Abstract
Chronic lymphocytic leukemia (CLL) is the most prevalent hematologic malignancy of adults in the western hemisphere. Involvement of the spleen is not uncommon, often presenting as diffuse enlargement due to infiltration of neoplastic cells. A case is presented of an 81-year-old woman with a past medical history of CLL who was incidentally found to have multiple, focal splenic infiltrates on sonography. The patient had a normal sized spleen, which is a less common occurrence with splenic invasion by CLL. As current CLL guidelines discourage imaging but rely on evidence of organ involvement, this finding was critical in staging the patient properly. The sonographic appearance of this rare condition is described, and the advantages of sonography in its evaluation are discussed.
Introduction
Chronic lymphocytic leukemia (CLL) is the most prevalent hematologic malignancy of adults in the western hemisphere. 1 With a slight male predilection and a peak incidence ranging from the fifth to the eighth decade, this disease is responsible for more than 15 000 new cases and an estimated 4500 deaths per year. 2 Splenic involvement is a common finding and most frequently presents as diffuse enlargement due to infiltration of neoplastic cells. Imaging of the spleen most commonly demonstrates either generalized splenomegaly or miliary, low-attenuation, nodular foci. Very rarely, CLL can manifest as focal lesions within the spleen and is usually accompanied by splenic enlargement. A case is presented of an 81-year-old female with a past medical history of CLL in whom sonography incidentally showed multiple splenic infiltrates. As splenic involvement without splenomegaly is a rare occurrence, the use of sonography was helpful in staging the patient correctly.
Case Report
An 81-year-old female was referred to the emergency room because of a gradual onset of dysphagia and shortness of breath. She first noted difficulty swallowing medications and solid foods over the three weeks preceding her visit. Two weeks prior, she noted shortness of breath but denied fever, chills, cough, or sputum production. The patient’s medical record showed a history of type 2 diabetes mellitus, hypertension, and hyperlipidemia. She was also noted to have a history of CLL that had been stable for close to a year. The physical examination showed right anterior and posterior cervical adenopathy but was otherwise negative.
Laboratory tests were done and were significant for an increased white blood cell count of 116.7 × 103/μL (normal range, 4.5-10.0 × 103/μL), decreased hemoglobin of 11.2 g/dL (normal range, 12-15 g/dL), decreased hematocrit of 34.9% (normal range, 36%-44%), and a normal platelet count. In the presence of diagnosed CLL, this could indicate infiltration of bone marrow by leukemic cells resulting in an increased number of circulating lymphocytes and decreased production of healthy red blood cells. Medical renal disease was also suggested by an elevated creatinine of 1.69 mg/dL (normal range, 0.5-1.4 mg/dL), which had been normal at the patient’s last outpatient visit.
An abdominal sonogram was ordered to evaluate the patient for renal disease, as this had not been noted in the past. Her kidneys were normal sized but were echogenic bilaterally, suggesting medical renal disease. Incidentally, multiple well-defined masses were noted within a non-enlarged spleen, each with a hypoechoic halo, giving a targetoid appearance (Figures 1-4). Given the patient’s laboratory results, this was believed to represent splenic infiltration by lymphocytic cells.
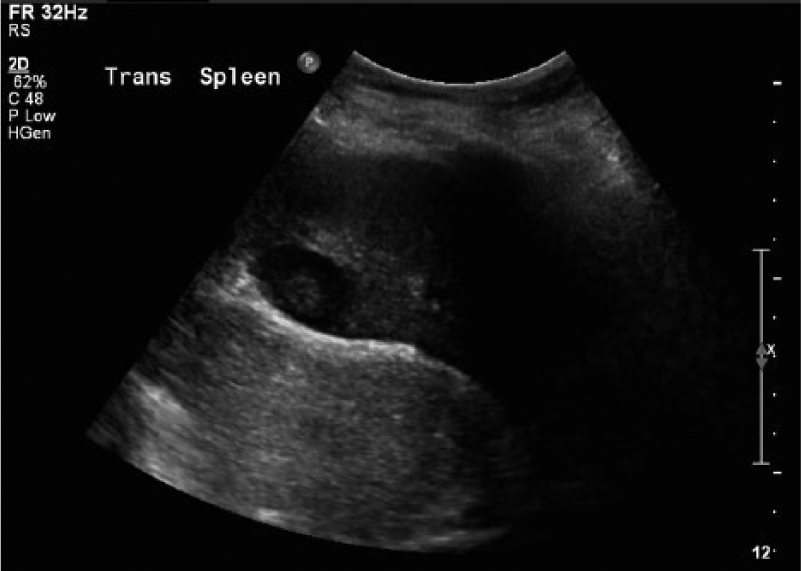
Transverse gray-scale image of the spleen showing a typical hypoechoic targetoid lesion.
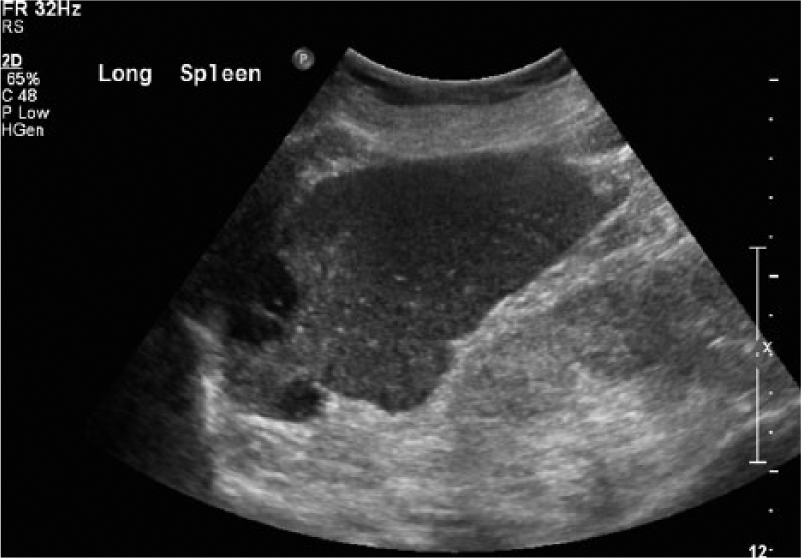
Long axis gray-scale image of the spleen showing multiple hypoechoic lesions.
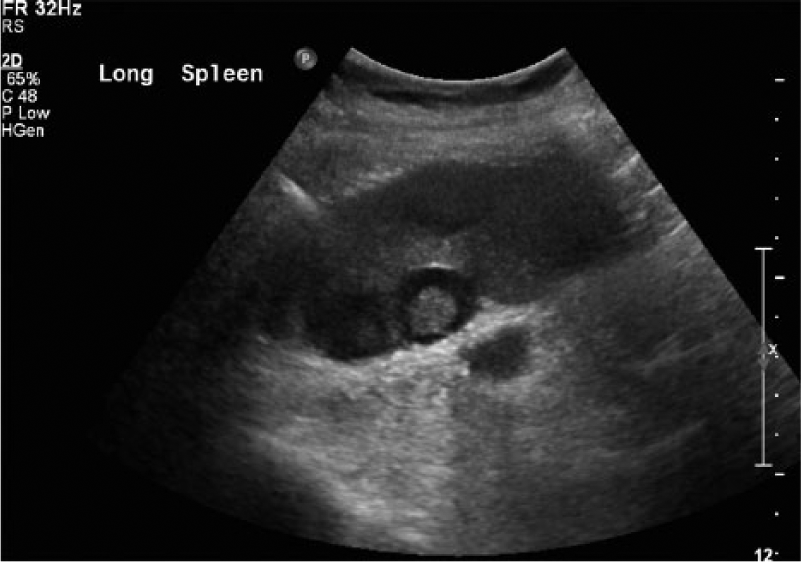
Long axis gray-scale image of the spleen showing multiple hypoechoic lesions; the central lesion shows a pronounced targetoid appearance.
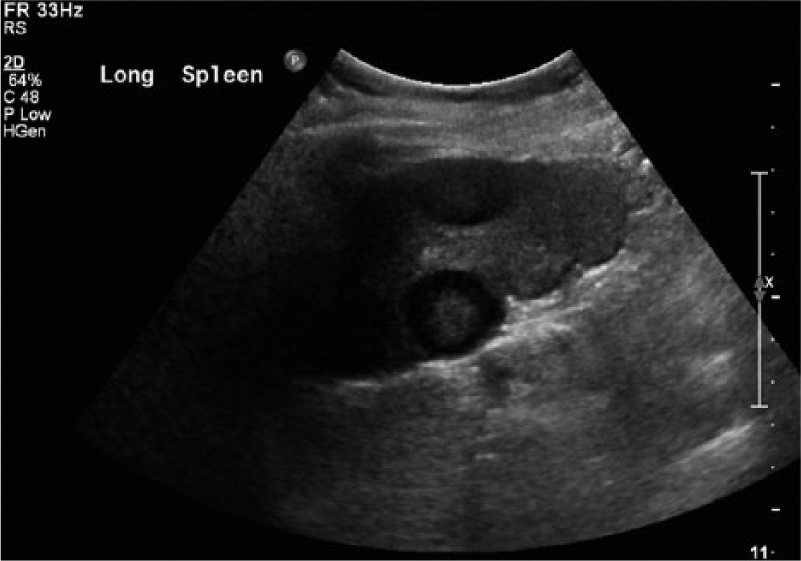
Similar long axis gray-scale image of the spleen showing multiple hypoechoic lesions with the central lesion showing a pronounced targetoid appearance.
The masses were further evaluated by a computed tomogram of the abdomen without contrast (Figure 5). A rounded 2 cm lesion was noted in the anterior spleen, and a rounded 3 cm lesion was noted in the posterior spleen. Both lesions were of lower density as compared to the surrounding parenchyma and were not present on previous examinations. Without contrast, the masses were unable to be completely characterized but were thought to represent lymphomatous infiltration of the spleen. In addition, a number of newly enlarged lymph nodes were noted to be present in the posterior mediastinum and the retroperitoneum as well as near the right heart border, inferior vena cava, and the splenic vein.
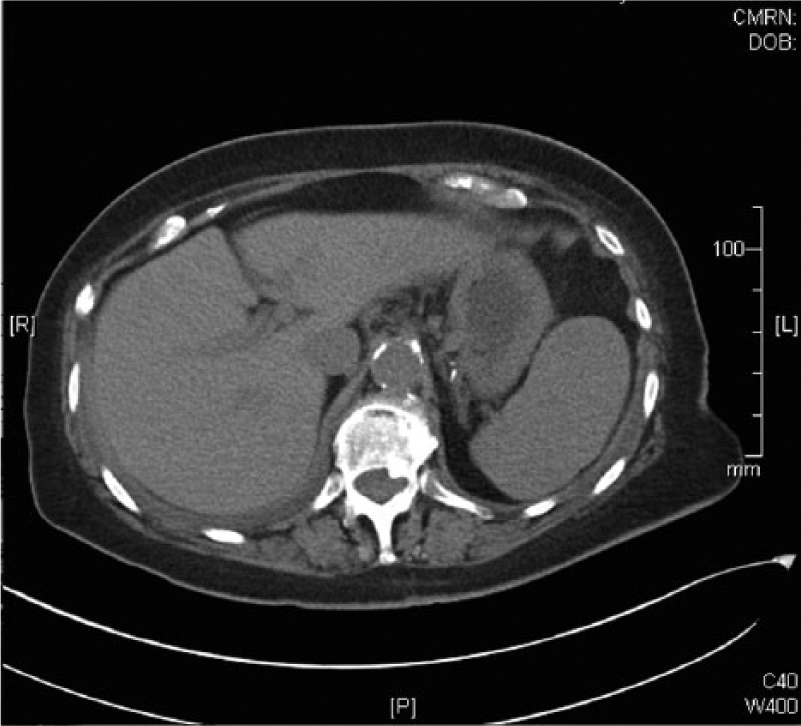
Axial computed tomographic scan of the spleen and liver showing a large hypoechoic lesion in the anterior aspect of the spleen.
Discussion
The etiology of chronic lymphocytic leukemia is currently unclear; however, genetic and familial factors have been implicated. This is evidenced by a 2% to 7% percent higher risk in first-degree family members when compared to the general population. 3 While no genetic mutation is considered specific for CLL, deletions of the long arm of chromosome 13 are seen in more than 50% of cases. 3 CLL is characterized by the accumulation of a neoplastic population of mature B-lymphocytes in the peripheral blood, bone marrow, and lymphatic tissues. Patients with this condition can be completely asymptomatic, whereas others can suffer from fatigue, weight loss, and adenopathy. As CLL progresses, adenopathy, hepatomegaly, and splenomegaly can develop, leading to anemia, thrombocytopenia, and autoimmune phenomena. 4 Mortality is often related to secondary infections from immune suppression related to bone marrow effacement and malignant transformation to diffuse large B-cell lymphoma. 5
Currently, the two most common methods to stage CLL are the modified Rai et al. 6 and Binet et al. 7 systems. The modified Rai et al. system categorizes patients into three different risk categories based on an increased number of lymphocytes or leukemic cells in the blood, anemia (using a cutoff of 11 g/dL), thrombocytopenia (platelet count less than 150 × 103/μL), or swelling of the lymph nodes, liver, or spleen. The Binet et al. system also categorizes patients into three different risk categories based on swelling of the lymph nodes, liver, and spleen as well as the presence of thrombocytopenia and anemia (using a cutoff of 10 g/dL). 8 These tests are performed clinically, relying only on the physical examination and laboratory tests.
Splenic involvement is a more prominent feature of the chronic leukemias and most commonly presents as diffuse, homogenous enlargement.9,10 The spleen can grow to a rather large size in CLL and is only surpassed in average size in patients with chronic myelogenous leukemia (CML). A study by Heiberg et al. 11 found splenic enlargement in 27 of the 84 cases of leukemia studied, with 7 cases demonstrating the spleen extending below the iliac crest. 11 The increased size of the spleen results in a propensity for nontraumatic rupture, which classically presents as abdominal pain referred to the left shoulder. However, the focal, targetoid lesions seen in the patient presented are a rare presentation of CLL and are more commonly seen in lymphoma. 12
Computed tomography (CT) in CLL can demonstrate a number of patterns: diffuse splenomegaly, low-attenuation nodules scattered throughout the liver and spleen, multiple nodules greater than 1 cm, and rarely, focal splenic lesions. 13 Related findings can include bacterial abscesses from immune suppression as well as occlusive and hemorrhagic infarcts. 14 While an incredibly useful tool, Heiberg et al. 11 noted that CT examinations of the spleen were more likely to reveal the organomegaly of more advanced disease. They concluded that CT was likely to be insensitive in identifying the subtle textural changes of early, occult disease. In addition, acute renal insufficiency can be seen in CLL due to leukemic invasion of the kidneys. 15 This limits the use of intravenous contrast in the evaluation of these patients, decreasing the specificity of findings and making CT less useful in the patient with CLL. 11
The sonographic appearance of CLL can also show several different patterns of disease, including diffuse splenomegaly, miliary nodules, multiple nodules greater than 1 cm, and solitary focal lesions. 16 The nodular and focal patterns are typically hypoechoic and rarely show the targetoid appearance noted in this patient. Occasionally, larger focal lesions can demonstrate internal necrosis, producing a cystic appearance. A study by Siniluoto et al. 17 compared the diagnostic accuracy of CT versus sonography when evaluating the spleen and found that sonography was superior to CT when evaluating malignant lesions of the spleen.
Current guidelines for the diagnosis and staging of CLL discourage the use of imaging and rather rely on the physical examination and laboratory testing. However, a randomized controlled trial by Tamayo et al. 18 showed wide interobserver variability in the detection of splenomegaly by physical examination and considered it to be relatively insensitive. Thrombocytopenia can also be used as an indicator of splenic dysfunction, although this can be confounded by both invasion of bone marrow by leukemic cells and autoimmune phenomena. Prior to the sonography done in this case, the patient would have been classified as intermediate risk by the modified Rai et al. 6 criteria and low risk by the Binet et al. 7 system. After the sonogram, the patient would have remained in the same risk class by the modified Rai et al. criteria, but she would have been classified as intermediate risk in the Binet et al. system. The patient’s oncologist responded to this by escalating the patient’s therapy, which has resulted in the normalization of the patient’s laboratory findings.
Conclusion
Sonography has been shown to be effective in differentiating malignant lesions of the spleen, and it is an important diagnostic adjunct to the physical examination when assessing splenomegaly. In addition, it is especially useful in patients with medical renal disease that precludes administration of intravenous contrast, which decreases the diagnostic efficacy of CT. Taking into account its lack of ionizing radiation, portability, lower cost, and ability for same day turnaround, it is likely that sonography could be a useful tool in the assessment and staging of CLL. The patient presented was staged accurately thanks to the incidental sonographic findings, and it is likely that other cases could similarly benefit. Future research could better elucidate the benefits and pitfalls of this approach.
Footnotes
Acknowledgements
The authors would like to thank University of Missouri Health Care System and the Department of Radiology for their help in providing the records information and images for this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
