Abstract
Compared with the assessment of most fetal anatomic structures, the assessment of the fetal heart during routine obstetric screening represents a diagnostic challenge for sonographers and interpreting physicians. This is due to a number of factors: the structural complexity of the heart, the rate at which it moves, the position of the fetus relative to the transducer, and variations in assessment protocols used by sonographers and physicians. The fetal heart is a relatively small and complex structure, as seen in an early second-trimester sonogram. In some forms of congenital heart disease, the abnormality may be detectable within only a relatively small fraction of the heart volume. Congenital heart disease represents a range of structural defects, a number of which have specific sonographic features that can be identified during routine assessment. Many approaches have been proposed to improve the assessment of the fetal heart, including specific still images, color Doppler, 3D imaging techniques, and cineloops.
The normal fetal heartbeat ranges from 120 to 160 beats per minute; each beat represents a systolic and diastolic phase equivalent to an effective unidirectional movement of 240 to 320 cycles per second. 1 The size of the fetal heart in the early part of the second trimester is small, as depicted in Figure 1. Critical anatomic structures, such as the ventricular outflow tracts, are only 2 to 6 mm in diameter in the second trimester. 2 Furthermore, the impact of congenital heart disease (CHD) varies widely, depending on the specific form that it takes. In some cases, the chambers of the heart may be entirely normal in appearance, and the defect may be observed only in the outflow tracts of the heart. For example, CHD relating to the outflow tracts, such as dextrotransposition of the great arteries, may be detected only at their origins and are not frequently detected by the four-chamber view (see Figure 2). Since still images of the fetal heart represent only very thin slices, it is important that the fetal heart is assessed in real-time and interrogated completely in contiguous slices. The diversity and relative complexity of congenital heart defects demand considerable skill of the sonographer and interpreting physician to ensure a complete examination.
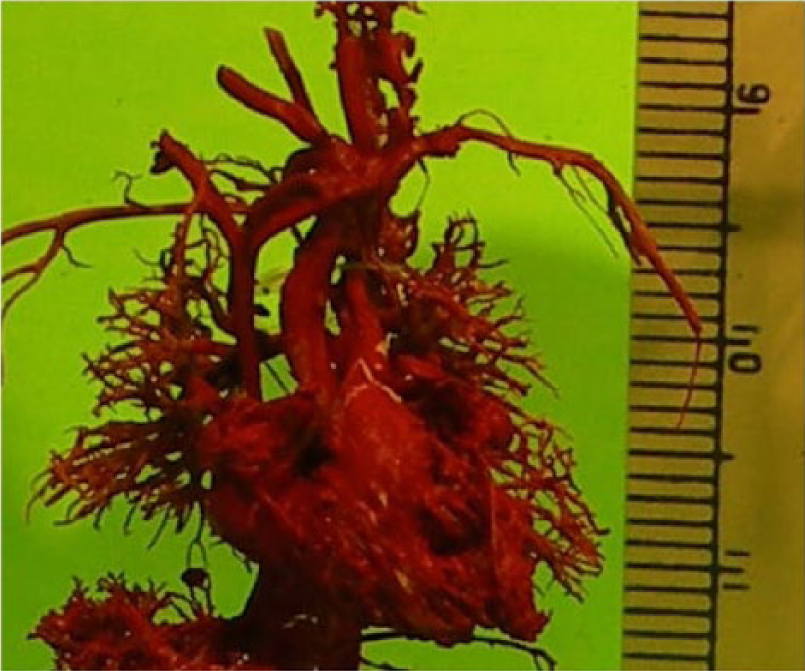
Cast of a fetal heart in early second trimester, obtained from the McMaster University anatomy laboratory with the assistance of Glenn Oomen.
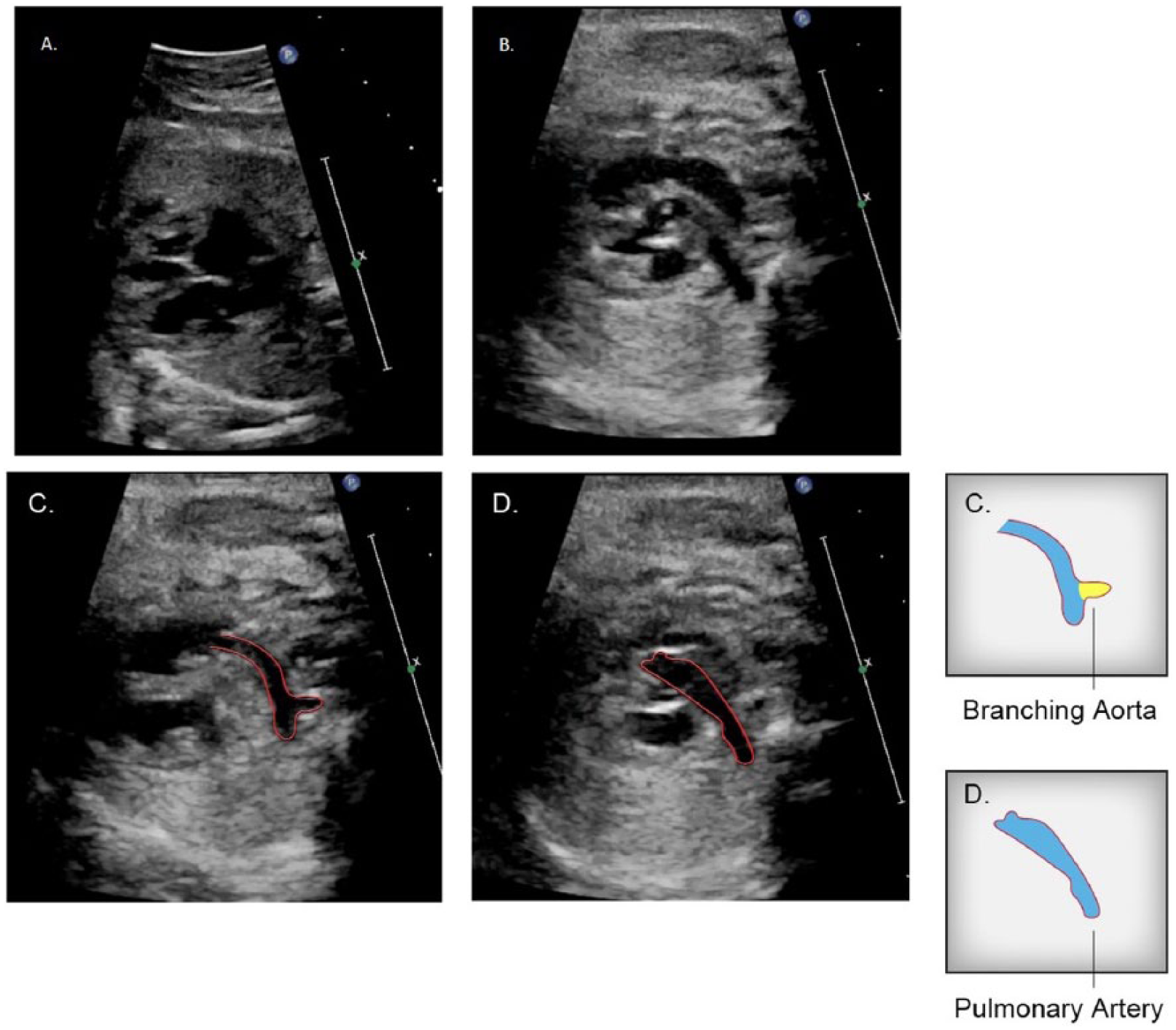
The four-chamber view appears normal in image A, but in image B, sweeping into the outflow tracts, it reveals a parallel orientation of the left and right ventricular outflow tracts, indicating a dextrotransposition of the great arteries. Additional images extracted from the same cineloop sweep demonstrate the branching associated with the aorta (C) arising from the right ventricle and a pulmonary artery arising from the left ventricle in (D). Line drawings provided by Ken K. Wong, graphic designer, Mohawk College.
According to a survey of 2758 referrals to the fetal cardiology unit at Guy’s Hospital, London, UK, the low-risk population is the source of 80% of CHD. 3 This underscores the importance of routine screening for detection of CHD. Routine screening of the fetus provides a more accurate measure of gestational age and an earlier detection of multiple pregnancies, and it may detect congenital malformations.
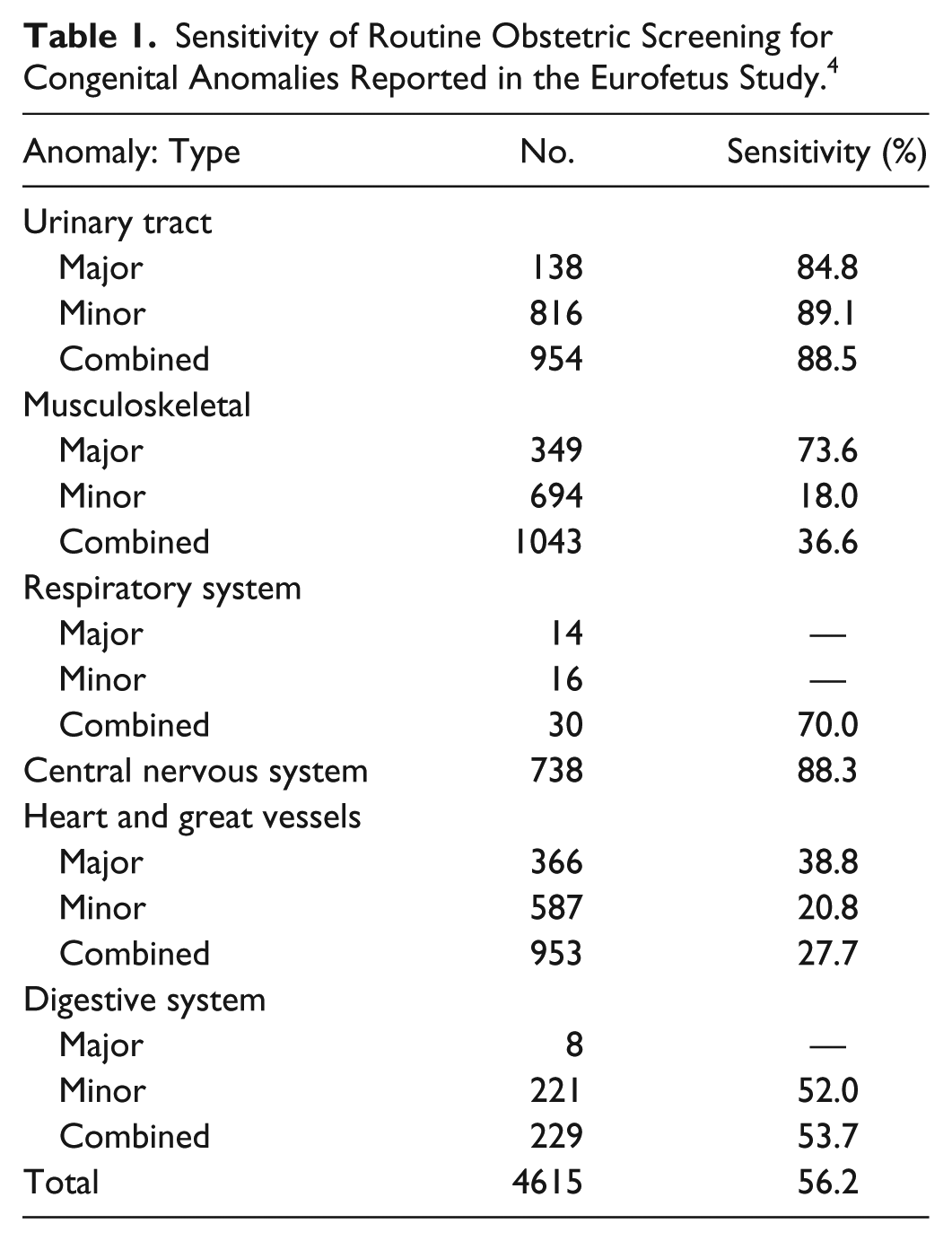
The Eurofetus Study identified marked differences in sensitivity for detection of systemic malformations (see Table 1). Abnormalities of the urinary system and nervous system were most frequently detected, with sensitivities of 88.5% and 88.3%, respectively. 4 In this study, cardiac abnormalities were not detected in most cases, with the sensitivities for major and minor cardiac defects being 38.8% and 20.8%, respectively, with a combined sensitivity of 27.7%. 4 The assessment of the fetal heart during routine obstetric screening has not yet been shown to match the sensitivity of screening for most other fetal pathologies.
Sensitivity of Routine Obstetric Screening for Congenital Anomalies Reported in the Eurofetus Study. 4
Prevalence and Classification of CHD
In an early large prospective study, Mitchell et al. defined a congenital heart defect as a “gross structural abnormality of the heart or intrathoracic great vessels that is actually or potentially of functional significance.” 5 Abnormalities of systemic veins and arteries, along with arrhythmias unassociated with structural malformations, are also excluded. A patent ductus arteriosus is considered normal for the first 14 days of the neonatal period. CHD can also be defined by the potential impact on fetal viability and practical implications for survival. A critical CHD is one that requires invasive intervention within the first month of life. 6
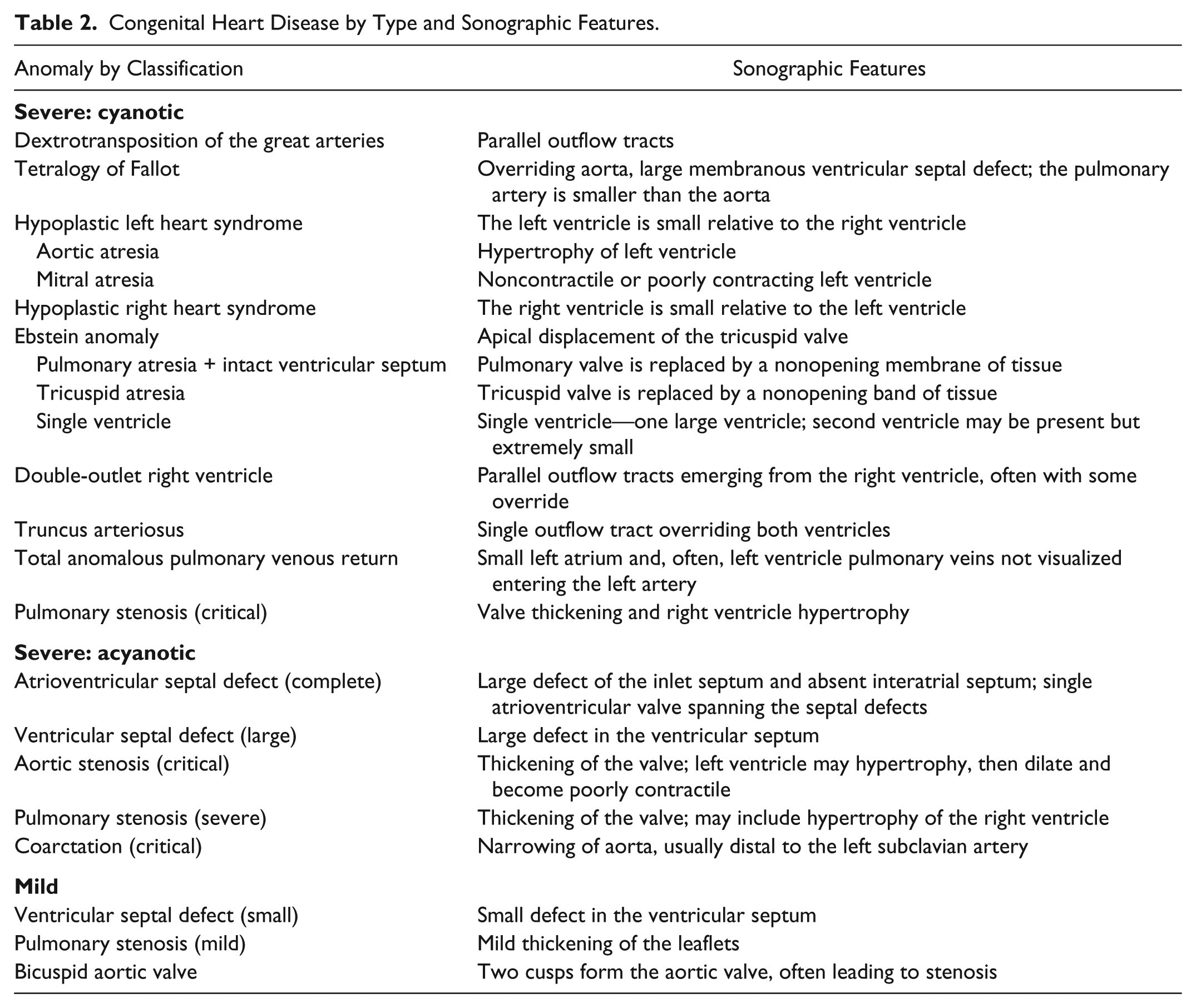
Typically, the incidence of disease is defined by the number of newly affected individuals, expressed as a unit of time or per population. Within the context of CHD, this is usually defined by thousands or millions of live births in a year. The prevalence is defined as the difference between those born and living with the disease and those who have died. 7 Estimating the incidence of CHD to a high degree of accuracy is quite challenging since some mild forms will go undetected and those with more severe forms may die early after birth and not be diagnosed via autopsy. 7 Incidence is usually estimated according to one of two approaches: (1) large numbers of live births, with the number of those born with CHD recorded; (2) smaller numbers of live births, usually from a single regional center. In the first case, recording all cases of CHD is unlikely to occur, leading to potential underestimation. In the second case, smaller numbers within the sample may be inadequate to accurately represent the true incidence of CHD within the population. 7 According to a systematic review by van der Linde et al., the global prevalence of CHD has increased over time, from 0.6 per 1000 live births in 1930–1934 to 9.1 per 1000 live births after 1995. 8 Prevalence in Europe was significantly higher than that in North America: 8.2 versus 6.9 per 1000 live births. 8 The distribution of CHD can be defined as follows: severe—for those who will require expert care; moderate—for those who will require care; and mild—for those whose CHD may resolve spontaneously. From an analysis of 44 studies, Hoffman and Kaplan determined that the incidence of severe CHD is stable at 2.5 to 3 per 1000 live births and moderately severe for another 3 per 1000 live births and that 13 per 1000 live births will have bicuspid aortic valves that may eventually need care. 7 Severe forms of CHD that can be further defined as cyanotic and acyanotic. Cyanotic forms result in hypoxia at birth, usually due to an intact interventricular septum. Acyanotic defects are those where left-to-right mixing of oxygenated and deoxygenated blood is possible through the atrial or ventricular septum or a patent ductus arteriosus. Common severe and mild forms of CHD are listed in Table 2.
Congenital Heart Disease by Type and Sonographic Features.
Sonographic Features of Severe CHD
The transposition of the great arteries is one of the most common forms of CHD, occurring in 2.5% to 5% of all cases. 6 Transposition of the great arteries with an intact ventricular septum represents about 75% of these cases and results in parallel and separated circulatory paths between the pulmonary and systemic circulatory systems (see Figure 2). In the absence of significant mixing at the level of the ductus arteriosus and atrial septum, the potential for severe cyanosis is very high. The four-chamber view in the majority of these cases will appear normal. Detection of transposition of the great arteries is usually made on the basis of visualizing the outflow tracts in parallel, rather than in their characteristic crossing-over orientation. This form of CHD has been found to be frequently missed in prenatal screening. Yates, using data provided by the British Paediatric Cardiac Association, found that <20% of these cases are identified in prenatal screening. 9 This finding is in agreement with a study by Jaeggi et al. comparing the pattern, management, and outcome of pre- versus postnatally diagnosed major CHD. In this study, on average, only 6.5% of CHD cases that require visualization of the outflow tracts were detected before birth. 10
Tetralogy of Fallot is the most common form of cyanotic CHD, representing 3% to 5% of all infants born with CHD. 11 The tetrad of Fallot is defined as (1) right ventricular outflow obstruction, (2) right ventricular hypertrophy, (3) overriding aorta, and (4) ventricular septal defect (VSD). It was first attributed to Canadian Maude Abbott in 1924. 11 Identification of a VSD and overriding aorta are key sonographic features (see Figure 3). In addition, the pulmonary artery is usually smaller than the aorta. Inclusion of outflow tract views in addition to the four-chamber view greatly enhances the likelihood of prenatal detection.
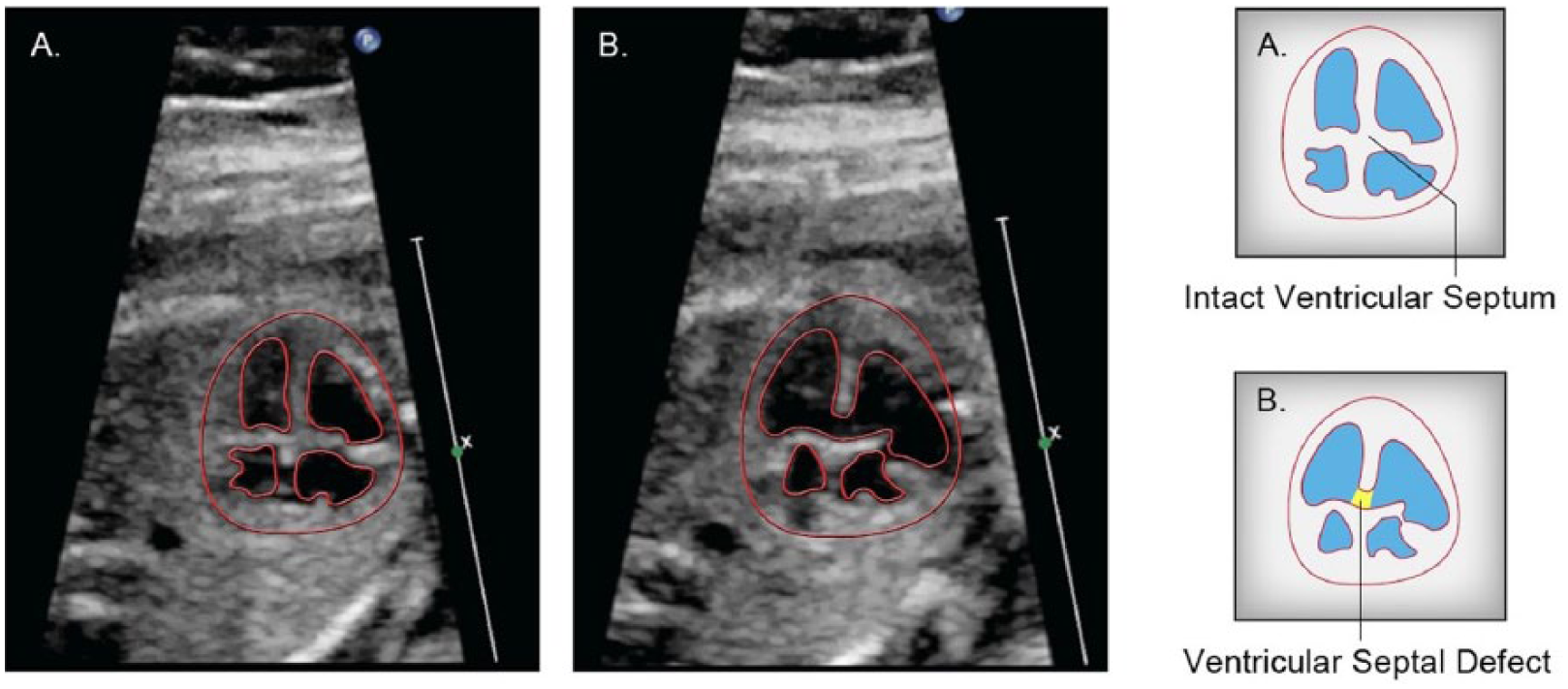
In this image pair (A + B), still images taken from the same cineloop sweep represent very different structural realities. Whereas a ventricular septal defect associated with tetralogy of Fallot is indicated by the shaded area in image B, the septum appears to be intact in image A, extracted from the same cineloop sweep. Line drawings provided by Ken K. Wong, graphic designer, Mohawk College.
Hypoplastic left heart syndrome (HLHS) is one of the most common and easily diagnosed severe forms of CHD. It is better described as a spectrum of hypoplasia where one end of the spectrum is defined by a slit-like left ventricle and atretic mitral and aortic valve. The opposite end of the spectrum is defined by a patent mitral valve, aortic valve stenosis or atresia, and a thick-walled ventricle, calcified and enveloped in endocardial fibroelastosis. 12 The standard four-chamber view demonstrates a small left heart or an echogenic left ventricle due to endocardial fibroelastosis. A small or difficult-to-visualize left ventricular outflow tract is usually present (see Figure 4). 13 Alternatively, an enlarged left atrium with a noncontractile large blood-filled left ventricle may present, which does not grow as the pregnancy progresses, leaving the left ventricle hypoplastic at birth. 14
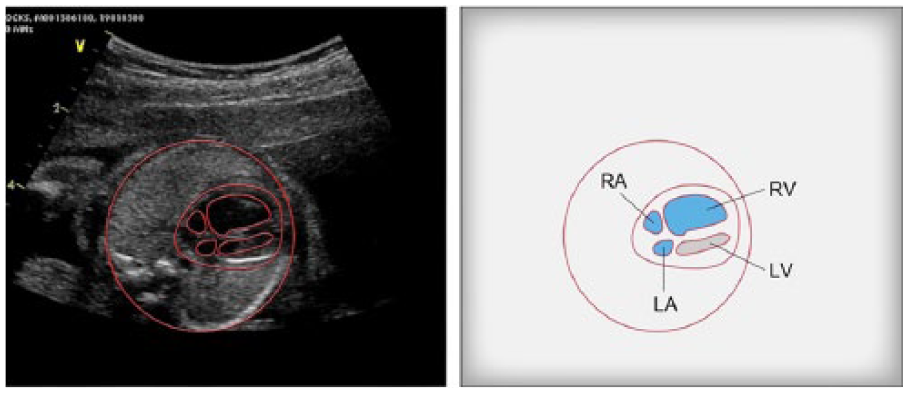
In this image, the asymmetry in the chamber size is defined in the four-chamber view. This is an example of hypoplastic left heart syndrome. Line drawings provided by Ken K. Wong, graphic designer, Mohawk College. LA, left artery; LV, left ventricle; RA, right; RV, right ventricle.
Hypoplastic right heart syndrome is less common than HLHS: it is defined by an underdeveloped right side of the heart, including the ventricle, pulmonary artery, and tricuspid and pulmonary valves. 15 Most commonly, this is due to pulmonary atresia. Sonographic features include a small hypertrophied right ventricle with a small or absent pulmonary artery or valve.
A single ventricle is usually (85%) the morphologic left ventricle and represents 2% of congenital heart defects. 15 There is often a second chamber, but it is a small rudimentary ventricle that is nonfunctional. Alternative diagnoses include large VSD, HLHS, or hypoplastic right heart syndrome.
A condition where most of the aorta arises from the right ventricle along with the pulmonary artery and a VSD is known as double-outlet right ventricle. It may be mistaken for tetralogy of Fallot or transposition of the great arteries with a VSD. The aorta is frequently parallel and to the right of the pulmonary artery. 15 Visualization of the outflow tracts is very important to enable identification of this defect.
A common arterial trunk arising from the base of the heart is commonly known as truncus arteriosus. Typically, a truncus overrides a VSD, and in the most frequently seen form, type 1, the pulmonary arteries arise from the trunk. It can be difficult to differentiate sonographically between truncus arteriosus and pulmonary valve atresia with VSD. 14
Total anomalous pulmonary venous return represents a rare form of CHD that, in its obstructed form, presents as a surgical emergency for the newborn. 16 The anomalous circulation is characterized by all four pulmonary veins returning to the right atrium rather than to the left atrium. This results in a postnatal closed pulmonary circulation where oxygenated blood recirculates between the right and left heart but never to the systemic circulation. A patent foramen ovale may allow some mixing of oxygenated and deoxygenated blood. 14 This anomaly is very difficult to detect in utero and can most reliably be excluded through documentation of pulmonary venous flow into the left atrium. 14
Pulmonary stenosis is difficult to diagnose sonographically and may show thickening of the valve leaflets and elevated blood flow velocities across the valve. Consequences of the stenosis may include right ventricle hypertrophy and a dilated pulmonary artery considered poststenotic dilatation.
A common atrioventricular junction in lieu of separate mitral and tricuspid valve orifices is described as a complete atrioventricular septal defect. It is also referred to as an endocardial cushion defect or atrioventricular canal defect. The sonographic features are defined by a common atrioventricular valve and by absence of the normal offset of the tricuspid valve, as well as an inlet VSD and a primum atrioventricular septal defect (see Figure 5). 15
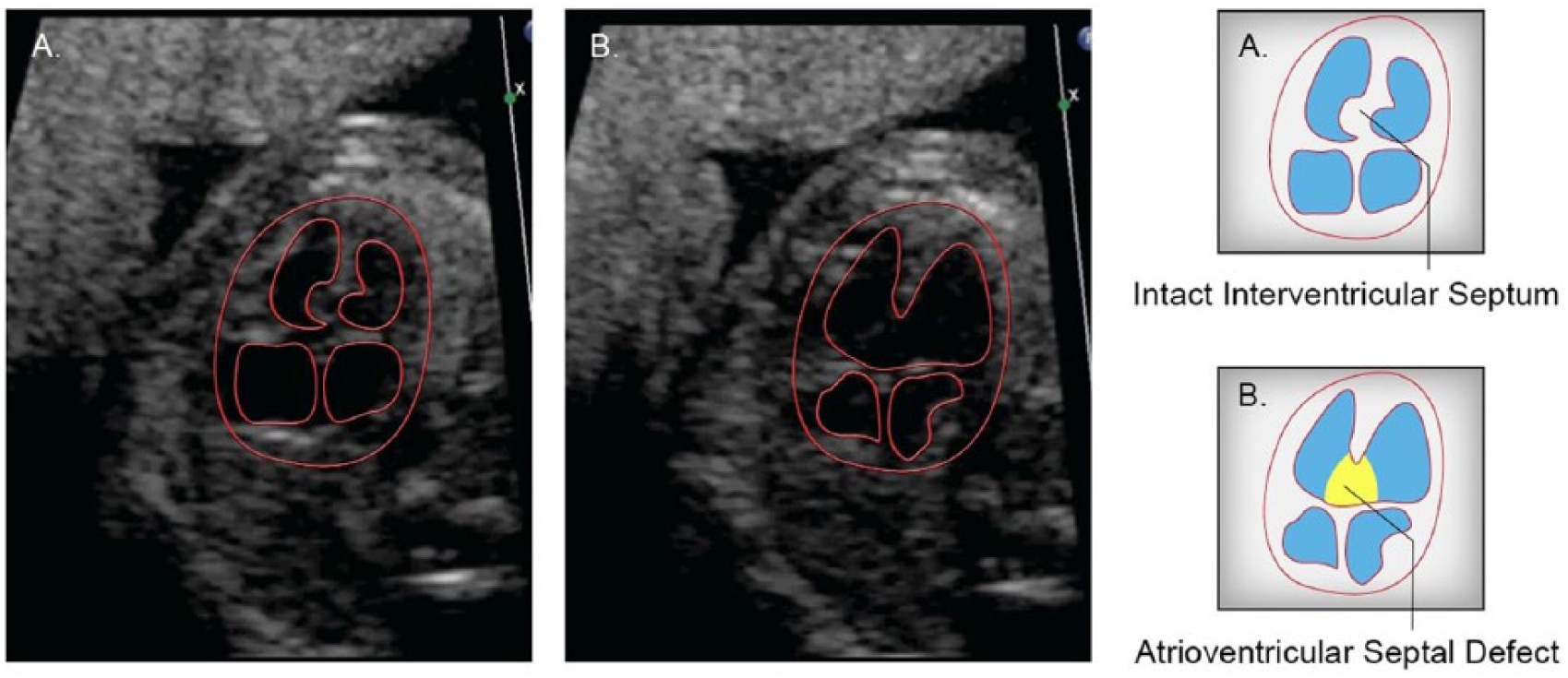
In this image pair (A + B) taken from the same cineloop sweep, the interventricular septum appears intact in image A but not in image B. This represents an atrioventricular septal defect. Line drawings provided by Ken K. Wong, graphic designer, Mohawk College.
Coarctation of the aorta is a discrete narrowing of the aortic arch distal to the left subclavian artery. It can be difficult to diagnose, while the ductus arteriosus is open, as it is in the fetus, as this masks the severity of the narrowed arch. Direct visualizations of the transverse arch and aortic isthmus are the best sonographic imaging planes for predicting severity, although indirect evidence includes dilatation of the right heart versus the left heart. 14 The ascending aorta is small, and the coarctation typically progresses in utero with gestational age. 15
Early Detection
Prenatal detection of CHD provides a number of advantages relative to postnatal identification: (1) counseling and the continuance of a viable pregnancy, (2) monitoring of pregnancy and treatment, (3) assessment for extracardiac disease and genetic associations, and (4) a decrease in fetal morbidity in some cases and a reduction in fetal neurologic impact. Some of the benefits of routine obstetric screening include maternal psychological effects, as well as increased connection with the unborn child and an enhanced maternal-fetal bond. 17 The visualization of the fetus along with a normal screening result has been found to decrease maternal anxiety and increase happiness. 17 Conversely, an abnormal result has the effect of decreasing maternal happiness and increasing anxiety. As stated by Menahem et al., “diagnosis was made at 17 weeks. The baby was active.” As such, the attachment had already begun between the mother and her unborn infant. As one father in the same study observed, “my wife loved the baby so many months and worked hard to keep it, to find out things were not right—is difficult beyond belief.” 18
In the long term, as compared with women who were not diagnosed prenatally, there was a more favorable impact on maternal happiness. The mother felt less responsibility for the CHD and tended toward an improved relationship with the infant’s father after prenatal diagnosis. 17 The early detection of CHD ensures that there will be an opportunity to provide a clear and accurate picture of the prognosis and outline treatment options to the parents. The clinician must be able to base the prognosis on a number of factors and not solely on the diagnosis. These factors include the security of the diagnosis, the stage in gestation, the potential for change, and the association with extracardiac malformations and known results of treatment. 19 The counseling opportunity provided by prenatal detection expands the range of options to parents, which may include continuance of the pregnancy, continuance with therapy, or termination of the pregnancy. Gestational age at the time of diagnosis is one of the strongest predictors of pregnancy termination. An early diagnosis is associated with termination in up to 70% of cases, and the probability of termination is inversely related to the percentage of early diagnosis in a population. 20
For those parents who choose to continue the pregnancy, early detection provides the opportunity to meet with perinatologists and cardiac specialists to engage in relaxed discussions related to prospective interventions. These discussions may provide the parents with a greater sense of control, while the health care team may be in a better position to minimize potential complications, adjusting the timing of the delivery by induction, caesarean section, or the use of tocolytic agents to allow greater maturation of the lungs if required. 16 Often, the infant is able to make a much less disruptive entry into postnatal life through the provision of prostaglandin infusions, respiratory support, and intensive monitoring as required. 16
A number of severe forms of CHD have been found to benefit from prenatal diagnosis. Tworetzky et al. reported that surgical outcomes for HLHS improved after fetal diagnosis. In a study of 88 patients, all 33 who were diagnosed prenatally survived surgery, whereas only 25 of 38 who were postnatally diagnosed survived. 13 Without prenatal diagnosis, most infants born with HLHS experience delays in diagnosis and appropriate resuscitation. This may lead to systemic hypoperfusion, shock, and organ damage, hindering the long-term survival prospects. 13 These findings are supported by Satomi et al. in their study of HLHS, which discussed finding improvements in preoperative conditions and avoidance of ductal shock. 21
Bonnet et al. determined that prenatal detection of transposition of the great arteries reduced neonatal morbidity and mortality. A 10-year study comparing 68 neonates with prenatal diagnosis and 250 neonates without prenatal diagnosis identified significant delays in access to the pediatric cardiology unit, increased postoperative mortality, and longer postoperative stays for those diagnosed postnatally. 22 Prenatal diagnosis of coarctation of the aorta was found by Franklin to improve survival and reduce morbidity. 23 An examination of 217 neonates with HLHS and 422 with transposition of the great arteries found that those with a prenatal diagnosis who underwent surgery had objective indicators of lower severity of illness preoperatively. 24 Although the prenatally diagnosed group experienced improved preoperative condition, in this study, postoperative outcome did not differ between the two groups. 24
Current Techniques
The type of CHD and the medical practice performing the screening are identified as key factors by a number of studies.10,25,26 Not surprising, a number of studies have shown increased detection of CHD at tertiary care centers as compared with community screening centers.4,10,25,27 The screening of CHD can range from a basic examination to an extended examination, to include additional views, such as the three-vessel view and the inclusion of Doppler and 3D imaging techniques.
Still Images
The basic examination consists of recording the four chambers of the fetal heart as a cross-sectional image. As noted by Chaoui, this approach has not led to widespread improvement in the detection of heart defects. 28 In his analysis, he identified four reasons for this outcome, the primary one being that many defects cannot be visualized in the four-chamber view. 28 In an extended effort to identify conotruncal defects, the extended examination includes views of the left and right outflow tracts in an effort to identify conotruncal defects.29,30 Yagel et al. proposed the acquisition and review of five cross-sectional images as a means of improving and simplifying the screening process. 31 This approach ensures that many of the key anatomic structures are represented, beginning with the abdominal situs and ending with a view of the three vessels: the pulmonary trunk, the aorta, and the superior vena cava. 31 In a similar line of reasoning, Yoo et al. proposed the inclusion of the three-vessel view as an element of every screening examination. 32 The three-vessel view is very effective in identifying many abnormalities associated with the outflow tracts and great arteries.33–35
Although the use of multiple cross-sectional planes and the three-vessel view have the potential to demonstrate a great deal of the fetal heart anatomy, these views have not been adopted by a majority of screening centers. Acquiring still images of the outflow tracts has proven to be more difficult than acquiring the four-chamber view, where all of the anatomy can be seen in one plane. 36 Furthermore, still images do not provide a real-time perspective of the fetal heart, and this remains a significant limitation of still images. Sklansky proposed a modified approach using cineloops that may address some of the challenges associated with acquiring multiple still images. 36
Importance of Cineloop Sweeps
The sonographic features of severe CHD can usually be captured as still images once the disease is identified. However, in many cases, it is a challenge to identify CHD using only still images. In Figure 2, the presence of parallel outflow tracts is documented as a series of still images. It is worth noting that in isolation, some of these still images may appear normal, whereas in the context of a cineloop, the aggregate set of images defines a relationship between the outflow tracts that is more difficult to establish as still images only. The four-chamber image in this set is normal in terms of the situs, axis, and relative size of the fetal heart as compared with the thorax. The image of the left ventricular outflow tract of Figure 2D could also be misinterpreted as normal; the key still images are depicted in Figure 2B and 2C. Should these images be unavailable, the identification of CHD could be missed.
Another example where a cineloop provides more value as compared with only a set of still images is illustrated in Figure 3. In this example, it is clear that the VSD is evident in the anterior aspect of the chamber and not the posterior aspect. This could be easily misrepresented as a normal case if only the still image is provided for interpretation, as in Figure 3A. Similarly, the atrioventricular septal defect apparent in Figure 5B could easily be overlooked if only Figure 5A is provided for interpretation. By sweeping the sound beam through the fetal heart and acquiring ≥30 images per second, this approach provides a more complete set of images for interpretation, with additional opportunities to assess the relationships among fetal cardiac structures. In addition, the cineloop sweeps provide an opportunity to assess the dynamics of the fetal heart, myocardial contractility, valve motion, and the synchrony of the chambers.
Color Doppler
The inclusion of color Doppler imaging in every fetal cardiac examination was proposed by Chaoui and McEwing to increase the speed and accuracy of the examination. 35 In their review article, they identified three cross-sectional planes to simplify the color Doppler examination: the four-chamber view, the five-chamber view, and the three-vessel view. 35 Copel et al. found that in 29% of cases, the use of color Doppler was essential for an accurate diagnosis. 37 In a pilot study, Scott et al. examined the potential for including a color Doppler cineloop of the three-vessel view in a routine obstetric screening protocol. 38 The results of this study determined that the color cineloop lengthened the examination time significantly and adversely affected the quality of the gray-scale features due to bleeding of color and to challenges associated with optimizing the color Doppler controls. 38
3D Imaging
The spatiotemporal image correlation (STIC) technology enabling the 3D imaging of the fetal heart as a cineloop volume has been commercially available for approximately 10 years. 39 The STIC methodology uses a slow, sweeping acquisition of many image planes over a 7.5- to 15-second interval. The mean heart rate is calculated, and the images are reformatted according to their correlation to the cardiac cycle.40,41 This technique generates a cardiac volume that can be reviewed as a cineloop at any speed, and images can be generated from the volume in any plane.40–42 The capability of interrogating the fetal heart in any plane within a moving cineloop provides a number of advantages when compared with 2D imaging, including the ability to review unlimited images, correlation of images between orthogonal planes, and access to novel planes, such as the en face view of the cardiac valves obtained through the coronal plane. 39
A number of closely related innovations have been designed to enhance the relationships among anatomic structures, particularly the outflow tracts. Those that leverage the STIC technology include Doppler and Doppler angio- and tomographic imaging. Doppler imaging relies on the phase shift associated with the movement of reflectors within the path of the sound beam. 43 The phase shift is angle dependent: reflections from moving reflectors at 90 degrees relative to the sound beam axis do not shift the phase of the frequency and are typically not visualized with Doppler processing. 43 In contrast, power Doppler and Doppler angiography techniques process the magnitude of the phase shift and do not determine directionality of the reflectors. The loss of directional information is compensated by significantly enhanced sensitivity to the presence of moving reflectors. 44 In 2009, Tonni et al. examined the feasibility of including 3D and Doppler angiography of the great arteries in the second-trimester examination. 45 They found that 3D imaging and Doppler can enhance the visualization rate of the great vessels, although it may be premature to include these techniques in routine examinations. The technique should be reserved for targeted cases where the 2D examination is incomplete. Tomographic imaging leverages the unique volume data set provided by the STIC technology. Tomographic imaging provides the sonographer with the ability to visualize an array of image slices represented as a panel, with a reference image available to correlate the position of the slices relative to the image volume. 46 Espinoza et al. provided a standardized method to reproduce the most clinically relevant and valuable images through commercial available software processing. 46 In a step further to minimize operator training and dependency, Abuhamad et al. evaluated an automated software program that demonstrated the capability of auto-generating tomographic images from standardized volumes. 47 In this evaluation, the software demonstrated a successful depiction of tomographic slices in >90% of each of the three cardiac planes, the five-chamber view, the aorta, the right ventricular outflow plane, and the pulmonary artery, along with the abdominal circumference and the plane capturing the stomach. 47
Summary
The detection of CHD during routine obstetric screening remains a diagnostic challenge. Unfortunately, improvements in sonography instrumentation and systems, including color Doppler and 3D imaging techniques, have not resulted in improvements in the detection of CHD. 48 In some cases, early detection has a significant impact on the morbidity of neonates born with CHD.21–23 For parents, early detection can provide an opportunity for counseling and improved understanding of the potential viability of the pregnancy. Early detection also provides an opportunity for improved management of the delivery and the potential to minimize complications.
A number of methods have been proposed to improve the detection rate of CHD, including the three-vessel view by Yoo et al., the use of color Doppler as proposed by Chaoui and McEwing, and a variety of 3D imaging techniques.32,35,49 Unfortunately, widespread adoption of these techniques has been limited. Access to 3D sonography systems is still limited, and the optimal use of these systems to consistently acquire 3D images of the fetal heart requires significant additional training.45,50
The reliance on still images for the assessment of the fetal heart imposes a significant limit to the screening of CHD during routine obstetric examinations. Using still images only, the interpreting physician will not be able to assess the function or anatomic relationships among cardiac structures. This means that, for practical purposes, the sonographer must identify the presence of CHD on behalf of the interpreting physician. Alternatively, using cineloop sweeps provides the sonographer and the interpreting physician with the opportunity to perform an assessment of the fetal heart in real-time. 51
The use of cineloops and real-time assessment of the fetal heart has been suggested by a number of authors as a means of improving the screening process.36,52 The use of cineloop sweeps will provide many more images of the fetal heart, establishing critical anatomic relationships and cardiac dynamics. Furthermore, this approach has the potential to improve the detection of CHD during screening by reducing the variability in the image data recorded and by standardizing the assessment of the fetal heart. 51
The acquisition of cineloop sweeps through the fetal heart in lieu of only still images has the potential to increase the detection of CHD without imposing additional requirements for sonographer training or additional imaging techniques, such as color Doppler and 3D imaging. Standardizing the assessment of the fetal heart and evaluating its functional characteristics during routine screening may provide significant advantages when compared with the interpretation of still images only.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
