Abstract
Acute pulmonary embolism is the third most common acute cardiovascular disease, with about 600,000 cases annually in the United States. Pulmonary embolism requires a multimodality diagnosis and immediate treatment. Although computed tomography and ventilation perfusion scans are the most commonly used modalities to diagnose pulmonary embolisms, many supplemental tests are necessary. Treatment options for pulmonary embolism include anticoagulation therapy, thrombolytic therapy, or insertion of an inferior vena cava filter when anticoagulation is contraindicated. The long-term benefits of thrombolytic therapy have made it an increasingly popular option in many institutions. The following case study describes a patient who presented to the hospital with shortness of breath for five months and was found to have extensive pulmonary embolisms upon admission. The patient underwent three days of thrombolytic therapy that significantly reduced his pulmonary arterial pressures and resulted in an almost complete resolution of his pulmonary embolisms.
Introduction
Pulmonary embolism (PE) and deep venous thrombosis (DVT) are leading causes of preventable death and disability in the United States. Acute PE is the third most common acute cardiovascular disease, with about 600,000 cases annually. Untreated PE is fatal in up to 30% of patients. Risk of death from acute PE is greatest within the first 30 days. 1 However, if PE is diagnosed and treated within a short timeline, the mortality rate drops to 2% to 10%. 2
Although PEs are relatively common, they remain difficult to diagnose. The clinical presentation is often nonspecific in that patients know “something is wrong” but cannot pinpoint specific symptoms. 3 Because patients cannot adequately describe their symptoms, they may delay seeking medical care. If symptoms are too vague, it is possible patients may be diagnosed with generalized anxiety rather than PE. 4 In a recent survey of literature from 1945 through 2002, PE was unsuspected or went undiagnosed before death in 84% of patients who had PE discovered at autopsy. 5
Case Report
This patient was a middle-aged man who presented to his cardiologist with worsening dyspnea for five months. The patient’s cardiologist performed an electrocardiogram in the office, which revealed T-wave inversions, and decided to send the patient to our emergency room for further evaluation due to increasing dyspnea.
Once the patient was in the emergency room, a lung ventilation and perfusion (V/Q) scan was ordered as the primary test. Ventilation images were taken using xenon-133 11.3 mCi. Anterior and posterior projections of the lungs were imaged during inhalation, equilibration, and washout of 133Xe gas. Perfusion images were taken using technetium-99m MAA 4.8 mCi. Anterior, posterior, posterior oblique, and lateral projections of the lungs were imaged during perfusion. Ventilation images demonstrated normal ventilation bilaterally. Perfusion images showed markedly abnormal lung perfusion, with almost complete absence of perfusion of the right lung except for a small segment near the apex (Figure 1). Small to moderate-size perfusion defects were present in the left lung apex and in the left midlung. The V/Q scan was read as “high probability of pulmonary embolism, primarily on the right.” A front chest radiograph, transthoracic echocardiogram, venous duplex, and computed tomography (CT) of the chest were ordered (per hospital protocol) due to the high probability of PE on the V/Q scan. Chest radiograph revealed no acute processes (Figure 2). The echocardiogram revealed a dilated inferior vena cava (2.52 cm; normal is 2.1 cm) as well as a dilated right atrium and ventricle with mildly reduced systolic function of the right ventricle. Pulmonary hypertension was demonstrated with tissue Doppler index of the lateral tricuspid valve annulus of 19 cm/s (normal limits [NL] > 10 cm/s) and tricuspid annular plane systolic excursion of 4.2 cm (NL ≥ 1.6 cm) (Figures 3-6). Venous duplex revealed no DVT in the bilateral lower extremities. CT revealed multiple pulmonary emboli with central emboli in the right upper and lower lobe bronchi and in the left lower lobe bronchus with multiple smaller emboli extending into peripheral branches. A large clot burden was noted with flattening of the interventricular septum (Figures 7 and 8).
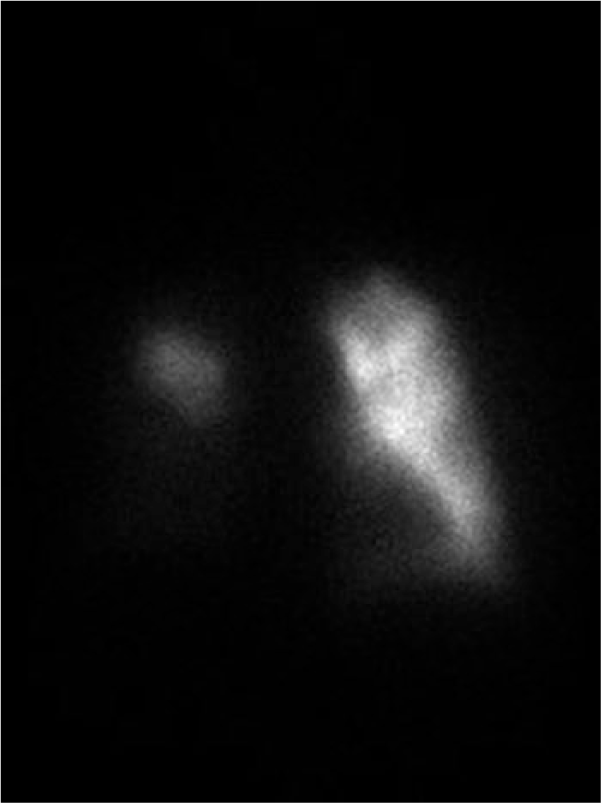
Perfusion images showing almost complete absence of flow in the right lung and small defects in left lung.
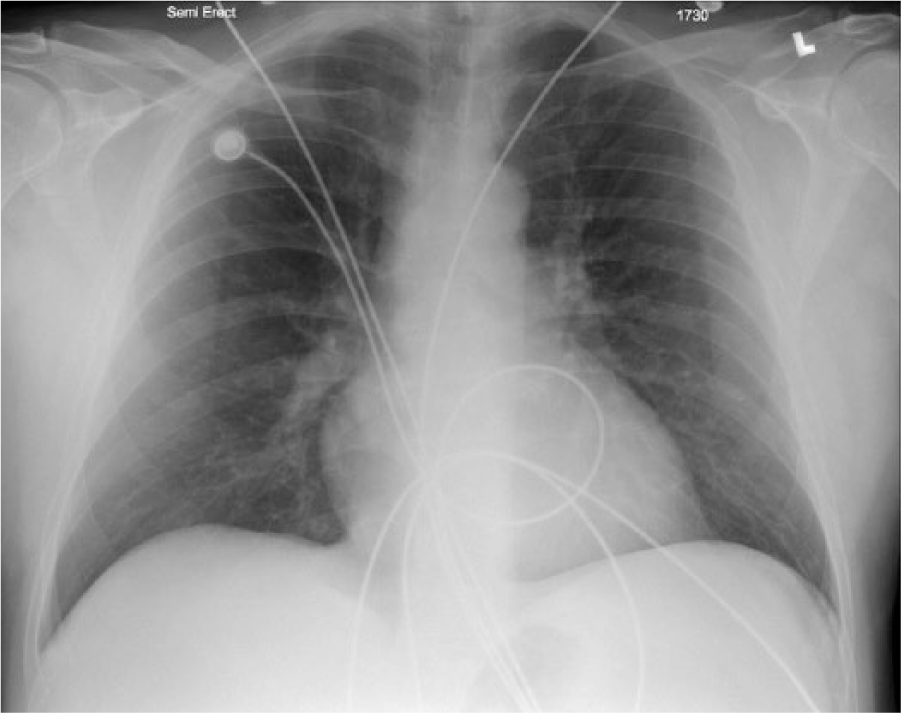
Normal chest radiograph of the patient being treated.
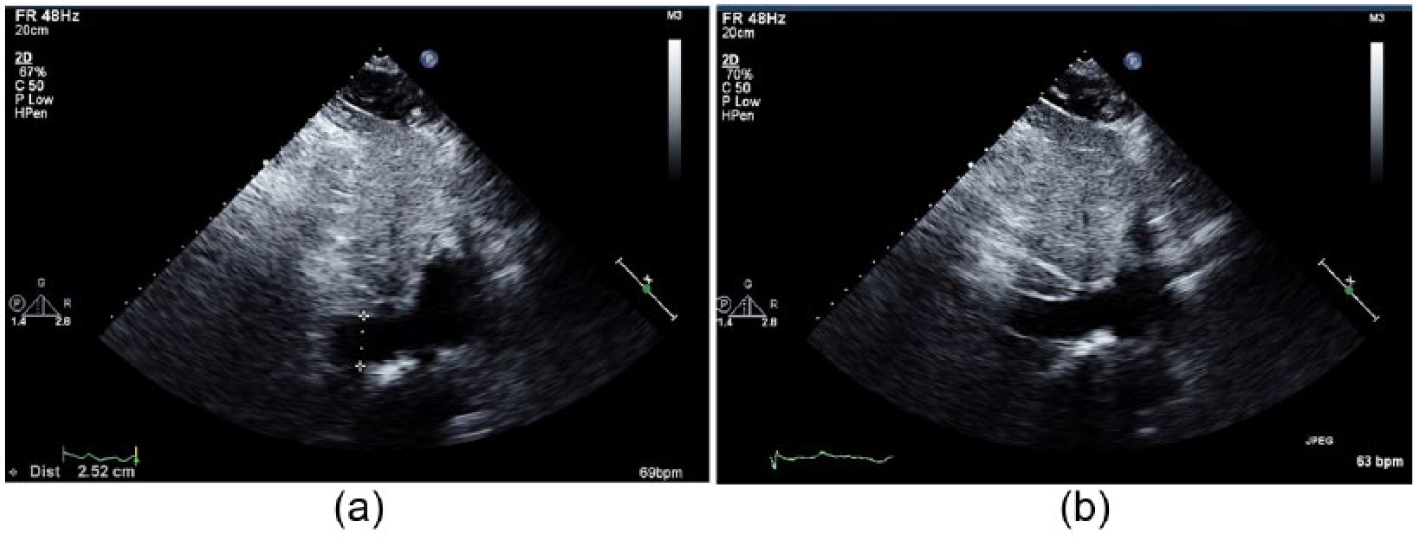
The patient’s echocardiogram demonstrating a dilated inferior vena cava.
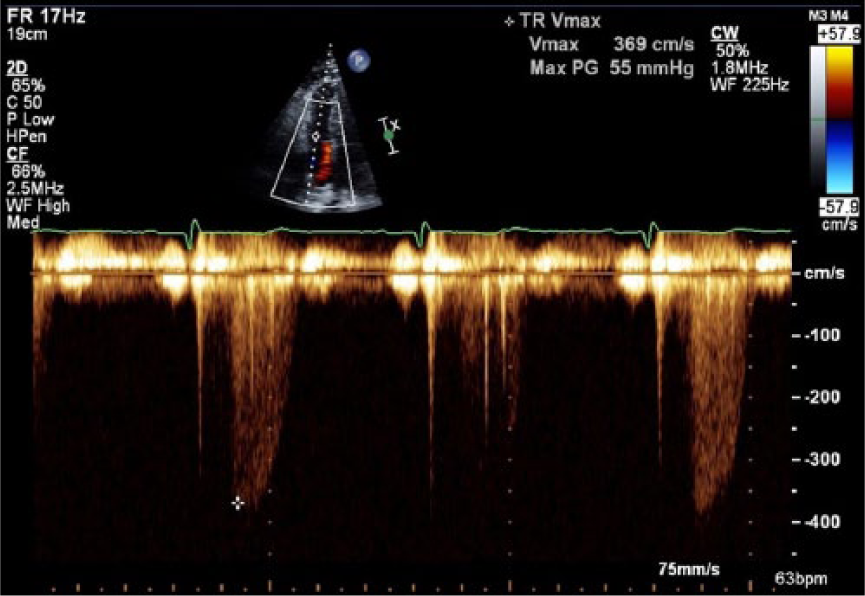
Echocardiogram image demonstrating a high tricuspid regurgitation gradient measured at 55 mmHg. Because the inferior vena cava is dilated (Figure 3a, 3b) but has normal respiratory variation, add 8 mmHg for a total of 63 mmHg. A total of 63 mmHg suggests severe pulmonary hypertension.
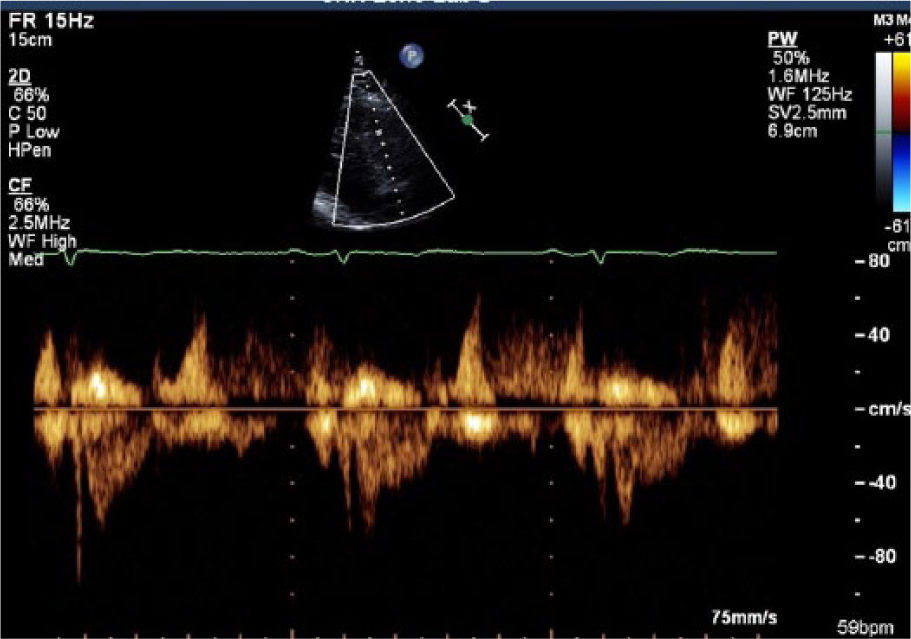
A “dagger-shaped” profile at right ventricular outflow tract shows high vascular resistance in the lungs (high pulmonary pressure).
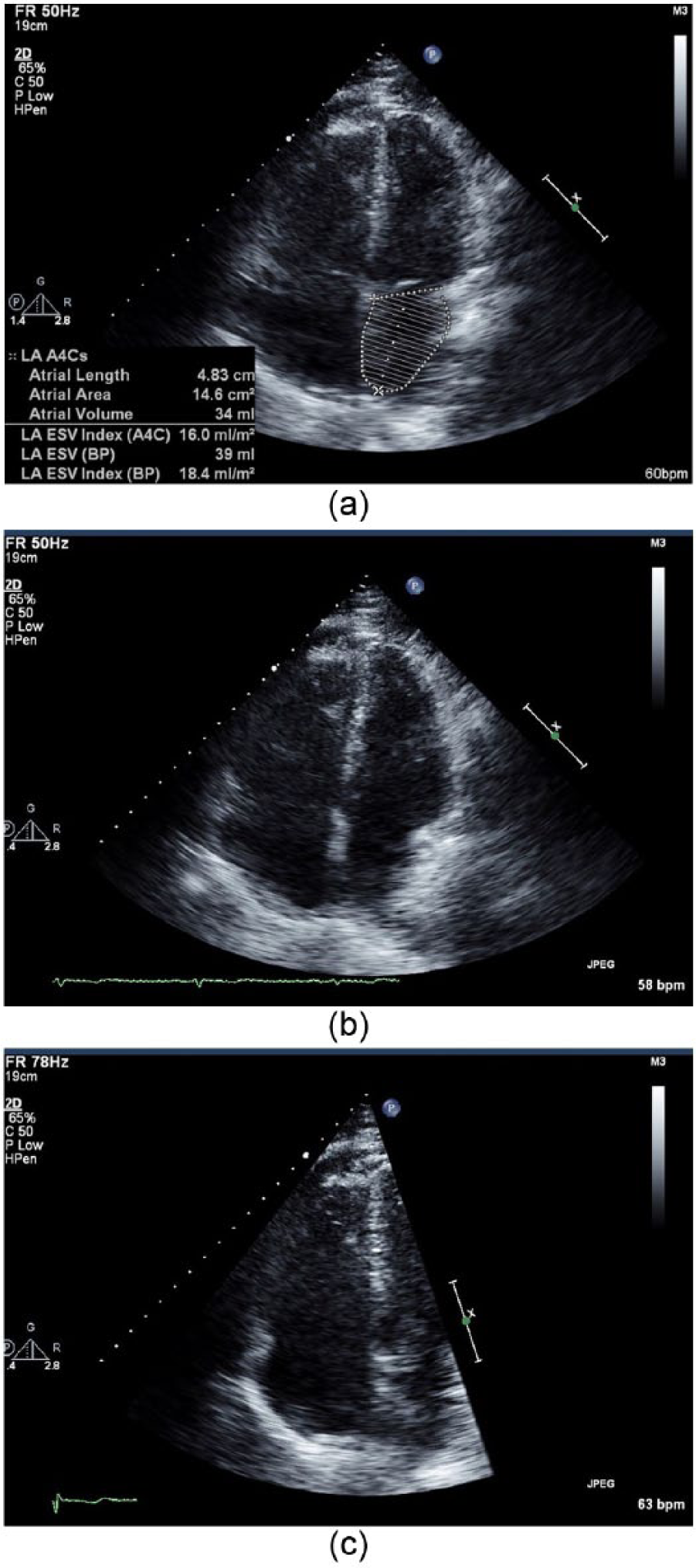
(a) Echocardiogram demonstrating right atrium dilatation (left atrium being measured here). (b, c) Echocardiogram demonstrating the patient’s right heart dilatation.
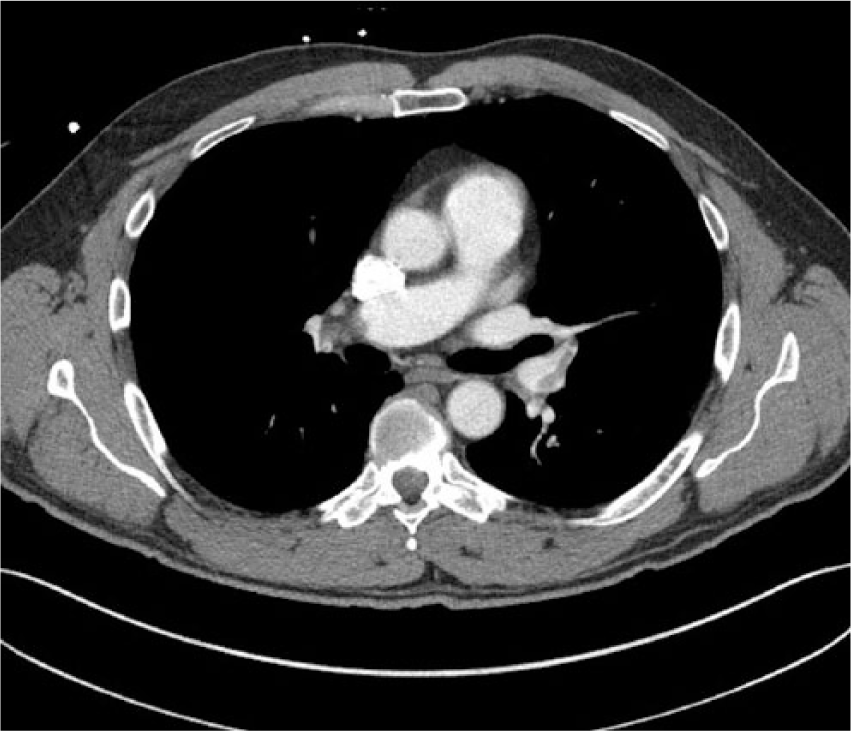
Computed tomography image showing filling defect of the patient’s right pulmonary artery.
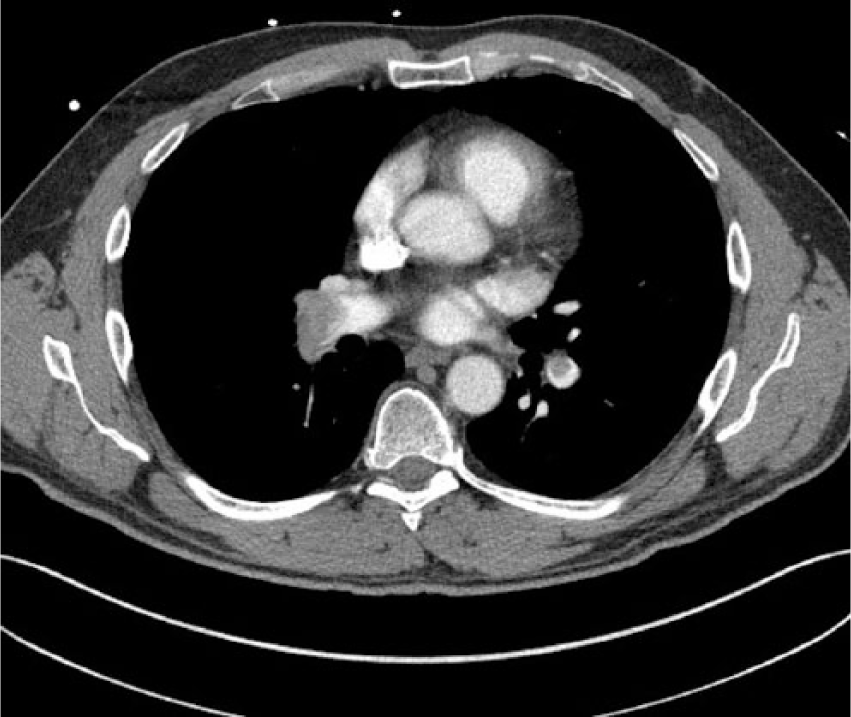
Computed tomography image showing filling defect of the patient’s right pulmonary artery.
Due to the severity of the patient’s PE, the decision was made to admit the patient. The interventional radiology service was consulted as a result of the pulmonary hypertension revealed on the echocardiogram. The interventional radiology staff recommended a pulmonary arteriogram and possible lysis (pending results of the arteriogram).
The patient was taken to the radiology suite the following morning, where he was prepared for the procedure and conscious sedation was administered. Ultrasound guidance was provided to ensure patency of the right common femoral vein and to provide guidance for the catheter wire. Pulmonary artery pressures were measured in the right pulmonary artery first and then the left. Pulmonary pressure measurements revealed severe pulmonary hypertension, 55/16 mmHg for the right pulmonary artery and 55/16 mmHg for the left pulmonary artery (normal pulmonary artery pressures are 15-25 mmHg). Following the pressure measurements, pulmonary arteriography was performed (Figure 9). Pigtail catheters were negotiated into the bilateral interlobar pulmonary arteries, where the catheters were then exchanged for EKOS infusion thrombolytic catheters (18 cm on the right, 12 cm on the left). Ultrasound accelerated thrombolysis was then delivered via the bilateral EKOS catheters. Nurses were instructed to deliver 1.0 mg of tissue plasminogen activator (TPA) per hour for the first six hours in the right catheter and then reduce the TPA to 0.5 mg per hour. Left infusion catheter was to be delivered at a constant 0.5 mg per hour. The patient was instructed to return to the radiology suite in the morning for a follow-up.
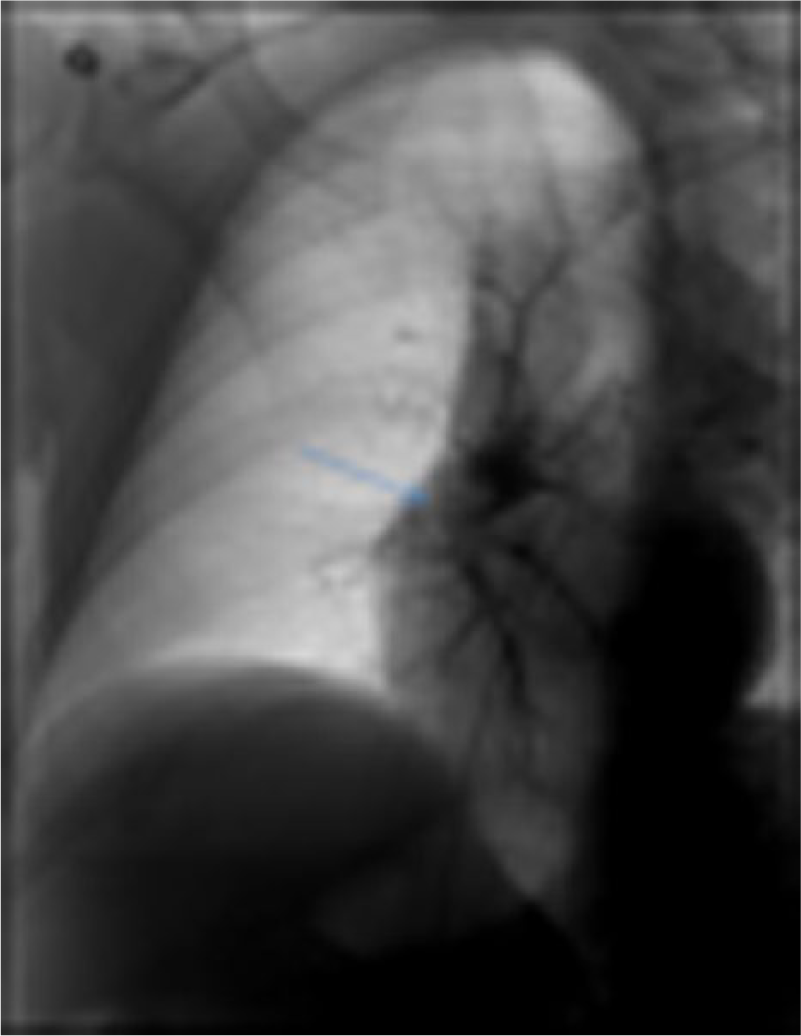
Initial radiological procedure image, showing no filling in the right pulmonary artery.
The patient returned to the radiology suite the following morning, where an improvement in bilateral pulmonary artery pressures was discovered. The left pulmonary artery pressure was 44/7 mmHg and the right was 44/7 mmHg. Left pulmonary arteriography showed markedly improved flow with near-complete resolution of clot burden. Right pulmonary arteriography demonstrated reduced but residual clot in the interlobar artery. The decision was made to discontinue left pulmonary artery catheter–directed thrombolysis but to continue thrombolysis on the right pulmonary artery and to image the patient again the next day.
When the patient returned to the radiology suite the following day, he had undergone a total of 44 hours of thrombolytic therapy. Pulmonary arteriogram demonstrated decreased clot burden with a single eccentrically based residual filling defect in the inferior lobar right pulmonary artery (Figure 10). Pulmonary arterial pressures had further decreased to 34/8 mmHg bilaterally. Due to the further improvement in arterial pressures, decrease in clot burden, and clinical improvement of the patient, thrombolytic therapy was discontinued. The patient was discharged the following day with no complaints of shortness of breath.
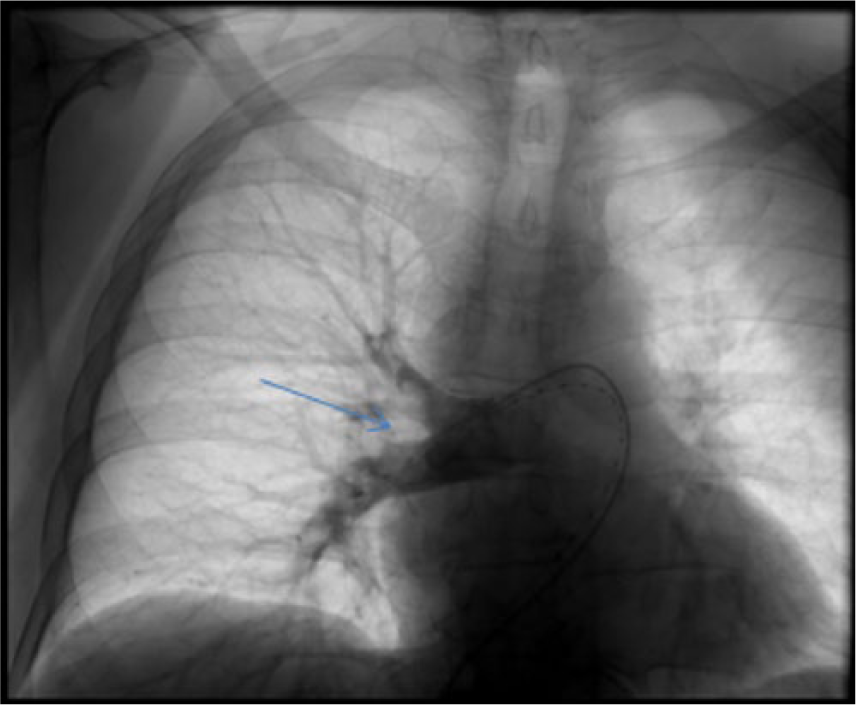
Final radiological procedure image, showing full filling of the right pulmonary artery.
Discussion
PEs are typically classified as acute minor PE, acute massive PE, or subacute massive PE. The severity of a PE will determine the patient’s symptoms. In cases of acute minor PE, emboli obstruct less than 50% of the pulmonary circulation, and the patient will most likely be asymptomatic. If more than 50% of the pulmonary circulation becomes suddenly obstructed, patients are considered to have an acute massive PE. 6 Massive acute PE will lead to hemodynamic instability and hypotension. When obstruction occurs, the impaired pulmonary circulation causes diminished blood flow to the right cardiac ventricle and patients will experience a sudden onset of symptoms. 7 Unexplained chest pain and/or dyspnea is present in about 97% of patients with acute PE. 7 Other symptoms may include unilateral swelling of an extremity, fainting or syncope, and/or hemoptysis. 5 Subacute massive PEs can be caused by multiple small or moderately sized emboli that accumulate over several weeks. Because the right ventricle and pulmonary system have time to adapt, these patients will experience a gradual onset of symptoms. 6
Risk factors for PE include, but are not limited to, older age, previous DVT or PE, cancer, neurologic disease with extremity paresis, surgery, prolonged bed rest, use of oral contraceptives, and hereditary factors (factor V Leiden, acquired thrombophilia). 2 Risk factors such as pregnancy have been debated, but recent studies have revealed that a woman’s risk of PE increased five-fold during pregnancy. 7 Up to 30% of PEs are determined to be idiopathic. 2
Although sonograms of the extremities cannot confirm or deny the presence of a PE, sonography remains a key part of a patient’s plan of care in cases of suspected PE or attempts to prevent PE. Recent publications report that as many as 50% of patients with sonographically documented DVT did not report typical symptoms (pain, swelling, warmth). Approximately 40% of patients with extremity DVT develop an asymptomatic (silent) PE. 8 Silent PEs are more likely to occur in patients with proximal DVT as opposed to infrapopliteal DVT. 9 Duplex sonography is a critical tool in diagnosing DVT in the extremities. Sonography is 89% to 96% sensitive and 94% to 99% specific for diagnosing proximal (common femoral, femoral) DVT. 10 An estimated 70% of patients diagnosed with PE are found to have extremity DVT when imaged. 2 If a duplex imaging study of the extremities is interpreted as negative, it is often assumed that the entire thrombus has detached and embolized. 6 Sonography is also critical for surveillance of known DVT. If the risk of using anticoagulants outweighs the benefit, physicians may choose to leave a calf DVT untreated. However, if left untreated, calf DVT has a 20% to 25% risk of propagating into the proximal leg. 11 Therefore, the physician treating the patient will typically order serial duplex sonograms to monitor the DVT. For patients with minor acute PEs and distal DVT with no propagation, anticoagulation therapy can be safely omitted. 6
Sonography is also helpful when attempting to differentiate between acute and chronic DVT. Acute DVT appears on a sonogram as an enlarged vein with anechoic or homogenous intraluminal echoes. Little or no color flow may be present. Chronic DVT appears on sonography as a vein with echogenic intraluminal echoes or webbing with evidence of recanalization. Veins may be atrophied, and collaterals may be present. In cases of chronic DVT with recanalization, continuous flow may be noted.
Diagnosing PE requires multiple modalities. Using single diagnostic tests in isolation can lead to mismanagement of suspected PE. 7 Several different laboratory tests and diagnostic imaging studies may be ordered on a patient presenting with suspected PE. These tests may include drawing D-dimer serum and/or performing V/Q scintigraphy scan, pulmonary computed tomography angiography, and transesophageal echocardiography. 2
Due to its high negative predictive value, a normal (negative) D-dimer draw can be used to exclude PE (without further testing) in patients with a low pretest probability. 12 In fact, patients with a low pretest probability and normal D-dimer have a 99% negative predictive value for DVT.
The two main imaging modalities for diagnosing PE are pulmonary CTA and V/Q scan. V/Q scans should include both ventilation and perfusion. In cases of PE, perfusion defects seen on V/Q are due to a blockage in blood flow due to embolism. V/Q scans are most helpful when used in combination with chest radiographs. 13 V/Q scans are useful for baseline imaging. If a patient presents with recurring symptoms, a new V/Q scan can be compared with the baseline to determine whether a PE is old or new. 2 Additionally, V/Q scans result in fewer allergic reactions than CTA. 9 CTA has become the gold standard for imaging PE in recent years. 12 Due to the higher sensitivity of CTA, PE disease diagnosed with CTA is usually milder than disease diagnosed on V/Q scans. In fact, the odds of death among patients with PEs diagnosed with pulmonary CTA were approximately one-half of the odds for those diagnosed with V/Q. 14 CTA has a low rate of “nondiagnostic” results and is readily available. In addition to detecting PEs, CTA can diagnose other conditions such as acute pneumonia, lung abscess, pleural abscess, pleural or pericardial effusion, aortic disease, cardiovascular disease, esophageal rupture, and/or malignancy. As well as ruling out PE, diagnosing these disease processes via CTA can expedite appropriate treatment. 9 CTA has greater than 90% specificity in diagnosing PE in the main, lobar, and segmental pulmonary arteries. 6 Studies have shown an increase in the risk of patient death within the first 30 days after diagnosis with CT findings of increased clot burden and septal bowing toward the left ventricle. 13 The most common supplemental test ordered with a CTA (for suspicion of PE) is sonography (lower extremity and/or echocardiogram). 12 Echocardiograms are useful in differentiating heart failure from PE and assessing right ventricular function in patients with large clot burden. 12 Although it is rare to see a PE on an echocardiogram, thrombus may be seen in the right ventricle or atrium. 6 Echocardiograms and transesophageal echocardiography can provide rapid, real-time, portable tests in cases of critically ill patients who are at an increased risk of death. 11
Treatment for PE involves anticoagulation therapy unless contraindicated. Anticoagulation therapy entails risks (hemorrhage, interactions with other medications), patient inconvenience (side effects, blood draws), and cost. 3 Patients with idiopathic embolism or low risk of recurrence of embolism are typically anticoagulated for 3 to 6 months. Patients with high risk factors or high probability of recurrence may be anticoagulated indefinitely. 15 The benefits of anticoagulation versus a patient’s risk of complications must be considered before any treatment regimen is begun. Mild PEs are managed with anticoagulants, while severe PEs may be managed with thrombolytic therapy. Historically, PEs have been noted to resolve faster in the main and lobar pulmonary arteries than in the segmental branches. 13
Thrombolytic therapy has demonstrated superior clot breakdown compared with a combined treatment of heparin and warfarin. 13 Early benefits of thrombolytic therapy, aside from clot resolution, include improved right ventricular function and preservation of the diffusing capacity of the lung. 16 Thrombolytic therapy has also proven especially useful for preventing postthrombotic syndrome when used simultaneously for DVT lysis in the lower extremities. Postthrombotic syndrome is a long-term complication of DVT that involves issues caused by venous insufficiency. Postthrombotic syndrome results when DVT enlarges a vein, resulting in a decrease in venous wall contractility and damage to the valves. When a patient is standing, the patient’s venous pressure increases and the decreased contractility and incompetent valves cause the development of reflux and varicosities. Symptoms of postthrombotic syndrome include swelling, pain, discoloration, and ulceration. This syndrome significantly impairs the patient’s quality of life and occurs in 25% to 50% of patients with proximal DVT. 17 Recent studies showed a 72% lower extremity patency rate at 6 months in patients who underwent thrombolytic therapy versus 12% in anticoagulated patients. 18 Other long-term benefits of thrombolytic therapy include improvements in hemodynamic stability, decreases in recurrent PEs, maintenance of the patient’s pulmonary reserve, and, possibly, prevention of the development of pulmonary hypertension. 16 Contraindications for thrombolytic therapy include known bleeding issues, renal or hepatic failure, uncontrolled blood pressure, pregnancy, and recent stroke. 18
In cases where anticoagulation is contraindicated, or in patients experiencing recurring PEs despite adequate anticoagulation, a filter may be placed in the inferior vena cava to prevent future PEs. Filters do not prevent or stop the blood from clotting, but because they are placed in the inferior vena cava below the level of the renal veins, they will stop any mobile thrombus from reaching the pulmonary circulation. 7 Recurrence of PE is three times more likely if the initial event was a PE and not DVT. Preventing recurrent PE is important as recurrent PE may cause chronic cardiopulmonary impairment. 15
A thorough clinical assessment and time are of the essence when treating patients presenting with potential PE symptoms. A patient’s outcome is dependent upon competency and cooperation between the medical team and the imaging departments. Accurate imaging is critical when PE is suspected, because both false-positive and false-negative results can have fatal consequences. The treatment used for PE, like all healthcare decisions, requires the weighing of risks and benefits. The severity of the PE typically dictates how it is treated; less severe cases are treated with anticoagulants and more severe cases are treated with thrombolytic therapy. In cases such as the patient described here, a prompt diagnosis and use of thrombolytic therapy can save lives and preserve the quality of life.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
