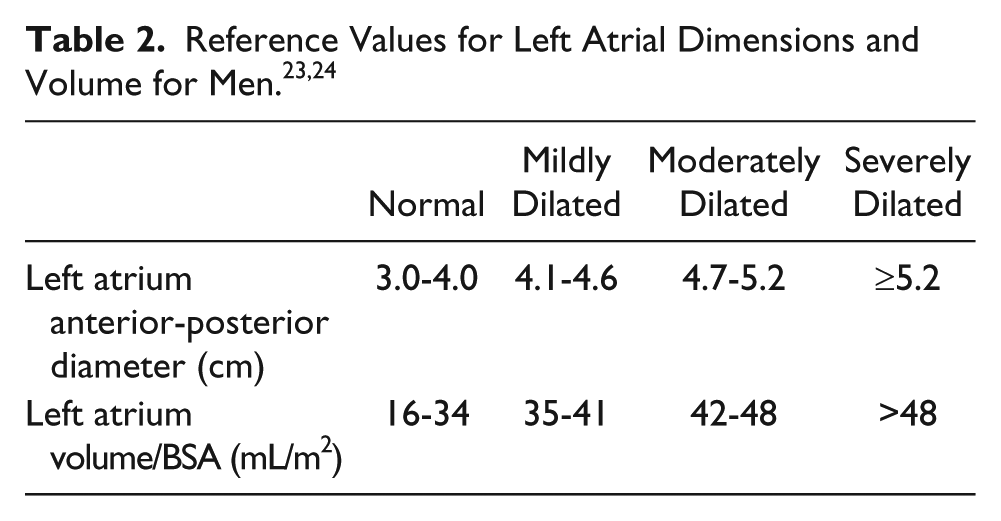
The loss of coordinated atrial contraction that is characteristic of AF results in decreased diastolic filling and blood stasis, which can lead to thrombus formation (particularly in the left atrial appendage; LAA) (Figure 1). 17 Atrial fibrillation is considered the cause of almost half of all thromboembolic events with a cardiac source. 18 Transesophageal echocardiography (TEE) is the gold standard for detecting LA or LAA thrombus prior to radiofrequency or cryoablation for AF treatment (Figure 2). 19 Transesophageal echocardiography provides an excellent sensitivity and specificity for detecting thrombus in the left atrium, of approximately 95% to 100%. 18 Use of a microbubble contrast agent during TEE imaging, such as Definity or Optison, can also improve thrombus detection. A study performed by Jung et al. 20 found that contrast imaging could definitively exclude a LAA thrombus in a greater percentage of patients (83.3% vs. 66.7%). In the setting of an atrial arrhythmia, the American Society of Echocardiography recommends sweeps of the atria and atrial appendages as well as 3D imaging with a high-frequency transducer to rule out thrombus. 21 This procedure is an important step in confirming the absence of clot in the LA and can potentially determine which AF treatment will be utilized in the short term. However, transthoracic echocardiography (TTE) is a less invasive assessment of cardiac function and offers additional information on which treatment may be preferred.
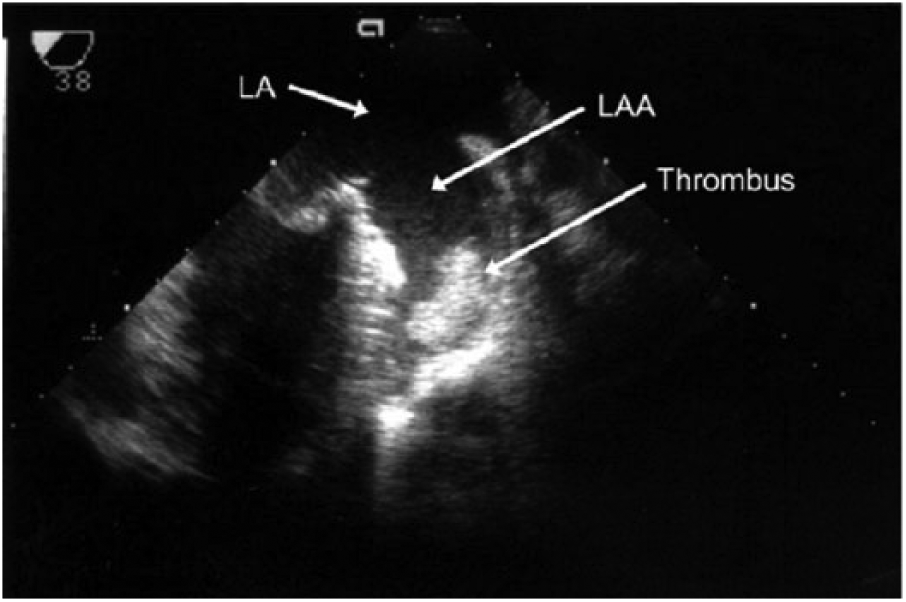
This transesophageal echocardiography image shows a thrombus in the left atrial appendage. 16
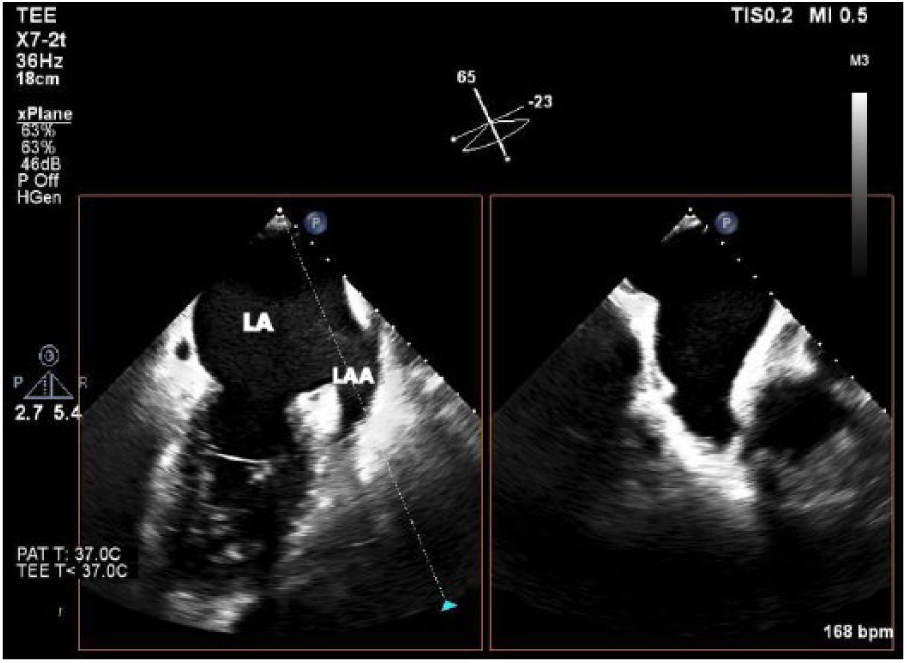
This transesophageal echocardiography image shows an X-plane through the left atrial appendage (LAA). X-plane allows for simultaneous biplane imaging through orthogonal planes.
Role of Transthoracic Echocardiography
Transthoracic echocardiography provides an essential evaluation of left atrial and left ventricular size and function while potentially revealing any underlying heart disease that may be a risk factor for AF, such as rheumatic heart disease, mitral valve disease, or ischemia. 22 Left atrial size is a common predictor for the effectiveness of an ablation; a 5 mm increase in anterior-posterior (AP) dimension for the same patient between exams is associated with a 39% greater risk for developing AF for that patient, even status post ablation (Tables 1, 2). 18 However, AP measurements can easily underestimate an oblong left atrium, and LA volume is currently the recommended standard for a more accurate measurement. 18
Left atrial volume is typically measured using the ellipsoid model, which takes both the area and length of the atrium in an apical two and four chamber and calculates a prolate ellipse (Figures 3, 4). 23 If the left atrium is severely enlarged (greater than 5.2 cm AP for men, 4.7 cm for women or a volume greater than 79 mL for men, 73 mL for women), an ablation may not be recommended; most of the studies performed excluded patients with severely dilated atria (Figure 5). As the LA dilates with recurrent AF, the relative position of the pulmonary veins and the body of the left atrium may shift closer to the esophagus, increasing the risk of creating an atrial-esophageal fistula; this risk is due to the relative relationship of these structures in the thorax (Figure 6). 25 However, if an ablation is possible, a cryoballoon ablation is a safer option as it carries a decreased risk of esophageal perforation compared to RF ablation. 3
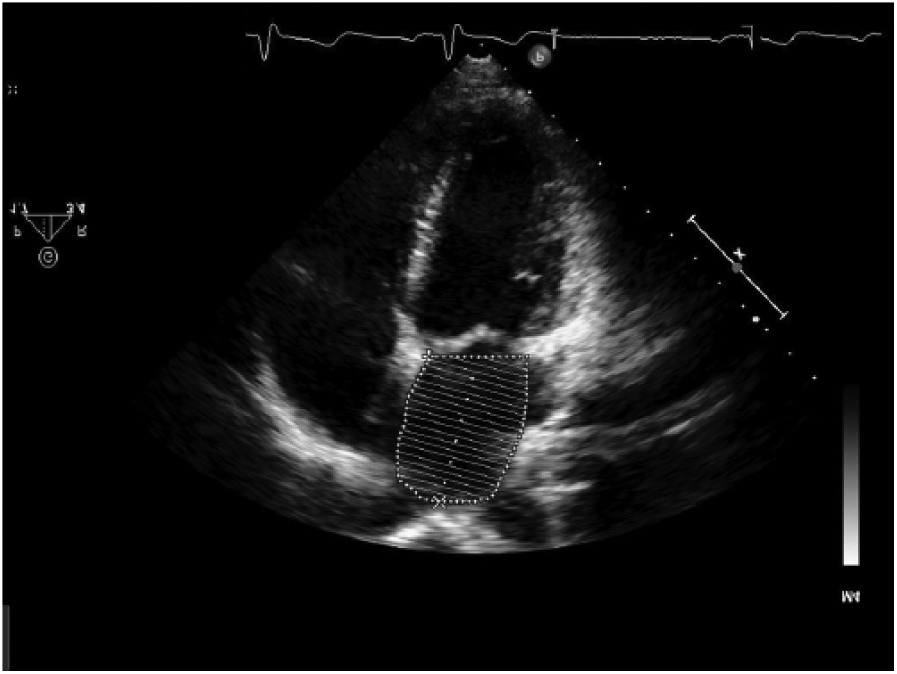
This transthoracic image shows a left atrial volume taken in the apical four chamber view.
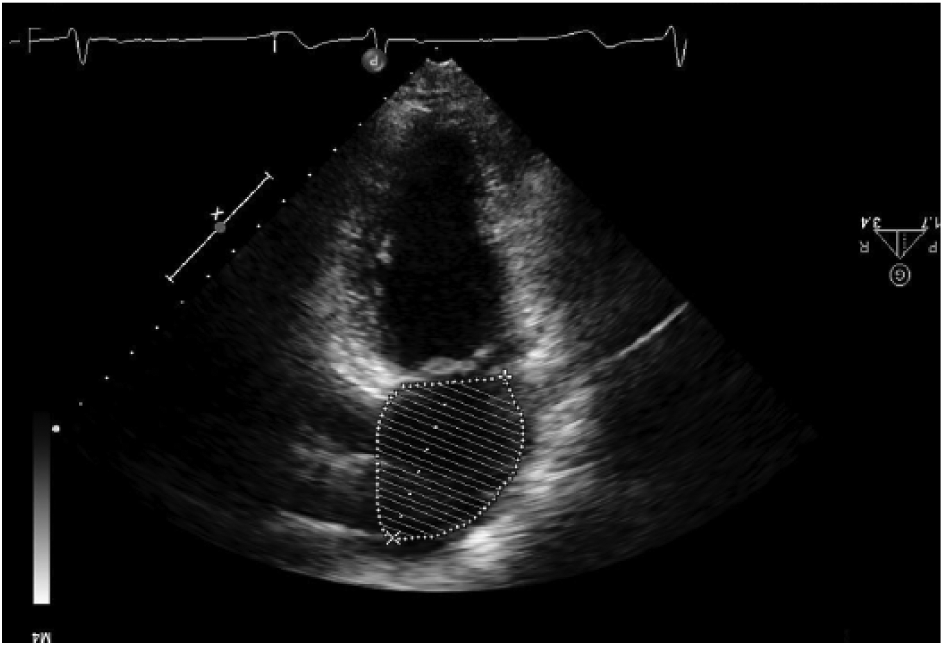
This transthoracic image shows a left atrial volume taken in the apical two chamber view.
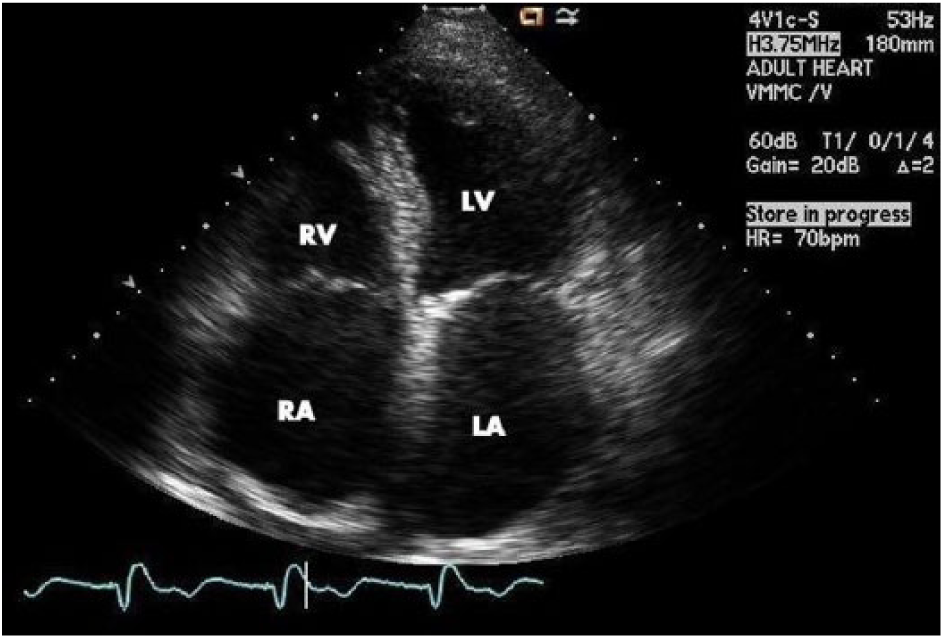
This transthoracic echocardiogram shows a grossly enlarged left atrium (LA) from an apical four chamber view.
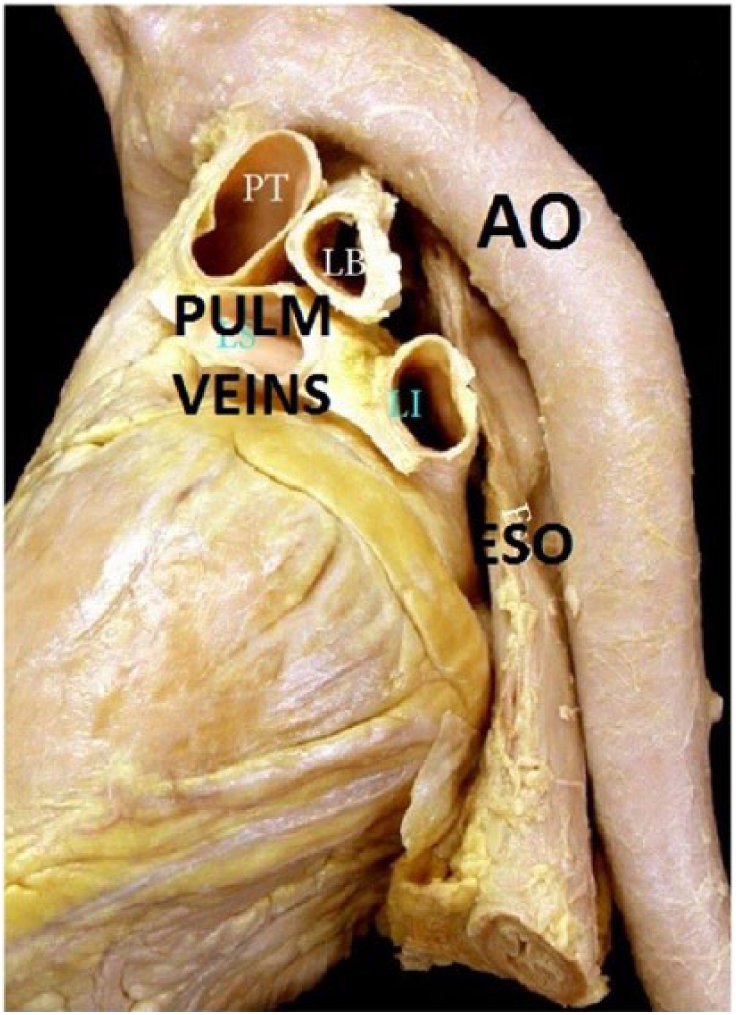
This image shows the relative position of the left atrium to the aorta (Ao), esophagus (Eso), and pulmonary veins. 23
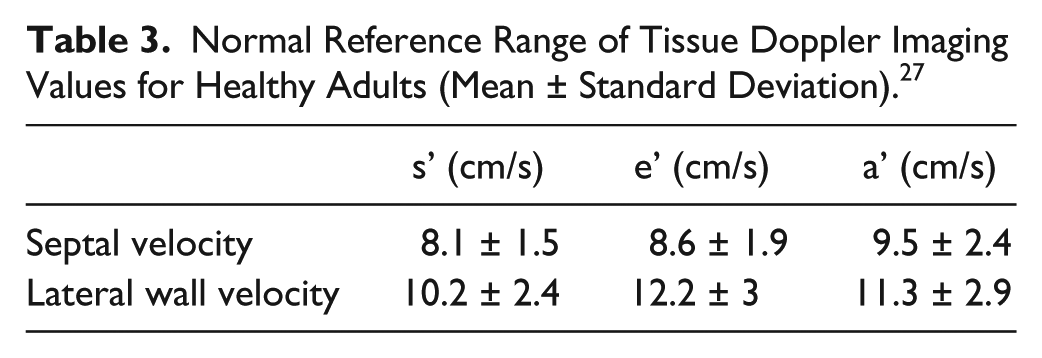
Another consideration prior to ablation is left atrial function, which has been historically difficult to quantify in the setting of AF due to the lack of atrial waveform in mitral inflow patterns. Tissue Doppler imaging (TDI) has emerged as a rhythm-independent method of measuring atrial function. 18 Tissue Doppler imaging quantifies the low-velocity signals created by myocardial tissue movement, which is typically used to assess diastolic left ventricular function in adults (Table 3).26,27 The waveforms associated with TDI represent much of the same information gathered from mitral inflows; the e’ wave shows early diastolic relaxation of the myocardium, the a’ wave represents atrial contraction, and a positive systolic velocity shows myocardial contraction (Figure 7). 26 These parameters can change with age, restrictive physiology, or decrease in ventricular or atrial function (Table 4). A mitral annular a’ wave of less than 4 cm per second is a proven indicator of decreased LA function, which leaves the patient at an increased risk of recurrent AF status post ablation. 28 Given this possibility, radiofrequency ablation is likely a preferred treatment; if a patient has a greater probability of reverting to AF, repeated RF ablations have actually been shown to improve left atrial function over time. 29 Since cryoablation is typically performed only once due to the risk of extensive fibrosis, a patient with decreased left atrial function is not the best candidate for a cryoballoon procedure.
Normal Reference Range of Tissue Doppler Imaging Values for Healthy Adults (Mean ± Standard Deviation). 27
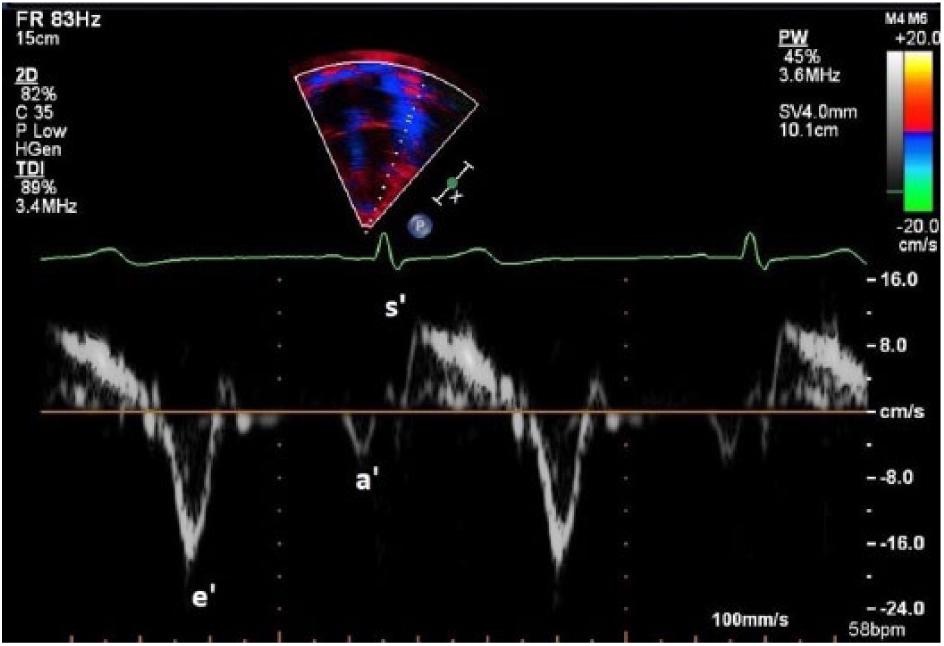
This image of Tissue Doppler imaging from an apical four chamber shows the e’, a’, and s’ waves.
Average s’, e’, and E/e’ Ratio Values (Averaged From Annular and Lateral Tissue Doppler Imaging; Mean ± Standard Deviation) for Different Age Ranges. 30
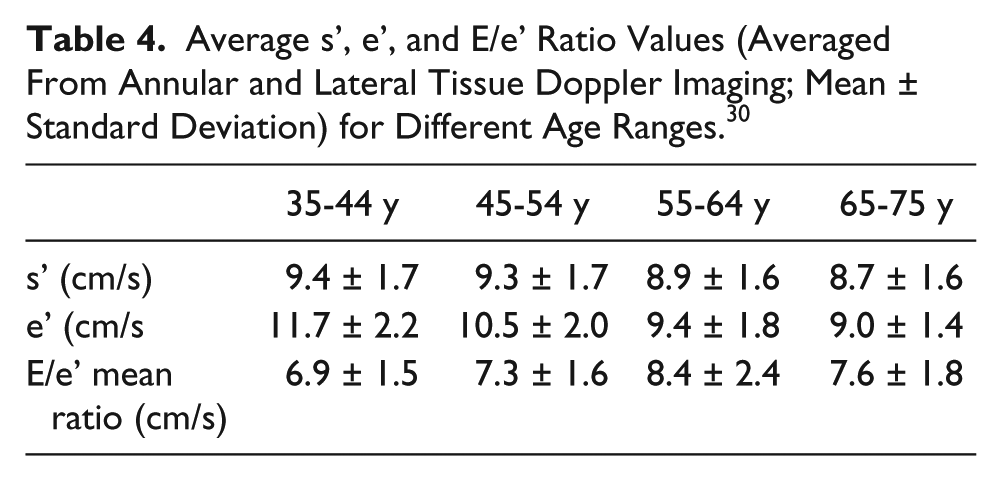
A third echocardiographic assessment for AF ablation is left ventricular diastolic dysfunction, which can be another indicator of AF recurrence in patients undergoing catheter ablation. Many of the same TDI parameters used to quantify left atrial function can give information about diastolic function since left atrial pressure and left ventricular diastolic function are closely related; for example, an E/e’ ratio of greater than 15 cm/s represents abnormally high left ventricular filling pressures. 30 A 2014 study used mitral annular TDI e’ wave velocity and mitral inflow E wave deceleration time as a way to quantify LV diastolic function prior to RF ablation. 31 The study found that rhythm-independent measurements of diastolic dysfunction are associated with an increased risk of recurrent AF after ablation. 31 Although this study is flawed in that it only considers one method of ablation, a patient at high risk for recurrent AF is best treated with RF ablation, as opposed to cryoablation, which can only be performed once.
Conclusion
At present, echocardiography is considered a reliable and effective imaging modality for determining the most appropriate treatment in the clinical setting of atrial fibrillation. If an ablation is necessary in a patient with symptomatic AF, radiofrequency and cryoablation have both been proven to be safe and effective in the management of AF. Small variations in echocardiographic appearance may indicate a preference for one technique over another; however, these changes do not occur in a vacuum. Medical professionals should consider the larger picture of clinical presentation when determining the most suitable treatment for the management of arrhythmias.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.