Abstract
Objective
The coronavirus disease 2019 (COVID-19) pandemic illustrated the relationship between cardiac arrhythmias and pro-inflammatory states. Pro-inflammatory cytokines, including interleukin-6 (IL-6), have significant effects on cardiac conduction. Atrial or ventricular arrhythmias occurring while infected results in a doubling of mortality. Tocilizumab, a monoclonal antibody that blocks the IL-6 receptor, is associated with improved mortality and is believed to be related to immune modulation of the COVID-19–related hyperinflammatory state.
Methods
A single-center retrospective review of all patients with severe COVID-19, defined as admission to an intensive care unit or requirement of respiratory or circulatory support, from March 2020 through March 2022, was conducted. Patients who received or did not receive tocilizumab were grouped into the treatment and control groups, respectively.
Results
Four hundred seventy-three patients were reviewed and 400 met the criteria for inclusion in our study. There were 305 patients (age, 63 ± 13 years, 58% male) in the control group and 95 (age, 57 ± 15 years, 51% male) in the treatment group. In-hospital mortality was greatly reduced with tocilizumab compared with controls (44.2% vs 85.9%, p < 0.001) and new-onset atrial fibrillation (AF) showed a statistically significant reduction (17.8% vs 29.5%, p = 0.019). New-onset wall motion abnormalities, potentially related to myocarditis or acute coronary syndrome, also trended toward significance with tocilizumab (7.7% vs 15.7%, p = 0.10). Deep vein thrombosis, pulmonary embolism, stroke, and sustained ventricular arrhythmias did not meet statistical significance.
Conclusion
As expected, tocilizumab did show significant improvement in mortality. Tocilizumab also showed a significant reduction of new-onset AF. Other cardiac structural endpoints did not reach statistical significance.
A preliminary version of this research was presented during a regional conference at the Mid-Atlantic Capital Cardiology Symposium (MACCS) on 19 November 2023.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 viral infection, has resulted in millions of deaths around the world. 1 COVID-19 has been associated with a hyperinflammatory state, or cytokine storm, which results in predominant respiratory symptoms. It also has implications for other organ systems, most notably the cardiovascular system. Cytokine storm is an exaggerated release of cellular proteins or cytokines. They act as signaling molecules for inflammatory cell recruitment in order to increase the cellular defense response to infections. 1 Examples of cytokines include interleukin-1 (IL-1), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and others. These cytokines have been found to have multiple effects on the cardiovascular system and cellular homeostasis including the propagation of arrhythmias. 1 Cardiac arrhythmias have been reported in approximately 20% of infected patients.1,2 Atrial fibrillation (AF) is the most common arrhythmia observed in patients with COVID-19, and it has been independently associated with increased mortality.3,4
The propensity for increased atrial and ventricular arrhythmias is believed to be secondary to metabolic stressors from the infection, cytokine-induced ion channel changes, abnormal cellular calcium regulation, direct viral myocyte injury and hypoxemia. 4 Additional cardiac manifestations include myocarditis, heart failure, or acute coronary syndrome from plaque rupture. 5
The mechanism of cardiac arrhythmias with acute COVID-19 includes direct cellular damage by cytokines, including IL-6 and interleukin-10 (IL-10), altered cellular ion channel expressivity and tissue remodeling/fibrosis.6,7 Higher levels of both IL-6 and IL-10 were also found to be predictive of new-onset arrhythmias in patients with COVID-19. 8 In this study, both IL-6 and IL-10 levels were associated with the severity of COVID-19 illness and morbidity/mortality. 8 Inflammation likely contributes to both the degree of illness with COVID-19 as well as arrhythmic risk through similar underlying pathophysiology.
Non-targeted immunosuppressants, notably dexamethasone, were found to be associated with reduced mortality but not with a significant difference in major cardiac arrhythmias (defined as AF/atrial flutter, supraventricular tachycardia, ventricular tachycardia [VT], ventricular fibrillation [VF], or atrioventricular block). 9
More targeted immunosuppressants, such as tocilizumab, an IL-6 receptor antagonist, have also been found to reduce mortality in patients with severe COVID-19, but its cardiac benefits have not been fully elucidated. 10 Tocilizumab has been used to successfully treat COVID-19–induced myocarditis with reduced ejection fraction, but no studies on arrhythmic benefits have been conducted.11,12 We aimed to evaluate the cardiovascular effects of patients with severe COVID-19 with respect to atrial arrhythmias, ventricular arrhythmias, atrioventricular block, and structural heart complications.
Methods
This was a single-center retrospective chart-based review of all patients admitted with severe COVID-19 in our institution from March 2020 through March 2022. We conducted our study in accordance with the Helsinki Declaration of 1975, as revised in 2024. This study was exempt from institutional review board (IRB) approval, given its retrospective nature and given that all patient data had been de-identified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13
Severe COVID-19 was defined as a patient with serologically confirmed COVID-19 based on COVID-19 antibody tests, admitted to the intensive care unit (ICU), or requiring respiratory and/or circulatory support with mechanical ventilation or extracorporeal membrane oxygenation as part of the care for COVID-19. Patients were included in the treatment arm if they were treated with at least one dose of tocilizumab 8 mg/kg (maximum 800 mg) for COVID-19. The majority of patients included in this study were admitted to the ICU for mechanical ventilation or high-flow ventilation, with very few patients requiring extracorporeal membrane oxygenation treatment. The exclusion criteria included age under 18 years, admission to the ICU not related to COVID-19, and prior use of tocilizumab treatment before hospitalization. All other patients who met the inclusion criteria but were not treated with tocilizumab for COVID-19 were included in the control group. While we cannot determine the clinical rationale for tocilizumab treatment, the control patients may have been younger, deemed more likely to survive by the clinician or have higher IL-6 values, which prompted treatment with tocilizumab during a time of limited accessibility. A total of 95 patients were enrolled in the treatment arm and 305 patients in the control arm.
The technology team created a data collection tool within a secure intranet site using REDCap, and all the study-related material was stored on a shared drive within the institution's intranet. All members of the study team had access to the data and assisted in data collection and interpretation. The data collected included patient-related demographic characteristics and COVID-19–related data including laboratory tests, imaging, electrocardiography, and echocardiography data.
Statistical analysis was conducted with continuous variables presented as mean ± SD or median (interquartile range) and categorical variables as percentages. T-tests or Mann–Whitney U‐tests and chi‐squared tests were used for between‐groups comparison accordingly. Statistical analyses were performed using R4.2 (https://crain.r-project.org/).
The IRB reviewed and confirmed that access to protected health information (PHI) was minimal. The PHI used in this study involved minimal risk to the participants and in addition, the research could not practically be done without access to and the use of PHI. This study was therefore issued a full waiver of the Health Insurance Portability and Accountability Act approved by the Ethics Committee and granted approval by Carilion Clinic.
According to the IRB (approval number: IRB-21-1483, approval date: 15 December 2021), informed consent was not required from the participants.
Results
Four hundred seventy-three patients were reviewed, and 400 patients met the criteria for inclusion in our study. The principal criteria for exclusion were related to ICU admission for other reasons, with incidental COVID-19 noted. There were 305 patients (age, 63 ± 13 years, 58% male) in the control group and 95 (age, 57 ± 15 years, 51% male) in the treatment group. Notable inter-group differences include an older age, increased frequency of chronic obstructive pulmonary disease and higher frequency of baricitinib treatment in the control group compared with the treatment group (Table 1). The control group also had a higher baseline prevalence of AF, which may be related to their older age and comorbidities. During statistical analysis, both new-onset AF (in patients without a history of AF) and recurrent AF (including all patients with or without a history of AF) were analyzed separately.
Baseline demographic characteristics of patients who received tocilizumab and patients who did not receive tocilizumab.
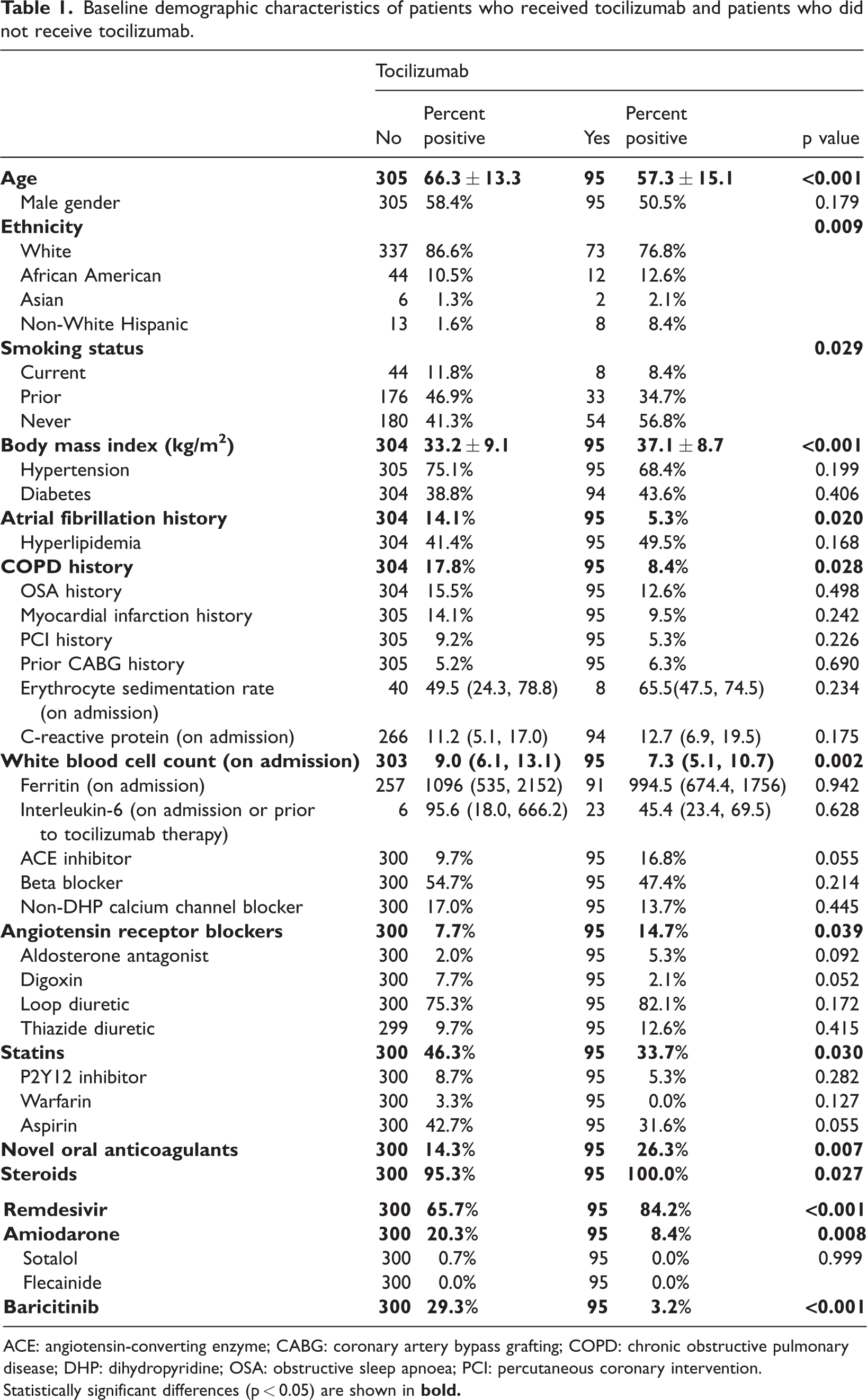
ACE: angiotensin-converting enzyme; CABG: coronary artery bypass grafting; COPD: chronic obstructive pulmonary disease; DHP: dihydropyridine; OSA: obstructive sleep apnoea; PCI: percutaneous coronary intervention.
Statistically significant differences (p < 0.05) are shown in
In-hospital mortality was greatly reduced with tocilizumab compared with controls (44.2% vs 85.9%, p < 0.001) and new-onset AF showed a statistically significant reduction (17.8% vs 29.5%, p = 0.019) with tocilizumab (Table 2, Figure 1).
In-hospital mortality and arrhythmic complications in the control and treatment (tocilizumab) groups.

AF: atrial fibrillation; VF: ventricular fibrillation; VT: ventricular tachycardia.

The cardiovascular effects of tocilizumab therapy in patients with severe COVID-19. Selected arrhythmic and structural and thromboembolic complications are shown. * indicates a statistically significant difference with tocilizumab therapy (p < 0.05). AF: atrial fibrillation; Aflutter: atrial flutter; COVID-19: coronavirus disease 2019; DVT: deep vein thrombosis; PE: pulmonary embolism; VF: ventricular fibrillation; VT: ventricular tachycardia.
Other cardiac arrhythmias, including VT or VF and high-grade atrioventricular block, showed no statistically significant difference between groups (Table 2, Figure 1). New-onset wall motion abnormalities, potentially related to myocarditis or acute coronary syndrome, trended toward significance with tocilizumab (7.7% vs 15.7%, p = 0.10). Other echocardiographic parameters, including ejection fraction <50%, severe valvular disease or structural abnormalities, showed no significant difference (Table 3, Figure 1). Thromboembolic complications can also be increased during COVID-19 due to immobility during mechanical ventilation, thrombogenic state due to inflammation and predisposition to in-situ thrombus formation, particularly in the lungs due to endothelial damage or direct viral injury. Thromboembolic complications, including deep vein thrombosis, pulmonary embolism, stroke, or other peripheral embolization, showed no significant difference between groups (Table 4, Figure 1).
Echocardiographic results for the control and treatment (tocilizumab) groups.
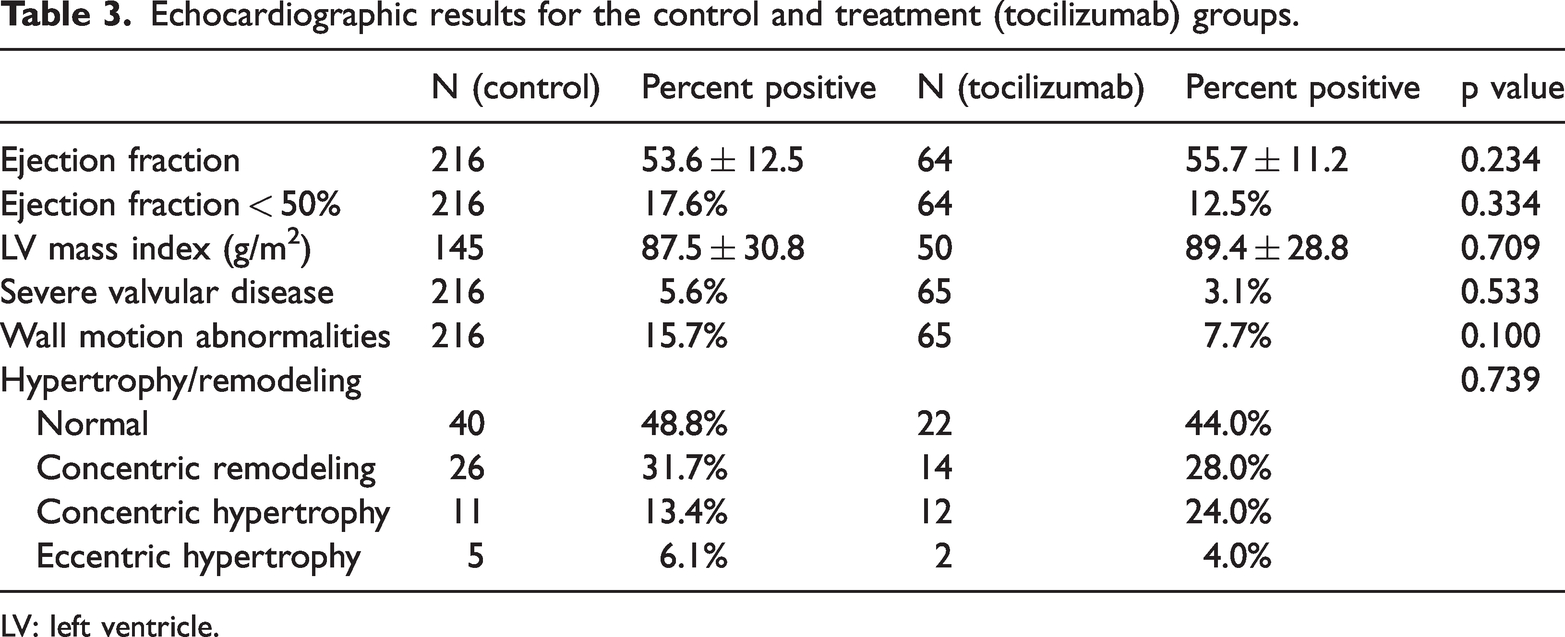
LV: left ventricle.
Thromboembolic complications in the control and treatment (tocilizumab) groups.

DVT: deep vein thrombosis; PE: pulmonary embolism.
Notably, in-hospital mortality was significantly increased in patients with both recurrent AF (95.2%) compared with controls (73.6%, p = 0.002) and with new-onset AF (91.3%) compared with controls (67.2%, p < 0.001, Figure 2). New-onset wall motion abnormalities were also associated with increased mortality (92.3% vs 77.6%, p = 0.034, Figure 2). Other arrhythmias, thromboembolic complications and structural heart disease presence exhibited no statistically significant differences in mortality (Figure 2).

In-hospital mortality for patients treated with the presence or absence of selected cardiovascular variable. * indicates statistically significant difference (p < 0.05). AF: atrial fibrillation; Aflutter: atrial flutter; AV: atrioventricular; DVT: deep vein thrombosis; PE: pulmonary embolism; VF: ventricular fibrillation; VT: ventricular tachycardia.
Discussion
Tocilizumab, a potent IL-6 receptor antagonist, is associated with reduced new-onset AF and mortality in patients with severe COVID-19. New wall motion abnormalities, potentially from myocarditis or acute coronary syndrome, showed a trend toward significance with tocilizumab treatment, but other arrhythmias and structural heart disease showed no significant changes with tocilizumab treatment.
Systemic inflammation as an arrhythmic trigger is an emerging field of study. A review of retrospective studies found that patients with COVID-19 and new-onset AF had increased IL-6 levels and was associated with higher mortality. 14 However, the study also found similar trends in patients hospitalized with influenza infection, suggesting the AF is not related to COVID-19 specifically, but instead to the generalized inflammatory cascade of severe viral illness. 14 There have been some prior studies before the COVID-19 pandemic suggesting a link between inflammation and AF, but the pandemic has certainly highlighted this association and the need for therapeutic options.15,16
Thus far, no therapeutics targeting inflammation have been approved for the treatment of atrial arrhythmias. Our study illustrated that tocilizumab, a targeted IL-6 immunosuppressant, not only reduces mortality but also decreases new-onset AF, which has independently been associated with increased mortality. There was also a trend toward reduced new-onset wall motion abnormalities, which may be indicative of a protective effect against myocarditis or acute coronary syndrome.
Limitations of our study include the single-center retrospective nature of our study, as well as a statistically significant decrease in the age of the tocilizumab group, which may have resulted from selection bias and could have inflated the mortality benefit of the medication. The significantly higher percentage of patients with chronic obstructive lung disease in the control group may also have contributed to worse outcomes in this population or contributed to selection bias, given worse outcomes for patients infected with COVID-19 with chronic lung disease. The small sample size of the tocilizumab group (95 patients) may have led to reduced power to detect statistically significant differences between groups as well. The anti-inflammatory properties of a single dose of tocilizumab have a half-life of around 1–2 weeks, so its ability to impact a more chronic fibro-inflammatory cardiovascular process may be limited.
We cannot exclude the possibility that the statistically significant differences in AF propensity between the groups may also be due to confounding and selection bias. Selection bias for the tocilizumab group may result in the younger age (10 years) given the supply limitations of the medication at the time, and age is a known strong predictor of AF prevalence. In addition, weight was slightly lower in the control group, and statin medications were slightly higher in the control group, which could lead to alterations in AF prevalence as well. The antiarrhythmic medication amiodarone was used more frequently in the control group (potentially to treat AF or other ventricular arrhythmias), and the immunomodulator baricitinib was used more frequently in the control group, which could have blunted the observed effect. These confounders could be further investigated in larger multicenter studies to determine their full effects. Our study illustrates the potential cardioprotective effects (reduction of mortality and new-onset AF) of tocilizumab during acute severe viral infection with the SARS-CoV-2 virus. Tocilizumab is intravenously or subcutaneously administered and is currently indicated for the treatment of several chronic rheumatic diseases when other disease-modifying agents are ineffective. It is, however, associated with an increased risk of serious infection, hepatotoxicity, cytopenias, and allergic reactions. For arrhythmia prevention, an anti-inflammatory medication must have a favorable side effect profile and preferably be available orally. Colchicine has anti-inflammatory properties related to the inhibition of microtubule depolymerization. A recent meta-analysis indicated that colchicine showed positive benefits in reducing both post-cardiac surgery AF and AF recurrence after pulmonary vein isolation. 17 There are several phase 3 clinical trials in development or recruiting that plan to assess the effectiveness of colchicine in maintaining sinus after direct current cardioversion to prevent recurrence (COLFIB ClinicalTrials.gov identifier: NCT05928728 and COLECTRO-AF ClinicalTrials.gov identifier: NCT05890664). Another trial will study the effect of colchicine after transcatheter aortic valve implantation to prevent new-onset AF and atrioventricular conduction disturbances that may require a pacemaker (Co-STAR; ClinicalTrials.gov identifier: NCT04870424).
Understanding the mechanism of AF pathophysiology is critically important for drug and device development. Our current medical therapies for AF primarily target sodium and potassium channels and reduce the excitability of cardiac tissue. These therapies, however, remain inadequate, and current antiarrhythmic drugs are limited by a lack of atrial selectivity with substantial pro-arrhythmic side effects. While the optimal medical therapy to prevent arrhythmia generation remains elusive, there is growing evidence, as supported by this study, that inflammation is a worthy therapeutic target.
Abbreviations
atrial fibrillation coronavirus disease 2019 deep vein thrombosis intensive care unit interleukin-6 pulmonary embolism protected health information ventricular fibrillation ventricular tachycardia
Footnotes
Acknowledgements
We would like to acknowledge the Cardiology Department at Carilion Clinic for support of this manuscript, and we would like to acknowledge the patients who helped contribute to our further understanding and improvement of patient care.
Author contributions
All authors have read and approve of the submission of this manuscript. MSB was involved with data collection and interpretation and manuscript generation. YW performed the statistical analysis. All other authors reviewed and made edits to the manuscript.
Conflict of interest disclosures
The above authors listed have no financial or other disclosures which involve the conduction or publication of this research.
Data availability statement
The data will be made freely available upon request to the corresponding author.
Declaration of conflicting interests
All authors report no conflicts of interest with the generation or publication of this manuscript.
Funding statement
There was no funding provided for the conduction or publication of this research.
