Abstract
Endometrial ablation procedures are growing in popularity for the treatment of menorrhagia and dysmenorrhea. Sonography is the modality of choice in the evaluation of patients prior to ablation and as a follow-up for any postprocedure complications. After ablation, conditions including hematometra, postablation tubal sterilization syndrome, postablation endometriosis, and pregnancy complications have been documented. Sonographers should be aware of the conditions associated with endometrial ablations and the variety of sonographic findings that may be present.
Introduction
Endometrial ablations are widely used to treat menorrhagia instead of the more costly and invasive hysterectomy.1,2 Radiofrequency ablation is a second-generation technique that uses radiofrequency waves to destroy endometrial tissue, resulting in a significant decrease in menstrual bleeding or, in some cases, amenorrhea.1,3,4 According to the American College of Radiology Appropriateness Criteria, sonography is the imaging modality of choice for abnormal vaginal bleeding in the evaluation of patients prior to ablation procedures, as well as for follow-up of potential problems postprocedure. 5 Endometrial ablations have been associated with long-term complications such as hematometra, postablation tubal sterilization syndrome (PATSS), postablation endometriosis, and pregnancy complications. 2 Sonographers should be aware of these consequences and the associated sonographic findings.
Case No. 1
A 37-year-old woman, gravida 5, para 2-0-3-2, with a primary complaint of increasing right-side pain was evaluated sonographically. The patient indicated that the symptoms had begun over a 3- to 4-month period and were cyclical in nature. In addition to pain, the patient complained of nausea and near syncope during these episodes. The patient’s obstetric/gynecologic surgical/procedure history included two cesarean sections, two dilation and curettages (D & Cs), and methotrexate for a right tubal ectopic pregnancy. A permanent tubal sterilization procedure using small coils inserted into the fallopian tubes (Essure®; Bayer, Whippany, NJ) was performed 3 years prior, and a radiofrequency endometrial ablation (NovaSure®; Hologic, Marlborough, MA) was completed 7 months prior in an attempt to treat menorrhagia. A transvaginal sonogram was performed using a Samsung UGEO WS80A system (Samsung Neur-Logica, Danvers, MA) with a transvaginal V5-9 probe. Imaging revealed two cystic areas within the uterine fundus. The area on the right contained low-level echoes and measured 1.2 cm × 0.99 cm (Figure 1). The area on the left was more anechoic in appearance and measured 0.82 cm × 0.61 cm (Figures 2 and 3). Three-dimensional imaging also demonstrated intrauterine cystic structures (Figure 4). These areas were believed to represent hematometra. Both Essure coils were visualized and appeared to be in communication with the respective cystic areas. A total hysterectomy with salpingectomy procedure was recommended and performed laparoscopically. The pathology report documented a 1.9-cm-diameter cystic structure filled with red to brown viscous fluid in the right superior portion of the uterus, uterine serosal adhesions, and small intramural focus, suggesting adenomyosis. After surgery, the patient reported complete resolution of symptoms.
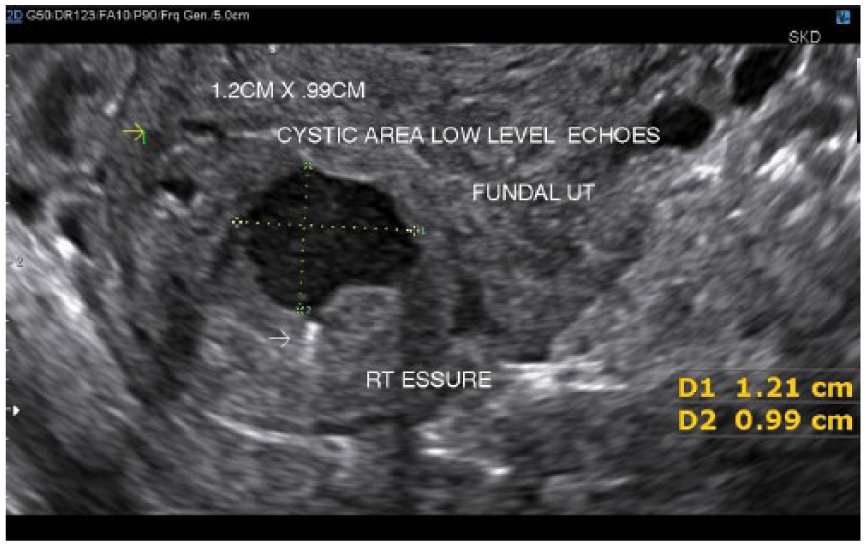
Transvaginal transverse gray-scale image of the uterus demonstrating the 1.2 cm × 0.99 cm cystic area with low-level internal echoes. The right sterilization coil can be seen (white arrow) in contact with the cystic area.
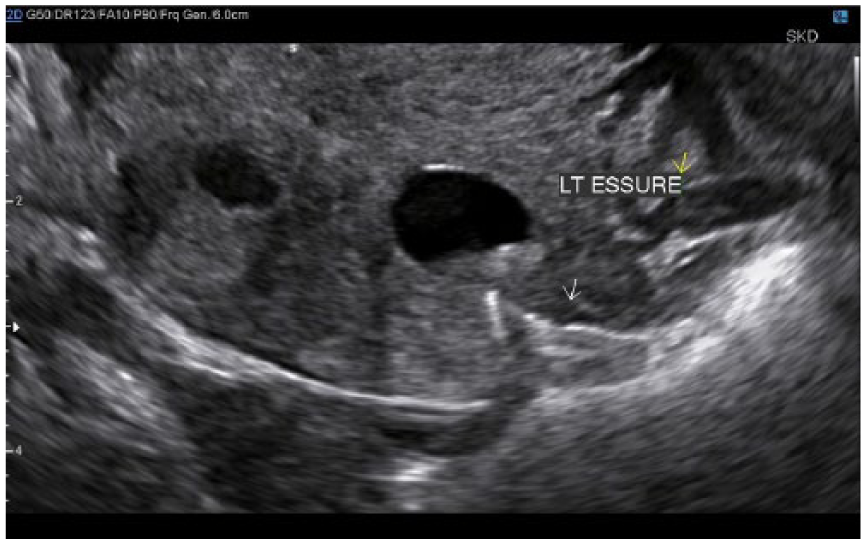
Transvaginal transverse gray-scale image of the uterus showing the cystic area on the left side of the fundus. The left sterilization coil can be visualized (white arrow).
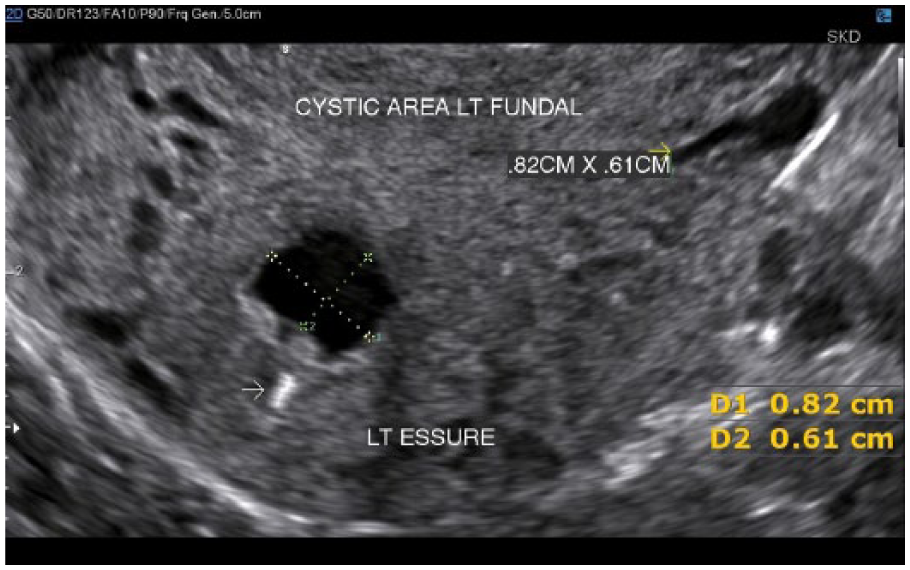
Transvaginal transverse gray-scale image of the uterus showing the 0.82 cm × 0.61 cm cystic area on the left side of the fundus. The left sterilization coil can be visualized (white arrow).
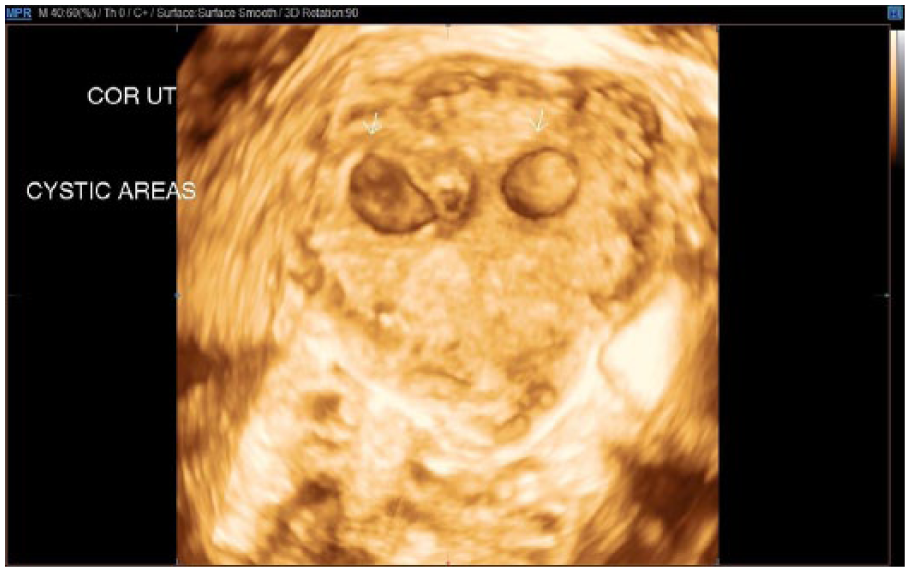
Transvaginal coronal 3D image of the uterus demonstrating both cystic areas (white arrows) in the fundal region.
Case No. 2
A 39-year-old woman, gravida 2, para 2-0-0-2, presented with complaints of increasing abdominal pain and abnormal bleeding over a 2- to 3-month period. Obstetric/gynecologic surgical/procedure history included a left-side ruptured ovarian cyst with referred right-side pain, two natural childbirths, a tubal ligation, and an endometrial ablation to treat menorrhagia and dysmenorrhea. Magnetic resonance imaging of the pelvis was performed using a 1.5 Tesla GE scanner. Coronal T1-weighted and sagittal T2-weighted images were obtained, demonstrating a retroverted uterus with fluid within the endometrium and free fluid posterior to the uterus (Figure 5). A comparison of sagittal T1-weighted and T2-weighted images showed bright fluid within the endometrium consistent in appearance with that of chronic blood (Figures 6 and 7). 6
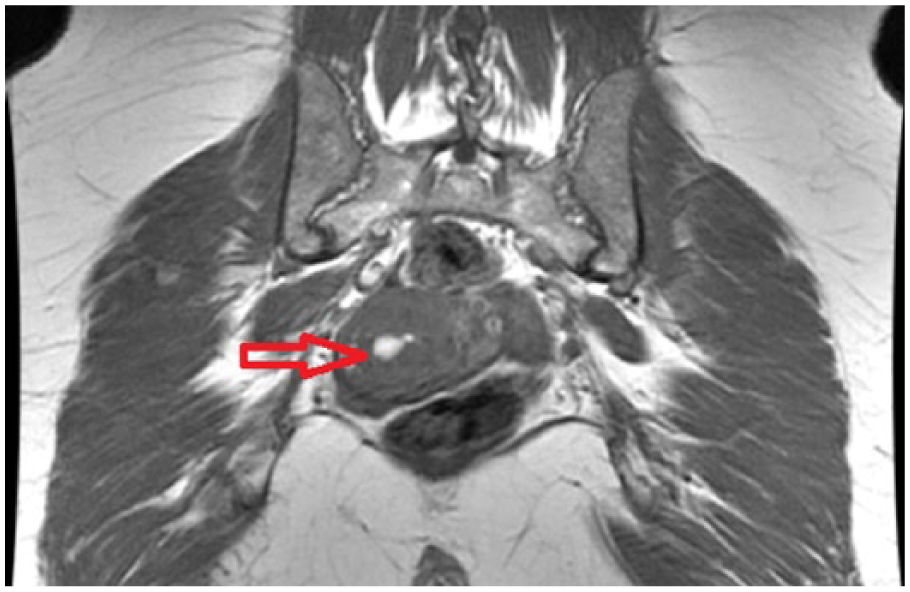
Coronal T-1 weighted image of the pelvis. Bright fluid is seen within the uterus consistent with blood (red arrow).
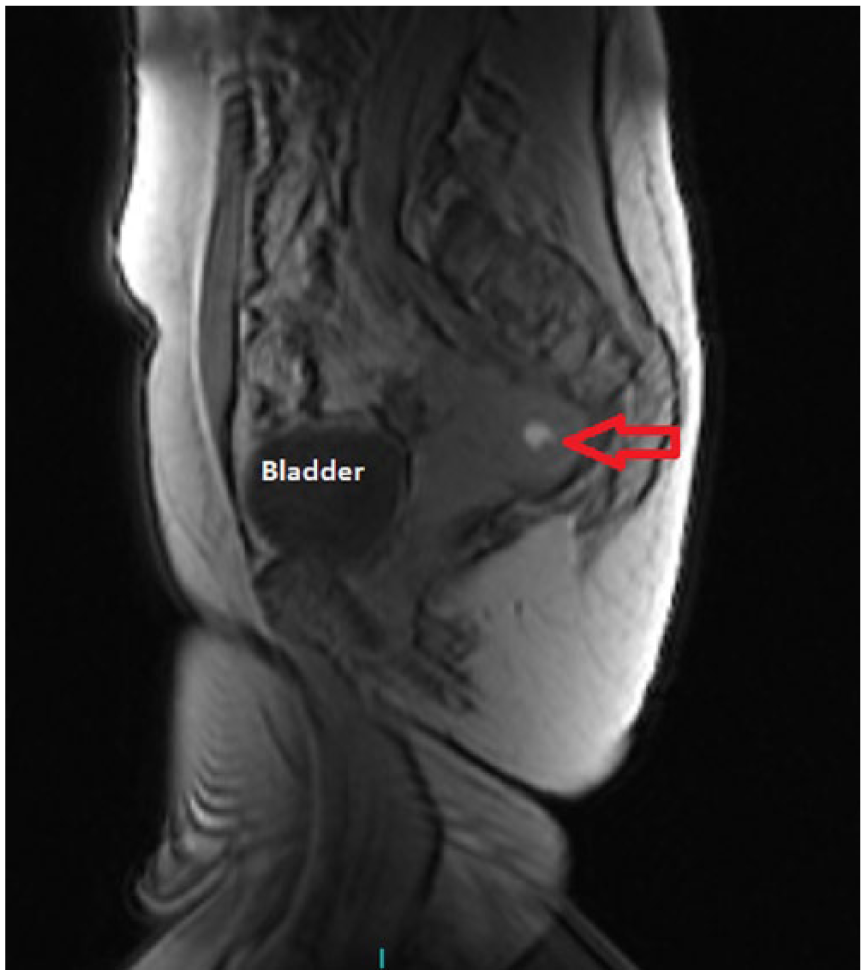
Sagittal T-1 weighted localizer image of the pelvis showing a retroverted uterus and bright fluid within the endometrium consistent with blood (red arrow).
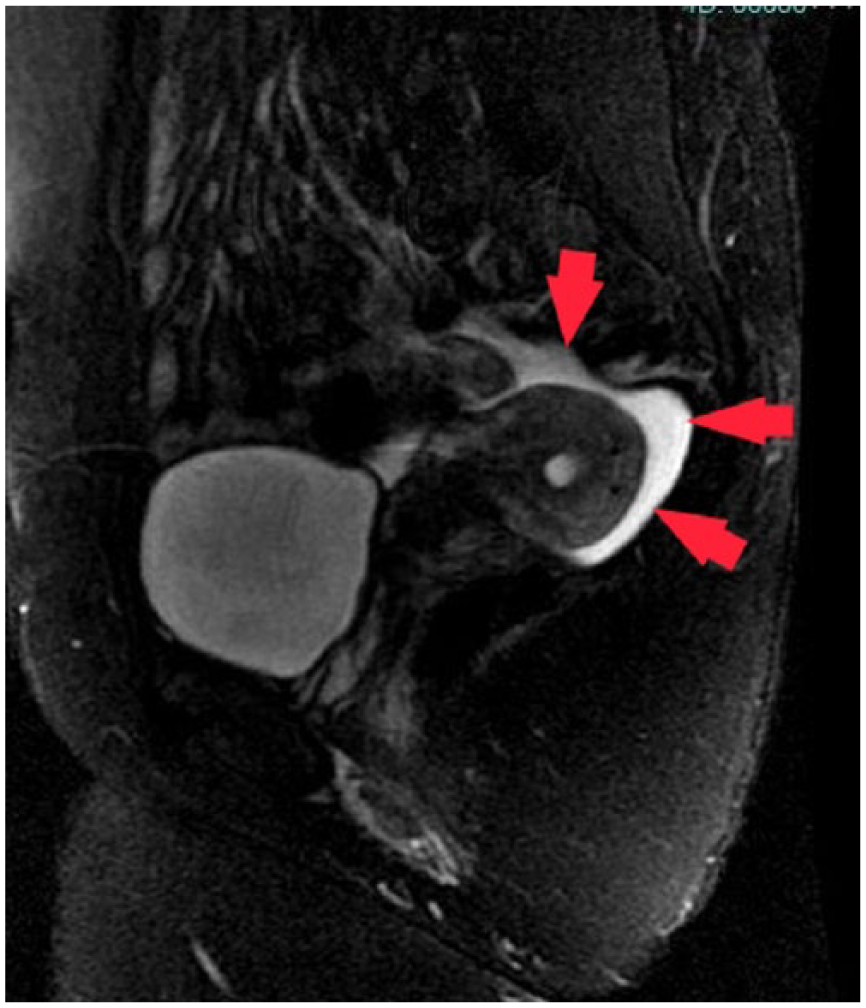
Sagittal T-2 weighted image with fat saturation demonstrating retroverted uterus and bright fluid within the endometrium and surrounding pelvis consistent with blood (red arrows).
A transvaginal sonogram was performed using a GE Voluson 8 system (GE Healthcare Ultrasound, Waukesha, WI) with a transvaginal RIC5-9 probe. The retroverted uterus (Figure 8) was seen with endometrial tissues measuring 1.38 cm (Figure 9). Fluid and debris were seen located within the endometrial cavity consistent with central hematometra (Figure 10). Also, free fluid was noted within the pelvis (Figure 8). A laparoscopically assisted vaginal hysterectomy with left salpingo-oophorectomy was performed. Surgical notes indicated that one area within the pelvis was suspicious for endometriosis. This finding would explain the free fluid in the pelvis seen by sonogram and the bright fluid corresponding to blood on the magnetic resonance images. As with the previous case study, the patient reported complete resolution of symptoms following surgery.
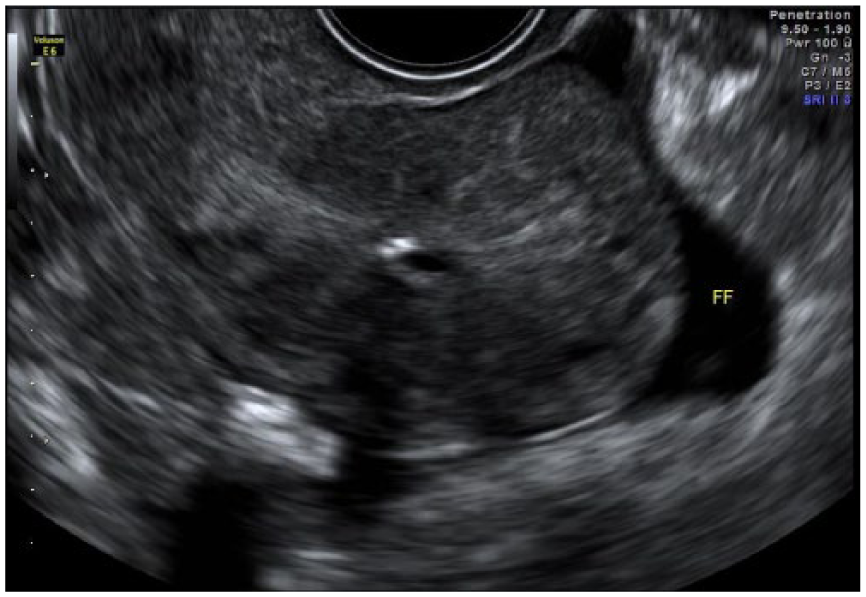
Transvaginal sagittal gray-scale image showing a retroverted uterus. Free fluid (FF) is seen in the pelvis.
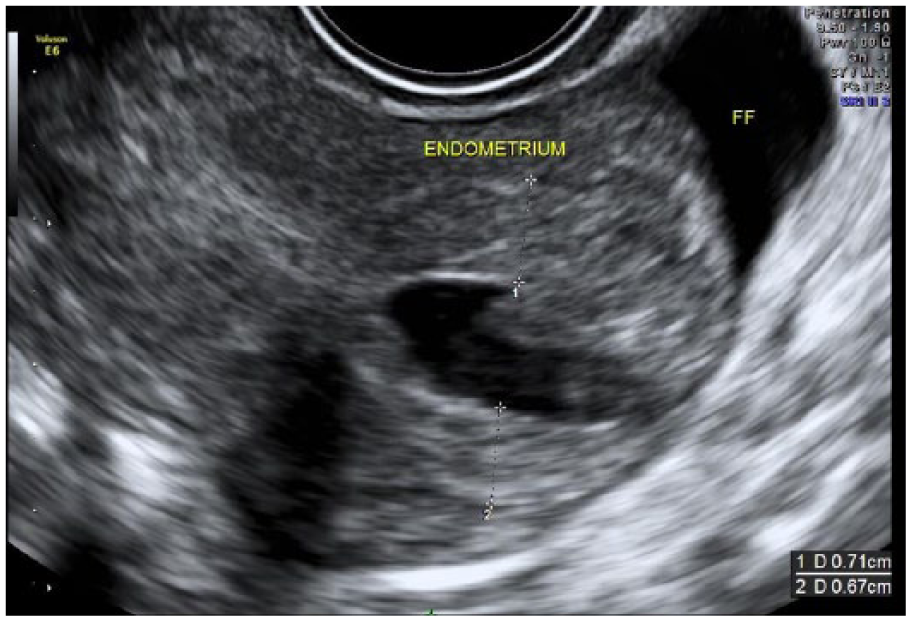
Transvaginal sagittal gray-scale image of the uterus demonstrating an endometrial stripe of 1.38 cm.
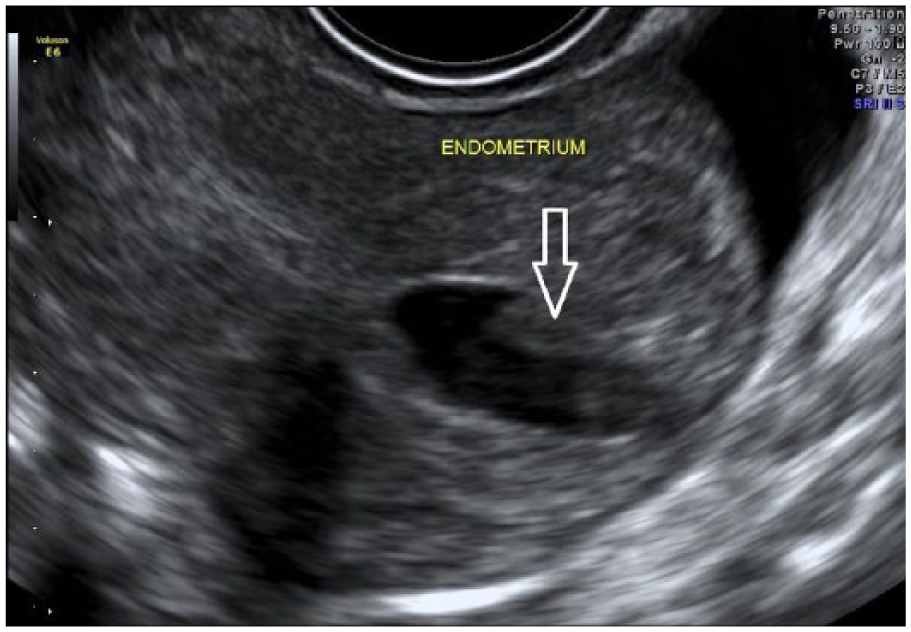
Transvaginal sagittal gray-scale image of the uterus demonstrating fluid and debris within the endometrium (white open arrow).
Discussion
Endometrial ablation techniques have been used since the late 1980s to relieve symptoms of menorrhagia or heavy menstrual bleeding. 3 First-generation procedures were referred to as resectoscopic or hysteroscopic and required the physician performing the procedure to have significant skill with hysteroscopic procedures. These included procedures such as laser fiber, loop, and rollerball ablation techniques.2,7
Second-generation procedures, global endometrial ablation (GEA) techniques, eliminated the need for hysteroscopy and began to be used in the 1990s.1,8 These procedures were viewed as easier for the physician to perform. Global endometrial ablations include procedures such as microwave, cryotherapy, hydrothermal, thermal balloon, and radiofrequency ablations.2,7 The ultimate goal for all endometrial ablation techniques is to remove the endometrial tissues, thereby eliminating or significantly reducing menstrual bleeding.
Both patients presented in these case studies were treated with the radiofrequency ablation technique. Radiofrequency ablation, a second-generation GEA, has been in use for more than 14 years and has treated approximately 2 million women. Although hysterography is not a required part of the procedure, it is often performed to assess for intrauterine pathology and to measure the cavity length. 1 Carbon dioxide (CO2) is injected to pressurize the uterus and rule out uterine wall perforation prior to treatment. 8 This technology relies on bipolar radiofrequencies for charring of the endometrial tissue, resulting in decreased menstrual bleeding or amenorrhea. 9 During the procedure, a triangular mesh is inserted through the cervix into the uterus. The device expands to fit the contours of the endometrial cavity. One of the unique features of radiofrequency ablation is its compatibility with endometrial cavities that are irregular in shape. 9 Once the device is in place, radiofrequency energy ablates the endometrial tissue. Typical ablation time is 90 seconds with the maximum treatment time being 120 seconds.8,9 It is convenient that the procedure is not affected by the specific stage of the menstrual cycle at the time of ablation. 1
Positive Outcomes of Ablation Procedures
Overall, GEAs have a good record of patient satisfaction. In a study by Kalkat and Cartmill, 1 94% of patients experienced improvements in their menstrual symptoms 6 months after the radiofrequency ablation procedure. Basinski et al. 10 reported an amenorrhea rate of 61%, whereas Claas et al. 8 reported amenorrhea in 51% of patients treated with radiofrequency ablation (Table 1).
Positive Outcomes of Ablation Procedures.

Complications Following Ablation Procedures
Global endometrial ablations have relatively few short-term complications, but long-term complications often lead to further intervention techniques. 2 Complications associated with endometrial ablation include hematometra, PATSS, postablation endometriosis, and postablation pregnancy complications (Table 2).
Complications Following Endometrial Ablation.

Hematometra is a collection of blood within the uterus that becomes trapped. Hematometra may present as cornual, as seen in Case 1, or central, as seen in Case 2. After an ablation procedure, adhesions or synechiae can form during healing. These adhesions will not create an issue unless endometrium either begins to regenerate or is not completely ablated during the procedure. 2 Cornual hematometra will present with pain located more on one side or the other as was seen in Case 1. In contrast, patients may present with generalized abdominal pain in cases of central hematometra as seen in Case 2. 2
Postablation tubal sterilization syndrome may occur in patients who have previously undergone a tubal sterilization procedure. Both patients in the study had a history of tubal sterilization procedures; however, they did not suffer from PATSS. This condition is not common and is seen more often with the first-generation ablation techniques and very rarely with GEAs. 7 Patients with PATSS present with severe unilateral or bilateral pain. Sonographic imaging on patients with PATSS may demonstrate hydrosalpinges and may be seen in combination with hematometra. 7
Postablation endometriosis can occur in patients without a history of tubal sterilization procedures who are experiencing retrograde bleeding into the pelvis as a result of uterine adhesions. 2 The adhesions do not allow shedding of endometrial tissue to pass out of the uterus. As a result, the tissues and fluids are pushed retrogradely through the open fallopian tubes into the pelvis where they can deposit and continue to grow and shed, resulting in pain. It is important to note that some patients undergoing endometrial ablation may also have endometriosis before the procedure and a definitive diagnose of endometriosis as a result of ablation may be difficult.
If pregnancy should occur postendometrial ablation, there are significant risks involved, including pregnancy loss, ectopic pregnancy, placental attachment issues, uterine rupture, and preterm delivery. 2 Patients undergoing ablation techniques should be aware that these procedures are not a form of birth control and should not be performed if the patient may consider a future pregnancy. 9 Contraception options need to be discussed, including permanent sterilization of the patient or her partner.
It is important to note that these conditions (hematometra, PATSS, and postablation endometriosis) may be difficult to diagnosis if imaging studies such as sonograms and magnetic resonance imaging are not performed while the patient is symptomatic. The body may reabsorb blood and fluid when not in the menstrual phase, thereby making these conditions difficult to detect with imaging. 2
Treatment Options After GEAs
Women who continue to experience problems following endometrial ablation may need further intervention. The most common patient complaints leading to reintervention are pain, bleeding, or a combination of pain and bleeding.2,11 Several studies have attempted to link predisposing risks to ablation failures. A younger age at the time of ablation seems to point to a greater chance of failure.11,12 El-Nashar et al. 12 found patients younger than 45 to be at a greater risk of procedure failure. This study also indicated that prior tubal ligation or history of dysmenorrhea prior to ablation leads to a greater chance of a poor outcome postablation. Increased reintervention in patients who had previously undergone a cesarean section was noted by Shavell et al. 11 (Table 3).
Increased Risk of Postablation Complications.

If further treatment is needed, options available would include undergoing another endometrial ablation technique or hysterectomy. Bansi-Matharu et al 3 studied 114 910 women following endometrial ablation and found that 3 times as many women undergo a hysterectomy as opposed to a second ablation procedure. In another study of 1169 women by Shavell et al., 11 13.4% reported undergoing a hysterectomy following an ablation procedure. Of the women undergoing hysterectomy, 60% occurred in the first 2 years postendometrial ablation, with 80% occurring within 3 years postprocedure. 11
Conclusion
Sonographers should be aware of a possible link between ablation and associated complications with resulting hysterectomy. For unknown reasons, the patients presented within this article were not notified of the risks specifically associated with endometrial ablations prior to the procedures being performed. An analysis of previous research and case studies should be conducted and consideration given to the inclusion of hysterectomy on the list of risks to the ablation procedure.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
