Abstract
Umbilical vein varix (UVV) is defined as a focal enlargement of the umbilical vein and represents approximately 4% of fetal umbilical cord malformations. The reported neonatal outcome of fetuses with UVV varies widely due to its rarity, hence the small sample sizes of the case series in the literature. Earlier studies reported high fetal mortality, but more recent reports have demonstrated no association between UVV and intrauterine fetal death. A recent study has described a possible association between UVV diagnosed prenatally and child developmental delay. The present study of fetuses with UVV was done to evaluate and compare the levels of triple test serum biomarkers used for Down syndrome screening (human chorionic gonadotropin, α-fetoprotein, and unconjugated estriol) between a group of fetuses with uneventful obstetric outcome versus a subgroup of children with developmental delay.
Umbilical vein varix (UVV) is defined as a focal enlargement of the umbilical vein and represents approximately 4% of fetal umbilical cord malformations.1–3 This rare anomaly, which was first described in autopsies of stillborn infants, 4 can occur either intra- or extra-abdominally.1–3 Varicose dilation of the umbilical vein may be identified by sonographic examination, appearing as an anechoic cystic mass between the abdominal wall and the inferior part of the liver. Its vascular nature should be confirmed by Doppler ultrasonography to differentiate it from other intra-abdominal cystic lesions, such as urachal or choledochal cyst, distended gallbladder, and others.
There is no single universally accepted criterion for the in utero diagnosis of UVV. Some suggest an umbilical vein diameter of >9 mm or an index portion of the umbilical vein that is at least 50% wider than the nondilated portion. Others have defined it as a measurement that is more than two standard deviations above the mean for gestational age. 5 Sepulveda et al 6 defined UVV as a focal dilatation of the intra-abdominal portion of the umbilical vein, with a transverse diameter at least 1.5 times greater than the diameter of the intrahepatic umbilical vein. Using this criterion (which avoids the umbilical vein diameter relationship to gestational age), the authors identified cases of UVV as early as 22 weeks’ gestation, with a median gestational age of 27 weeks.
The reported neonatal outcome of fetuses with UVV varies widely due to its rarity, hence the small sample sizes of the case series in the literature. 4 Earlier studies reported high fetal mortality as frequent as 44%.2,5–7 However, more recent studies have demonstrated no association between UVV and intrauterine fetal death.3,8–11 The most common related sonographic abnormalities to UVV were malformation of the cardiovascular system,6,7 fetal hydrops,5,7 fetal anemia-related complications,7,11 umbilical vessel abnormalities,7,11 and intrauterine growth restriction.11,12 Trisomy 21, 18, and 9 and triploidy have also been described in association with UVV.3,5–8,12–15
The maternal triple serum markers conducted in the second trimester for Down syndrome (DS) screening are human chorionic gonadotropin or its free β-subunit (βhCG), α-fetoprotein (AFP), and unconjugated estriol (uE3). Elevated levels of maternal serum hCG (MShCG) and low levels of AFP and uE3 have been found to be associated alone or in combination with an increased risk for fetal DS.16,17 However, unexplained elevations of maternal serum AFP (MSAFP) and/or hCG exist in approximately 1% of the obstetric population and are associated with an increased risk of adverse pregnancy outcome, including miscarriages, low birth weight, preterm labor, abruptio placenta, preeclampsia, intrauterine fetal death, and a wide spectrum of fetal and placental malformations.18–20
While reviewing the literature, we found only one anecdotal case report describing a combination of extra-abdominal UVV, elevated MSAFP of a 2.9 multiple of the normal median (MoM), and mesenchymal dysplasia of the placenta verified on postpartum histopathological examination. Obstetrical and postnatal courses were uneventful. 21 Recently, our group described a possible association between UVV diagnosed prenatally and child developmental delay. 22 In the present study, we have attempted to evaluate and compare the levels of triple test biomarkers between a group of fetuses with uneventful obstetric outcome versus a subgroup of children with developmental delay. All fetuses were diagnosed with UVV prenatally. To our knowledge, this profile of the triple test serum markers in patients diagnosed as having UVV has not been previously reported.
Methods
The study design, patient population, methods, and baseline characteristics have been previously reported, 22 and the study was approved by the institutional Clinical Research Committee prior to initiation. The study included fetuses with UVV that were referred to our Ultrasound Unit between 2005 and 2011. All underwent a detailed anomaly scan in the community. For the purpose of this study, only singleton pregnancies that were diagnosed with fetal intra-abdominal UVV were included. Prenatal workup of this referral group was composed of fetal echocardiography, a targeted ultrasonographic examination for possible associated malformations, and genetic counseling. Follow-up included a biweekly fetal well-being assessment using ultrasonography with Doppler flow studies, paying particular attention to fetal growth and vascular flow patterns within the variceal component for evidence of turbulence or thrombi. During the first years of our study, planned delivery was conducted at 36 to 37 weeks’ gestation after lung maturity was proven by amniocentesis. Later, induction of labor or planned delivery was conducted only upon obstetric indication and not because of the presence of the varix.
After delivery, all newborns were assessed by a pediatric specialist to rule out any additional anomalies or dysmorphic features. For the current study, we retrieved data from the patients’ medical records on the second-trimester triple test results. Data regarding patient demographics, pregnancy, and obstetrical outcome as well as infant and child follow-up were obtained from the medical records in the obstetric, pediatric, and child developmental clinics and by telephone interview to the parents. 22
Developmental delay was assessed by a telephone interview using a questionnaire based on the Ages and Stages Questionnaire. If the child’s score was below the cutoff in one or more domain(s), the families were offered a formal cognitive assessment by our pediatrician and developmental psychologist team. 22
Screening Tests
The second-trimester screening test was derived from the combination of triple serum markers and maternal age, and results were calculated using commercial software. The maternal serum markers evaluated in this study were maternal serum hCG or its free β-subunit (FβhCG), AFP, and uE3. The serum samples were tested in a routine analytical run together with regular maternal serum samples, all in the same prenatal Down syndrome screening program. Testing was carried out in a manner that was blinded to group classification; that is, samples from our study cases and those of other pregnant women were assessed in the same laboratory. The measured marker levels were expressed as multiples of the gestation-specific normal medians (MoM). Median values for each serum analyte were calculated against completed menstrual weeks and adjusted for maternal weight. We compared results with reference MoM values that were calculated from our own local population as established in Zer Medical Laboratories (certified and authorized by the Ministry of Health, Israel).23,24
Statistical Analysis
Standardized kurtosis showed that the data were derived from a normal distribution and were expressed as a mean and standard deviation. Frequencies were expressed as percentages. Statistical analysis was performed using the Fisher exact test for intergroup comparison of developmental delay. Student t test was used to compare the second-trimester markers between different groups. AFP and hCG concentrations were logarithm transformed to follow normal distributions; uE3 showed normal Gaussian distribution.
A one-sample t test was applied to compare the results among the study and control groups to normal population values. A P value <.05 was considered significant. Calculations were performed in the statistical laboratory at Tel Aviv University using SPSS software (version 13; SPSS, Inc, an IBM Company, Chicago, Illinois).
Results
Overall, 36 fetuses were identified with UVV in our patient population. Of these, 31 (86%) were included in our study, excluding five (14%) twin pregnancies. UVVs were identified at a range of 20 weeks’ to 36 weeks’ gestation (mean ± SD, 30.5 ± 4.5 weeks) (Figure 1). The average UVV diameter was 11.9 mm (range, 8–17 mm). Mean maternal age of the UVV group was 31.9 ± 4.3 years (range, 23–44 years). Gestational age at delivery was 37.2 ± 2.1 weeks (range, 32–41 weeks’ gestation). Newborn birth weight was 2812.7 ± 526.7 g (range, 1710–3810 g). Apgar scores at 1 and 5 minutes were 8.8 ± 12 and 9.8 ± 0.6, with a range of 2 to 10 and 7 to 10, respectively. Child age at follow-up evaluation was 2.5 ± 1.8 years (range, 0.2–6.8 years).
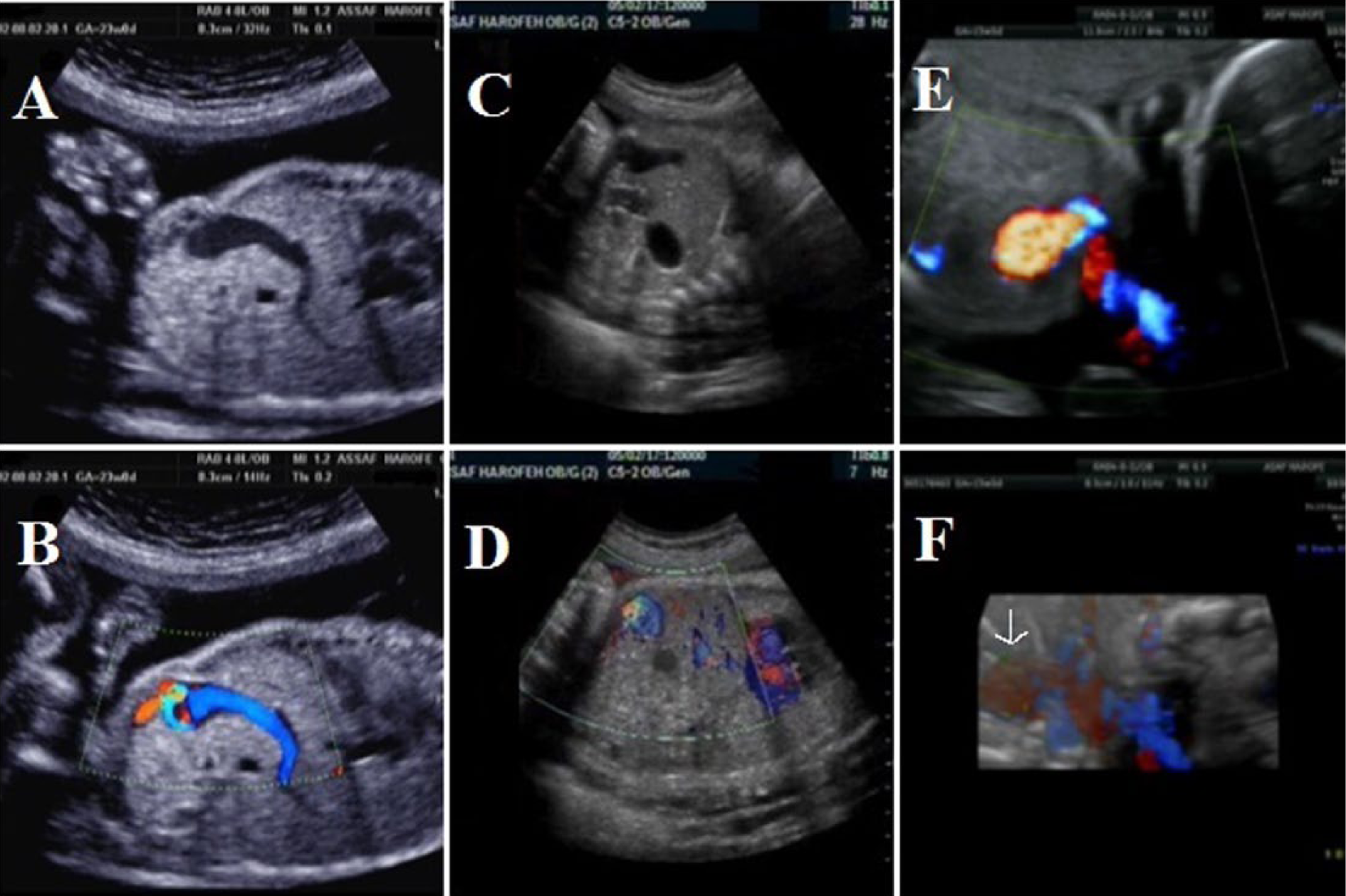
Plate of three different cases of intrahepatic umbilical vein varix (UVV). (A, B) Ultrasonographic image (transverse view) of UVV having a 12-mm diameter at 23 weeks’ gestation and the corresponding color Doppler flow image. (C, D) A second case of a transverse image of UVV and the corresponding color Doppler image. A third case (E, F) at 23 weeks’ gestation showing the color Doppler image and a reconstructed 3D image showing the enlarged umbilical vein (arrow).
In six (16.7%) cases of UVV, other anomalies were detected on ultrasonographic examination. Mild to moderate hydronephrosis was the most common finding, accounting for three (50%) of these abnormalities. One case of UVV was diagnosed with DiGeorge syndrome, one with an atrial septal defect/ventricular septal defect, and one with a single umbilical artery. There were no cases of intrauterine fetal or neonatal death.
Median hCG, AFP, and uE3 levels are shown in Table 1. The median hCG, AFP, and uE3 levels of women in the entire UVV study group were not significantly different from the control reference group. In addition, these markers were not significantly different between the two subgroups of developmental delay versus normal development cases as well as compared with the overall and control laboratory data.
Comparison of Triple Test Screening Markers Between UVV Study Group and Children With Developmental Delay and Normal Pregnancy Outcome Based on Findings at the Telephone Interview, All Compared With the Reference Laboratory Values (P = NS).

For AFP and hCG: comparison of mean log10 for each marker. For uE3: comparison of the mean. Superscripted letters indicate the following: P value between a and d = .198, P value between e and h = .146, P value between i and l = .125, P value between b and d = .4, P value between f and h = .052, P value between j and l = .424, P value between c and d = .317, P value between g and h = .739, and P value between k and l = .052. AFP, α-fetoprotein; hCG, human chorionic gonadotropin; MoM, multiple of the median; NS, not significant; uE3, unconjugated estriol; UVV, umbilical vein varix.
Discussion
With the improvements in image quality and Doppler capabilities in ultrasonography over the past years, the frequency and awareness of the diagnosis of UVV have increased substantially. In the current study, the MoM levels of triple test biomarkers in fetuses diagnosed with UVV were not significantly different among a group of children with uneventful pregnancy outcome compared with a subgroup of children with developmental delay as well as in comparison to a normal reference group.
Some studies have suggested that placental pathology may be associated with increased levels of hCG or elevated AFP. Hypoxia increases hCG overproduction in trophoblastic cells cultured in vitro. 25 Placental pathology at delivery, such as infarction, ischemic changes, villitis, and intervillous thrombosis, has been associated with increased hCG concentrations. 26 All of these placental pathologies may cause inadequate trophoblastic remodeling of the maternal uterine vasculature with an absence of physiologic changes in the spiral arteries, leading to placental hypoxia and hCG overproduction.
Other studies have suggested that placental pathology may permit a more rapid diffusion of AFP from the fetoplacental compartment to the maternal compartment, resulting in elevated AFP levels in maternal serum.27,28 This hypothesis is further supported by Mulch et al, 21 who presented a case report where UVV was associated with an elevated maternal serum AFP on prenatal testing and mesenchymal dysplasia on pathological evaluation of the placenta. In the patient’s case report, they found long-standing mural thrombi in the varix on postpartum histopathological examination that were not detected on prenatal ultrasound. Vascular flow, however, was never fully compromised by the thrombi, as verified by Doppler flow studies and normal fetal growth. Their study emphasized the fact that a meticulous examination of the placenta and placental structures should undergo careful examination, both gross and microscopic, following delivery.
Developmental delay is a term that generally refers to children who do not show the expected developmental properties according to their age. The US Census Bureau has reported a 4.5% rate of developmental disability among children up to five years of age. 29 The precise mechanism by which UVV may be associated with postnatal developmental delay abnormalities in apparently normal fetuses is yet unknown. Recently, our group proposed various possible hypotheses. 22
Some of the proposed mechanisms that may cause in utero fetal death may also be relevant for postnatal developmental delay. UVV predisposes to thrombosis, due to significant abnormal flow patterns within the varix such as flow separation, turbulent blood flow, and shearing stress.7,30 One hypothesis suggests an early episode in which fragmentation and dispersal of the microthrombi occurs with paradoxical embolization through a patent foramen ovale. This can even be supported by the paradoxical embolism phenomenon, in which systemic passage of thrombi through an interatrial conduit (right-to-left shunt) is thought to be the mechanism of stroke in patients with patent foramen ovale (PFO). Small blood clots and microaggregates of venous origin may cross the PFO, thereby escaping lysis in the lungs and leading to clinically significant sequelae in the brain. The shunting of these particles could occur after a Valsalva maneuver or in relation to a chronic condition causing increased pulmonary artery pressure. 31 An association between thrombosis of the UVV and disseminated intravascular coagulation also has been recently reported, suggesting that postnatal developmental delay abnormalities may be a result of consumption of coagulation factors on the thrombogenic surface of the UVV segment, with the subsequent occurrence of micro-infarctions in various areas throughout the brain.32,33
Another hypothesis by which UVV may be associated with postnatal developmental delay abnormalities may mimic one or several of the mechanisms by which a varicocele (an abnormal dilation of the spermatic veins within the scrotum) can cause abnormalities of the testis and semen parameters by increased scrotal temperature, reflux of toxic metabolites, decreased volume of blood flow, and anoxia. 34
Despite the above hypotheses relating pre- and postnatal compromise to the fetal varix formation and its sequelae, early imprints of the effects of the varix were not found in the mid-gestation triple test profile in our entire study population or in the subgroup of the developmentally delayed children. This may be explained by the supposition that some of the adverse mechanisms previously mentioned develop later in gestation after this test is conducted or that there is simply no relation between the two events.
In conclusion, this is the first study to evaluate and compare the levels of triple test biomarkers between fetuses diagnosed with UVV prenatally and later found to have developmental delay in postnatal life and a subgroup of children with UVV and uneventful obstetric outcome. Overall, the entire study cohort, as well as the two subgroups, had triple test results similar to those found in general population. Additional studies should be conducted to verify our results.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
