Abstract
Congenital coronary artery fistula (CAF) is a rare anomaly of the heart presenting in 0.002% of the general population. This case study presents an adult patient with an undiagnosed heart murmur and symptoms of chest pain, fatigue, and lightheadedness that demonstrated an incidental finding of CAF diagnosed by transthoracic echocardiography. Transthoracic echocardiography revealed an aneurysmal, tortuous right coronary artery terminating in a dilated coronary sinus. Sonographic findings were compared with angiography and computed tomography. This study highlights the potential of transthoracic echocardiography to assess heart structure and physiology and detect a CAF.
Keywords
Congenital coronary artery fistula (CAF) is a rare cardiac malformation in which a coronary artery drains into a chamber or vessel having bypassed the myocardial capillary bed.1,2 Coronary artery fistula presents in 0.002% of the general population and accounts for 13% of all congenital coronary anomalies.3,4 The diagnosis of CAF is most commonly identified by angiography or computed tomography (CT); however, this case study presents the incidental finding of a large, uncomplicated congenital right coronary artery (RCA) to coronary sinus fistula in an adult diagnosed by transthoracic echocardiography.
Case Report
A 42-year-old male presented with an undiagnosed murmur and symptoms of chest pain, fatigue, and lightheadedness. Physical examination revealed a low-pitched blowing plateau holosystolic murmur of grade 3/6 at the upper right sternal border, and medium-pitched harsh crescendo early systolic murmur of grade 2/6 at the upper left sternal border. The patient’s vital signs were stable, and electrocardiogram, chest radiograph, and laboratory findings were unremarkable.
The patient reported having a transthoracic echocardiogram (TTE) and exercise stress echocardiogram (ESE) performed by an external provider 4 years prior. The TTE reported presence of trace mitral valve regurgitation but no significant abnormalities. The ESE reported achievement of 99% of the predicted target heart rate, no evidence of stress-induced wall motion abnormalities, normal sinus rhythm with nonspecific ST-T wave changes, and hypertensive response to exercise. The patient’s overall medical history was unremarkable.
The second TTE was performed using a General Electric Vivid q ultrasound system with a M4S phased array transducer. Sonography revealed enlargement of the right ventricle and atrium; dilated proximal coronary sinus measuring 1.95 cm (Figure 1A) with prominent, disturbed flow entering into the right atrium (Figure 1B); and an anomalous, large vessel-like structure alongside and circumscribing the heart in the atrioventricular groove (Figures 2A–2D). The origins of the coronary arteries were well visualized and appeared unremarkable. The unusual presentation of right-sided enlargement, an anomalous vessel, turbulent flow from a dilated coronary sinus, and a cardiac murmur suggested the presence of a fistulous connection.5,6 Accordingly, a Qp:Qs ratio was performed to determine the presence of a shunt. The ratio was 2.4:1, which indicated existence of a large left-to-right heart shunt. Differential diagnoses were coronary sinus aneurysm or dissection, partial anomalous pulmonary venous return, persistent left superior vena cava, and sinus venosus defect. The echocardiogram also revealed normal left ventricular size and function with an ejection fraction of 68%; moderate left atrial enlargement; trace mitral, pulmonic, and tricuspid regurgitation; and a right ventricular systolic pressure of 38 mmHg. Compared to the previous TTE, there were multiple different findings. Since the previous reported no evidence of chamber enlargement or coronary sinus dilation, the new findings were suspected to be due to progressive severity of the shunt. For further evaluation, the cardiologist referred the patient for cardiac catheterization and coronary CT angiography.
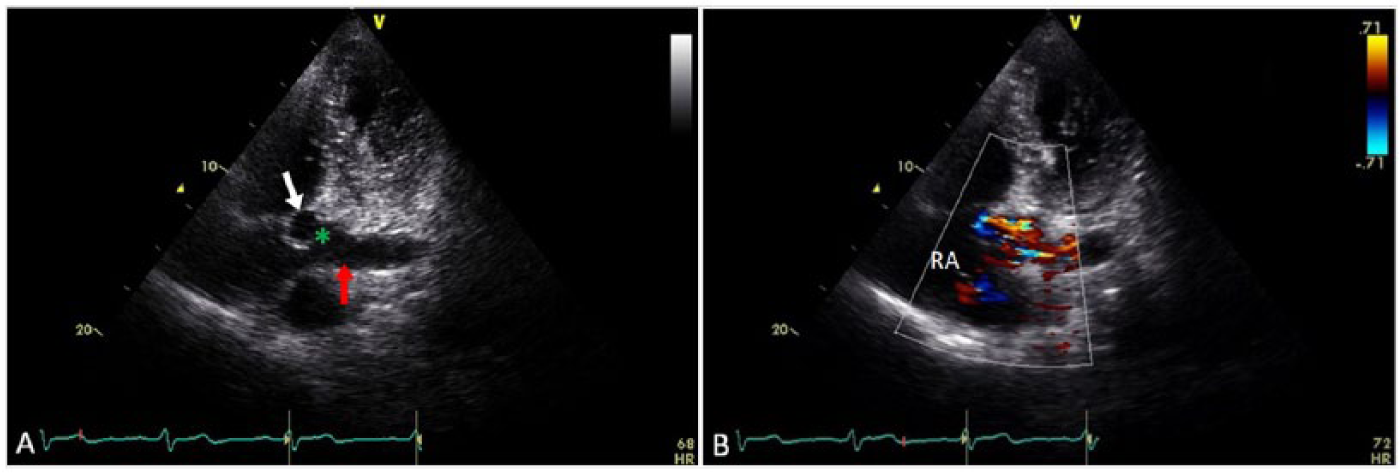
Echocardiogram modified apical four-chamber views of the right coronary artery (RCA) to coronary sinus fistula. (A) The distal RCA (white arrow) terminates (green asterisk) into the proximal coronary sinus (red arrow) adjacent to the atrial junction. (B) Turbulent flow from the fistula is shown entering the coronary sinus and right atrium (RA).
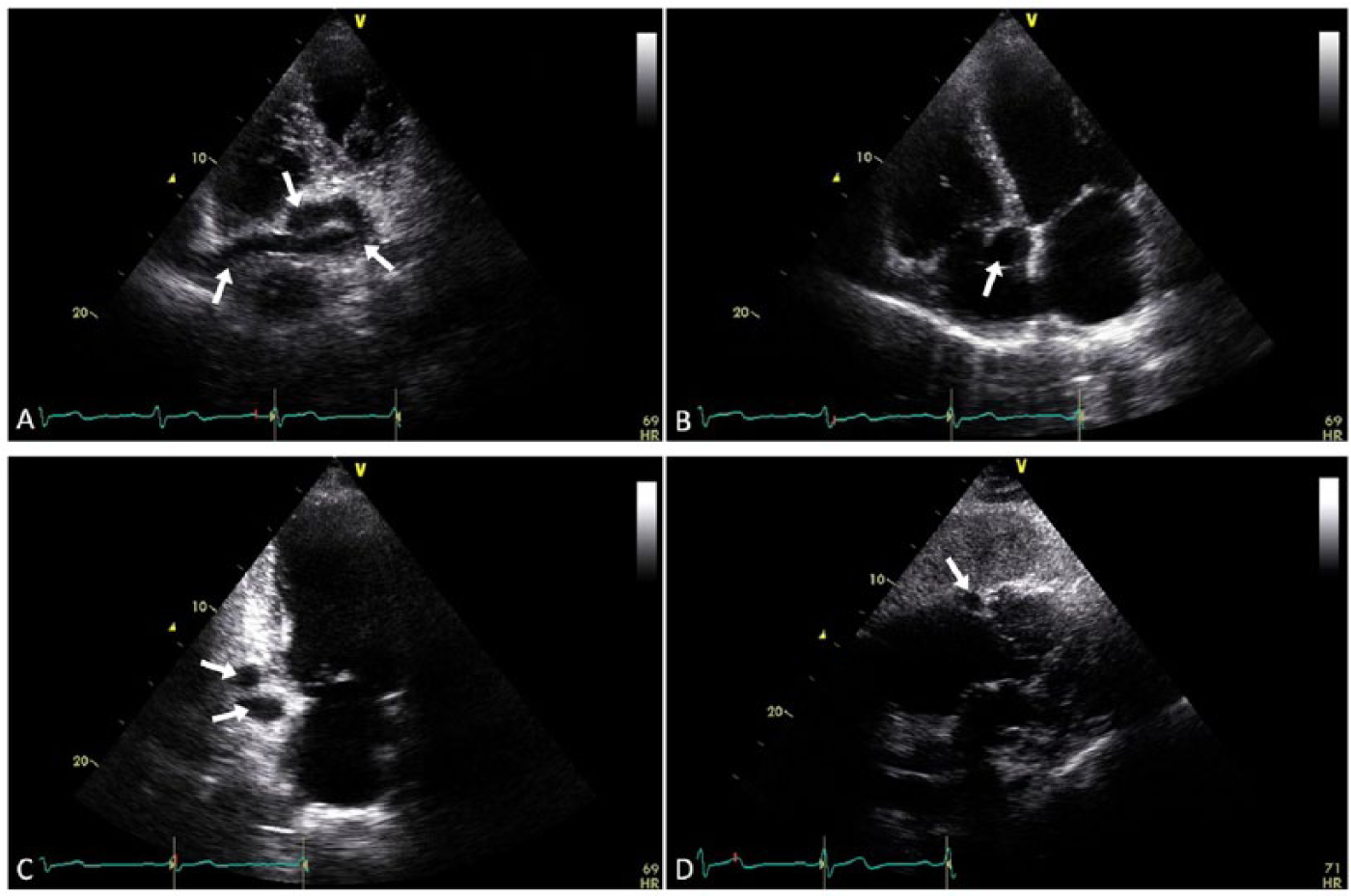
Echocardiography of the right coronary artery (RCA). (A) Modified apical four-chamber view demonstrates the tortuous RCA (arrows) in longitudinal plane circumscribing the right heart. (B) Apical four-chamber view demonstrates the distal RCA (arrow) diameter and lumen patency in transverse plane. (C) Apical two-chamber view demonstrates dilation and tortuosity of the RCA (arrows). (D) Subcostal five-chamber view demonstrates the dilated RCA (arrow) within the atrioventricular groove.
Cardiac catheterization with angiography revealed an aneurysmal RCA measuring 10+ mm in diameter (normal is 3 mm) with clear drainage into the proximal portion of the coronary sinus as it entered the right atrium. The angiogram revealed no evidence of significant coronary artery disease or anomalous coronary drainage.
Coronary CT angiography demonstrated normal location and diameter of the RCA origin with dilation beginning at the proximal segment (Figure 3A). The mid to distal segments of the RCA were aneurysmal and tortuous and seen circumscribing the right heart within the atrioventricular groove (Figure 3B). The RCA was seen terminating into the proximal coronary sinus with the fistulous connection measuring 14 mm from the right atrial junction (Figure 3C). Three-dimensional volume rendering revealed a single, large, and uncomplicated serpentine RCA around the heart. Although the diameter of the RCA origin was unremarkable, enlargement of the RCA was present throughout the proximal to distal segments with multiple S-shaped tortuosities (Figure 4A). The fistulous connection to the coronary sinus was also identified (Figure 4B). The RCA was seen circumscribing the right heart in the atrioventricular groove and draining into an enlarged coronary sinus (Figures 4C and 4D). All other coronary arteries appeared unremarkable in size. Findings from the catheterization and CT angiography confirmed TTE findings.
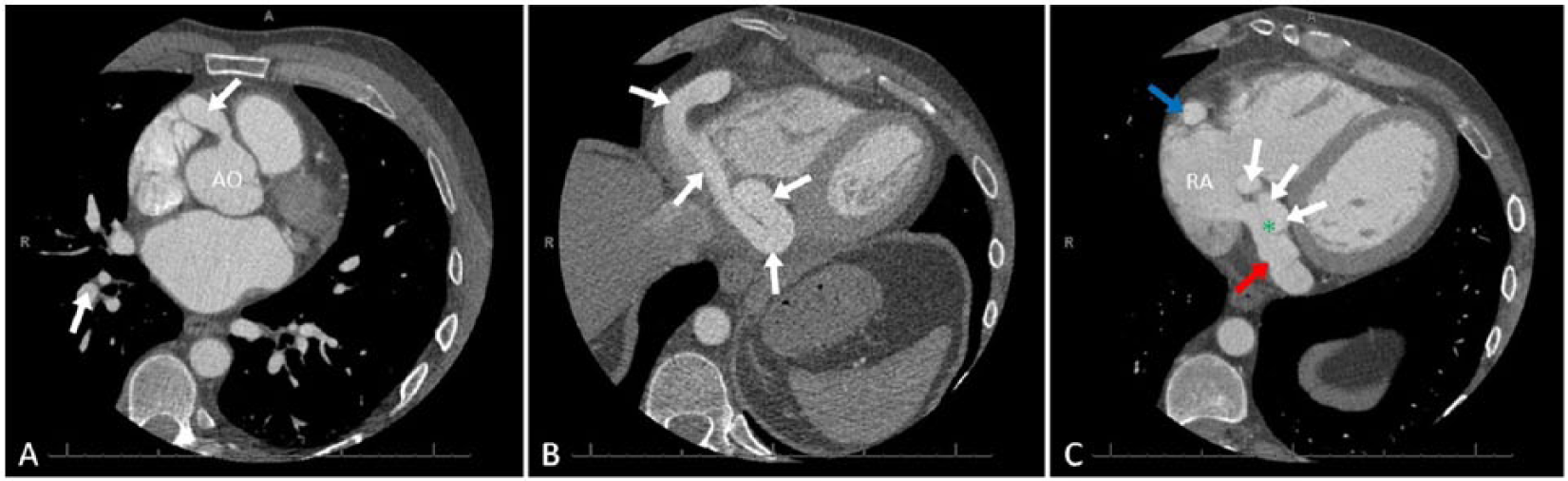
Multi-slice 2D computed tomography of the right coronary artery (RCA) and coronary sinus. (A) The proximal RCA (white arrow) dilates just after the origin in the axial view of the aortic sinus (AO). (B) The mid to distal segments of the RCA (white arrows) are shown as aneurysmal and tortuous and circumscribe the right heart in the axial view of the atrioventricular groove. (C) The distal RCA (white arrows) terminates (green asterisk) into the dilated coronary sinus (red arrow) adjacent to the atrial junction in the axial view of the coronary sinus. The RCA is also seen in transverse plane within the atrioventricular groove (blue arrow). RA, right atrium.
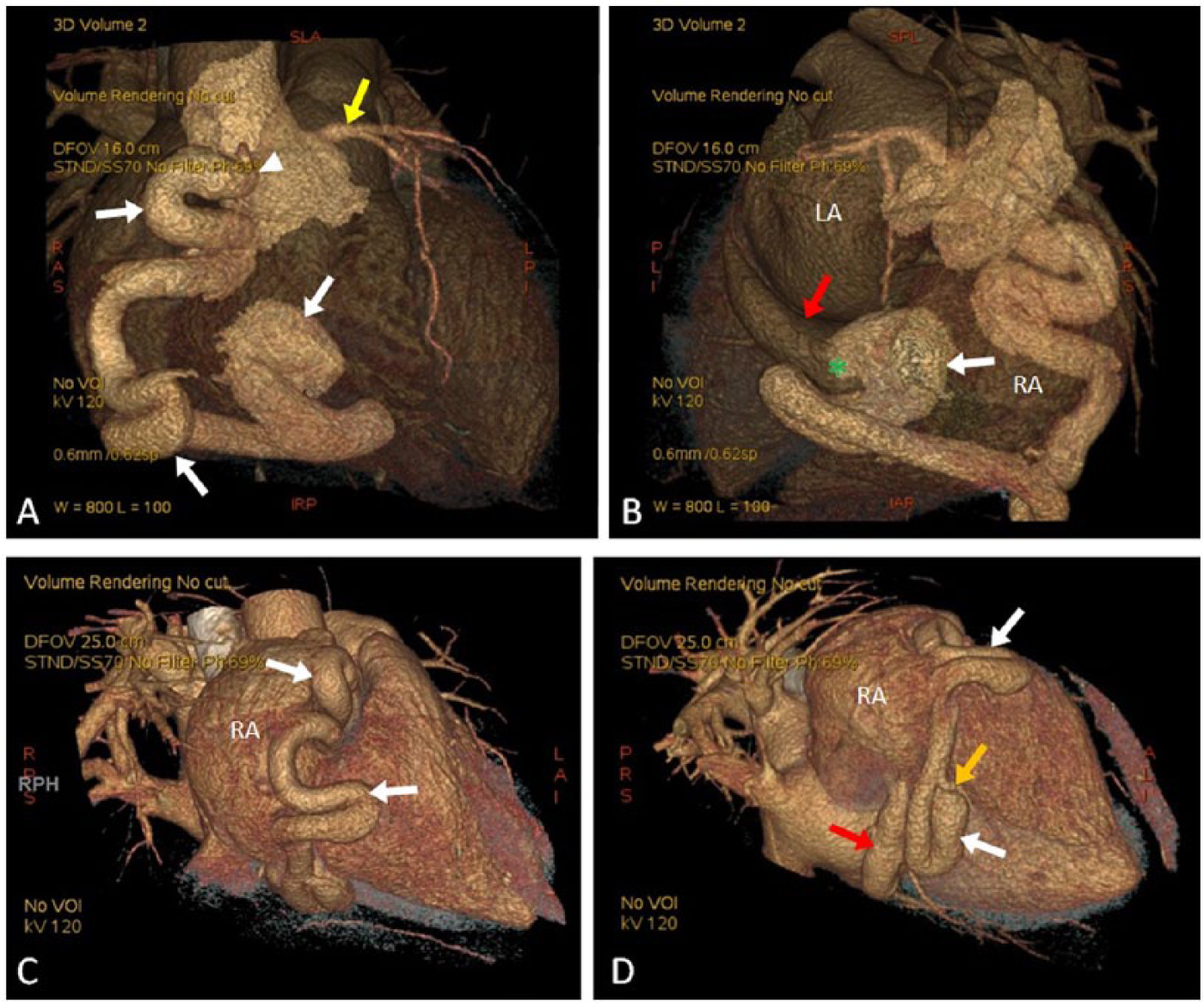
Volume rendered 3D computed tomography of the right coronary artery (RCA). (A) The anterior view demonstrates the RCA origin (white arrowhead) and S-shaped tortuosities (white arrows) of the vessel. An unremarkable left coronary artery (yellow arrow) is seen. (B) The posterior view demonstrates the RCA (white arrow) and fistula connection (green asterisk) to the proximal coronary sinus (red arrow). (C and D) Oblique posterior views demonstrate the RCA (white arrows) circumscribing the right heart, a dilated coronary sinus (red arrow), and an unremarkable posterior descending artery (orange arrow). LA, left atrium; RA, right atrium.
Discussion
Congenital CAFs can go undetected in utero and at birth. 3 Normally, during embryologic development, two coronary arteries arise from the aortic root and taper progressively as they branch to supply the cardiac parenchyma. Coronary artery fistulas develop when there is a failure of normal development and persistence of primitive intratrabecular spaces, or sinusoidal connections, allowing a developing artery to communicate with a vein or chamber bypassing the myocardial capillary bed.1–2,7
There is a wide spectrum of CAF anatomic variants—isolated or multiple, vessel-to-vessel or vessel-to-chamber, straight or tortuous, and normal sized or aneurysmal. Isolated, or uncomplicated, anomalies present in 55% to 80% of cases with 74% to 90% having a single origin and 67% having a single termination.5,8–11 Coronary artery fistulas can involve the RCA (50%), left coronary artery (42%), or both coronary arteries (5%) with drainage into the right ventricle (40%), right atrium (26%), pulmonary artery (17%), coronary sinus (7%), left atrium (5%), left ventricle (3%), or superior vena cava (1%).2,5,9–10,12 More often, CAFs in adults are large (5–20 mm diameter) and tortuous.1,5,9–10 This case study presents a large, uncomplicated CAF arising from the most common vessel, the RCA, but terminating in one of the least common drainage sites, the coronary sinus.
Clinical presentation is dependent on the severity of the shunt and presence of other cardiac conditions. 13 The most common clinical presentation in a patient with a CAF is a continuous murmur, which is usually caused by turbulent flow through the fistula.2,13 Symptoms present in less than half of patients and are typically uncommon for patients younger than 20 years. Symptoms tend to appear later in life when a shunt increases in size and becomes severe. Eighty percent of patients older than 50 years will develop symptoms and complications, such as angina pectoris, fatigue, orthopnea, palpitations or dyspnea caused by arrhythmia, atherosclerotic disease, congestive heart failure, endocarditis, myocardial ischemia or infarction secondary to steal phenomena, pulmonary hypertension, or rupture or occlusion secondary to thrombus formation.2,13–15
The size of the shunt is determined by the differences in flow rerouted by the fistula and pressure between the coronary artery and chamber into which the fistula drains. A right-sided fistulous connection can lead to right heart overload, including the pulmonary vascular bed and left chambers. A left-sided connection will affect only the left atrium and ventricle bypassing the pulmonary bed.5–6 Therefore, shunts are generally large when the fistula drains into the right side.
In echocardiography, shunts are determined by the Qp:Qs ratio. Qp represents the volume of pulmonary blood flow and Qs is the systemic blood flow. In a left-to-right shunt, Qp will be increased due to the additional blood flow bypassing the systemic system. In the absence of a shunt, the ratio is 1, which indicates that equal volume of blood flow is being pumped in both the pulmonary and systemic systems. A ratio of 1.5 or less indicates a small shunt, 1.5 to 2.0 indicates a moderate shunt, and greater than 2.0 is a large shunt. 16
Ninety percent of CAF cases have a left-to-right shunt. 10 Although dilation is common, degree of dilation does not depend solely on shunt size, such as in the presence of concomitant disease. 5 Approximately 20% to 45% of cases will be associated with congenital heart lesions that may affect shunt severity, such as atrial septal defect, tetralogy of Fallot, patent ductus arteriosus, ventricular septal defect, and pulmonary atresia.5,8–10,13,17 Therefore, due to the diversity of variants, severities, and concomitant conditions, fistulas can have different presentations.
Coronary artery fistulas are seen in 0.2% of echocardiographic studies and 0.05% to 0.8% of angiographic studies.3–5,13,15,18,19 However, the true incidence is difficult to evaluate because most cases may be asymptomatic, small, or undetectable. Further imaging may include transesophageal echocardiography, angiography, magnetic resonance imaging (MRI), nuclear MRI, or contrast-enhanced CT. 11
Computed tomography angiography is the gold standard for diagnosing CAF and other coronary anomalies because it reliably identifies the origin, size, and anatomic features of the fistula and can be both diagnostic and therapeutic. 2 Diagnosis is usually determined by identification of the origin, course of the vessel, dilation, and site of drainage. However, TTE has proven to be a useful tool for detection. Transthoracic echocardiography has identified anomalous origins of coronary arteries, abnormal termination sites, vessel dilation, multiplicity, and abnormal flow into chambers, but it has low sensitivity and specificity for detecting distal CAF sites or coronary flow abnormalities.10,15,20
Diagnosis by TTE is achieved by visualization of an enlarged coronary artery and fistula drainage site by 2D echocardiography, and abnormal flow from the fistula by color flow mapping and spectral pulsed Doppler. Although there is no standard protocol for assessment, CAF should be suspected when an anomalous vessel-like structure is seen alongside or circumscribing the heart, chambers are enlarged, and a murmur is present.5–6 Multiple echocardiographic windows should be used to follow the course of the vessel in any standard or off-axis planes to determine size, patency, presence of branches, and location and number of fistulous connections. Extent of artery dilatation can be determined from its origin using the parasternal short axis view at the level of the aortic valve. Proximal to distal segments can be assessed using apical and subcostal windows. High-volume flow may be detected by color flow mapping at the origin, along the length of the vessel, or site of drainage, as well.
This case demonstrates the potential of TTE to diagnose and characterize CAF. Multiple TTE views were comparable to CT multi-slice 2D views (Table 1). Comparison revealed that the TTE accurately identified and described characteristics of the RCA, location and size of the shunt, and drainage into the coronary sinus. Thus, this case highlights the utility of TTE to contribute to a CAF diagnosis. However, there are limitations of echocardiography—position of the probe, cardiac motion, and curvilinear nature of the vessel—that may limit visualization. 6 In addition, abnormal flow may not be detected or specific to CAF. False-positive and false-negative CAF diagnoses have occurred previously. 21
Comparison of Transthoracic Echocardiogram and 2-Dimensional Computed Tomography Views Used to Identify and Characterize the Right Coronary Artery Fistula.
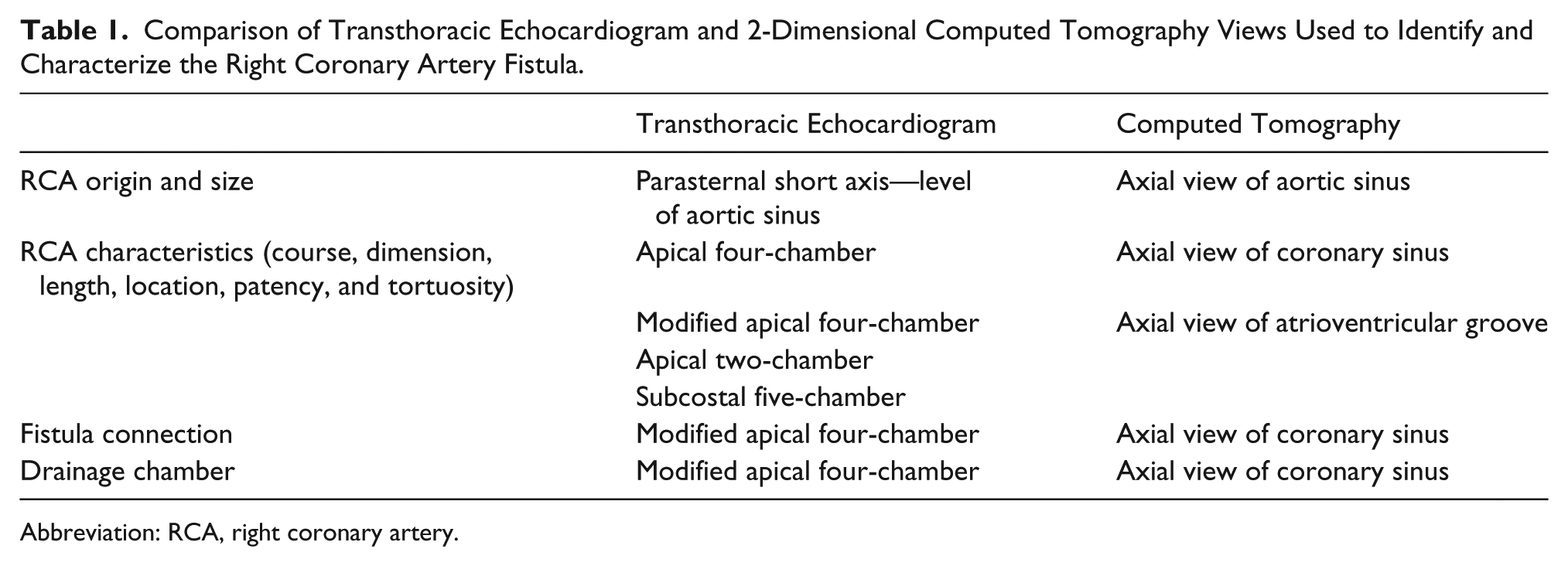
Abbreviation: RCA, right coronary artery.
Treatment for CAF is dependent on the anatomy and size of the fistula, presence of symptoms, and concomitant cardiovascular disease. Small, asymptomatic CAFs in adults do not need transcatheter or surgical intervention and are usually managed conservatively. They can remain benign or close spontaneously.2,13,22 Large, tortuous, complex, symptomatic CAFs are treated with transcatheter embolization or surgical ligation. 13 This method is safe and effective with a high closure rate and survival, but periprocedural myocardial infarction, fistula recurrence, and induced arrhythmia have been described. 23 Large, complex CAFs with multiple origins or terminations would not be suitable for a transcatheter approach and would require surgical correction. Symptomatic candidates eligible for surgical treatment are similar to those with left-to-right shunts; Qp:Qs is greater than 1.5 and presence of right ventricular volume overload.5,13 Although the patient in this case study was eligible for surgical intervention by both of these criteria, the patient opted not to proceed with elective surgical closure since he had an isolated CAF. The patient chose conservative management with the cardiologist.
Conclusion
Coronary artery fistula is a rare coronary artery malformation in which an artery drains into a chamber or vessel rather than the normal arterial circulation. Coronary artery fistula should be considered when the heart size is enlarged, or unusual vessel-like structure(s) and turbulent flow are seen by TTE. This report underscores the importance of careful delineation of heart structure and physiology by the sonographer during routine TTE. In addition, this case study demonstrates the cross-correlation and diagnosis of CAF by multi-modality imaging.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
