Abstract
Umbilical artery catheterization is considered the standard of care for arterial access in neonates. Studies have shown, however, that umbilical artery catheterization carries a high risk of aortic thrombosis, which can be fatal. Neonatal aortic thrombosis can be difficult to treat appropriately, in part because of the absence of reliable data for this patient population and nonstandardized reporting of the condition. Sonography of the abdominal aorta in neonates provides a noninvasive means of diagnosing the presence and extent of aortic thrombus, which assists in managing the patient’s plan of care. An important case also has been made for long-term follow-up studies using sonography as the diagnostic tool in patients who received umbilical artery catheters as infants.
Keywords
Sonography provides a rapid, reliable, noninvasive diagnostic method to assess the entire abdominal aorta and determine the presence of thrombus or foreign bodies within the lumen. 1 Neonatal aortic thrombosis in particular can be problematic, as it may be fatal, and treatment can be difficult because of a lack of studies and reliable data for the neonatal population.2,3 Studies have shown that an overwhelming number of neonatal patients with aortic thrombus, up to 78%, have a history of previous umbilical artery catheterization.4-6 Umbilical artery catheterization is considered the standard of care for arterial access in neonates in the neonatal intensive care unit (NICU), and nearly 7% of newborns in the United States in 2008 were admitted to a NICU, an increase from 6% in 2004.4,7 The number of NICU beds in the state of Texas alone, including levels I through IV, has increased by 74 percent in the 10-year period from 2000 to 2010.4,8 A case of neonatal aortic thrombus is presented here, highlighting the role of sonography in diagnosis and management.
Case Report
A 3-month, 6-day-old premature male in the NICU was referred for sonographic examination of the abdominal aorta following an echocardiogram. No prior sonographic studies were available for comparison, nor were any specific signs or symptoms noted in the patient. With the patient lying supine, sonographic images were obtained of the abdominal aorta including the proximal, middle, and distal portions to the level of the bifurcation of the left and right common iliac arteries using a Philips iU22 ultrasound instrument (Philips US, Bothell, Washington) and a C8-5 broadband curved array transducer. The sonographic examination included gray-scale, color Doppler, and spectral Doppler images in orthogonal planes. Imaging showed visualization of a possible line cast in the descending aorta demonstrated by the sonographic appearance of a hyperechoic linear structure within the vessel lumen (Figures 1 and 2). Adjacent to the hyperechoic linear structure, thrombus was noted at the superior portion, as can be seen in Figure 1. This hyperechoic linear structure along the posterior wall within the proximal to distal abdominal aorta, extending into the bifurcation, measured approximately 3.4 cm in length (Figure 3). Figures 4 and 5 show color Doppler images in long-axis and cross-section showing patency of the aorta with flow seen around the linear structure and thrombus. In Figure 4, a segment of thrombus can be seen to fill the lumen of the aorta just superior to the hyperechoic linear structure with color flow above and below the thrombus. The appearance was concerning for remnants of a catheter that had been placed into the umbilical artery in the NICU following premature birth. The findings were discussed with the nurse practitioner caring for the patient, and a follow-up examination to monitor the abdominal aortic thrombus was performed 5 days later. The follow-up study was limited by bowel gas, particularly visualization of the middle and distal portions of the abdominal aorta. No new thrombus was noted within the lumen of the aorta at that time. The patient was transferred for further care shortly after the follow-up study, and no further data are available.
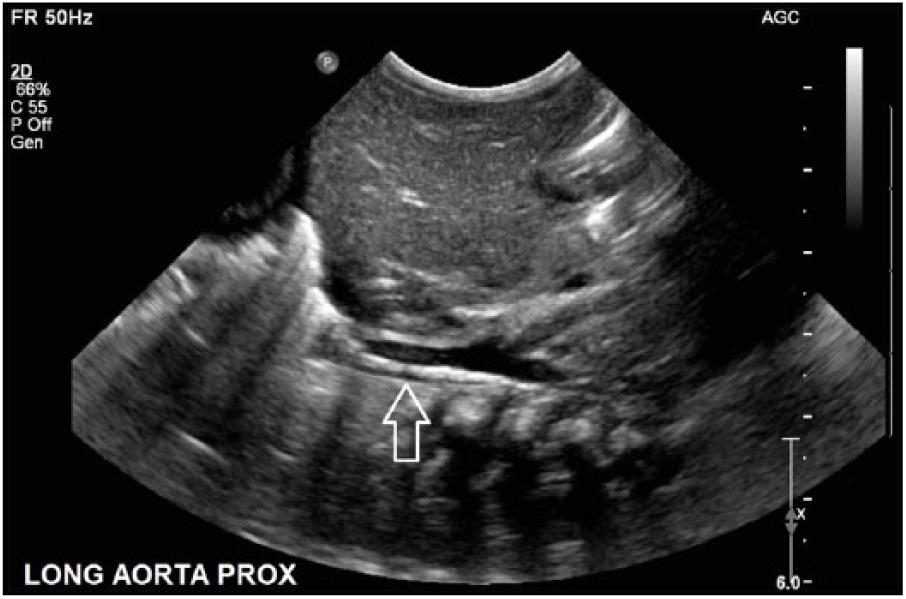
Gray-scale long-axis image of the proximal abdominal aorta, showing a hyperechoic line echo (arrow) with more hypoechoic echoes just proximal, suggesting aortic thrombus.
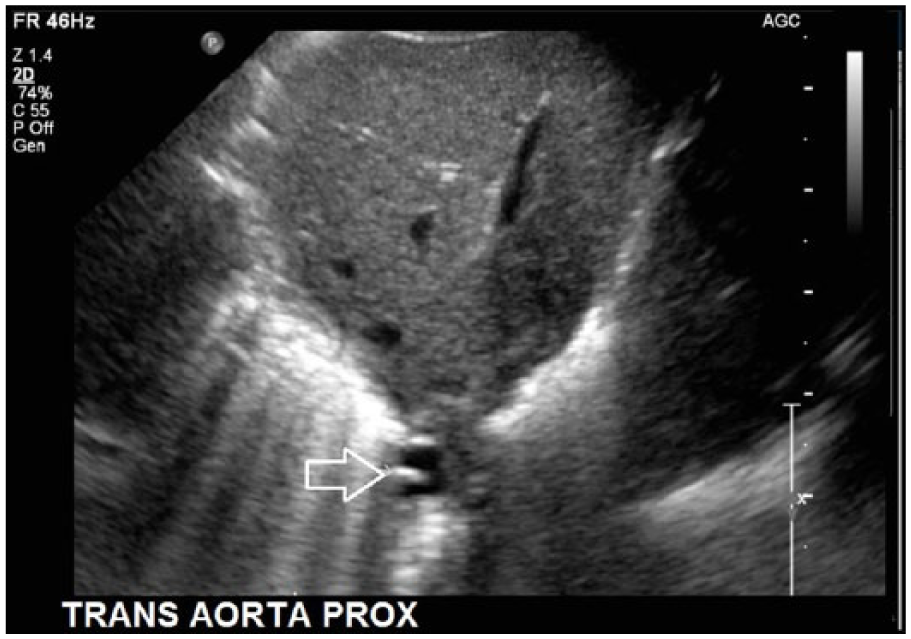
Gray-scale cross-sectional image showing the hyperechoic line echo (arrow) in the right posterolateral aspect of the vessel lumen.
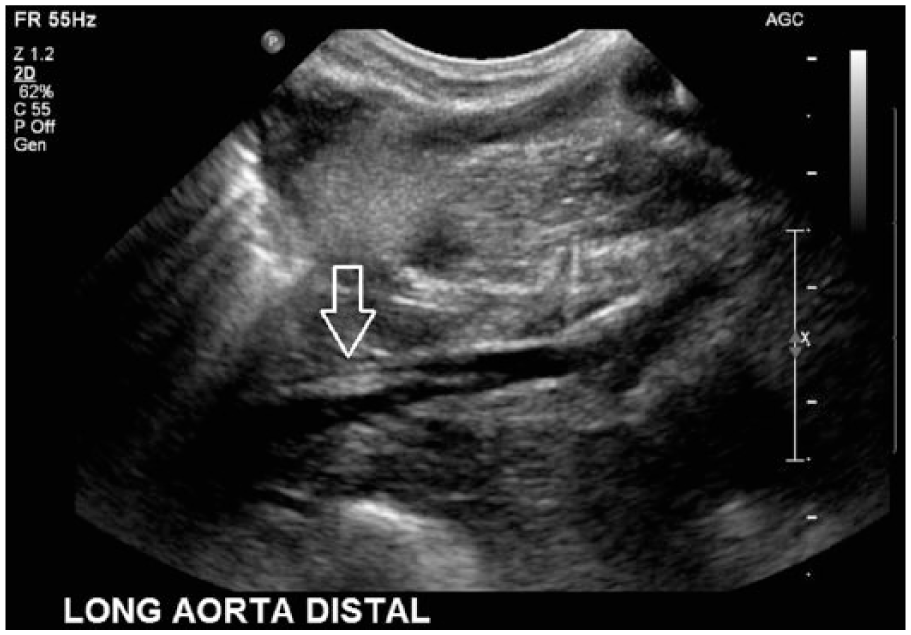
Gray-scale long-axis image of the abdominal aorta extending distally to the aortic bifurcation showing the extent of the hyperechoic line echo (arrow).
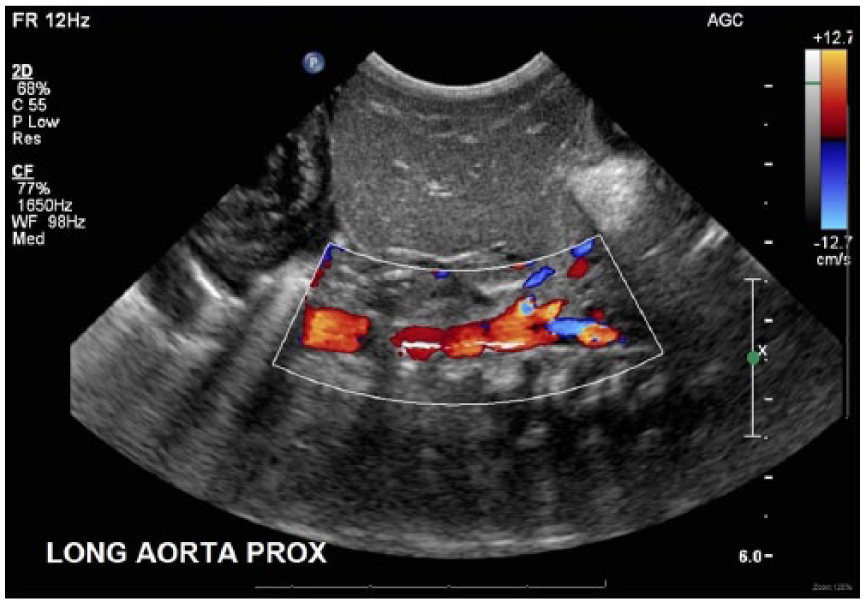
Color Doppler long-axis image of the abdominal aorta showing patency of the vessel with the nonocclusive hyperechoic line echo and color filling above and below the more hypoechoic proximal thrombus.
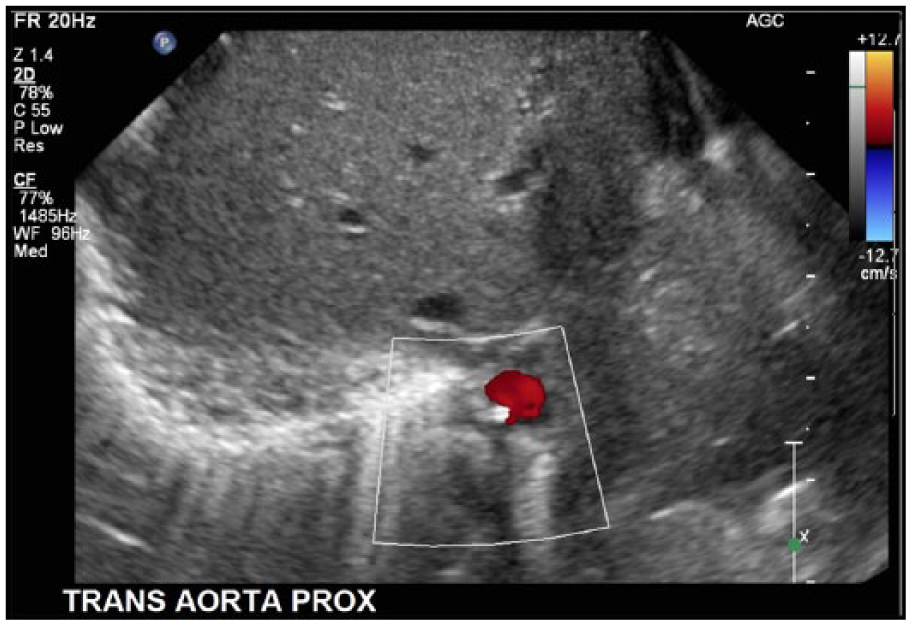
Color Doppler cross-sectional image of the abdominal aorta showing patency of the vessel with the nonocclusive hyperechoic line echo in the right posterolateral aspect of the lumen.
Discussion
The first umbilical artery catheterization was documented in the late 1950s. 9 The umbilical artery can be used for arterial access during the first 5 to 7 days of life but is rarely used beyond 7 to 10 days. Umbilical artery catheters (UACs) are routinely used to obtain arterial blood for diagnostic and investigative purposes as well as for intravascular fluid therapy in an infant. Another common use is monitoring blood glucose levels, often frequently in the first few hours of life, as well as monitoring arterial blood pressure.10,11 Typically, a 3.5F catheter (diameter of approximately 1.1 mm) is used in neonates weighing less than 1500 g and a 5F catheter (diameter of approximately 1.6 mm) in neonates weighing greater than 1500 g. Standardized tables are used to determine the insertion depth (length) of a UAC based on the neonate’s weight (Table 1). A UAC can be inserted in either the high position, with the catheter tip above the diaphragm, celiac artery, superior mesenteric artery (SMA), and renal arteries between T6 and T9, or the low position, with the catheter tip above L4 to L5 where the aorta bifurcates, between L3 and L4 near the origin of the inferior mesenteric artery (IMA). Placement in the high position is associated with lower complications.9-13 For example, a neonate weighing 1.5 kg would require a 5F UAC, and the catheter length in the high position would be 13 cm. Table 2 correlates neonatal weight with catheter length (insertion distance) for high positioning. Catheter position should be confirmed by noncontrast radiography following insertion. Umbilical artery catheterization is contraindicated in patients with omphalocele, omphalitis, cord anomalies, peritonitis, necrotizing enterocolitis, or vascular compromise to the kidneys or lower limbs.2,10,11
Calculation of Length (Insertion Depth) of Umbilical Artery Catheters (UACs).

High Position Umbilical Artery Catheter Placement: Distances at Which Catheters Must Be Inserted to Reach the Diaphragm.

The placement of a UAC is an essential technique in the care of ill neonates, such as those with respiratory distress and/or congenital malformations. When left in place for 5 to 7 days and then removed, UACs have been associated with thrombotic complications, which may include partial or complete occlusion in the aorta.11-15 These short- and long-term risks are well known, and the most severe include stroke, hypertension caused by renal artery thrombosis, and death.2,12,14 Because some of these risks persist long after catheter removal, the case has been made for the importance of long-term follow-up studies of patients who received UAC as infants.12,14 Possible treatments include anticoagulation therapy and surgical aortic thrombectomy.16-18
Studies regarding the incidence of aortic thrombus associated with umbilical artery catheterization indicate the utility of sonography as an initial means for interrogation of the neonatal abdominal aorta.5,13,14,19,20 The sonographer can perform a swift yet detailed survey of the entire anatomy, which can help identify and diagnose thrombus as well as foreign bodies. This will allow more specific patient care and potentially a better outcome for the individual. Sonographic interrogation of the abdominal aorta can be improved through patient fasting and use of a high-frequency transducer. If color Doppler filling is not apparent within the vessel lumen, power Doppler imaging should be used, as it is more sensitive to any slow flow that may be present, indicating nonocclusive thrombus. Thrombus varies in sonographic appearance from isoechoic to hypoechoic to hyperechoic, in part depending on whether the process is acute or chronic, although even acute thrombus may appear hyperechoic.5,19
A differential diagnosis that should be kept in mind is coarctation of the aorta, a congenital heart defect defined as a narrowing of the aorta. This may appear as a long linear segment or can be tubular, and it typically occurs in the aortic arch. Some investigators have reported that patients presenting with symptoms of coarctation were instead found to have aortic thrombus associated with umbilical artery catheterization.1,21,22 A pseudo-dissection of the aorta, an additional differential, would appear with a hypoechoic thrombus near the outer margin of the aorta with echogenic, laminated clot. Color Doppler evaluation would demonstrate disturbed, possibly turbulent, flow, and no intimal flap would be visualized as is common in a true dissection.1,12,20
Sonography is the primary imaging modality that has been used to identify neonatal aortic thrombosis, but its validity has not been rigorously established.12,20 In addition to sonography, aortography has been used to identify and diagnose vascular problems such as thrombosis.23,24 It is thought to be a safe, simple, and reliable technique for the early detection of thrombotic complications of UACs, and it is considered by many to be the gold standard for diagnosis.12,24 Computed tomographic angiography and magnetic resonance angiography can be used to detect thrombus,12,15,19 and in some cases there may be a role for radionuclide flow studies in diagnosing major aortic occlusion as well as for follow-up. 12 Further standardization of reporting and documentation of the complications of UAC as well as aortic thrombosis of the neonate are needed to provide the necessary data to create a standard of care for treatment.3,12 One proposed classification scheme is shown in Table 3. 3
Proposed Classification Scheme for Neonatal Aortic Thrombosis 3 .

Conclusion
Sonography provides a noninvasive, accurate diagnostic method to assess the entire abdominal aorta in neonates and can be used to determine the presence of thrombus or foreign bodies within the lumen. Because of the high risk of thrombus development within the neonatal abdominal aorta after umbilical artery catheterization and the resulting significant complications, sonographers must be able to identify this problem and differentiate its associated sonographic appearance from other possible diagnoses. In follow-up, sonography can be a useful adjunct for correlation with other imaging modalities.
Footnotes
Acknowledgements
The author acknowledges the assistance of Jennifer Bartley, RDMS, RVT, and Lynn Schluns, RDMS, RVT, in the preparation of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
