Abstract
Severe brachiocephalic artery stenoses are relatively rare obstructions of the extracranial arteries and can cause significant changes to hemodynamics and spectral Doppler waveform patterns, complicating diagnosis. This case study presents disease progression from stenosis to occlusion and resulting complex steal phenomena extending to the contralateral upper extremity, which has not been widely reported. The sonographic findings of disease progression and collateral pathways were confirmed by magnetic resonance angiography and contrast-enhanced computed tomography.
The brachiocephalic artery (BCA), also called the brachiocephalic trunk or innominate artery, is the first branch of the aortic arch. It divides into the right subclavian artery (SA) and common carotid artery (CCA) supplying oxygenated blood flow to the right arm, head, and neck. Atherosclerotic disease is the most common cause of lesions in the BCA; however, severe stenoses are relatively rare.1–3 The presence of a hemodynamically significant stenosis in the BCA typically results in compensatory changes to flow in the ipsilateral vertebral artery (VA) and CCA. However, these collateralized arteries may not provide adequate perfusion, and compensatory changes may appear in other arteries as well.1–3 This case presents progression of a severe stenosis to occlusion and its impact on hemodynamics and Doppler waveforms. In addition, this case examines the unique presence of multiple steal phenomena—right subclavian-vertebral steal, right subclavian-carotid presteal, right external-internal carotid partial steal, and left vertebral-subclavian partial steal—resulting in complex collateral pathways, including the unusual extension of collateralization from the contralateral upper extremity, which has not been widely reported.
Case Report
A 72-year-old female patient with a medical history of extracranial cerebrovascular disease was referred to our cardiovascular laboratory in 2011 to establish care. A carotid duplex examination performed in a hospital setting the year prior reported tardus-parvus waveforms in the right CCA with a peak systolic velocity (PSV) of 12 cm/s, retrograde waveforms in the right VA, and suspected stenosis of the proximal right SA. The BCA was not insonated during the examination. The patient’s medical history also included abdominal aortic aneurysm repair, cerebral infarction, chronic obstructive pulmonary disease, coronary artery bypass grafting, hypertension, hyperlipidemia, mild aortic stenosis, peripheral arterial disease, transient ischemic attack, and tobacco abuse (half pack-a-day smoker for 45+ years).
In the following three years, our outpatient laboratory examined the patient biannually and performed a bilateral extracranial cerebrovascular duplex at each visit. In the first visit of late 2011, the patient presented with expressive aphasia and dyspnea on exertion with no complaints of bilateral arm pain. Physical examination revealed a left brachial systolic blood pressure of 118/70 mm Hg, radial artery pulse of 60 on the left, and absent on the right. Patient discomfort prevented blood pressure assessment in the right arm. Auscultation revealed a soft pansystolic bruit in the left mid-neck and faint sounds on the right.
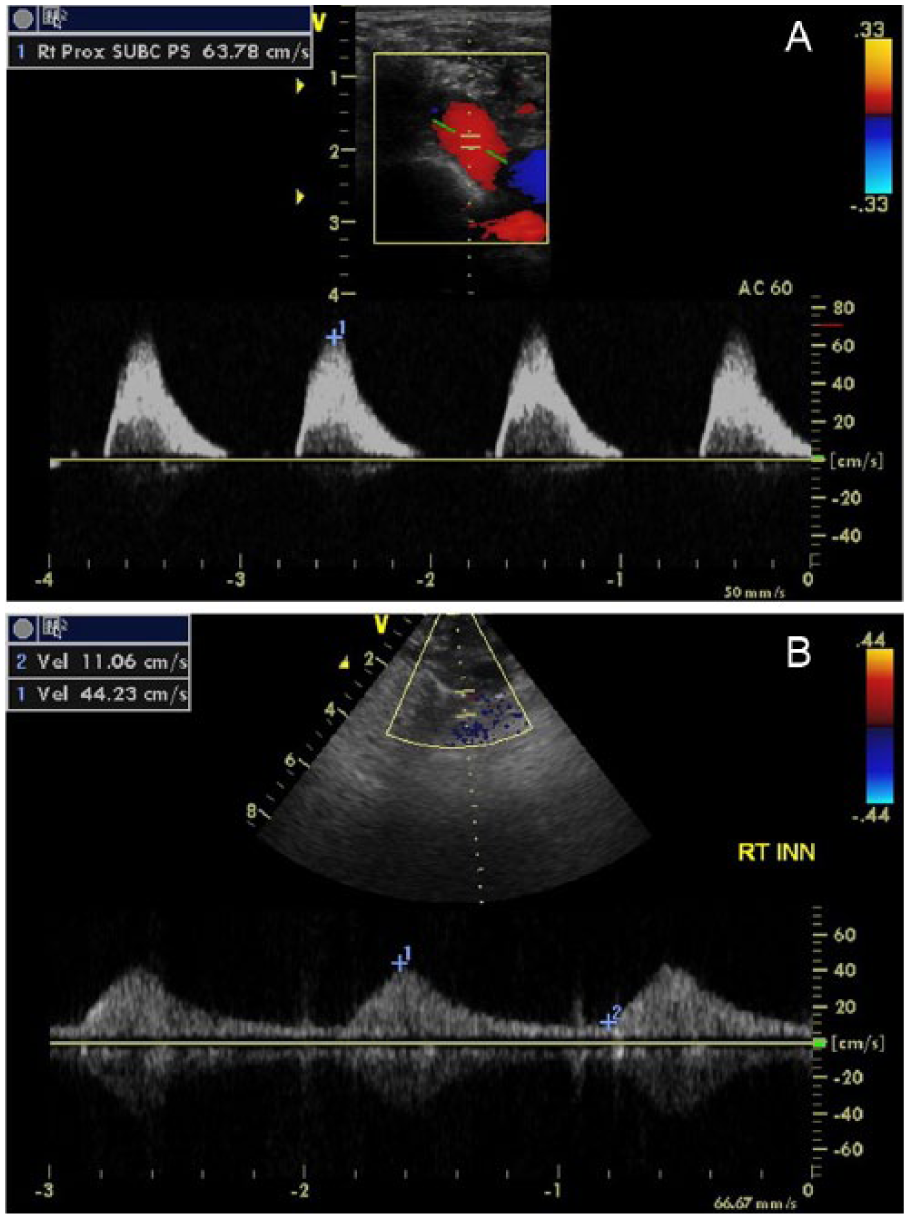
The duplex examination was conducted on a General Electric Vivid q (GE Healthcare, Milwaukee, WI) ultrasound system with a 7.5-MHz linear array transducer and a M4S phased-array transducer using gray-scale, color, and pulsed-wave Doppler imaging. Sonography of the right side revealed abnormal findings in all arteries. The CCA demonstrated transient (presteal) waveforms with late-systolic flow reversal (Figure 1). The internal carotid artery (ICA) demonstrated alternating (to-and-fro) waveforms with significant retrograde rather than antegrade flow (Figure 2). Atherosclerosis was seen in the ICA, and the degree of stenosis was graded by the presence of plaque and lumen diameter seen in gray-scale, which were consistent with less than 50% diameter reduction. The external carotid artery (ECA) demonstrated tardus-parvus waveforms (Figure 3). ECA branches were not clearly identified, so the temporal tap technique was used to help confirm location of the vessel by assessing the degree of transient flow oscillations (Figure 4). The VA demonstrated retrograde waveforms (Figure 5). The SA demonstrated alternating waveforms in the proximal segment and antegrade flow distal to the VA origin (Figure 6). The distal SA and BCA segments demonstrated tardus-parvus waveforms (Figure 7). These findings indicated severe stenosis of the BCA with compensatory complete steal phenomenon of the VA, presteal phenomenon of the CCA, and partial steal phenomenon of the ICA and proximal SA. On the left side, gray-scale and Doppler interrogation revealed atherosclerotic disease in the ICA consistent with 50% to 79% diameter reduction and in the ECA consistent with greater than 50% diameter reduction. High-velocity flow was demonstrated in the left VA, which was noted as likely due to compensatory cross-collateralization rather than stenosis since atherosclerosis and turbulence were not seen. The SA was noted as unremarkable. A six-month follow-up examination and repeat duplex showed no changes in symptoms, physical presentation, or hemodynamics.
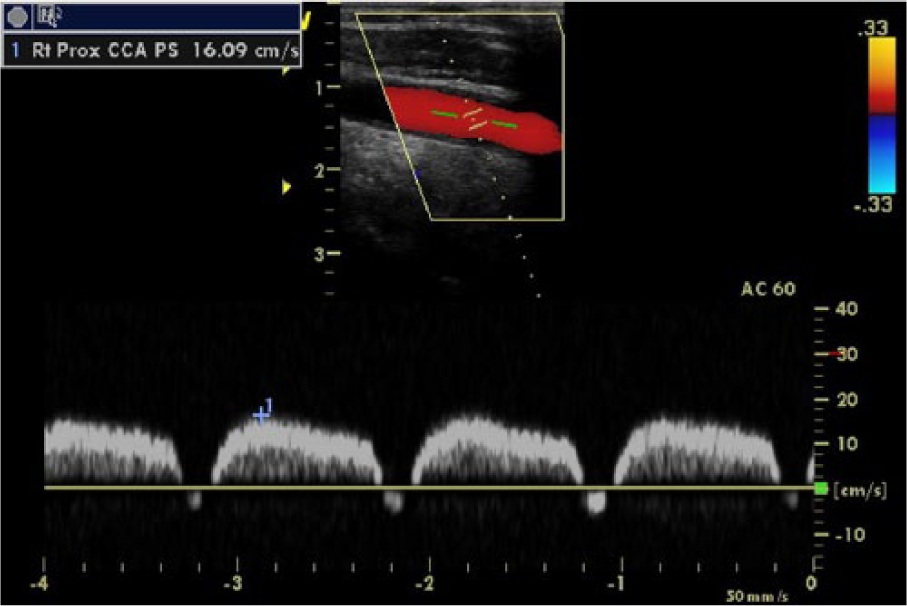
Duplex sonogram of right subclavian-carotid presteal phenomenon. The right common carotid artery demonstrating presteal waveforms. The mid-systolic increase is measured by a caliper.
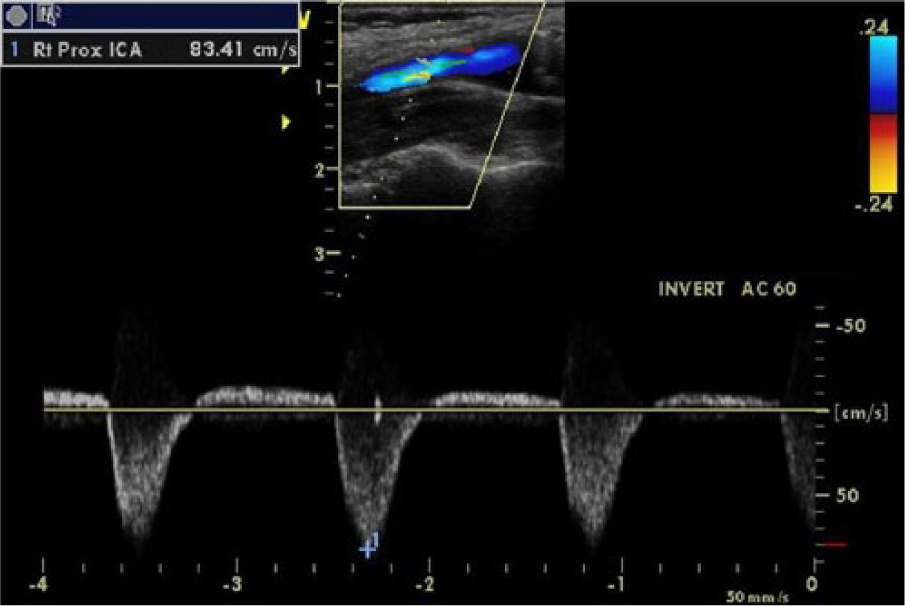
Duplex sonogram of right external-internal carotid partial steal phenomenon. The right internal carotid artery demonstrating alternating waveforms. The significant retrograde flow is demonstrated by color Doppler and measured by a caliper on spectral Doppler.
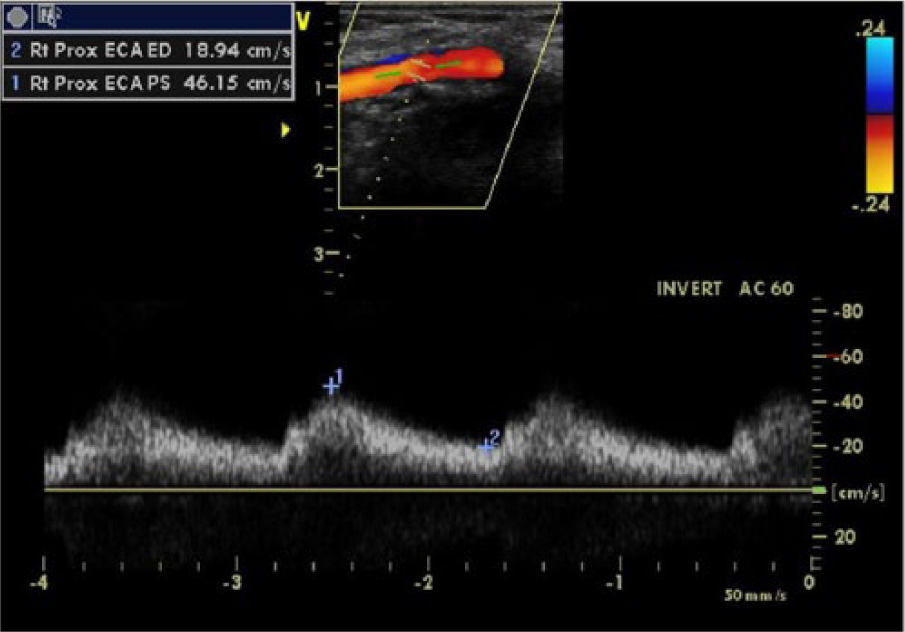
Duplex sonogram of the right external carotid artery demonstrating tardus-parvus waveforms.
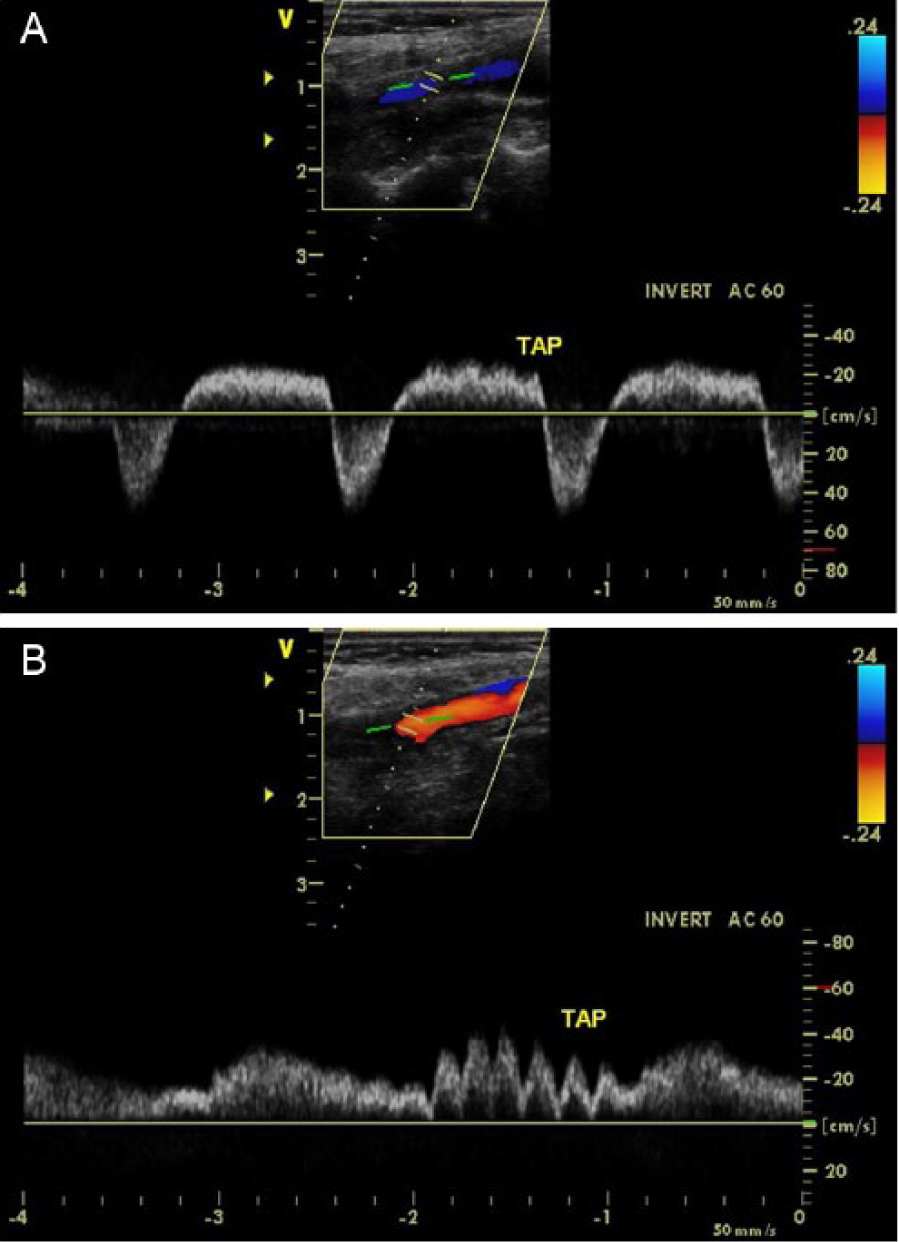
Duplex sonograms comparing degree of transient flow oscillations performed on the right internal carotid (A) and external carotid (B) arteries with the temporal tap technique.
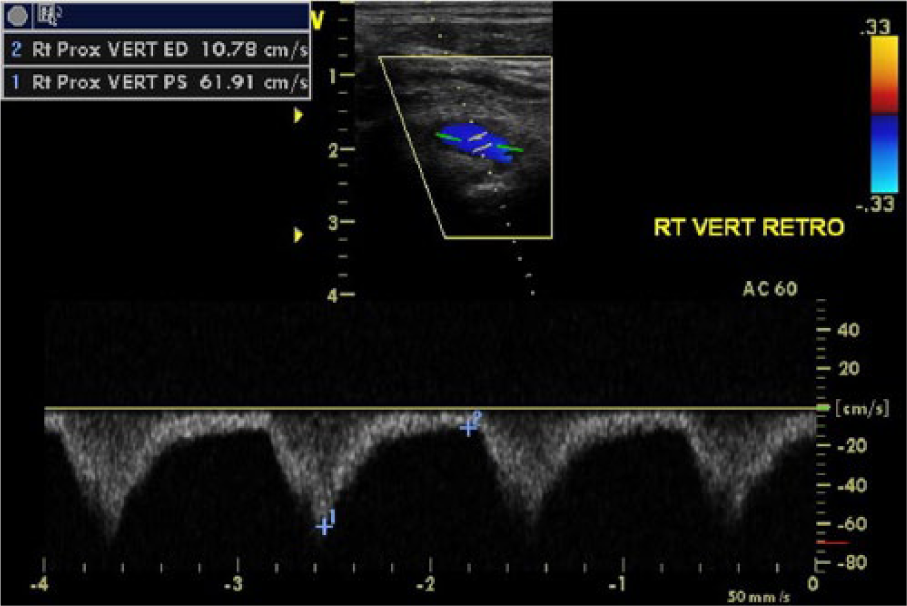
Duplex sonogram of right subclavian-vertebral complete steal phenomenon. The right vertebral artery demonstrating retrograde flow throughout systole and diastole.
Duplex sonogram of the right subclavian artery demonstrating retrograde flow in the proximal segment and antegrade flow distal to the vertebral artery origin.
Duplex sonograms of tardus-parvus waveforms in the right subclavian artery distal to the vertebral artery origin (A) and distal brachiocephalic artery (B).
In 2012, the patient returned for her next follow-up duplex, the third in our laboratory. The patient presented with aphasia, dizziness, and malaise but no complaints of arm pain at rest. She noted that she was hospitalized recently for unrelated reasons of marked thrombocytosis and mild leukocytosis, and her cerebrovascular system was not evaluated. Physical examination revealed a left brachial systolic blood pressure of 108/64 mm Hg and radial artery pulse of 68 on the left. Auscultation revealed a pansystolic bruit in the left neck and faint sounds on the right.
The duplex examination was conducted on the same General Electric Vivid q ultrasound system with a 7.5-MHz linear array transducer. The sonogram revealed changes in hemodynamics and stenosis grade compared to the first exam. On the right, the BCA demonstrated no flow with insonation, and the CCA demonstrated presteal waveforms with a decreased PSV. On the left, the VA demonstrated mid-systolic turbulence, and the SA distal to the VA takeoff demonstrated alternating waveforms with prominent mid-systolic retrograde flow (Figure 8). There was no evidence of significant subclavian arterial disease. These new findings indicated BCA occlusion and left VA partial steal phenomenon, which was reported as likely secondary to increased vertebrobasilar cross-collateralization. The subsequent three six-month follow-up examinations and repeat duplexes showed no changes in hemodynamics; however, the patient presented with worsening aphasia, dizziness, and generalized fatigue.
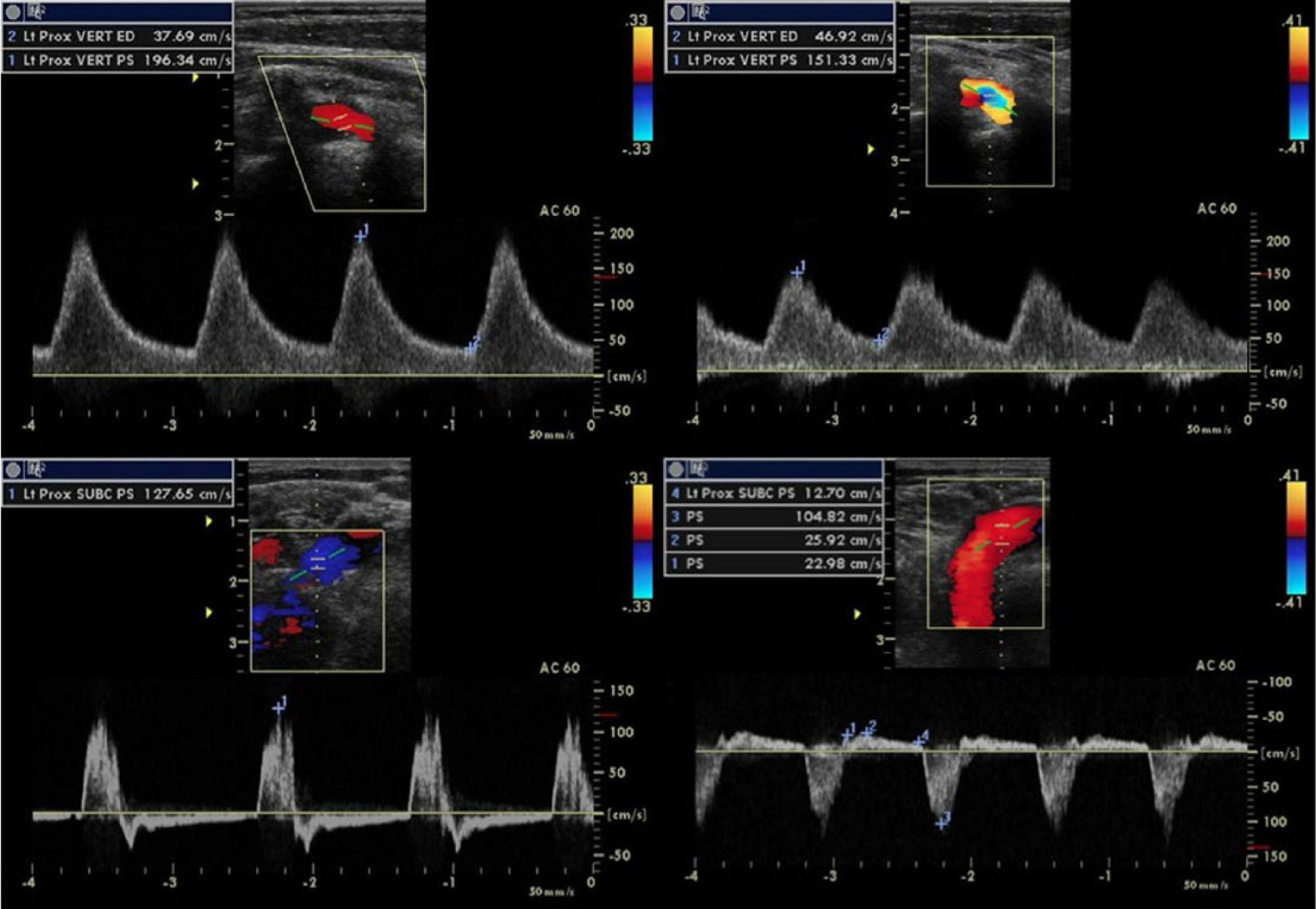
Duplex sonograms comparing left vertebral and subclavian artery waveform changes caused by right brachiocephalic artery occlusion and resulting in left vertebral steal phenomenon. After occlusion, the left vertebral artery demonstrated mid-systolic turbulence (top right), and the subclavian artery distal to the vertebral artery origin demonstrated significant mid-systolic reversal of flow with alternating waveforms indicating partial steal phenomenon (bottom right).
Due to complexity of the patient’s disease and hemodynamics, other imaging modalities were used for clinical correlation. Magnetic resonance imaging of the brain showed old lacunar infarcts and an old right-sided watershed infarct with no evidence of an acute stroke. Magnetic resonance angiography (MRA) showed occlusion of the BCA. The right CCA, ICA, and ECA also appeared extremely small and were noted as appearing “thready” (Figure 9). Nonobstructive atherosclerosis was noted in the carotid bulbs and proximal left SA. Computed tomography angiography (CTA) of the head and neck with and without contrast enhancement was also performed. Non-contrast-enhanced CTA demonstrated an old right-sided posterior fossa infarct and old left caudate head infarct with no other hemorrhage or lesions. Intracranial nonobstructive atherosclerosis was noted. Contrast-enhanced CTA demonstrated calcific atherosclerotic disease in the aortic arch and BCA. There was no evidence of significant stenosis in the left CCA or bilateral SAs. MRA and CTA confirmed sonographic findings and revealed adequate intracranial cross-collateralization networks through both the vertebrobasilar and circle of Willis pathways (Figures 9 and 10). At the time, surgical intervention was not recommended, and the patient agreed with conservative treatment.
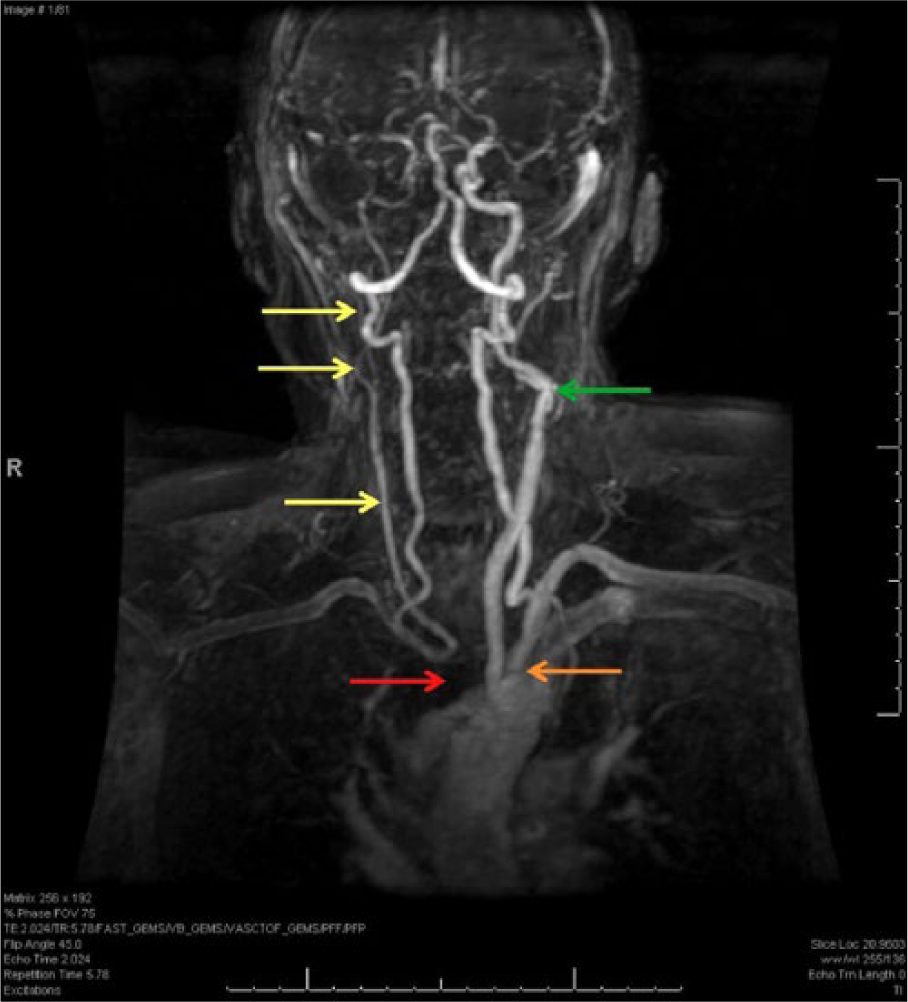
Contrast-enhanced magnetic resonance angiogram anteroposterior view demonstrating brachiocephalic artery occlusion (red arrow); small, “thready” appearing right carotid vessels (yellow arrows); mild left subclavian artery stenosis (orange arrow); and moderate stenosis of the left internal and external carotid arteries (green arrow). Note the size and patency of the vertebrobasilar vessels and left common carotid, which perfuse the right carotid system and intracranial vessels, respectively.
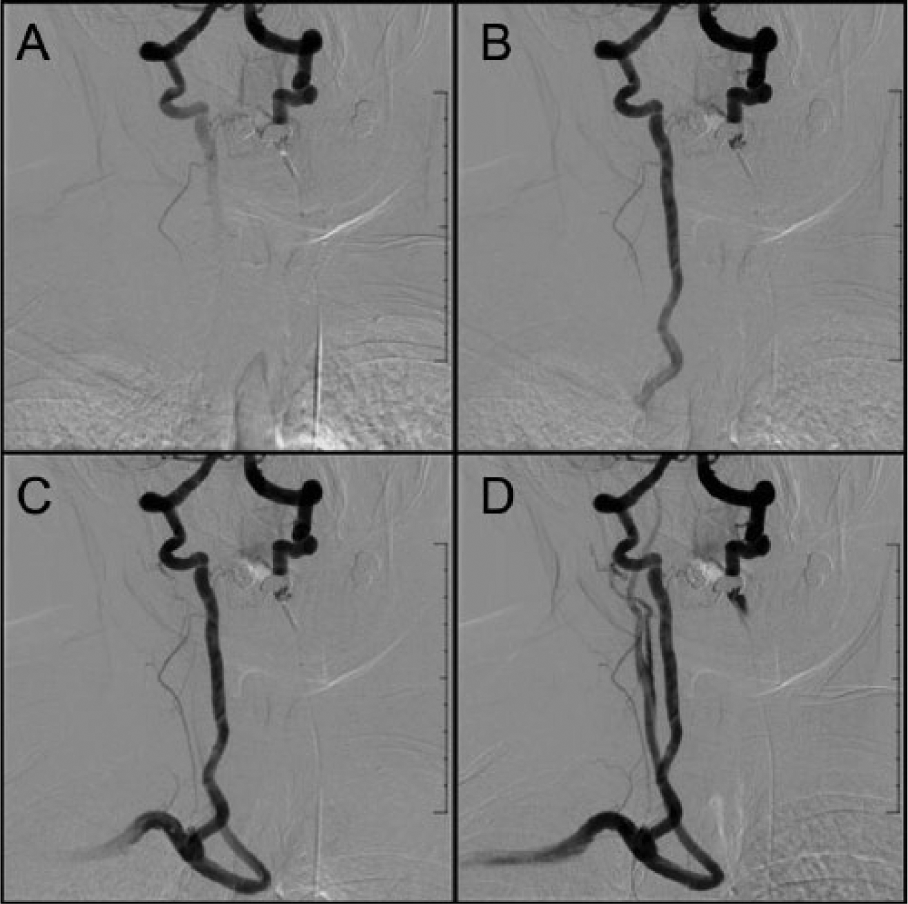
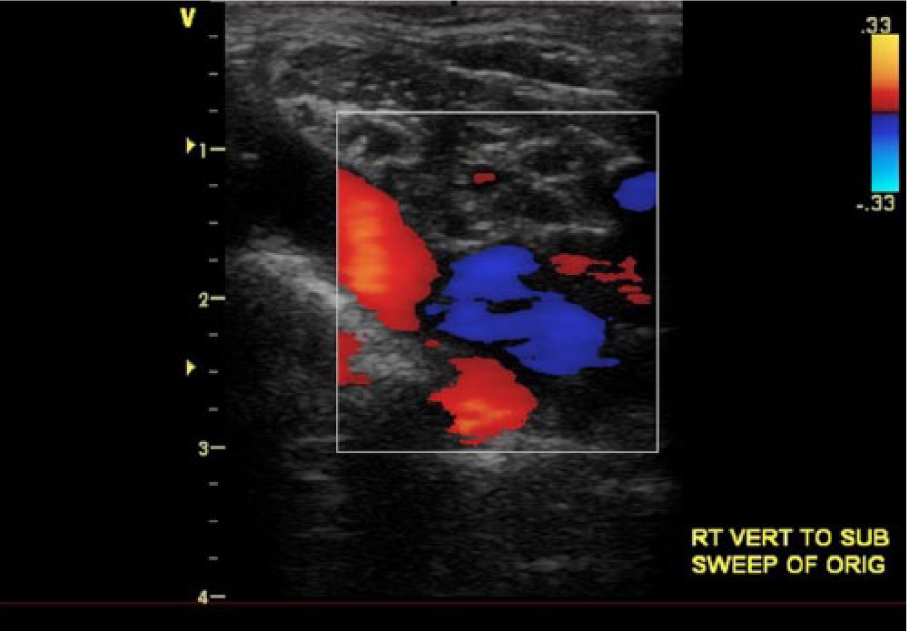
Subsequent computed tomography angiographic images demonstrating vertebrobasilar and right extracranial carotid cross-collateralization pathways after contrast injection in the left vertebral artery (VA). Contrast enhancement demonstrates blood flowing from the left to the right VA (A) and retrograde flow in a widely patent right VA (B) to the right subclavian artery (C) and carotid arteries (D).
Discussion
A hemodynamically significant BCA stenosis can greatly alter cerebrovascular perfusion pathways.1–3 Almost 20% of cases develop steal phenomenon in one artery.2,4 One percent to 2% of cases reported steal phenomena in two arteries, and two cases described involvement of three arteries in the literature.5,6 A PubMed search of “brachiocephalic artery,” “stenosis,” and/or “occlusion,” with “bilateral steal,” “contralateral steal,” “left steal,” or “quadruple steal,” at the time of this study revealed that few similar cases have been reported. The limited existence of similar cases with multiple steal phenomena makes comparative analysis unlikely. Therefore, this is an exceptionally rare case of a BCA lesion resulting in multiple complex steal phenomena not widely reported.
Hemodynamic changes occur in approximately 17% of BCA stenoses and usually involve the ipsilateral VA and CCA.2,3,7 The VA is typically collateralized first because it contributes the least perfusion, and the vertebrobasilar system is an easier cross-collateralization route than the circle of Willis pathway. 4 The CCA is also affected because of the decreased pressure gradient distal to the stenosis and low perfusion contribution of the VA. Changes in the CCA help differentiate the presence of BCA versus SA stenosis. The short length of the BCA can lead to poststenotic characteristics extending into the SA mimicking subclavian steal phenomenon; however, SA disease does not affect the CCA and is only associated with VA changes.1,3 For these reasons, hemodynamic changes seen in both the VA and CCA are distinctive of BCA disease.
In approximately 2% of cases, perfusion from the VA and CCA is inadequate, and hemodynamic changes can be seen in other nonadjacent arteries.1,2,5 Studies have shown a wide-ranging constellation of changes as patency and compensation are compromised. For example, involvement of the right ICA and ECA has been reported in cases with multiple concomitant stenoses and vertebrobasilar arterial insufficiency.2,3,5,6,8,9 This scenario was seen in the case of our patient. Compensatory flow from the right VA and CCA was inadequate, and hemodynamic changes further developed, affecting the right ICA, left VA, and left SA (Figure 11). Presence of left VA partial steal phenomenon is likely due to greater dependence on the vertebrobasilar system for perfusion to the right side since there was reduced patency of the left ICA and ECA. In addition, it is unusual that the left SA was not stenotic given the patient’s history of vasculopathies and the fact that the left SA is the most common branch of the aortic arch affected by atherosclerotic occlusive disease. 10
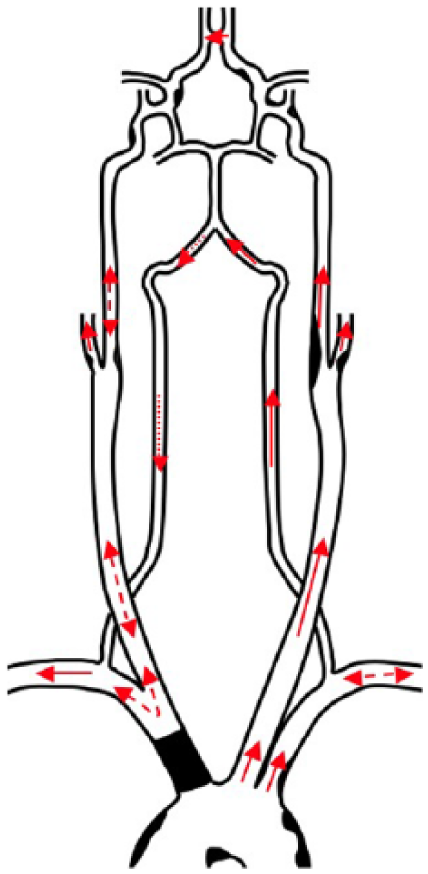
Diagram of atherosclerotic disease and hemodynamics of the third serial duplex sonogram depicting brachiocephalic artery (BCA) occlusion and steal phenomena: right subclavian-vertebral complete steal, right subclavian-carotid presteal, right external-internal carotid partial steal, and left vertebral-subclavian partial steal. Occlusive atherosclerotic disease is demonstrated in the BCA. Moderate stenosis is demonstrated in the left external and internal carotids. Nonobstructive atherosclerosis is seen in the aortic arch, left subclavian artery, right internal and external carotid arteries, and intracranial arteries. Complete steal phenomenon (dotted arrow). Partial and presteal steal phenomena (dashed/double-pointed arrows). Antegrade flow (solid arrows).
Waveform morphology on spectral Doppler can reveal hemodynamic changes. Direction, pattern, and velocity parameters help distinguish abnormal flow and characterization of the type of steal. Retrograde waveforms present as flow below the baseline throughout both systole and diastole. This is characteristic of complete steal phenomenon, which indicates a severe form of hemodynamically significant stenosis or occlusion. 2 Incomplete steal phenomenon is characterized as alternating and transient waveforms. Alternating, or to-and-fro, waveforms present as a pattern of both antegrade and retrograde flow, which occurs when there is too little poststenosis perfusion pressure to exceed systemic pressure during systole. This is characteristic of partial steal phenomenon, which indicates a moderate form of hemodynamically significant stenosis. Transient, or presteal, waveforms present as a pattern of decreased or increased PSV, immediate mid-systolic decrease, little to no late systolic reversal of flow, and restoration of antegrade flow in diastole. 7 The short-lived mid-systolic deceleration is attributed to increased systemic pressure reversing flow and sudden poststenosis pressure restoring antegrade flow. 2 This is characteristic of presteal phenomenon, which indicates a mild form of hemodynamically significant stenosis. 7 Tardus-parvus waveforms present as a pattern of prolonged systolic upstrokes with a rounded peak and decreased PSV. It does not indicate collateralization but rather damped flow caused by a more proximal high-grade stenosis and decreased peripheral resisitance. 11 This is typically seen distal to a BCA lesion in the trunk, right SA, or carotid system. 2 These variations of waveform morphology—retrograde, alternating, transient, and tardus-parvus—reveal hemodynamic changes associated with complete, partial, and presteal phenomena and hemodynamic stenosis in patent and collateral pathways.
Comparison of hemodynamic changes and waveform morphologies of our patient’s first and third sonograms clearly revealed disease progression, which also correlated with her development of vertebrobasilar ischemia symptoms. The third sonogram revealed changes in arterial pressure and degree of peripheral resistance, which were reflected by the low PSVs, tardus-parvus waveforms, and increased severity of cross-collateralization. When the lesion became occluded, it resulted in greater perfusion demand on the right with dependence on the left CCA to supply the intracranial system and left SA to supply the contralateral side. It is likely that the vertebrobasilar system preferentially supplied flow to the right side due to greater patency in the left VA than the left ICA and ECA, which had moderate stenoses and were smaller in size (Figure 10).
Quantification of steal from the vertebrobasilar and carotid systems by waveform morphology is not possible due to the presence of multiple steal phenomena, variations of each phenomenon, complexity of extracranial and intracranial collateral routes, and existence of concomitant stenoses. Classifications of waveforms have been created to categorize morphologies. Brunhölzl and von Reutern 8 proposed a phenomenological classification. Type I depicts alternating flow in the VA and minor waveform changes in the CCA. Type II depicts reversed flow in the VA and presteal waveforms in the CCA. Type III depicts alternating or retrograde flow in the VA and/or CCA. Kliewer et al. 12 similarly described a classification system to differentiate severity. Type 1 depicts antegrade flow with a transient mid-systolic decline creating two systolic peaks. Type 2 depicts a pronounced cleft between the two systolic peaks and low-velocity antegrade flow at end-diastole. Type 3 depicts antegrade flow with a mid-systolic decrease that reaches baseline, similar to a presteal waveform. Type 4 depicts flow below the baseline. Although abnormal waveforms can be associated with levels of disease severity, stenosis grade cannot be based on waveforms. Instead, waveform morphology reflects manifested collateral pathways and provides a subjective severity of hemodynamic change. Thus, these classifications are not widely used today. 2
The presence of symptoms is a more widely considered indicator to assess severity and need for surgical intervention. Symptoms appear in 2% to 10% of cases and usually only in the most severe.4,13 Symptoms are associated with head, neck, and arm ischemia, including ataxia, dizziness, extremity claudication, syncope, transient ischemic attack, vertigo, and visual disturbances.1,13 Symptoms can be aggravated by arm exercise with severity dependent on the amount of arterial vasodilatation, compensatory perfusion, collateral pathway adequacy, and exercise duration and intensity. 3 The low percentage of symptomatic lesions is attributed to the extensive cerebrovascular network, which readily allows multiple pathways for cross-collateralization.
Treatment of a severe BCA lesion may be aggressive or conservative. Surgical options include balloon angioplasty, endovascular stent placement, and bypass graft revascularization. 14 However, in this case, due to the patient’s high risks of morbidity and mortality, surgical intervention was not considered safe, and the physician and patient chose a conservative therapeutic route instead. Efficacy and long-term patency of the patient’s cerebrovascular system are unknown.
As shown by this case, sonography is an excellent tool for identifying extracranial cerebrovascular disease and supports why it is the most widely used noninvasive imaging modality and initial examination for evaluating cardiovascular disease. 2 Sonographers can visualize and insonate extracranial vessels and determine the degree of BCA stenosis with high sensitivity. Spectral Doppler waveform patterns associated with hemodynamic changes caused by a BCA lesion can be readily recognized by sonography and accurately depict collateralized pathways. Combination of sonography and physiologic testing can reveal upper extremity arterial insufficiency, further changes to hemodynamics, and neurologic consequences of vertebrobasilar ischemia. 3 Utility of sonography for diagnosing disease severity and progression on serial examinations makes it an important tool in the continuum of care.
There are two disadvantages of sonography for assessing a BCA lesion. The first is visualization of the BCA, which may be inadequate due to location under the clavicle and depth. The second is the high degree of user dependence in this modality, which relies on sonographer experience and skill. A sonographer’s skill set may be limited if BCA assessment is not regularly performed or a part of his or her laboratory’s standard protocol. Therefore, it is essential for sonographers to be aware of normal and abnormal waveform morphologies, discern when to interrogate the BCA, grade stenosis severity, identify collateralized pathways, and differentiate subclavian steal phenomenon.
Conclusion
A BCA occlusion resulting in multiple complex steal phenomena extending to the contralateral upper extremity is an exceptionally rare vascular abnormality that can be accurately diagnosed and evaluated by sonography. Typical appearance of a BCA lesion involves collateralization of the right VA and CCA but can involve other arteries for compensation. This case study presents a progressive lesion that significantly altered hemodynamics and waveform morphology. Multiple steal phenomena were well demonstrated by spectral Doppler in the right VA, CCA, and ICA and left SA. Study findings were confirmed with MRA and CTA.
Comprehensive knowledge of clinical and sonographic presentations of BCA lesions, extracranial and intracranial anatomy, pathophysiology, hemodynamics, and waveform morphology is important to adequately assess and accurately diagnose these unusual findings. Proficiency to demonstrate waveform morphologies and changes to hemodynamics are also imperative for proper diagnosis and comparison of serial studies. This case supports sonography as an excellent modality to assess and diagnose such a rare pathology.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
