Abstract
Dandy-Walker malformation and agenesis of the corpus callosum, although uncommon among all births, are commonly seen in association with one another due to the malformations’ effects on the central nervous system. Obstetric sonographers are likely familiar with the typical appearance of these abnormalities in utero, as sonography is routinely used to detect such congenital anomalies prenatally. However, sonography also plays a valuable role in the postnatal follow-up evaluation of the brain and other body systems affected by Dandy-Walker malformation and its associated abnormalities, thus making it imperative for all sonographers to be familiar with their postnatal sonographic appearance. The case presented demonstrates the use of neonatal sonography to evaluate and follow a case of Dandy-Walker malformation with associated agenesis of the corpus callosum that was originally diagnosed by prenatal sonography. The purpose of this case study is to highlight the pathologies and their coexistence, demonstrate their neonatal sonographic characteristics, exhibit corresponding magnetic resonance imaging appearance, and provide guidance for future patient cases.
Introduction
Dandy-Walker malformation (DWM) describes a continuum of abnormalities of the posterior fossa, which includes partial or complete agenesis of the cerebellar vermis. Dandy-Walker malformation is the most common congenital abnormality of the cerebellum and affects an estimated 1 in 10 000 to 30 000 newborns. 1 It is often accompanied by other congenital anomalies, with associated agenesis of the corpus callosum being the most common corresponding deficiency. Sonography, both antenatal and neonatal, is a widely used method for the detection and follow-up evaluation of these malformations. The present case serves as a relevant example of the characteristic neonatal sonographic findings of DWM with concomitant absent corpus callosum, along with correlating magnetic resonance imaging (MRI) findings.
Case Presentation
A 27-year-old G3P1 female patient presented to a high-risk maternal fetal medicine facility for a follow-up morphology sonogram at 20 weeks 2 days based on last menstrual period, after exhibiting abnormal results at an outside facility. She had a surgical history of cholecystectomy, cesarean section with her first child, and dilation and curettage following a first trimester spontaneous abortion. Sonographic evaluation of the fetal brain demonstrated ventriculomegaly, with the lateral ventricles measuring 1.44 cm. Sonographic findings consistent with DWM were also demonstrated, including splayed cerebellar hemispheres, absent cerebellar vermis, and dilated but within normal limits cisterna magna, which measured 8.3 mm. The fetal profile demonstrated a hypoplastic nasal bone and frontal bossing. A two-vessel cord was visualized. Abnormalities associated with skeletal dysplasia were detected, in which long bones measured 4 weeks behind, and hands and feet were observed to be abnormally positioned. Based on the severity of sonographic findings, an amniocentesis was recommended and performed under sonography guidance. Amnio-centesis results revealed normal karyotyping. A fetal echocardiogram was recommended to further evaluate the heart; however, due to fetal size and position, the examination was limited. Findings suggested potential atrial and ventricular septal defects.
Late in the second trimester, the expectant mother began to develop maternal hemolysis, elevated liver enzymes, and a low platelet count (HELLP). Complications such as hypertension, moderate thrombocytopenia, and proteinuria were reported, which resulted in premature delivery via cesarean section at 27 weeks 5 days, with the infant weighing 1.8 pounds. The neonate patient was assessed at 1, 5, and 10 minutes and Apgar scores were recorded as 2, 4, and 6, respectively. The patient was intubated at 3 minutes. Pulse oximetry levels rose to 98% and the patient exhibited pink color and some flexion by 10 minutes. Shortly after birth, a neurosonogram was ordered to rule out intraventricular hemorrhage (IVH). Sonography was performed using a Philips Epiq ultrasound machine with a C8-5 curvilinear array transducer (Philips Ultrasound, Bothell, WA). Sonographic evaluation of the neonatal brain via the anterior fontanelle revealed widely spaced ventricles that were slit-like anteriorly and dilated occipitally (Figures 1 and 2). The cavum septum pellucidum (CSP) was not visualized coronally or sagittally (Figures 1 and 3). There was no evidence of IVH. Sonographic evaluation via the mastoid fontanelle revealed splayed cerebellar hemispheres, absent cerebellar vermis, and a fourth ventricle that was continuous with a dilated cisterna magna, all consistent with DWM (Figure 4). The neurosonogram findings correlated with the antenatal sonographic findings, and routine follow-up evaluation was recommended. Weekly follow-up sonograms of the neonatal brain were performed, with serial examinations revealing a stable and unchanging brain.
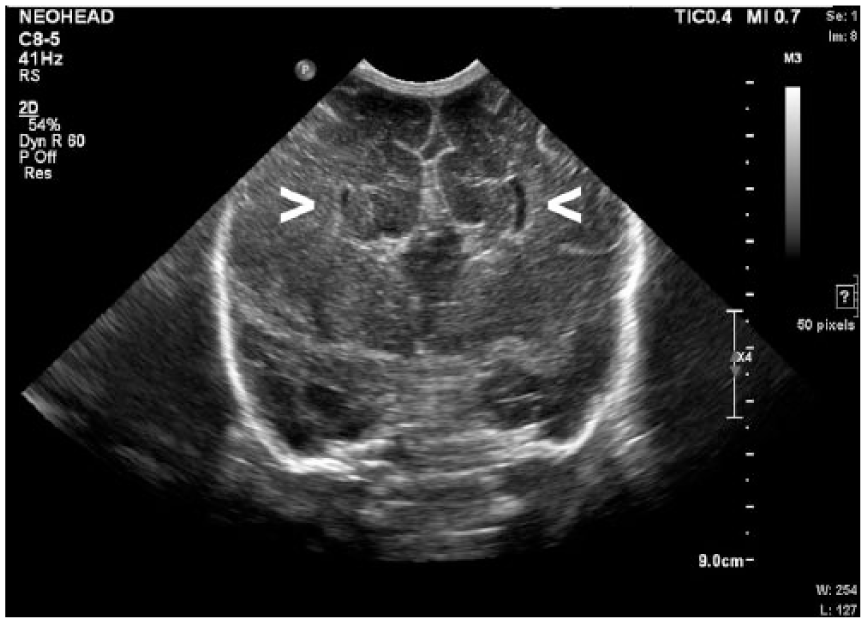
Coronal gray-scale sonogram of the neonatal brain demonstrating the “longhorn sign” with widely spaced slit-like lateral ventricles (arrows) and an elevated third ventricle, consistent with agenesis of the corpus callosum.
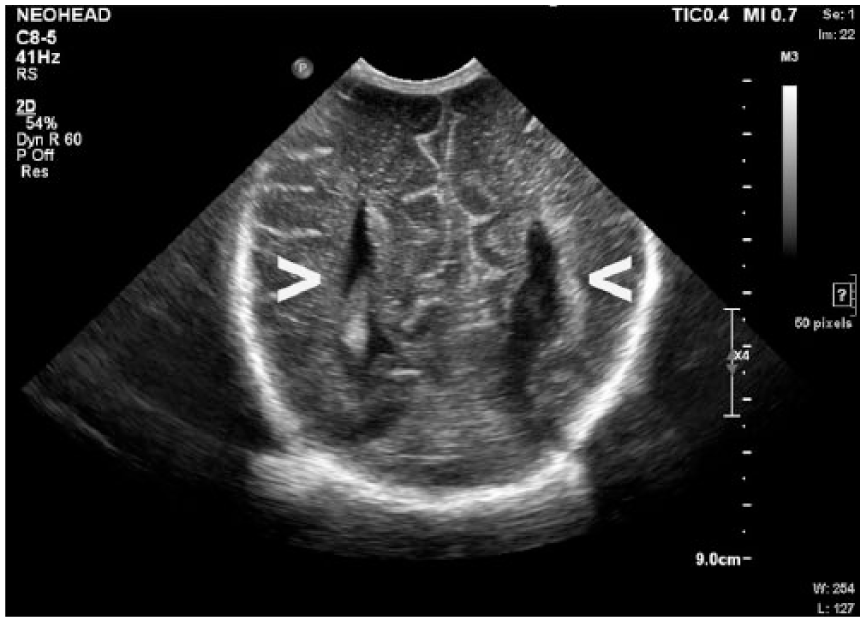
Coronal gray-scale sonogram of the neonatal brain demonstrating parallel configuration of the body of the lateral ventricles (arrows).
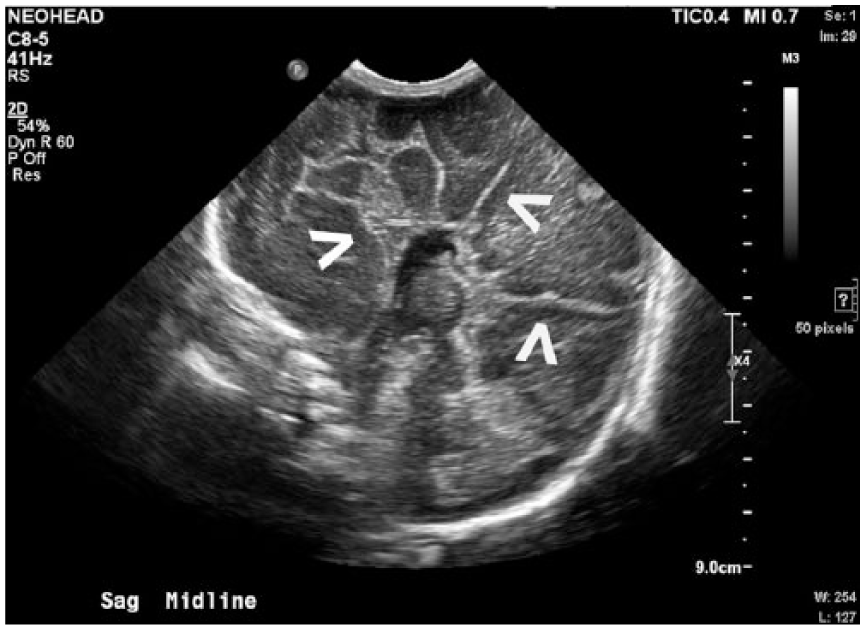
Sagittal midline gray-scale sonogram of the neonatal brain demonstrating the “sunburst sign” with sulci and gyri arranged radially about the third ventricle (arrows), along with absence of the corpus callosum and cavum septum pellucidum.
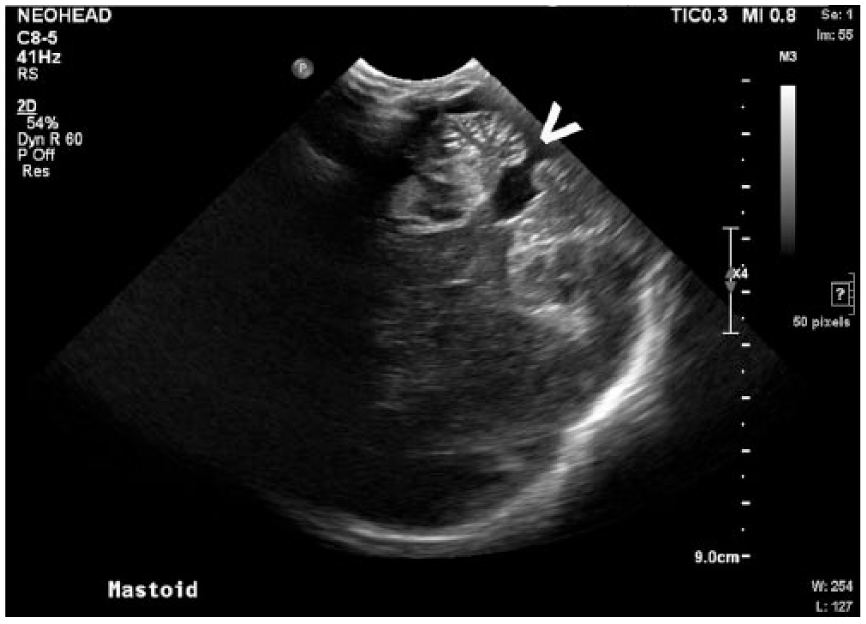
Obliquely modified axial gray-scale sonogram of the neonatal head via the mastoid fontanelle demonstrating characteristics of Dandy-Walker malformation including an absent cerebellar vermis (arrow), splayed cerebellar hemispheres, and prominent fourth ventricle that appears continuous with a dilated cisterna magna.
Based on the fetal echocardiography findings that suggested possible atrial and ventricular septal defects, a neonatal echocardiogram was performed at 1 day old. The echocardiogram was performed on a Philips iE33 ultrasound machine (Philips Ultrasound) and demonstrated evidence of congenital heart defects. An atrial septal defect was visualized due to a moderately patent foramen ovale, which exhibited a left to right atrial level shunt (Figures 5 and 6). A patent ductus arteriosus was also present, as well as a persistent left superior vena cava, which drained into the coronary sinus.
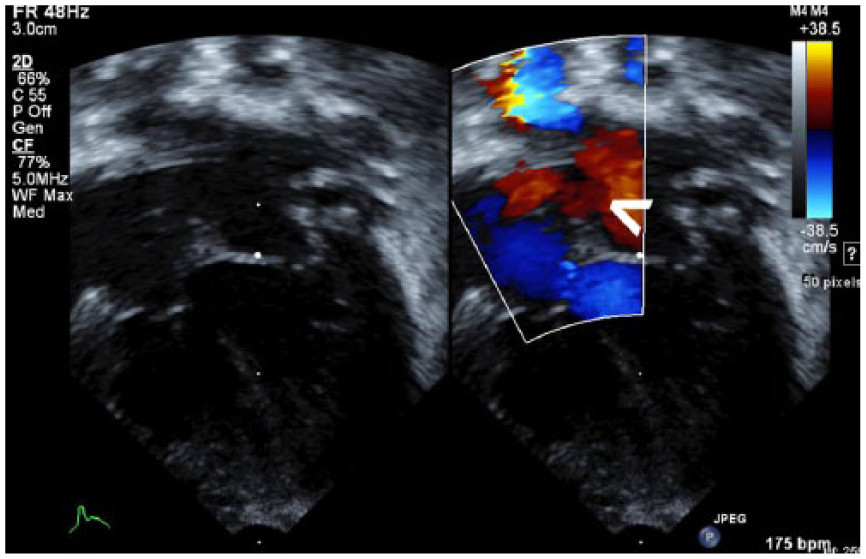
Gray-scale and corresponding color Doppler sonogram of the left ventricular outflow tract demonstrating left to right motion of atrial blood via the patent foramen ovale (arrow).
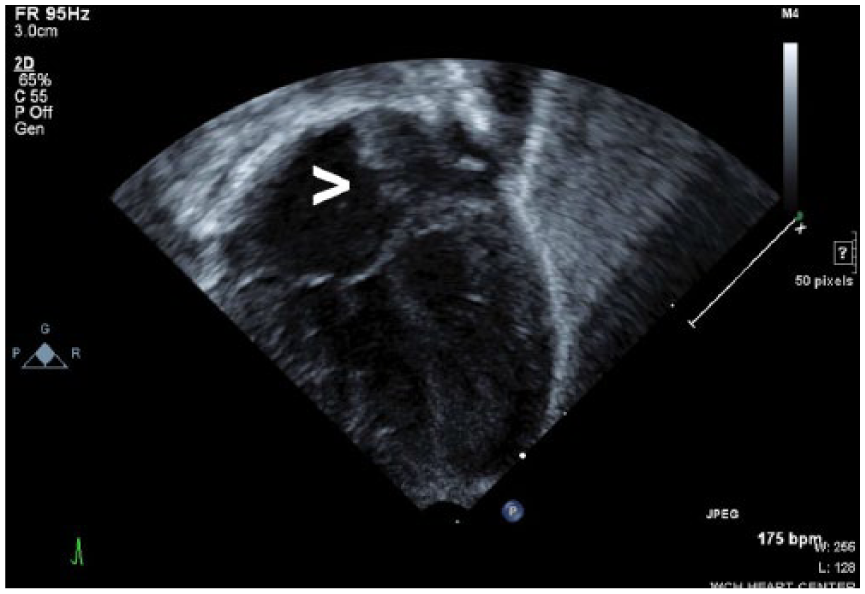
Gray-scale sonogram of the neonatal four-chamber heart, demonstrating an opening in the atrial septum (arrow) consistent with an atrial septal defect.
Neurology, as well as genetic counselors, recommended MRI evaluation once the patient reached an adjusted age of 40 weeks in order to investigate potential white matter disease. A neonatal MRI of the brain was performed and the initial diagnosis of DWM with associated agenesis of the corpus callosum was consistent with MRI findings. Magnetic resonance imaging demonstrated comparable “sunburst” and “longhorn” signs in the midsagittal and coronal views, respectively (Figures 7 and 8). Axial evaluation at the level of the posterior fossa displayed splayed cerebellar hemispheres and a dilated fourth ventricle, which is continuous with the cisterna magna (Figure 9). The MRI report also concluded the presence of polymicrogyria, particularly in the left lateral temporal occipital region. In addition, bilateral closed lip schizencephaly along the occipital horns was noted, although it was difficult to exclude subependymal heterotopia.
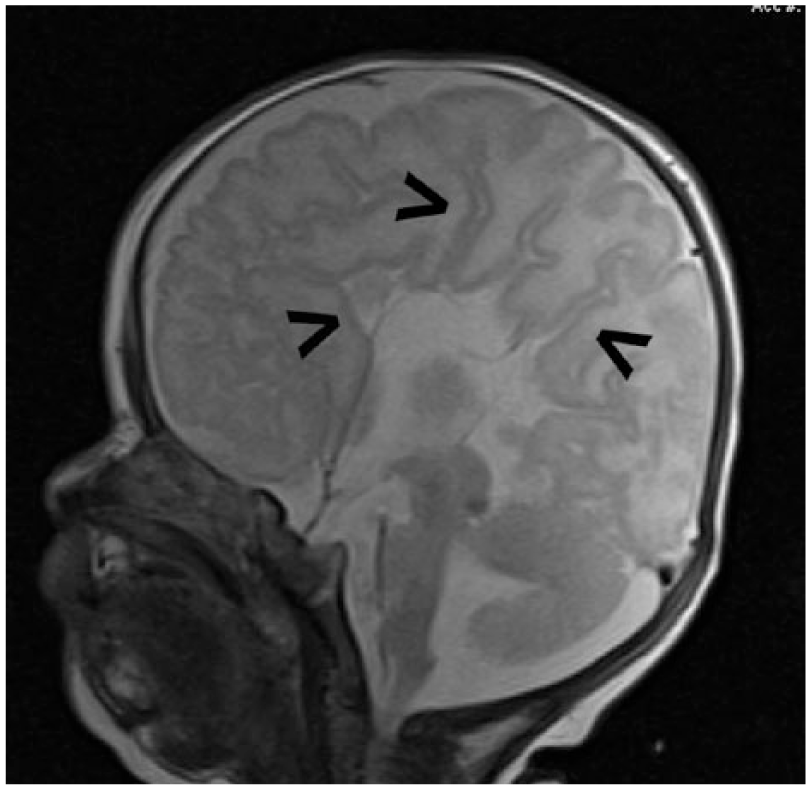
Midsagittal magnetic resonance image of the neonatal brain demonstrating the “sunburst sign” with sulci and gyri arranged radially about the third ventricle (arrows), along with absence of the corpus callosum and cavum septum pellucidum.
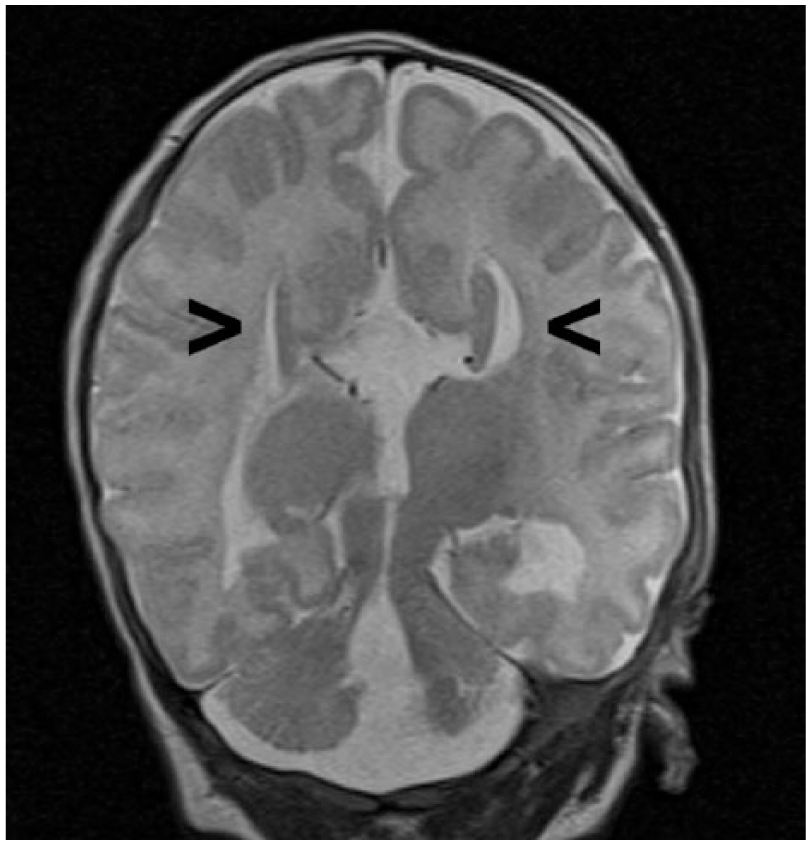
Coronal magnetic resonance image of the neonatal brain demonstrating the “longhorn sign” with widely spaced slit-like lateral ventricles (arrows) and an elevated third ventricle, consistent with agenesis of the corpus callosum.
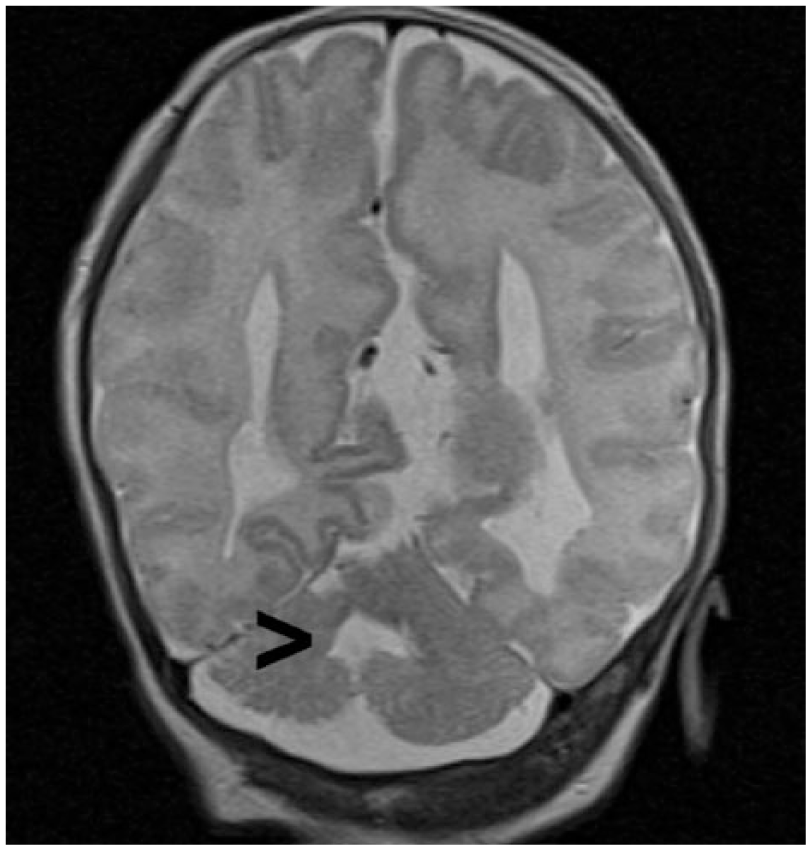
Axial magnetic resonance image of the neonatal brain demonstrating typical Dandy-Walker characteristics of the posterior fossa including splayed cerebellar hemispheres and dilated fourth ventricle (arrow).
Caffeine for neural protection was administered initially during treatment in the neonatal intensive care unit, but no surgical intervention was performed. The patient was discharged at an adjusted age of 43 weeks 1 day, weighing 7.3 pounds. At the time of discharge, the patient was on room air and taking all feedings by mouth. It was advised that her head circumference be monitored weekly, and a cardiac follow-up was recommended after 6 months. Neurologic consult reported that it was extremely likely—a 99% chance—that the patient would have special needs but that the degree of disability was unknown, and follow-up was recommended at 1 year of age. Future therapies were recommended, when necessary, including speech, physical, and occupational therapy.
Discussion
The brain is divided into three main parts: the cerebrum, the brainstem, and the cerebellum. The largest portion, the cerebrum, is divided into two hemispheres, which are connected by the corpus callosum. The corpus callosum consists of white matter nerve tracts that function to facilitate communication between the cerebral hemispheres. The corpus callosum is formed between 8 and 20 weeks gestation, beginning ventrally and extending dorsally, and is located midline, forming the roof of the lateral and third ventricles.2,3 The other two main components of the brain are the brain stem and cerebellum, which are located in the posterior fossa. This fossa also houses the fourth ventricle and the cranial nerves. The cerebellum is the center of the voluntary motor system, as well as balance and coordination, and is located inferior to the occipital lobe. It is separated from the occipital lobe of the cerebrum by a double fold of dura mater, termed the tentorium. The cerebellum consists of two halves joined by a central structure called the vermis. 2 The fourth ventricle is formed by a space between the cerebellar hemispheres and serves to receive cerebrospinal fluid (CSF) from the third ventricle via the aqueduct of Sylvius and drain CSF via the foramina of Luschka and Magendie to the subarachnoid space. Between the medulla and the cerebellum is the cisterna magna, which is a fluid-filled structure that communicates with the fourth ventricle. 2
Between 24 and 27 days gestational age, the primary division of the neural tube occurs and the three primary brain vesicles are formed: the prosencephalon, mesencephalon, and rhombencephalon. The latter divides into the myelencephalon, which forms the medulla oblongata, and the metencephalon, leading to the pons and cerebellum. The cerebellum is formed from the fusion of embryonic structures called rhombic lips. The rhombic lips join at midline, creating the vermis in the ninth gestational week, and 30 to 60 days later lead to the cerebellar hemispheres via neuroblastic proliferation. Organogenesis of the vermis and the cerebellum is complete by the end of the fifteenth week. 4
Dandy-Walker malformation is characterized by the fulfillment of three criteria: (1) the complete or partial absence of the cerebellar vermis, (2) the enlargement of the fourth ventricle, and (3) the cystic enlargement of the posterior fossa.2,4,5 Its frequency is between .003% and .01% of births.1,4 Dandy-Walker malformation is commonly associated with genetic abnormalities, with one review article stating that out of 78 cases of DWM reviewed by three studies, chromosomal abnormalities were present 43.6% of the time. Of the 78 DWM cases reviewed, trisomy 18, which is the most commonly associated trisomy, was present in 20 cases. 6 Trisomy 18 is generally fatal, with only 5% to 10% of infants living past the first year of life. 3 Whereas DWM may be associated with genetic deficiencies or trisomies 30% to 43.6% of the time, it is also suggested that it may be linked to environmental factors or modifiable risk factors such as smoking or stimulant use.1,3,6 A lack of significant correlation with maternal exposure suggests, however, that a genetic predisposition is more predominant. Some research has been conducted that identifies a correlation between maternal history of fertility treatment and twinning. 7 Dandy-Walker malformation is largely sporadic, occurring without prior family history; however, siblings of those with the anomaly are more likely to be affected.
Whereas Dandy-Walker malformation describes the classic appearance, the term Dandy-Walker complex offers an overarching term for the many variants that are similar to this malformation, including associated anomalies. One such association is hydrocephaly, which is found in 80% of DWM cases. Malformations of the cerebral gyri are another possible intracranial anomaly associated with DWM, such as the polymycrogyria present in the current case. Dandy-Walker malformation is also associated with gray matter heterotopia, which was a differential diagnosis in the current patient. 3 Another anomaly that is found in one-sixth of those with DWM is complete or partial agenesis of the corpus callosum (ACC), which is one of the most common anomalies of the brain found in humans. 4 When occurring singularly, those affected by ACC may live normal lives free from symptoms. However, additional central nervous system abnormalities generally lead to worse prognoses.3,8 Similarly, DWM alone has a better prognosis than if it were accompanied by additional intracranial or extracranial anomalies.
Dandy-Walker malformation is generally associated with developmental problems, especially in motor-related skills such as crawling and walking. Those affected will also have mental deficiency or learning disability. One of the most common symptoms is excess fluid in the posterior fossa, leading to hydrocephaly. The congenital malformation is generally treated with interventional therapies, allowing those affected to cope with mental and developmental disabilities. Associated hydrocephaly may require a more acute solution, which most often comes in the form of a ventriculoperitoneal or cerebral shunt. 9 These shunts allow the drainage of excess CSF from the ventricles, reducing intracranial pressure and allowing for necessary cognitive development. These shunts have been found to reduce morbidity and mortality in patients with DWM experiencing associated hydrocephaly. 9 The current patient, although displaying both DWM and ACC, did not exhibit hydrocephaly that was in need of surgical intervention.
Dandy-Walker malformation is most commonly diagnosed in utero, as a result of prenatal sonography. 1 Magnetic resonance imaging is also a sensitive and effective modality for the interrogation of the fetal and neonatal brain. The normal posterior fossa in a neonate is best viewed via the mastoid fontanelle. However, when DWM is diagnosed in utero, an axial view of the posterior fossa is used. The normal cerebellum appears hyperechoic and heart shaped, with an intact vermis connecting the two hemispheres. The fourth ventricle will appear as a small anechoic collection between the hemispheres. The normal cisterna magna will measure less than 10 mm. 10 The normal corpus callosum will appear best in the midsagittal view of the neonatal brain, forming the roof of the anechoic CSP.
Malformations of these structures create important sonographic characteristics imperative for diagnosis. First, the ACC is demonstrated by widely spaced lateral ventricles that appear slit-like ventrally and become dilated dorsally. 3 The spacing of the lateral ventricles, along with the third ventricle, creates a “longhorn” symbol in coronal imaging, with the third ventricle representing the face, and the lateral ventricles the horns of the bull, as demonstrated in Figure 1. Complete ACC leads to a resulting absence of the CSP. In addition, when viewed midsagittaly, the cerebral sulci and gyri are arranged radially about the third ventricle, causing the “sunburst sign,” 3 as demonstrated in Figure 3. Dandy-Walker malformation sonographically presents with an enlarged fourth ventricle, which communicates with an enlarged cystic collection in the posterior fossa. The cerebellar vermis is absent, which leads to splayed and hypoplastic cerebellar hemispheres, 3 as demonstrated in Figure 4.
Dandy-Walker malformation, and its characteristic excess fluid in the posterior fossa, is at times difficult to distinguish from other disorders. Other disorders with which it may be confused include mega cisterna magna or arachnoid cyst of the posterior fossa, which can also appear as fluid collections. Additional imaging may be required to determine if the fluid communicates with the subarachnoid space. 4
Conclusion
The case presented exhibited classic sonographic characteristics of both DWM and ACC under the scrutiny of neonatal neurosonography, along with the corresponding MRI characteristics. Antenatal sonography resulted in early detection of a variety of fetal abnormalities that led to appropriate fetal monitoring in utero. Upon birth, neonatal sonography confirmed the diagnosis of DWM and its most common corresponding deficiency, ACC, and was used for serial assessment of the condition. Although the infant is currently stable, an increase in fluid may indicate the need for surgical intervention. Sonography proved to be an important diagnostic tool in improving the management of the infant’s care as well as assisting in treatment planning and preparing the parents for expected obstacles as a result of this condition.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
