Abstract
Heterotaxy syndrome is a rare, complex disorder involving structural cardiac defects in combination with symmetrical development of abdominal viscera that are normally asymmetrical organs. The vast assortment of cardiac manifestations with accompanying abdominal visceral variations determines the prognosis of heterotaxy syndrome. This case study provides an example of the management of this disease, which is unique to the patient. This is a case report of heterotaxy syndrome diagnosed in utero at 29 weeks and 4 days.
Heterotaxy syndrome (HS) is defined as the abnormal arrangement or lateralization of the thoracic and abdominal viscera. In addition to a wide spectrum of cardiac and noncardiac defects, in most cases of HS, the spleen is affected, either with polysplenia or asplenia. The unusual arrangement of abdominal organs can have an impact on visceral performance, leading to systematic dysfunction. Abdominal manifestations of heterotaxy include liver situated in the midline, inconsistent stomach location, intestinal malrotation, pancreatic anomalies, biliary atresia, urogenital malformations, and ipsilateral position of the aorta and inferior vena cava (IVC). 1 An inconsistency between the location of the fetal stomach and cardiac apex can be the first indication that HS may be present. The etiology of HS is unknown and in most cases occurs sporadically, with an incidence of 1 in 10 000 to 40 000 live births. 2 Studies to determine risk factors have established that preexisting maternal diabetes is the most commonly noted maternal condition associated with HS. 2
Case Report
A woman in her mid-30s was referred to the maternal fetal medicine department at 24 weeks’ gestation. Her reasons for referral included hypoplastic left heart syndrome (HLHS) confirmed with fetal echocardiography prior to maternal fetal medicine consultation, advanced maternal age, and grand multiparity. All prior deliveries resulted in healthy, living children with no history of congenital heart disease. The patient was also noted to have a history of prediabetes and latent tuberculosis. A subsequent scan at 30 weeks’ gestation was again consistent with HLHS. However, during this evaluation, the situs was discovered to be abnormal.
A Medison V20 with a C2-61C transducer was used for two-dimensional, color flow, and pulsed-wave Doppler evaluation of the fetal heart. The fetus was in the cephalic position with the spine to the maternal left at the onset of the examination. In a normal situs arrangement, the left-sided structures would have been visualized on the posterior aspect of the image and the right-sided structures would be visualized on the anterior side. However, the stomach and heart were visualized on the right side on the abdomen and thorax, respectively (Figure 1). The bicaval view was also noted to be abnormal. Sagittal views of the IVC were suggestive of interruption with continuation via the azygous vein (Figure 2). A transverse cross section of the fetal thorax also demonstrated a dilated azygous vein (Figure 3). The four-chamber view showed significant size discordance between the right and left ventricles as well as a moderate-sized ventricular septal defect (Figure 4). Other than showing vessel size discordance between the pulmonary artery and aorta, the three-vessel view was also significant for a persistent left superior vena cava (SVC; Figure 5). The persistent left SVC appeared to terminate at a dilated coronary sinus (Figure 6). The hepatic veins appeared to drain directly into the right atrium as opposed to the intrahepatic portion of the IVC (Figure 7). The combination of complex single ventricular physiology, suspected interrupted IVC with dilated azygous vein, and abnormal situs was concerning for heterotaxy. It is important to note that this particular fetus did not present with any arrhythmia throughout the duration of the pregnancy.
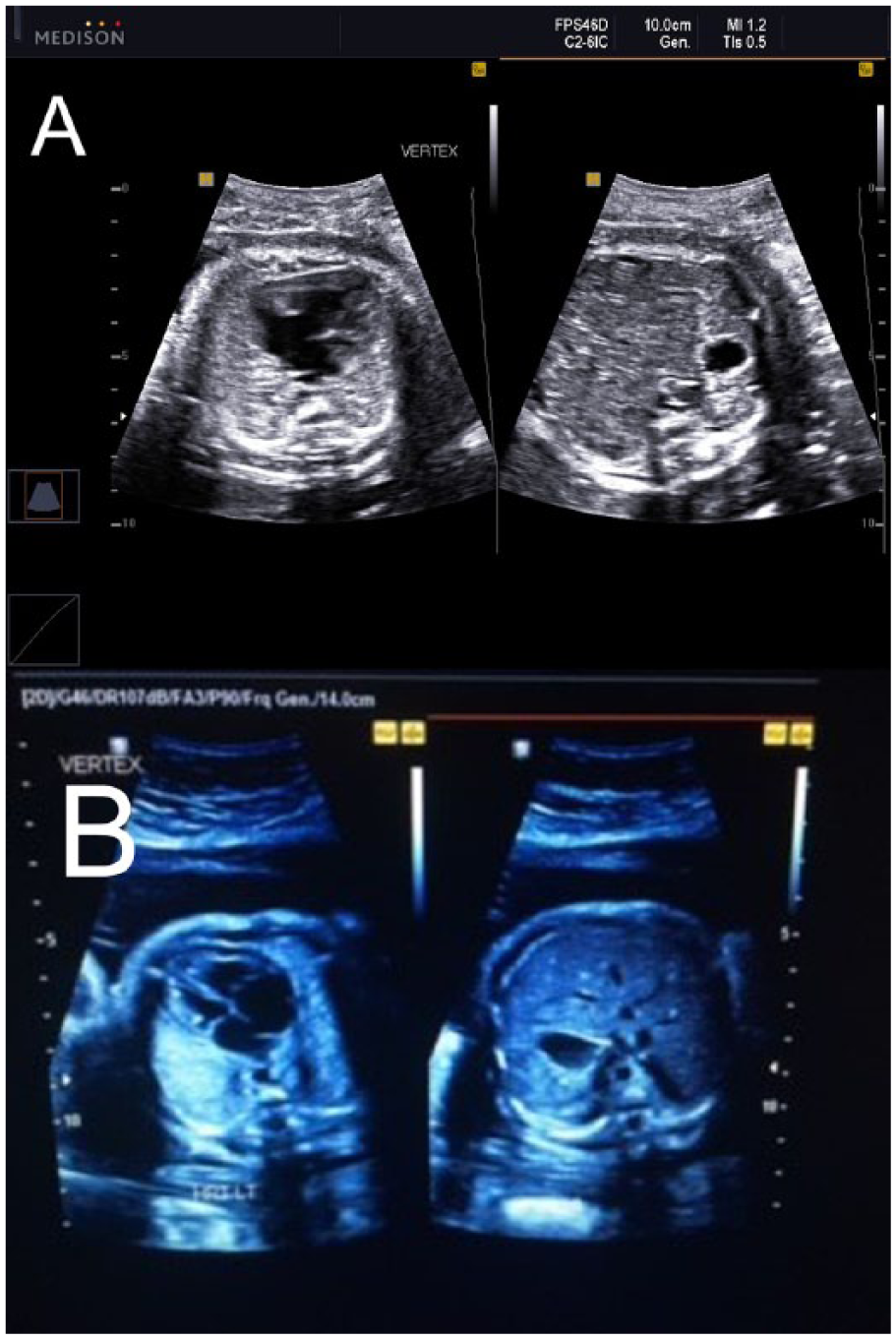
A comparison of normal versus abnormal situs. (A) A split-screen image demonstrating an abnormal situs arrangement. The fetus is positioned with the head in the lower uterine segment with the fetal spine posterior. In this image, both the stomach and heart are visualized on the right side of the fetus (maternal left). (B) This split-screen image demonstrates normal situs arrangement showing left-sided structures positioned to the maternal right.
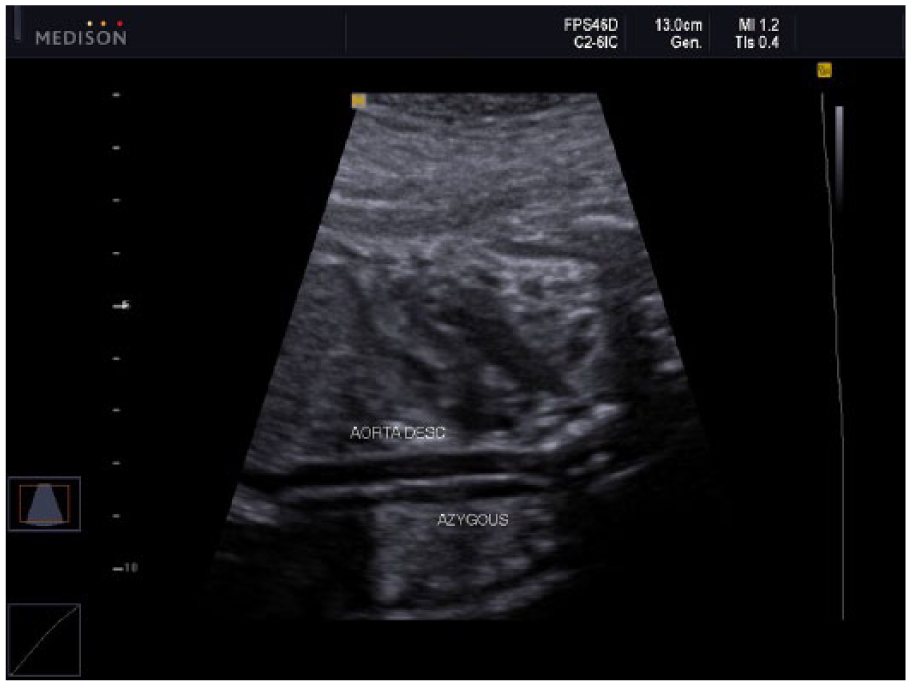
Dilation of the azygous vein is a typical finding with interruption of the inferior vena cava. In this image, the azygous vein is dilated and located posterior to the descending aorta.
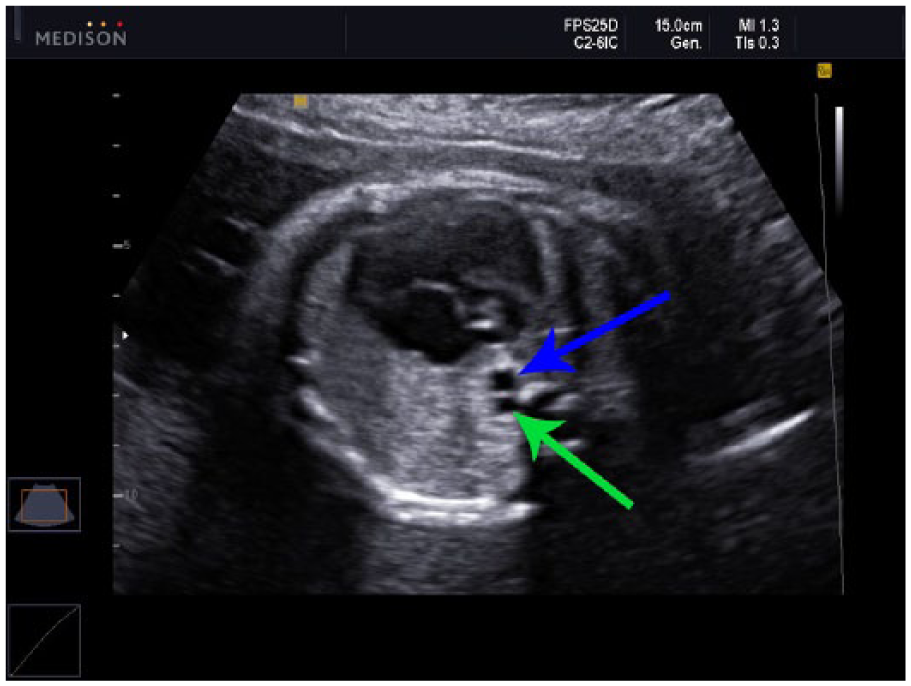
Cross section of dilated azygous vein (green arrow) and descending thoracic aorta (blue arrow) at the level of the four-chamber view.
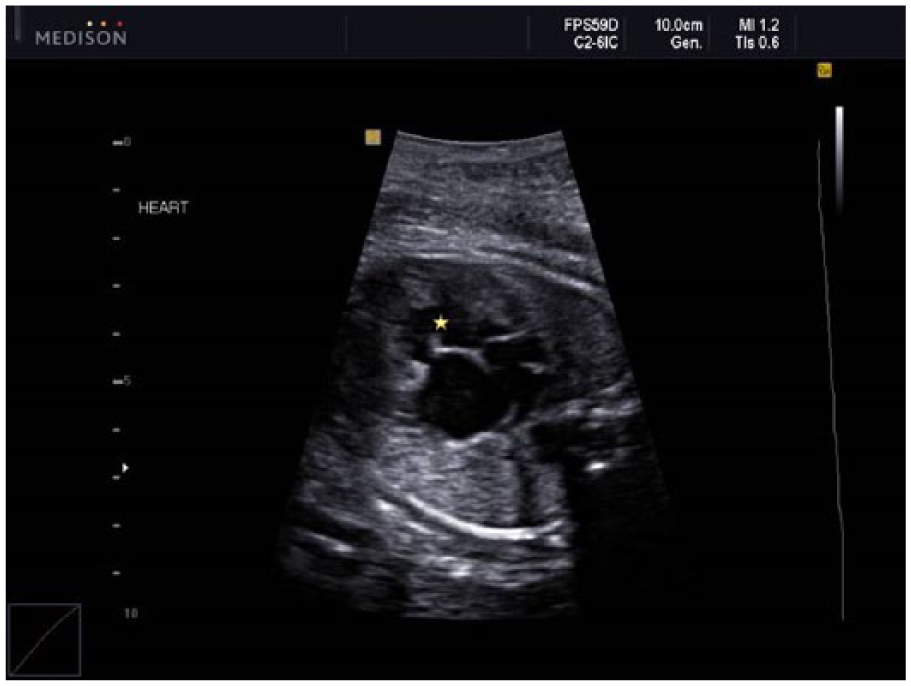
Abnormal four-chamber view showing discordance in ventricular sizes with the left ventricle being smaller than the right ventricle. Moderate-sized ventricular septal defect is noted (yellow star) with discontinuity of the interventricular septum between the cardiac ventricles.
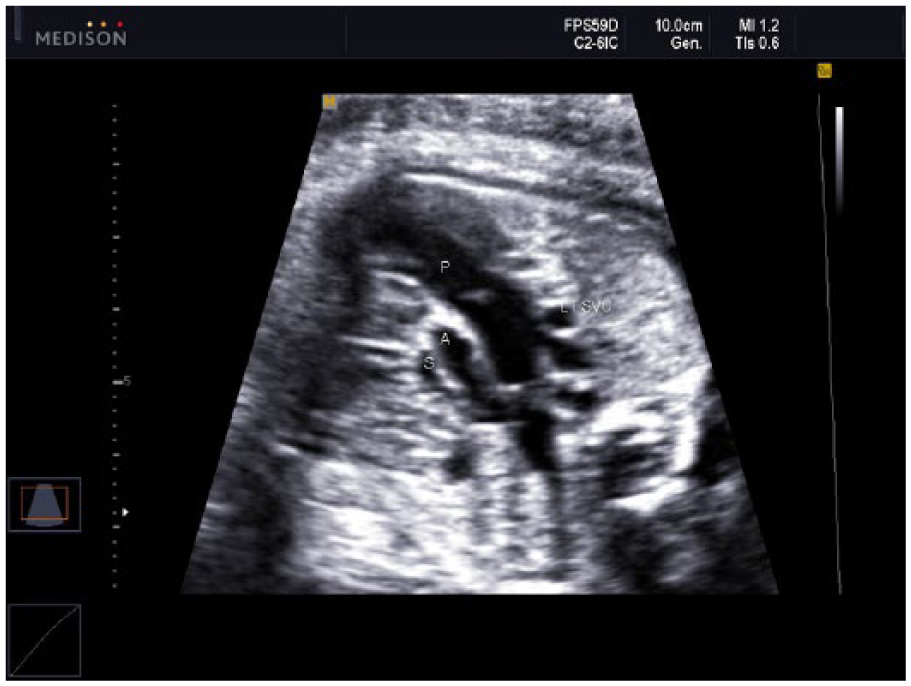
Three-vessel view demonstrating persistent left superior vena cava (LT SVC). The LT SVC is visualized as a cross section to the left of the pulmonary artery. In this view, the discordance between the pulmonary artery (P) and aortic vessel (A) size can also be appreciated. The normal right SVC is also noted on image (S).
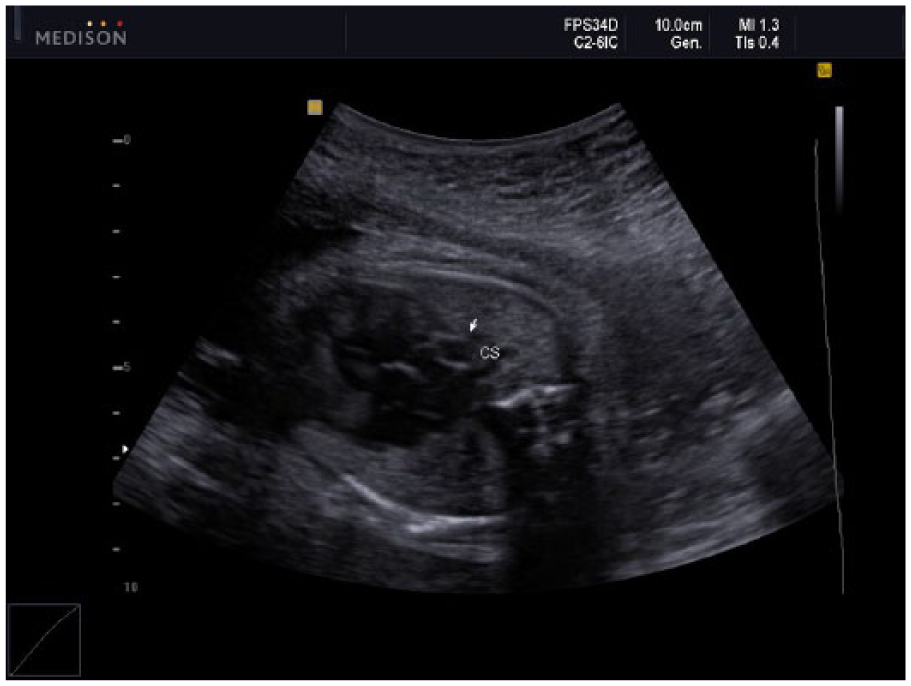
Four-chamber view demonstrating a dilated coronary sinus (CS). A dilated coronary sinus can be visualized in a fetus as a bubble-like appearance in the left atrium and is often indicative of a persistent left superior vena cava.
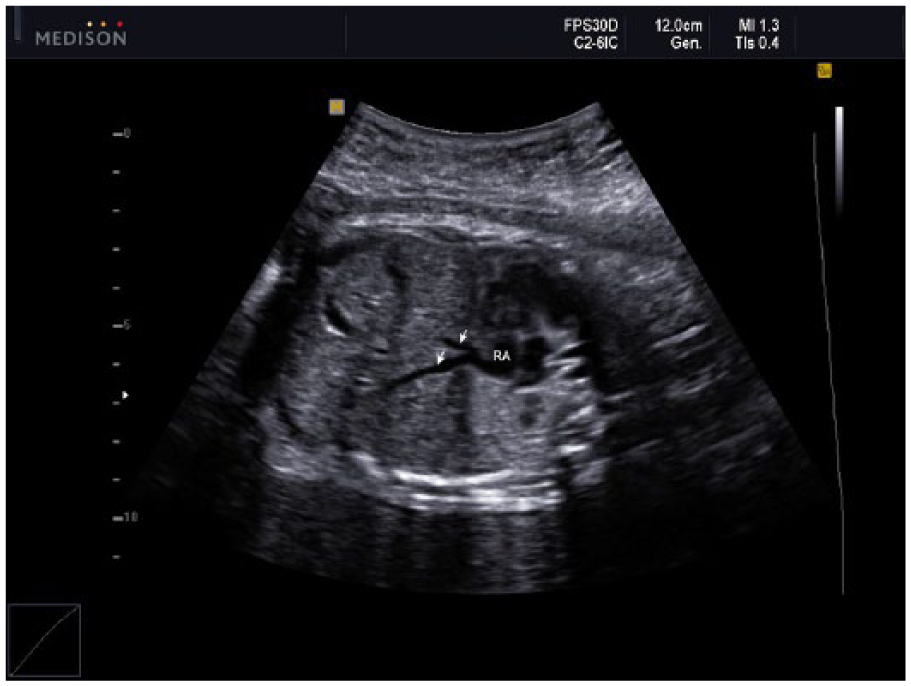
The white arrows in this image demonstrate two hepatic veins terminating directly into the right atrium (RA) due to the interruption of the intrahepatic portion of the inferior vena cava.
At a later point in the gestation, the patient was diagnosed with gestational diabetes mellitus requiring insulin. Subsequent complications of pregnancy also included elevated blood pressures, headaches, and proteinuria. These clinical findings, signs, and symptoms were consistent with mild preeclampsia. The patient was hospitalized at the onset of gestational hypertension for further evaluation and monitoring of preeclampsia. The fetus presented in a persistent breech presentation, creating an additional complication during the third trimester. The patient delivered shortly after successful external cephalic version at 38 weeks’ gestation. APGAR scores (appearance, pulse, grimace, activity, respiration) were eight and nine at 1 and 5 minutes, respectively.
A complete abdominal sonogram was performed on the newborn after delivery. Findings were significant for situs inversus, a dominant spleen with two smaller surrounding nodules (Figure 8), and likely absence of the intrahepatic portion of the IVC. Chest and abdominal radiographs revealed situs inversus, mild cardiomegaly, dextrocardia, and a fractured right clavicle. Cardiac magnetic resonance imaging (MRI) and an echocardiogram were also used to confirm findings from prior studies. Abnormalities from the sum of all diagnostic studies include HS with polysplenia, hypoplastic aortic arch with discrete coarctation, medium-sized membranous ventricular septal defects, large secundum atrial septal defect, interrupted IVC with azygous and hemi-azygous continuation, right and left SVC with left SVC draining into a dilated coronary sinus, mildly hypoplastic left ventricle (Figure 9), aberrant right subclavian artery arising from the pulmonary artery, and lastly, a large patent ductus arteriosus.
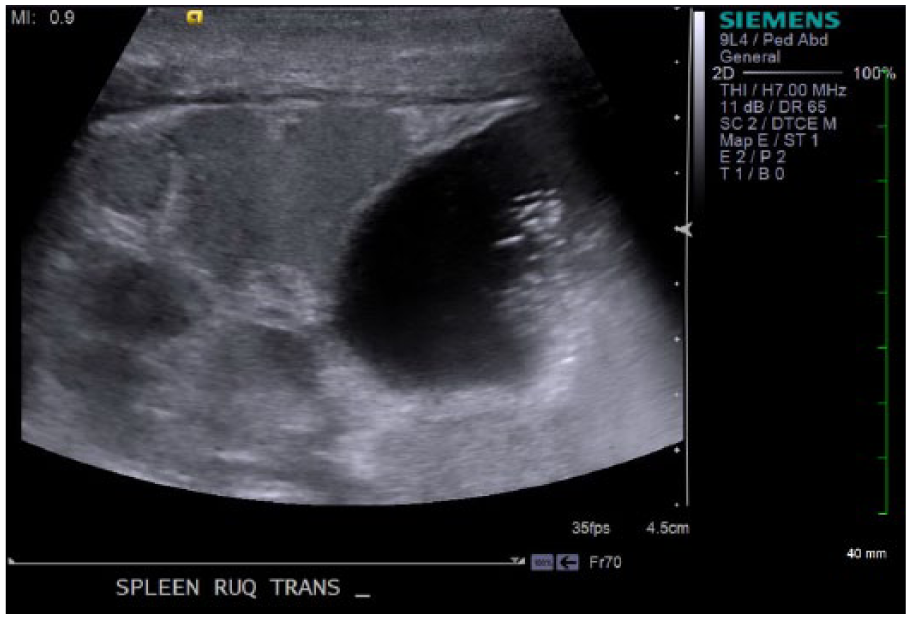
Abdominal imaging of the right upper quadrant revealing splenic nodules consistent with polysplenia and a fluid-filled right-sided stomach.
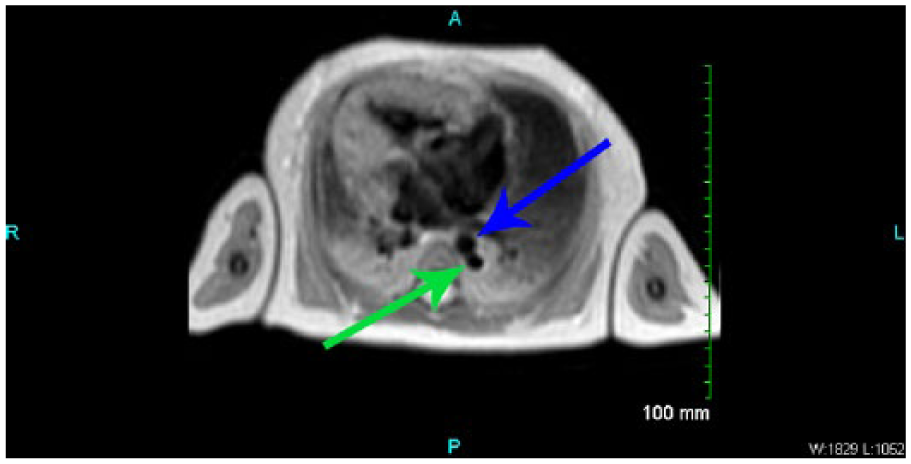
Cardiac magnetic resonance image demonstrating ventricular size discordance and dextrocardia. In the posterior aspect of the image, the dilated azygous vein (green arrow) can also be visualized posterior to the descending thoracic aorta (blue arrow).
This neonate has undergone a series of cardiac surgeries and procedures due to complex congenital heart disease. A modified stage 1 palliation was performed with Damus Kaye Stansel and Norwood procedures, a saphenous vein homograft Sano shunt, fenestrated atrial septal defect closure, and reimplantation of the aberrant right subclavian artery. The second stage of repairs included takedown of the right ventricle–pulmonary artery conduit, anastomosis of the right SVC to the right pulmonary artery (bidrectional Glenn), and a left-sided Blalock-Taussig shunt from the innominate artery to the left pulmonary artery. Finally, a pulmonary artery band was placed at the level where the respective bidirectional Glenn anastomosis and Blalock-Taussig shunt entered the right and left pulmonary arteries. Extracorporeal membrane oxygenation was initiated due to hypoxia postoperatively and was later discontinued once recovery ensued. Ideally, the patient will undergo a biventricular repair as the final staged procedure for the single ventricle physiology.
Some degree of intestinal malrotation is seen in about 70% of HS patients. 3 Distension, feeding intolerance, and vomiting further complicated the patient’s condition. Upper gastrointestinal imaging revealed findings suggestive of a duodenal web, in essence, functioning as a duodenal atresia. The patient underwent surgical correction of the duodenal web via laparotomy. Eventually, a gastrostomy tube was placed because of aspiration and difficulty with oral feeds.
Discussion
The incidence of congenital heart disease (CHD) is commonly reported as 8 in 1000 live births. 2 Abnormalities of situs, which refers to position or site, are among the least common varieties of CHD. Abnormalities of situs are considered a rare occurrence with the mechanisms of embryonic development.
The normal arrangement of viscera is termed situs solitus. In situs solitus, deoxygenated blood from the IVC drains into the venous atrium or the right atrium, located on the right side of the body. Typical right-sided visceral organs include the liver, gallbladder, and a trilobed lung. The left-sided organs in situs solitus include the pulmonary venous atrium or left atrium, spleen, stomach, aorta, and a bilobed lung. 2 A complete mirror image or reversal of organ positions is named situs inversus totalis. The organ orientation for totalis positions the IVC, venous atrium, liver, gallbladder, and trilobed lung on the left side of the body. Right-sided organs are the pulmonary venous atrium, spleen, stomach, aorta, and bilobed lung. The incidence of situs inversus totalis is approximately 0.01% of the population. 2
HS is an unusual, complex congenital condition that affects multiple organ systems, most specifically, the heart and visceral organs. The term heteros means “other” and taxis means “arrangement” or “order”; thus, the straightforward way to describe HS is a different or other arrangement of the thoracic and abdominal organs. HS is present when there is a combination of situs solitus and situs inversus or what clinicians term situs ambiguous. 2 It is important to note that HS does not have one specific finding; instead, there are considerable combinations of organ abnormalities and arrangements, making the study of this syndrome problematic.
Embryologically, cardiosplenic syndromes are thought to result from defects of the midline development field. Complicated malformations may occur with any disruption to the typically rhythmic, organized timeline of the developmental fields, resulting in defect of laterality or symmetry. The occurrence of these disruptions that result in cardiosplenic syndromes is believed to happen during days 28 to 35 of fetal development. Although most cases of heterotaxy are sporadic, familial occurrence has been reported. 4 Heterotaxy has been reported with cases of sibling and consanguinity, suggesting autosomal recessive inheritance occurs with this syndrome.
All cases in which HS is diagnosed involve the inaccurate lateralization of the visceral organs as well as complex heart anomalies with outcomes that range from minimal disease with an encouraging prognosis to congenital defects that can prove to be lethal. HS and resulting organ displacement has caused much confusion as to the proper or useful way to classify identifiable types of organ arrangement. The nomenclature commonly seen in the literature for isolating the subtypes of HS refers to the presence of extra splenic tissue (polysplenia) or absence of the spleen (asplenia). Other authors will incorporate bilateral right sidedness or right isomerism (asplenia) or bilateral left sidedness, also called left isomerism (polysplenia), for ease in recalling which organ systems are most commonly affected.1,5 Typical findings comparing right and left isomerism involving the heart have been well documented, leading some authors to further define the subtypes by isomerism of right atrial appendage (bilateral right sidedness/asplenia) or isomerism of left atrial appendage (bilateral left sidedness/polysplenia). 6 Table 1 summarizes common findings of left and right atrial isomerism.
Summary of Anomalies.
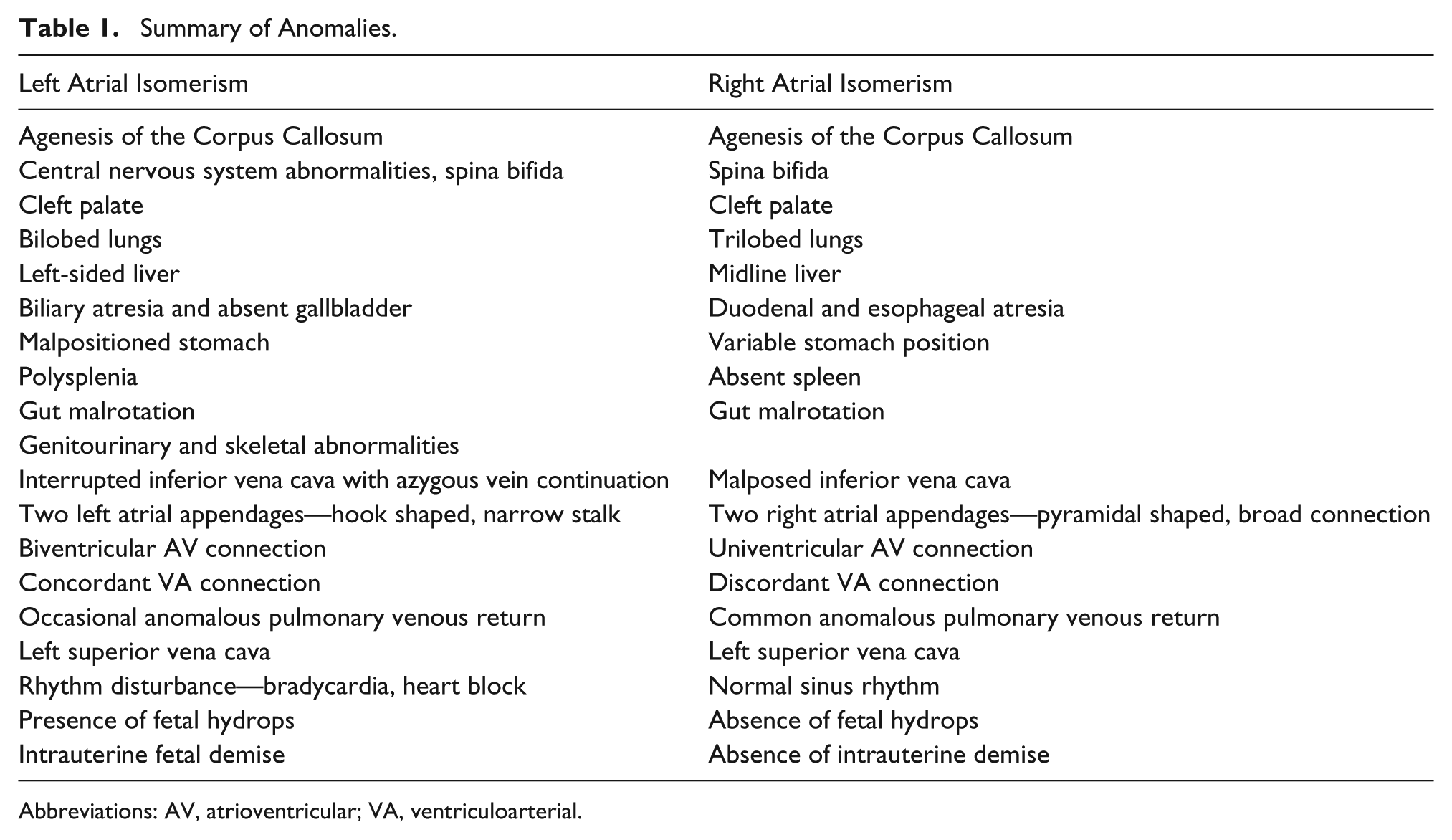
Abbreviations: AV, atrioventricular; VA, ventriculoarterial.
Patients with left isomerism tend to have fewer complex heart anomalies, allowing for a better prognosis, in the absence of heart block.7,8 Congenital heart block associated with structural heart disease and left isomerism is typically noticed during the first trimester, around the time a nuchal translucency sonographic examination is performed. 7 The occurrence of bradycardia can be detected without difficulty during antenatal examinations, either by sonography or auscultation. 11 Heart block detected in the first trimester strongly implies a congenital heart defect, typically a complete atrioventricular septal defect and/or left isomerism. 8 The occurrence of complete heart block has been reported in 50% of fetuses with left isomerism. 4 Fetal dysrhythmia in left isomerism can lead to the progression of nonimmune hydrops fetalis.5,10 Spontaneous fetal demise occurs more often in fetuses with left isomerism, likely credited to heart failure and the hydrops diagnosed in the first trimester.7,12 In contrast, intrauterine demise in the patient with right isomerism occurs with less frequency; however, the overall antenatal outcome for a patient with right isomerism can be much poorer than a fetus with left isomerism. 7
The associated findings with left isomerism include polysplenia, bilateral morphologic left atrial appendages, bilateral bilobed lungs, interruption of the IVC with azygos continuation into the SVC, midline symmetric liver, stomach on either right or left side of the abdomen, and absence of the gallbladder.9,10 Typical findings that accompany right isomerism include asplenia, bilateral morphologic right atrial appendages, bilateral trilobed lungs, bilateral SVCs, midline liver, and right- or left-sided gallbladder and stomach. In addition, most patients with right isomerism will have multiple and complex heart anomalies. Some examples include absent coronary sinus, common atrium, ventricular septal defect, transposition of the great vessels, total anomalous pulmonary venous connection to a systemic vein, or pulmonary artery stenosis/atresia.8,9
A sonographer needs to be familiar with the common sonographic findings in left isomerism, including the presence of multiple splenic structures (polysplenia), morphologic left atrial appendages bilaterally, IVC interruption with azygous continuation into the SVC, symmetric midline liver, stomach and absent gallbladder. Conversely, frequent sonographic findings of right isomerism include absence of the spleen, morphologic right atrial appendages bilaterally, bilateral superior vene cavae, midline liver, and stomach and/or gallbladder on the right or left side of the abdomen. In right isomerism, numerous and complex congenital heart defects are often present, such as atrioventricular canal defects and anomalies of pulmonary venous return. It is important to remember with both right and left isomerism that there is a vast amount of variation from case to case in associated cardiac defects, proving the importance of assessing visceral situs accurately and consistently during each obstetric examination.
The prevalence of major cardiac anomalies associated with HS has been estimated at 1.44 in every 10 000 live births. 12 The diagnosis of HS is morphological and typically not suspected clinically in most cases, as the patient’s exterior body structures show complete symmetry. Thus, the organ arrangement can be explored pre- and postnatally with imaging modalities or in postmortem investigations. 6 Patients with HS present major clinical difficulties to practitioners charged with their care. Therefore, early and accurate imaging by the obstetric sonographer is essential and may have a more favorable outcome for the fetus. During the routine obstetric sonographic examination, the sonographer should establish the position of the fetal stomach in relation to the cardiac apex. Using a transverse view of the fetal thorax and abdomen allows the sonographer to evaluate the fetal heart and its relative position to main visceral landmarks. 13 Any noted discrepancy or an apparent abnormally positioned fetal heart should raise suspicion that HS might be present. 1
Next, the subtype of HS may be established from the associated cardiac defects noted in the fetus. Visualization of the atrial appendages during fetal echocardiography would provide the most accurate diagnosis of HS. However, routine imaging has not been instituted as the appendages are very small, with inconsistency in configuration in HS, and they lie in an imaging plane outside the standard four-chamber view.7,10,14 Therefore, a thorough fetal examination in the suspected heterotaxy patient should include relational positions of the IVC, descending aorta, and azygos system in addition to the cardiac rhythm and visceral organ arrangement. 15 Noted cardiac abnormalities seen with sonography in patients with confirmed HS include double-outlet right ventricle, transposition of the great vessels, ventricular septal defect, and atrial septal defect. 2
According to the literature, the most frequently encountered anomaly associated almost exclusively with left isomerism was interruption of the IVC with azygos continuation to the SVC, which is rarely found under other circumstances.1,8,10,15 The incoherence of the IVC and the azygous vein positioned posterior to the descending aorta can be noted in fetal echocardiography without much difficulty and becomes an excellent indicator of left isomerism. A sonographer may find that using a transverse view at approximately the 10th thoracic vertebral body can assist in distinguishing the positional relationship between the descending aorta and the IVC when determining right versus left isomerism. 5
An important diagnostic feature of right isomerism is the ipsilateral position of the aorta and IVC. The aorta and IVC lie parallel on one side of the spine, with the IVC more anterior, instead of the normal arrangement of aorta to the left of the spine with IVC found on the right side of the spine. With both abdominal vessels positioned on the same side, the IVC enters the right atrium simultaneously with the hepatic veins, therefore not allowing for visualization in the four-chamber view posterior to the heart. 10 Anomalous pulmonary venous connection, more commonly associated with right isomerism, can be difficult to image in a fetus because of the negligible blood flow to the fetal lungs. Upward of two-thirds of fetuses with isomerism of the right atrial appendages will present with anomalous pulmonary venous connection, which occurs only sporadically in cases of isomerism of the left atrial appendages. 1
Extracardiac abnormalities are associated with HS and significantly affect the patient’s long-term outcome and clinical management. The spleen is the most commonly affected organ, either absent in right isomerism or with multiple, small, inadequately functioning lobes of splenic tissue seen with left isomerism. Using high-resolution sonographic imaging equipment, the ability to assess prenatally for splenic absence or presence of multiple splenules is promising. 16 Malrotation of the gut is present in a high percentage of both types of HS, leading to necessary surgery in the neonatal period to correct the deficiency.7,17 In patients with isomerism of right atrial appendage, anal atresia has been documented often, while 1 of 10 infants with isomerism of left atrial appendages has been diagnosed with biliary atresia. 1
The prognosis for a patient with HS varies significantly depending on the cardiac abnormalities and coexisting variable abdominal anatomy. A 50% or higher mortality rate is documented in patients with left isomerism, and for patients with right isomerism, the mortality jumps to 85% or greater. A neonate with suspected HS and comorbidities including complete heart block, pulmonary atresia, or biliary atresia will have the weakest prognosis. Evaluation of all visceral organ abnormalities in the HS neonate is necessary to evaluate for functionality, number, or absence. Establishing the presence or absence of functional splenic tissue is necessary as asplenic HS patients are often predisposed to bacterial infections, necessitating antibiotic therapy. Clinicians should evaluate for possible undiagnosed HS for infants found to have intestinal malrotation or biliary atresia. Multiple imaging modalities, including sonography, radiography, and fluoroscopy of the upper gastrointestinal tract, can be used to evaluate for gut malrotation. When considering treatment for patients with HS, the goal is typically palliative, as surgical repair to achieve normal anatomic arrangement is impractical. Some procedures necessary to treat patients with HS include biventricular repair, Fontan operations, and even cardiac transplant. 2
Conclusion
Heterotaxy is a complex syndrome with a diverse array of multisystem abnormalities, making prenatal diagnosis challenging. However, careful sonographic evaluation of fetal cardiac structures and situs arrangement can alert the sonographer that further detailed investigation is required. Recognizing and understanding common groupings of abnormalities can make prenatal diagnosis of HS possible. HS has been described under several different names, including polysplenia/asplenia, left atrial isomerism/right atrial isomerism, and situs ambiguous. Despite the perplexity in nomenclature, the important point to understand is that there is an unbalanced lateralization of left- and right-sided visceral organs. Left isomerism often presents with congenital heart block that results in hydrops, leading to poor intrauterine outcome. In the absence of congenital heart block, better postnatal outcomes have been reported. In contrast, right isomerism often results in poorer postnatal outcomes despite improved tolerance of intrauterine conditions.
For optimal management during the perinatal and postnatal time periods, both the sonographer and interpreting physician should play vital roles in recognizing abnormalities. A systematic approach to imaging the fetal heart structures and assessing visceral situs during routine obstetric imaging is the first step in the diagnosis of HS. With high-resolution imaging equipment and skilled operator expertise, early fetal cardiac imaging in the first trimester can be helpful in diagnosing left atrial isomerism earlier, as heart block can often be detected during this time. As imaging technology advances and continued medical education of sonographers increases, the fetal diagnosis of HS will continue to improve. Accurate prenatal diagnosis of HS is critical so that parents can make knowledgeable choices regarding pregnancy continuation and preparation for a complicated postnatal phase. However, because of the broad range of cardiac and noncardiac defects that range from minimal disease to severe anatomic and physiologic patterns not compatible with life, HS will continue to be associated with a high mortality rate.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
