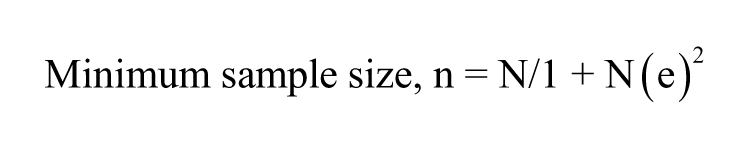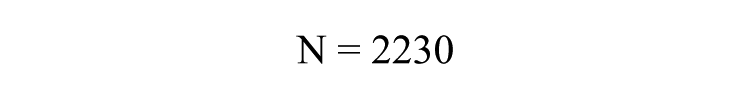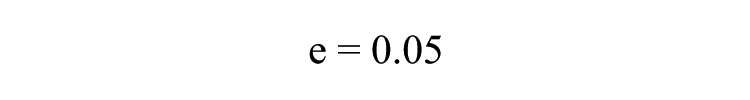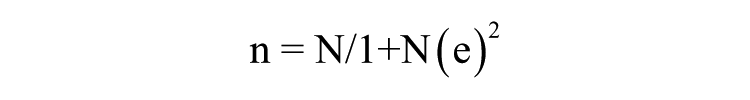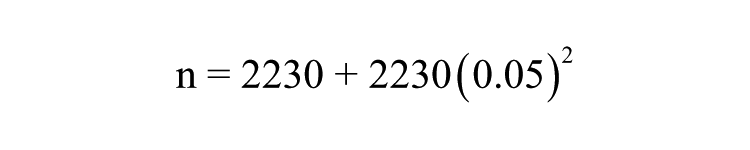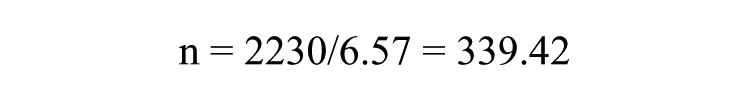Abstract
This study aimed to establish reference values of fetal transverse cerebellar diameter (TCD) in a Nigerian population. A cross-sectional convenience study was carried out between June 2013 and May 2014 in Enugu, Nigeria. The sonographic examinations were performed on 697 pregnant women with gestational ages between 14 and 40 weeks. The TCD measurements were obtained from the proximal outer margin to the distal outer margin of cerebellum. The women’s last menstrual period, femur length, biparietal diameter, head circumference, and abdominal circumference were also recorded. The mean (standard deviation) TCD increased from 13.6 (2.2) mm to 27.3 (2.6) mm and 28.9 (2.0) mm to 42.9 (2.0) mm in the second and third trimesters, respectively. The relationship of TCD with gestational age obtained from last menstrual period, femur length, biparietal diameter, head circumference, and abdominal circumference was determined. The TCD had a strong correlation with gestational age (r = .93; P < .05). Reference values for TCD were established for those patients who attended this clinic. This is a likely data set to use for future research that could focus on similar practices and in other regions of the country for possible generation of a nationwide nomogram.
Introduction
Accurate knowledge of fetal gestational age (GA) is important to facilitate the best possible prenatal care and a successful pregnancy outcome. Sonography is a useful modality for the assessment of GA and can greatly affect obstetric management and improve prenatal care. 1 The routine use of diagnostic sonography in obstetrics may be largely due to its noninvasiveness, affordability, and lack of ionizing radiation.
The cerebellum lies in the posterior cranial fossa, dorsal to the pons and the medulla, and is separated from them by the fourth ventricle. 2 The cerebellum appears in the embryo at the end of the fifth week of gestation and consists of a central portion called the vermis, which joins two lateral hemispheres. A fold of dura mater called the tentorium cerebelli separates the cerebellum from the cerebrum. Physiologically, the cerebellum assists in the coordination of fine motor movements, muscle tone, balance, and equilibrium in the body. 3 Sonographically, the cerebellum can be easily visualized from the second trimester onward via transverse views of fetal intracranial anatomy of the posterior fossa. The cerebellum is routinely imaged in obstetric sonography and transverse cerebellar diameter (TCD) measurement can be reliably obtained. 4
Proper assessment of age is of great importance in obstetric care and management of pregnancies considered overdue, which may be the result of an inaccurate assignment of GA. Correct GA assessment is influential in counseling patients regarding the option of pregnancy termination and detection of intrauterine growth restriction (IUGR). 5 Sonographic determination of fetal GA requires the measurement of several parameters such as crown rump length (CRL), biparietal diameter (BPD), head circumference (HC), abdominal circumference (AC), femur length (FL), and TCD. After the first trimester, CRL is not useful in fetal GA determination due to fetal posturing and size. 6 In addition, possible biological variations can skew results as well as advance GA, and fetal position often makes BPD inaccurate in fetal age determination, especially in the third trimester. 6 Fetal long bone length used in prediction of GA is best in early pregnancy and may be difficult for predicting GA in breech presentation. 6 However, previous research has demonstrated that TCD can serve as a reliable predictor of GA in the fetus and is a standard against which aberrations in other fetal parameters can be compared, especially when GA cannot be determined by the date of last menstrual period or an early pregnancy sonography. 7
A study by Pinar et al. 8 found that inadequate assessment of GA at later stages of pregnancy and an inability to determine fetal GA in the presence of IUGRs may be the result of not using the correct growth parameters. This may have motivated future researchers to explore the accuracy of fetal biometric GA parameters such as TCD, especially in growth-restricted fetuses. It is interesting that a study by Hashimoto et al. 9 found that the TCD of a growth-impaired fetus was lower than expected but still within the normal range. Similarly, Saifon et al. 10 found that the cerebellum size was less affected by deviation in fetal growth restriction or growth acceleration. Although there is a paucity of information on the sonographic reference values for fetal TCD in Nigerian populations, previous studies were done in other countries.10,11
The objectives of this study were, therefore, to determine the TCD nomogram in relation to the patient’s last menstrual period (LMP) and fetal FL, BPD, HC, and AC parameters; to establish a relationship between TCD and GA obtained from the patient’s LMP, fetal FL, BPD, HC, and AC parameters in a Nigerian population sample; and to compare the current mean TCD values from this study with those obtained from previous published studies.
Methods
This was a cross-sectional design that was carried out between August 2013 and May 2014 using a convenience sampling method in a university hospital in southeastern Nigeria. A total of 697 pregnant women who met the inclusion criteria had an ultrasound examination in the radiology department. The sample used for the study was calculated using the following formula by Yamane. 12 The formula can be used for the calculation of the minimum sample size for an infinite or unknown population so that results obtained from the sample size selected could represent the population studied:
where n = minimum sample size, N = population size, and e = percentage error (percentage error at 95% level of confidence = .05). Using the total number of patients who attended a prenatal clinic in 2012, a population sample of 2230 was obtained:
Therefore, an a priori sample of 339 was set for this study. The sample size was increased to 697 to address possible dropout participants and to oversample for the purposes of achieving the desired outcome.
Inclusion Criteria
Patients with the following criteria were included in the study to maintain the normal anatomy and size of the TCD: a confirmed singleton pregnancy, a known LMP, a GA between 14 and 40 weeks, and a normal pregnancy (an apparently healthy pregnancy).
Exclusion Criteria
Pregnant women with medical complications such as hypertension or diabetes, twin gestation, and first trimester pregnancy and women with unknown LMP were excluded from the study.
This study was given ethical clearance by the university teaching hospital’s ethics committee. Informed consent was obtained from each participant prior to the beginning of the study.
Equipment
A 2012 Mindray DP 2200 Plus ultrasound machine with 3.5 MHz curvilinear transducer was used for this study. The choice of the transducer frequency was made to ensure adequate penetration and resolution to the cerebellum measured. The measurements were taken using the electronic calipers of the ultrasound machine, making use of the freeze frame capacity.
Scanning Technique
The study was carried out by a sonographer with 12 years of experience in obstetric sonography. A transabdominal pelvic sonogram was completed with the patient in the supine position, after drinking water. A low frequency transducer was chosen to adequately penetrate the fetal cerebellum and obtain measurements. The TCD was measured from the transverse view of fetal intracranial anatomy, through the posterior fossa, that included visualization of anatomic landmarks such as thalamus, cerebellar vermis, cavum septum pellucidum, and cisterna magna. 10 The measured TCD was obtained by placing electronic calipers from the proximal outer margin to the distal outer margin of cerebellum. 10 The characteristic “butterfly appearance” of cerebellum appears as two lobules, on either side of the midline in the posterior cranial fossa. The sonogram of the TCD is shown in Figure 1.
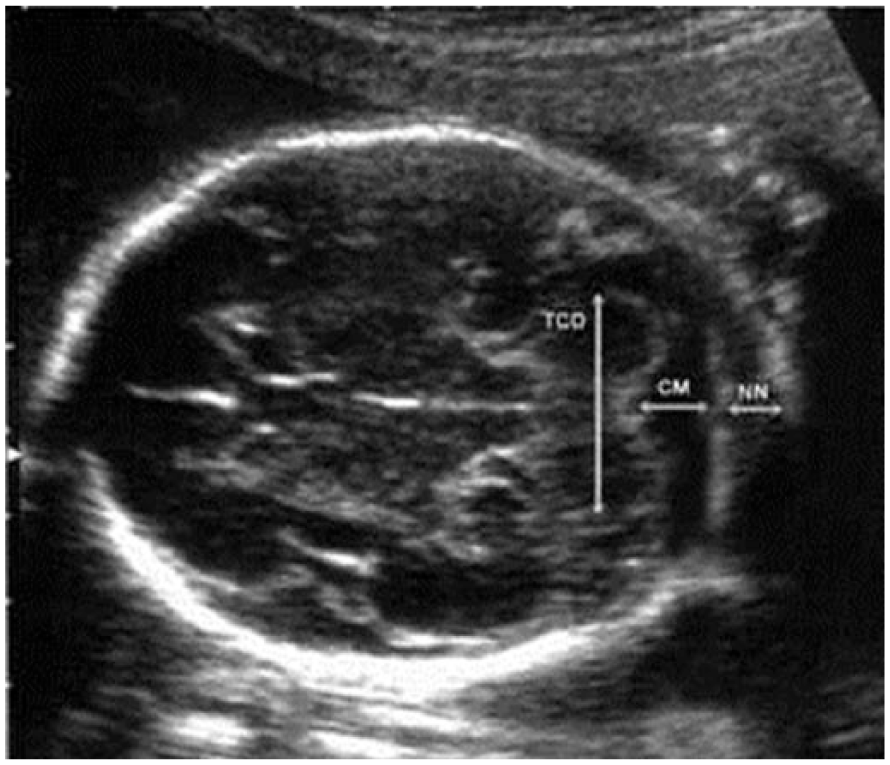
Sonogram showing fetal transverse cerebellar diameter. CM, cisterna magna; NN, nuchal fold; TCD, transverse cerebellar diameter.
Using guidelines published by Hadlock et al., 13 the BPD was measured as the distance between the outer edge of the cranium nearest to the transducer and the inner edge of the cranium distal to the transducer at the level of the paired hypoechoic thalami and cavum septum pellucidum. According to Chitty et al., 14 the HC was measured using the elliptical calipers over the four points of the BPD and occipital frontal diameter in the same plane as the BPD, between the leading edge of the frontal bone and the outer edge of the occiput. The AC was measured as the length of the outer perimeter of fetal abdomen at the level of the umbilical vein junction with the portal vein in a transverse plane perpendicular to the spine, 15 and the FL was measured as the length of the ossified diaphysis of the fetal femur from the greater trochanter to the femoral condyles. 16
Observer Variability Study
Prior to the main study, a pilot study was done involving 20 participants to determine observer variability in the measurement of the TCD. The pilot interobserver variation study involved making sure that the measurement of the TCD was taken on the same participants by two different sonographers, each having 12 years of experience in obstetric sonography. In addition, the measurement of TCD was taken 3 times and the average measurement recorded was made by the same sonographer with the same participant in the intraobserver variation study.
All sonographers graduated from an accredited sonography program and are registered with Radiographers Registration Board of Nigeria.
Statistical Analysis
Data were analyzed using SPSS version 16.0 (SPSS Inc., Chicago, IL). Descriptive statistics were used to determine the mean TCD in relation to the patient’s LMP and fetal FL, BPD, HC, and AC parameters. The correlation and relationship of the TCD with GA obtained from LMP, FL, BPD, HC, and AC parameters were established using a Pearson correlation and regression analyses. Student t test was used to determine the intrarater and interrater variability in the measurement of the TCD and also to compare the mean TCD values from the present study with the data obtained in previous studies (Nepalese 11 and Thai 10 populations).
Results
Table 1 shows that no statistically significant difference (P = .919) was found between the mean TCD values obtained by the same sonographer in the intraobserver variation study (P > .05). Similarly, the interobserver variation study shows that no statistically significant difference (P = .189) was found in the mean TCD values recorded between the two sonographers (P > .05).
Observer Variations in the Measurement of Transverse Cerebellar Diameter Studied (Pilot Study).
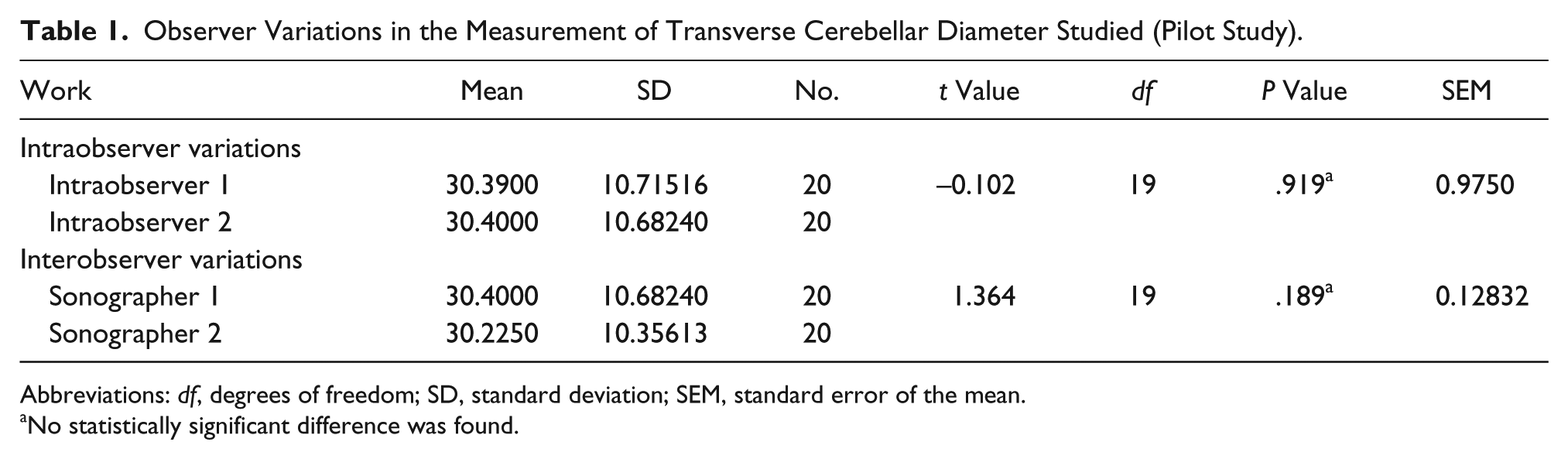
Abbreviations: df, degrees of freedom; SD, standard deviation; SEM, standard error of the mean.
No statistically significant difference was found.
Table 2 shows TCD reference values according to GA, obtained from the patient’s LMP in the second and third trimesters. The distribution of TCD according to GA by the patient’s mean LMP and the combination of mean FL, BPD, HC, and AC parameters in the second and third trimesters is shown in Table 3.
Distribution of TCD According to Gestational Age Obtained by Patient’s Last Menstrual Period.
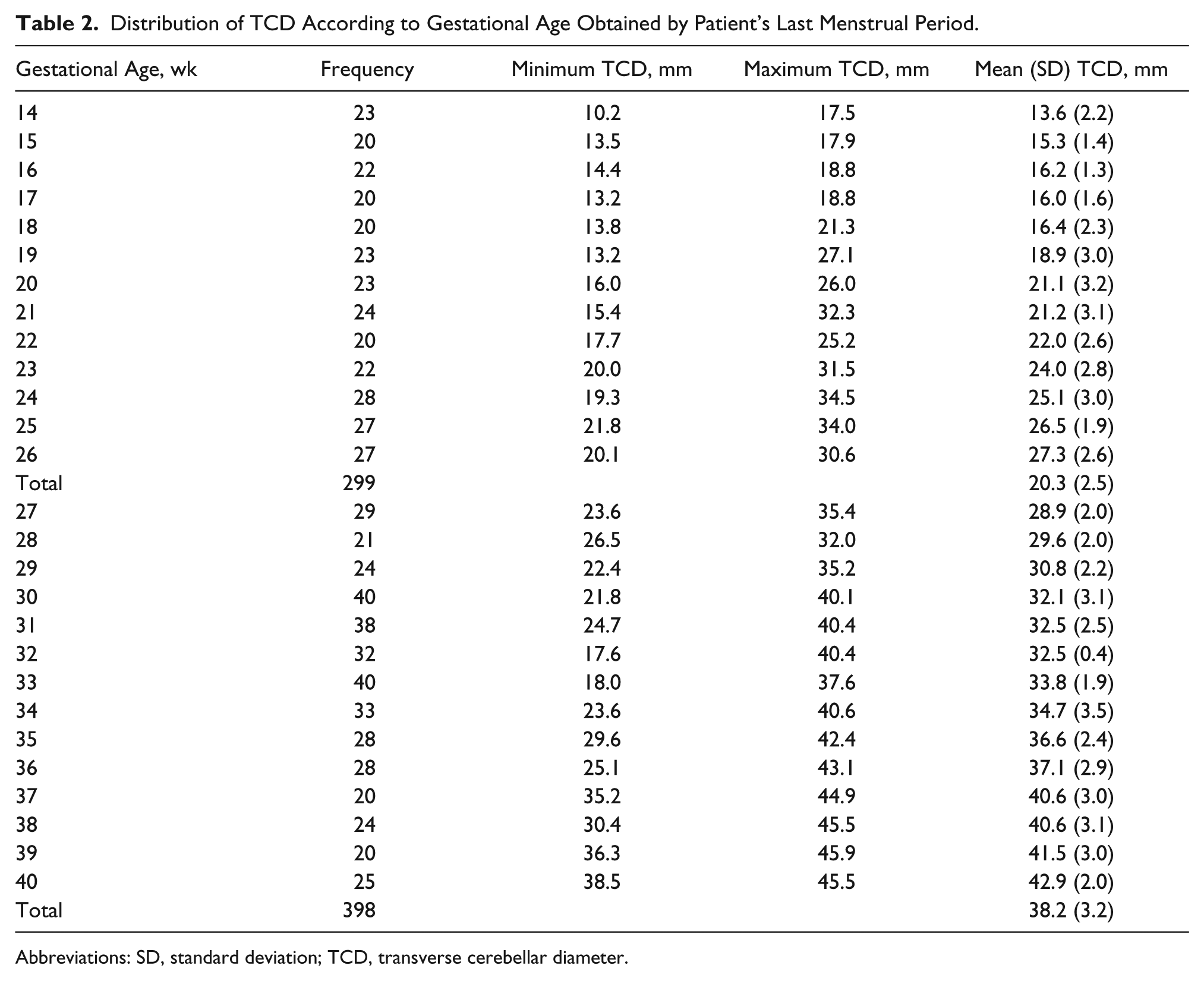
Abbreviations: SD, standard deviation; TCD, transverse cerebellar diameter.
Distribution of TCD According to Gestational Age by Patient’s LMP and the Combination of FL, BPD, HC, and AC Parameters.
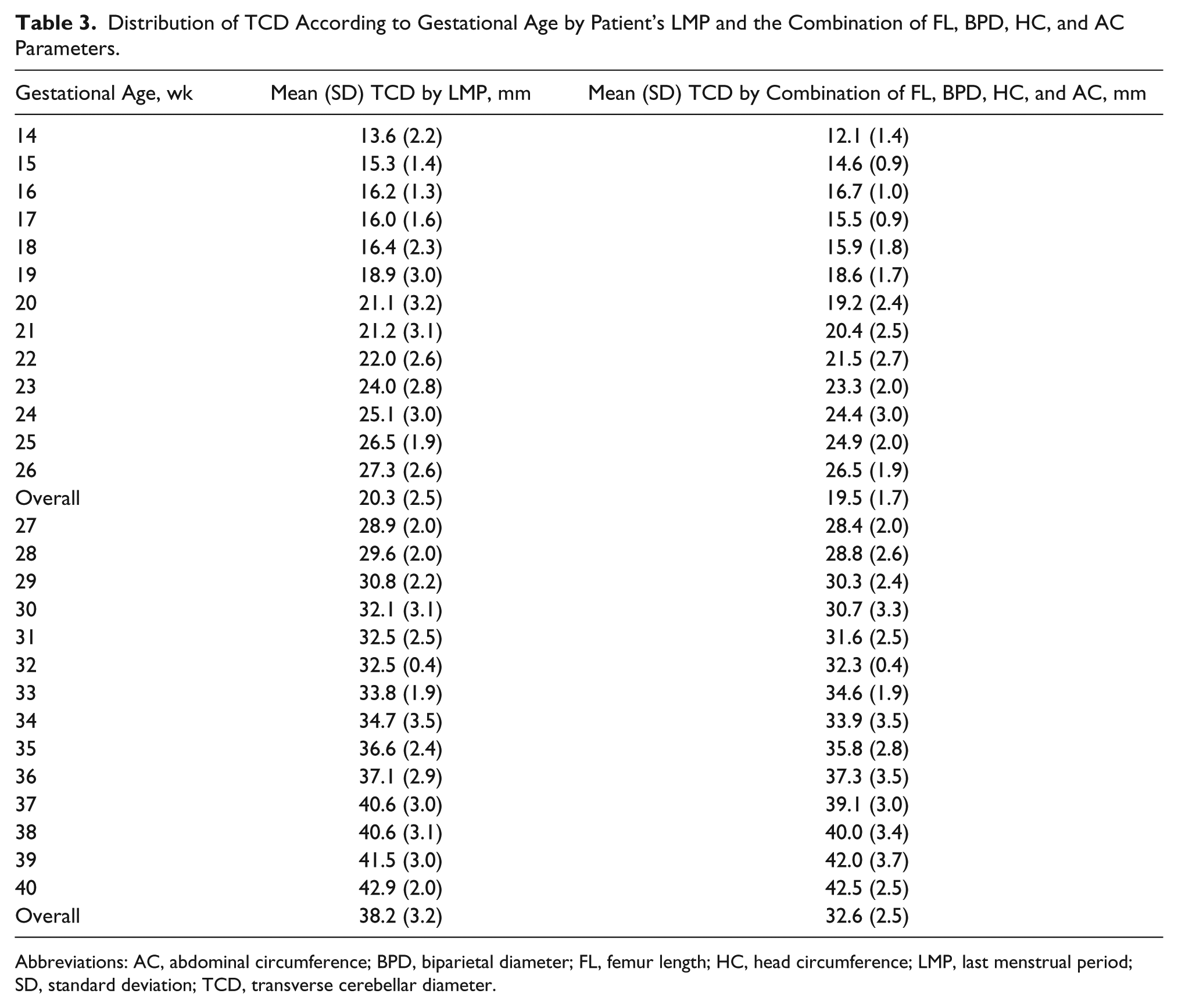
Abbreviations: AC, abdominal circumference; BPD, biparietal diameter; FL, femur length; HC, head circumference; LMP, last menstrual period; SD, standard deviation; TCD, transverse cerebellar diameter.
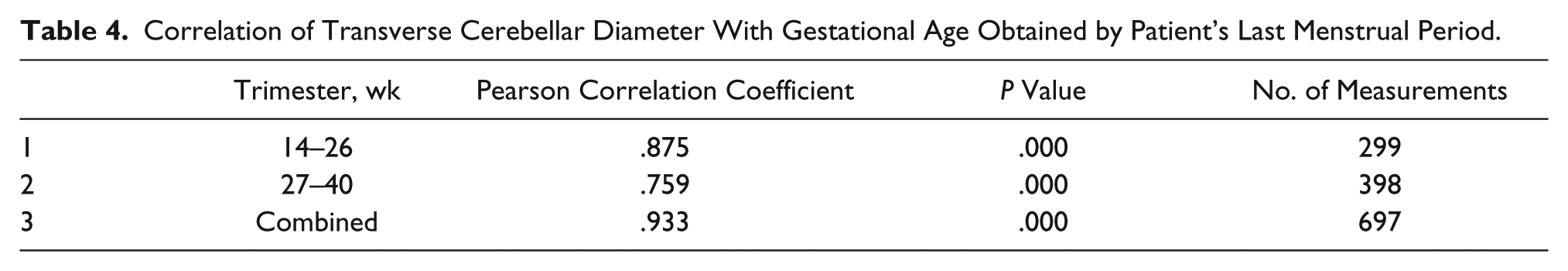
The TCD measurements had a positive correlation with GA in the second (14–26 weeks), third (27–40 weeks), and combined trimesters with r values of .875, .759, and .933, respectively (Table 4). Table 5 shows that TCD has a strong linear relationship with GA (P = .000, P < .05). The F statistic shows that this relationship was highly significant. The model equation for determining GA (Y) for any known TCD was obtained as Y = 0.63 (TCD) + 7.16; Y = 0.54 (TCD) + 14.32, and Y = 0.79 (TCD) + 4.91 in the second, third, and combined trimesters, respectively. The TCD has a positive relationship with FL, BPD, HC, and AC with r values of .957, .941, .940, and .949, respectively (Table 6) (P < .001). Table 7 shows that there was no statistically significant difference between this sample of patients and the data provided by Joshi 11 (Nepalese) and Saifon et al. 10 (Thai).
Correlation of Transverse Cerebellar Diameter With Gestational Age Obtained by Patient’s Last Menstrual Period.
Relationship Between TCD and Gestational Age Obtained From Patient’s LMP and Its Predicted Model Equations.
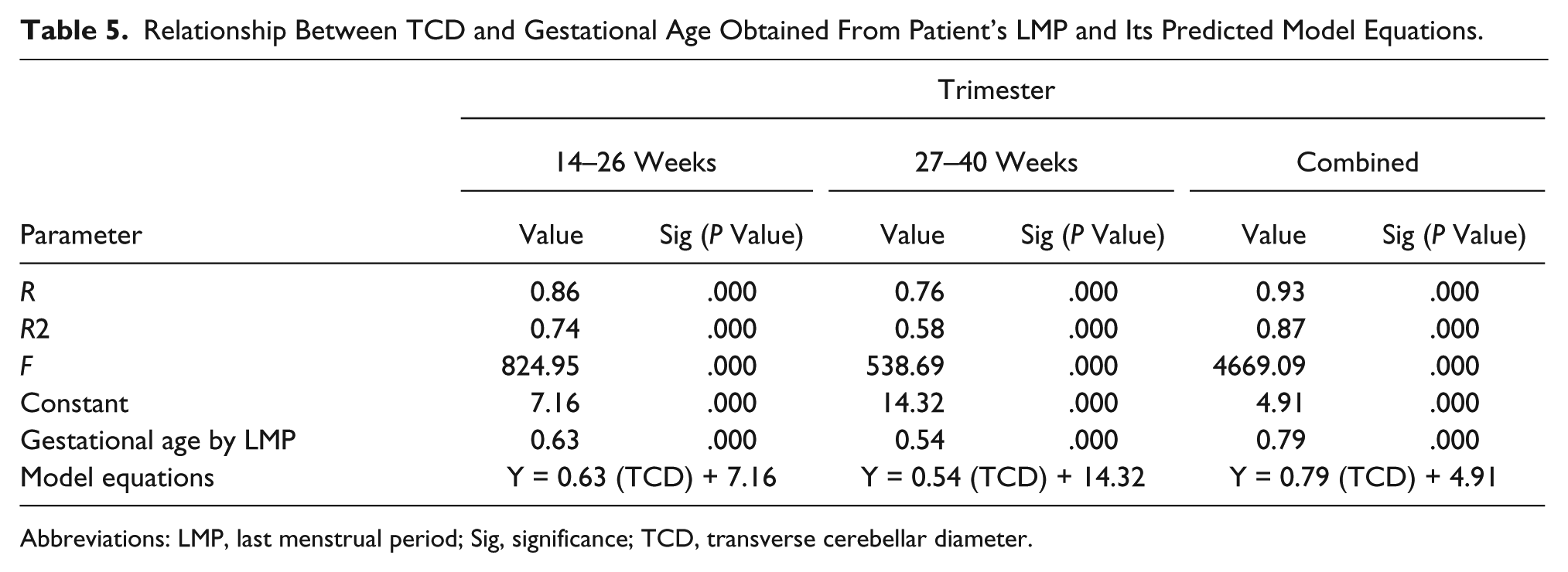
Abbreviations: LMP, last menstrual period; Sig, significance; TCD, transverse cerebellar diameter.
Correlation of TCD With Gestational Age Obtained From Last Menstrual Period and FL, BPD, HC, and AC Parameters.
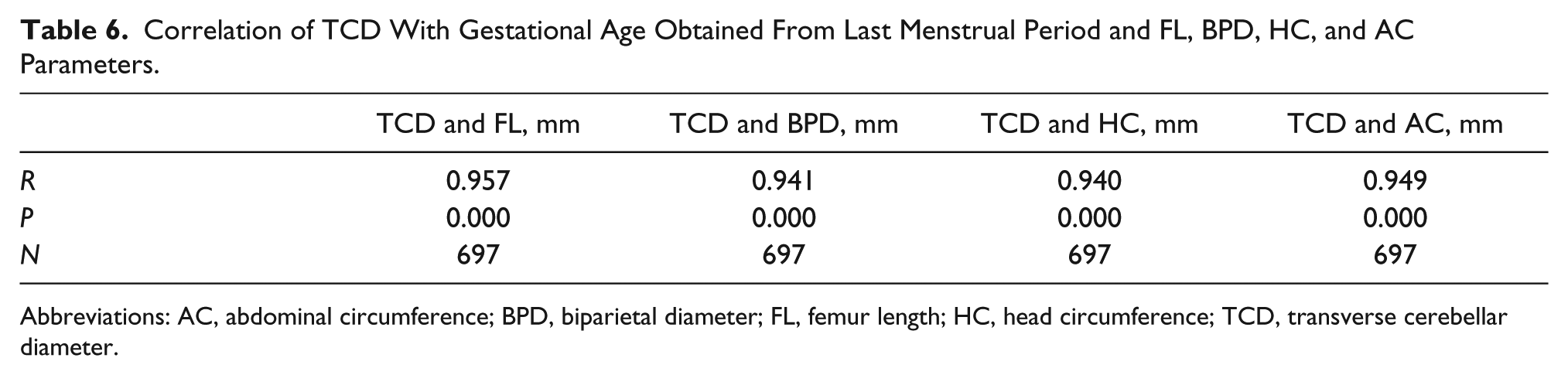
Abbreviations: AC, abdominal circumference; BPD, biparietal diameter; FL, femur length; HC, head circumference; TCD, transverse cerebellar diameter.
Comparison of Mean TCD Values From Present Study and Those Obtained From Previous Studies (Nepalese and Thai Populations).
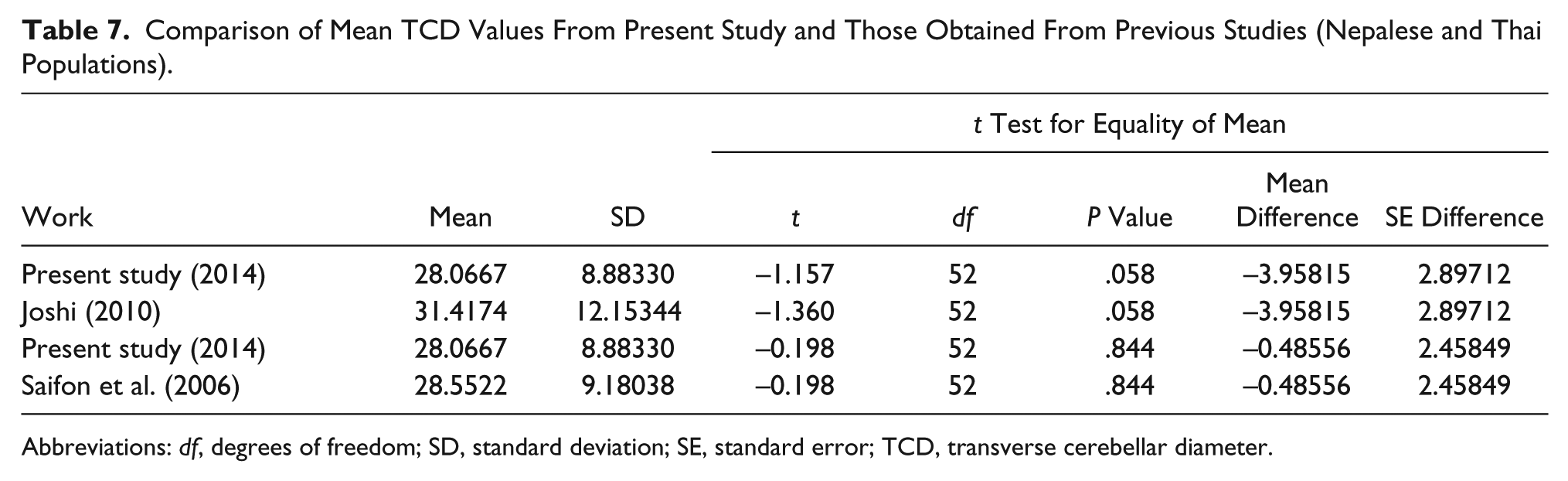
Abbreviations: df, degrees of freedom; SD, standard deviation; SE, standard error; TCD, transverse cerebellar diameter.
Discussion
There was no statistically significant difference (P > .05) between the measurements made by each sonographer or between different sonographers at varied points in the study. This would imply that there is reliability in the TCD measurements within and between sonographers. Furthermore, our results showed that this reliability in TCD measurements was only slightly greater for the intraobserver study. Thus, measurement of TCD may be useful in a clinical setting. It is important to note that two experienced sonographers carried out the determination of intraobserver and interobserver reliability of TCD measurements in our study. This may have affected our results in a positive way, as inexperienced sonographers could have a negative effect on the reproducibility of measurements. The use of sonographers who were both experienced and inexperienced might have made our results more realistic to clinical practice in this area of the world.
We had nonconsecutive enrollment of our participants due to 53 very active fetuses who were difficult to scan due to their activity level. Removing these fetuses due to inability to obtain the measurement of their TCD resulted in an attrition rate of 7.06%. This bias may have preselected an optimal scanning population, which could have resulted in a more favorable bias in these results. Thus, this will need to be a consideration for future studies that use a consecutive enrollment of participants. In addition, researchers in further studies would be well advised to instruct the pregnant women to eat less to reduce fetal activity. The hope would be that moderate fetal activity would increase the sample size and improve the accuracy of the TCD obtained. All pregnant women enrolled in this study were an uncontaminated sample of Nigerian women. In this study, the influence of fetal gender on the TCD measurements was not considered.
The maximum TCD obtained in this study was 45.5 mm, which is lower than the maximum TCD of 48.4 mm obtained by Goel et al. 4 in an Indian population. This difference may be due to racial variations. The mean (standard deviation [SD]) TCD value from 14 to 26 weeks was 13.6 (2.2) mm to 27.3 (2.6) mm and the mean (SD) TCD value from 27 to 40 weeks was 28.9 (2.0) mm to 42.9 (2.0) mm. These values show that TCD increases linearly with an increase in GA and at a faster rate in the third trimester in this population. Our results show that TCD has a strong positive correlation with GA in this sample of patients. This is similar to the findings of a previous report 17 in a Caucasian population. This could imply that TCD measurements have potential as a reliable predictor of GA and fetal growth in late pregnancy.
There was no statistically significant difference found between the mean TCD values obtained in this study (28.07 mm) and the values reported by Saifon et al. 10 (28.5 mm) in Thailand and those of Joshi 11 (31.42 mm) in a Nepalese population (P > .05). A possible explanation is that there may be no racial difference in the mean TCD. However, reliance on the mean value to make this suggestion may be misleading as it may not explain the spread of data points across each GA in both trimesters. Moreover, the sample sizes are not comparable between the studies. Thus, population-specific charts for TCD measurements may still be essential.
The following linear model equations produced by the regression analysis show the relationship between GA (Y) in weeks and TCD in millimeters: Y = 0.63 (TCD) + 7.16, Y = 0.54 (TCD) + 14.32, and Y = 0.79 (TCD) + 4.91 in the second, third, and combined trimesters, respectively. With the above equations, GA can be obtained by substituting the measured TCD in these equations. The larger values of R 2 in the combined trimesters show that the model equations fit quite well. The regression sum of squares is 33034.79 and the residual sum of squares is 4917.26. This implies that the model has a larger regression sum of squares. It also implies that in this large sample, TCD could be used as a good estimator of GA in the second and third trimesters.
A model equation showing the relationship of the four common fetal biometric parameters with TCD is TCD = 3.917 + 0.316FL + 0.088BPD – 0.008HC + 0.017AC. Thus, there is a significant linear relationship between TCD and FL, HC, BPD, and AC used in determination of fetal GA. Similarly, a previous study 18 concluded that there was a close relationship between TCD and GA (r = .94, P < .001). Naseem et al. 19 and Singhakom et al. 2 also found that TCD had a positive correlation with other fetal parameters in their studies, and Malik et al. 20 concluded that TCD gives an accurate estimate of GA in the third trimester. The r values of .957 (TCD and FL mm), .941 (TCD and BPD mm), .940 (TCD and HC mm), .949 (TCD and AC mm), and .933 (TCD and LMP) were obtained, respectively, from the Pearson correlation coefficient analysis. The value of TCD obtained from patients’ LMP is relatively lower than the rest, although it is not statistically significant (P > .05), probably because some women do not routinely document or remember correctly their LMP. This result is similar to the findings of Kalish and Chervenak. 1 The standard error of the TCD value (0.4) is lower than the standard error (0.5) of other fetal parameters (FL, BPD, AC, and HC) in the second and third trimesters. The size of the cerebellum is less affected by deviations in fetal growth restriction or growth acceleration. The cerebellum is resistant to chronic hypoxemia due to the brain sparing phenomenon, and in the fetus, cerebellar growth may be least affected by IUGR. 10 Holanda-Filho et al. 17 suggested that TCD is easier to obtain for the GA of the fetus in certain circumstances such as breech presentation and dolichocephaly (except in anencephaly), where other fetal parameters cannot be used. There is a need for replication of this research as well as a focus on reference values of fetal TCD in other regions of the country for possible generation of a nationwide nomogram.
Study Limitations
This study was limited due to the pre-experimental research design and the use of a convenience sample. Oversampling also is associated with a violation of statistical power, although it is advantageous for consenting of clinical patients. It is also noted that the sonographic measurement of the TCD in some very active fetuses was difficult and this further hampers the generalizability of the data. Determination of the intraoperator and interoperator reliability of TCD measurements was done in this study using only two experienced sonographers.
Assessment of TCD in this study was carried out on normal fetuses only; therefore, the effect of IUGR or fetal anomaly on TCD could not be assessed. Thus, the reliability of these reference values in a variety of cases may not be possible. A 3D ultrasound machine was not used for this study and may have facilitated measurements and results with 12 to 14 weeks of gestation. Transvaginal sonography could have decreased the limits of active fetuses; however, it was not used because its field of view decreases as pregnancy advances (second and third trimesters). It may be difficult to obtain the TCD when the fetal head is engaged in the maternal pelvis. This challenge can be overcome by instructing the patient to turn to the left or right side during the examination.
Conclusion
Transverse cerebellar diameter has a linear relationship with gestational age in this clinical sample. A significant relationship was found between transverse cerebellar diameter, last menstrual period, and other fetal biometric parameters like femur length, biparietal diameter, head circumference, and abdominal circumference. Reference values for fetal TCD were provided, given the limitations of this study. Sonographic assessment of TCD in other comorbidities among pregnant women is suggested to validate these reference values. It is important to note that this is one study performed in only one institution in southeast Nigeria. Therefore, further studies including multicenter studies in additional regions of the country are suggested to generate a reliable nationwide nomogram.
Footnotes
Acknowledgements
The authors give many thanks to Dr. Kalu Ochie for his useful critiques.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.