Abstract
Background and Aim:
We prospectively investigated, using sonographic methods, whether hepatitis C infection in compensated patients is related to endothelial dysfunction of the brachial artery, a potential indicator of subclinical atherosclerosis.
Methods:
Brachial artery flow-mediated dilatation (FMD) was used as a noninvasive test to measure the endothelial function of the brachial artery among the cases and the controls. The peak systolic velocity (PSV) and the resistive index (RI) were also measured. Multivariate statistics were used to compare the FMD, PSV, and RI in 28 cases (diagnosed with hepatitis C) and 14 healthy controls, all of whom were without cardiovascular risk factors or a history of cardiovascular disease.
Results:
Flow-mediated dilatation (baseline to 1 minute post-cuff inflation) was positively correlated with the patient’s body mass index (BMI) (Pearson’s r = .309, P = .025). When the BMI was accounted for using analysis of covariance (ANCOVA), there was no evidence for a significant difference between the mean FMD of the cases versus the controls (F = 0.11, P = .598). The mean FMD was 6.6% (95% confidence interval [CI], 3.1%-10.2%) among the cases and 7.3% (95% CI, 1.1%-13.6%) among the controls. Peak systolic velocity and RI (baseline to 3 minutes post-cuff inflation) were not positively correlated with BMI. The mean PSV was 0.57 m/s (95% CI, 0.51-0.66) for the cases and 0.64 m/s (95% CI, 0.56-0.72) for the controls; repeated measures analysis of variance (ANOVA) indicated no significant difference (F = 1.65, P = .208). Similarly, the mean RI was 0.87 (95% CI, 0.85-0.89) among the cases and 0.64 (95% CI, 0.82-0.88) among the controls, with no significant difference (P = .772).
Conclusion:
Our results show that hepatitis C positivity in compensated patients, in the absence of cardiovascular risk factors, does not appear to be independently associated with endothelial dysfunction when objectively analyzed by brachial artery flow-mediated vasodilatation.
Introduction
Several studies have suggested a link between atherosclerotic disease and persistent infection or seropositivity of certain micro-organisms. Specifically, a potential association between atherosclerosis and hepatitis C virus (HCV) has been suggested, but this is still controversial. Some studies have suggested that HCV seropositivity and the serum presence of the HCV core protein are associated with carotid artery plaques, elevated carotid intimal thickness, and increased coronary atherosclerosis.1–3 Others have indicated that seropositivity to HCV is associated with a favorable serum lipid profile, low cholesterol, diminished low density lipoprotein, and normal carotid intimal media.4,5 Because previous studies have not provided definitive evidence reflecting a link between atherosclerosis and hepatitis C, other sources of evidence need to be explored. Although it is known that endothelial dysfunction in arteries is a key process in the development of atherosclerosis,6,7 no links between endothelial dysfunction and compensated hepatitis C have yet been established. The purpose of this study was to prospectively investigate whether or not there is an association between hepatitis C infection in compensated patients and the early stages of pathogenesis of atherosclerosis, as indicated by measurements of the endothelial function of the brachial artery.
Normal arterial endothelium function is essential for the maintenance of vascular tone and participates in the regulation of blood flow. In response to physical and chemical stimuli, the endothelium regulates vascular tone through the release of vasodilators such as nitric oxide. The normal response of an artery to an increase in shear stress such as that produced by increased blood flow (hyperemia) is endothelium-dependent dilatation. The stronger the endothelial response, the greater the resulting vasodilatation. This phenomenon is termed flow-mediated dilatation (FMD) and its absence or a diminished response are potential indicators of subclinical atherosclerosis.8,9 Flow-mediated dilatation can be measured noninvasively using high frequency ultrasound 10 ; however, the interpretation of FMD measurements is difficult, as it may vary significantly depending on the duration and intensity of the hyperemia, the baseline diameter of the artery, and the ethnicity, sex, age, body mass index (BMI), and mood (overall vasomotor tone) of the patients.11–13 The peak systolic velocity (PSV) and the resistive index (RI) may also reflect endothelial dysfunction. The PSV identifies arterial blockage at a site of stenosis, with a higher PSV implying a greater degree of stenosis, and this may be associated with obesity. 14 The RI (defined as the PSV minus the end diastolic velocity / PSV) is a measure of the distal capacity for blood flow runoff and is another vascular parameter that might have value to detect endothelial dysfunction. 15
In light of previous studies concerning links between atherosclerosis and hepatitis C that have produced conflicting results, this study used sonographic techniques to prospectively investigate whether hepatitis C infectivity in compensated patients is related to the endothelial dysfunction of the brachial artery, expressed in terms of the variance in FMD, PSV, and RI. We compared a sample of cases (diagnosed with compensated hepatitis C infection) and healthy controls, all of whom were without cardiovascular risk factors or a history of cardiovascular disease. The possible confounding effects of age, sex, and BMI were also considered. The effects of BMI were considered to be particularly important because of the known positive associations between BMI and atherosclerotic risk factors.16,17
Materials and Methods
This prospective case-control study was conducted at the Community Regional Medical Center (CRMC) in Fresno, California. The protocol was approved by both the Institutional Review Board of the University of California, San Francisco, in Fresno and the University of California, San Francisco, in San Francisco. All patients gave written consent. The inclusion criteria for the cases were (a) adult patients (> 18 years of age) screened for cardiovascular risk factors from the gastroenterology clinics at CRMC between 2009 and 2011; (b) patients diagnosed with compensated chronic hepatitis C infection, having tested positive for either HCV antibody or for qualitative or quantitative HCV RNA, and/or those with a liver biopsy confirming the diagnosis of hepatitis C. The exclusion criteria were (a) patients with a history of heart disease (defined as prior diagnosis of cardiovascular or peripheral vascular disease; history of stroke, congestive heart failure, or acute coronary syndrome; uncontrolled hypertension defined as systolic blood pressure > 160 mmHg; dyslipidemia defined as triglycerides [TG] > 400 mg/dL; active current smokers; uncontrolled diabetes defined as hemoglobin A1C [HbA1C] > 8 mmol/mol); (b) patients with chronic liver disease from other causes, such as alcohol intake > 20 g/d, viral infections other than HCV, autoimmune hepatitis, or liver disease from medication use; (c) patients with decompensated cirrhosis, indicated by evidence of portal hypertension, including ascites, splenomegaly, active variceal bleeding, or evidence of synthetic liver dysfunction on laboratory evaluation, that is, international normalized ratio (INR) > 2, platelets < 50 000/mm3, hemoglobin < 8 g/dL, and low albumin < 2.5 g/dL; and (d) patients with chronic renal failure (creatinine > 3 mg/dL), cancer, pregnancy, HIV, or active infections/sepsis and those taking immune suppressants.
During the study period, 28 cases (hepatitis C infected patients) and 14 controls were enrolled and underwent the tests on their brachial arteries. All control subjects were healthy volunteers, with no history of HCV infection, chosen to match with the patients enrolled for age, sex, and BMI.
At the time of enrollment, clinical and anthropometric data were collected, including a brief medical history, history of current medications, a questionnaire regarding the patient’s alcohol intake and smoking history, blood pressure recordings, and BMI (kg/m2). A 12-hour fasting blood draw was required within 12 months prior to enrollment, which had to include a complete blood count, complete metabolic panel, triglyceride levels, HbA1C, insulin levels, and an INR.
Brachial artery FMD was used as a noninvasive test to measure the endothelial function of the brachial artery among the cases and the controls. In our study, reactive hyperemia was achieved by inflating a pneumatic cuff wrapped around the forearm to a blood pressure of 200 mmHg for 2 minutes, followed by a rapid deflation. This caused a transient decrease in distal peripheral vascular resistance and a period of hyperemic flow in the brachial artery. A high-resolution ultrasound system (iU22; Philips Healthcare, Bothell, Washington, USA) with a linear transducer (5-15 MHz) was used to quantify the arterial diameter changes. The arterial diameter was measured at baseline and at 1 minute and then 3 minutes after cuff deflation. The FMD was expressed as the relative (percentage) increase in brachial artery diameter during hyperemia, defined as 100 × (post-hyperemia diameter − basal diameter) / basal diameter. Angle-corrected spectral Doppler waveforms were recorded to determine blood flow velocity. The PSV (m/s) and RI were also measured pre-inflation and then at 1 minute and 3 minutes post-inflation to evaluate hemodynamics of the brachial artery.
Measurements were made on stored images offline using an Agfa HeartLab® System (Agfa Healthcare, Greenville, South Carolina, USA). To avoid observational bias, all the sonograms used for the study were performed and evaluated by a single expert ultrasonographer, head of the ultrasound department, and were read in a blinded fashion as to the patient group.
Data Analysis
No significant relationships could be found between the demographic factors (sex and age) and the vascular measurements (FMD, PSV, and RI), so sex and age were not considered as statistical confounders. Because the brachial artery diameters and FMD measurements were significantly correlated with the BMI of the patients, and BMI is associated with risk factors for atherosclerosis, the possible confounding effects of BMI were taken into account. Consequently, the measurements to compare the cases and the controls are presented as means ± 95% confidence intervals (CIs) partitioned with respect to three BMI (kg/m2) categories (normal weight ≤ 25; overweight = 26-30; obese > 30).
Statistical analysis was done using either analysis of covariance (a measure of how variables change together and the strength of the relationship between them, useful when there are dependent variables within levels of an independent variable) or analysis of variance (a more direct measure of the differences between groups), as appropriate. Repeated measures analysis of covariance (ANCOVA), with BMI held statistically constant, was used to compare the temporal variation in the mean brachial artery diameters within and between the controls and cases. ANCOVA (also controlling for BMI) was used to compare the mean FMD between the cases and the controls. Because BMI was not correlated with either PSV or RI, repeated measures analysis of variance (ANOVA, not controlling for BMI) was used to compare the temporal variation in the mean RI and PSV, within and between the cases and controls, partitioned by the three BMI categories. The significance level was set at α = .05. The effects sizes (η2 = the proportion of the variance explained) were also estimated, to provide indices of clinical significance. 18
Results
The baseline characteristics of the two study groups (n = 28 cases with compensated hepatitis C seropositivity and n = 14 healthy controls) are described in Table 1 and were noted to be matched. There were no significant differences between the cases and the controls with respect to age, BMI, or the proportions of males and females. Among both the cases and the controls, the mean levels of serum low density lipoprotein cholesterol, high density lipoprotein cholesterol, TG, albumin, bilirubin, creatinine, and HbA1C were within normal ranges.
Baseline Characteristics of Cases with Hepatitis C versus Healthy Controls.
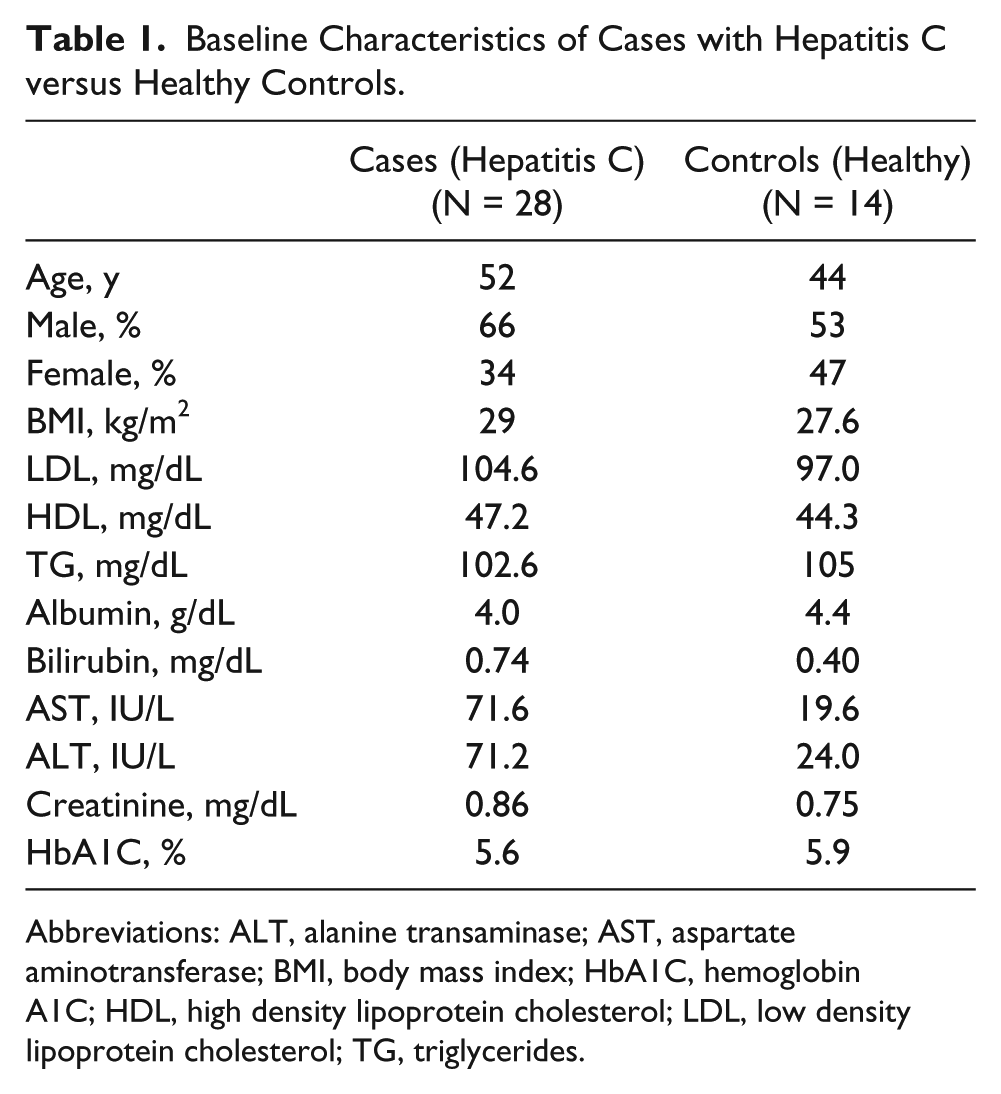
Abbreviations: ALT, alanine transaminase; AST, aspartate aminotransferase; BMI, body mass index; HbA1C, hemoglobin A1C; HDL, high density lipoprotein cholesterol; LDL, low density lipoprotein cholesterol; TG, triglycerides.
The temporal variation in the mean brachial artery diameter ± 95% CI from 0 to 3 minutes (post-cuff inflation) with respect to BMI among the cases and controls is illustrated in Figure 1. The brachial artery diameter was positively correlated with BMI (Pearson’s r = 224, P = .023), justifying the use of a repeated measures ANCOVA, holding BMI constant. The within-subjects effects were dominated by a significant time × BMI interaction (F = 3.84, P = .034, η2 = .194). The combined effects of time and BMI were indicated by the mean brachial artery diameter remaining relatively constant among the normal weight controls (3.92-3.98 mm) and cases (4.70-4.90 mm). In contrast, among the obese controls, the mean diameter increased from 4.30 to 4.70 mm, whereas among the obese cases, the mean diameter increased from 4.41 to 5.06 mm. There was also a significant between-subjects effect between the cases and controls (F = 7.25, P = .011, η2 = .164). When averaged over time for 3 minutes, after controlling for BMI, the mean brachial artery diameter among the cases (M = 4.94 mm; 95% CI, 4.76-5.13) was significantly higher than the controls (M = 4.33 mm; 95% CI, 4.10-4.56).
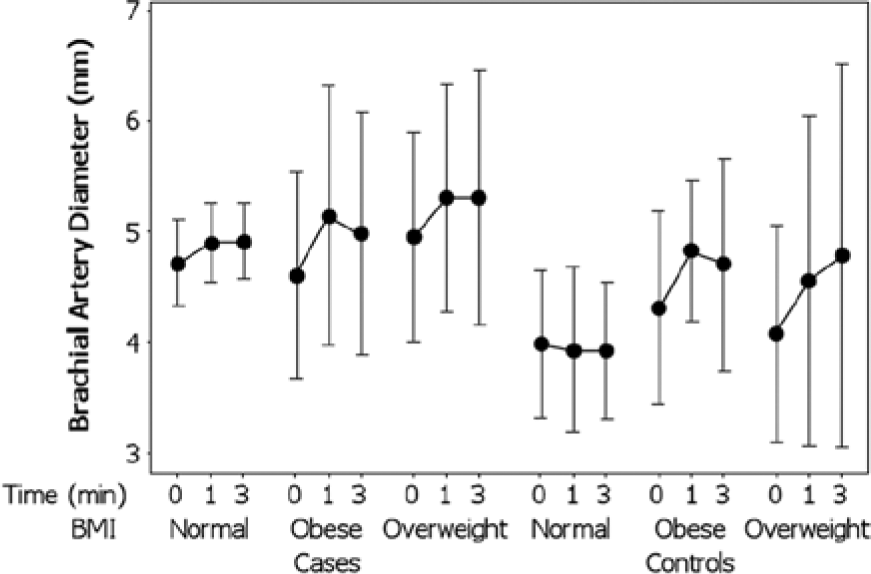
Temporal variation (post-cuff inflation) in mean brachial artery diameter ± 95% confidence interval with respect to body mass index among cases and controls.
The variation in mean FMD ± 95% CI with respect to BMI among the cases and controls is illustrated in Figure 2. The FMD measurements (i.e., the brachial diameters at 1 minute post-cuff inflation, minus the baseline diameters at time 0, expressed as percentages) were positively correlated with the BMI of the patients (Pearson’s r = .309, P = .025). Because BMI and FMD were correlated, the differences in the FMD measurements between the groups (cases vs. controls) were analyzed using ANCOVA, with BMI held constant. The effect of BMI on FMD was significant (F = 8.10, P = .007). The mean FMD varied from a minimum of 2.9% (95% CI, –1.2% to 6.9%) among the normal weight patients, to 8.9% among the overweight patients, reaching a maximum of 12.2% (95% CI, 7.2%-17.3%) among the obese patients. There was, however, no evidence for a significant difference between the mean FMD of the cases versus the controls (F = 0.11, P = .598) with no interactions between the groups. The mean FMD was 6.6% (95% CI, 3.1%-10.2%) among the cases and 7.3% (95% CI, 1.1%-13.6%) among the controls.
Variation in mean flow-mediated dilatation ± 95% confidence interval with respect to body mass index among cases and controls.
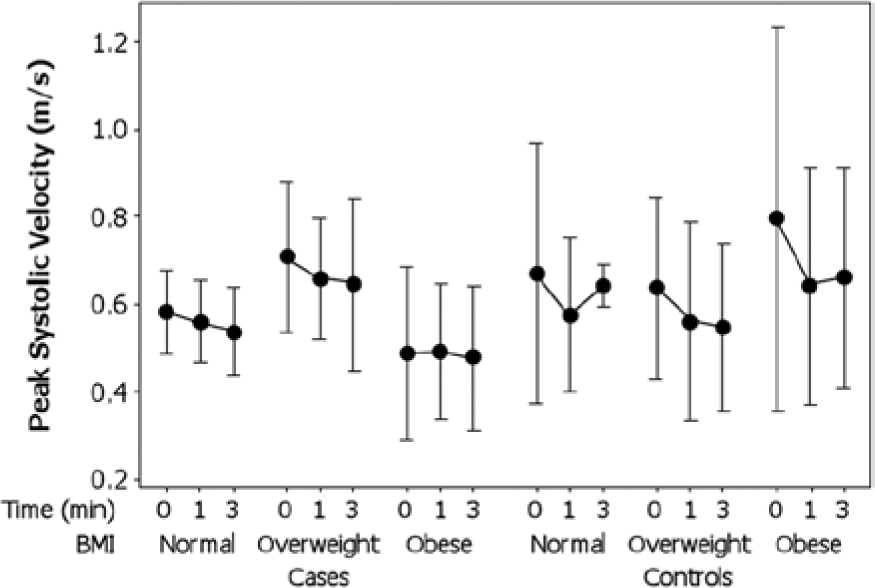
There were no significant correlations between the PSV versus the brachial artery diameters or BMI. The temporal variation in the PSV ± 95% CI (0-3 minutes post-cuff inflation) with respect to BMI among the controls and cases is illustrated in Figure 3. Among the normal weight patients, the mean PSV varied from a minimum of 0.56 m/s (95% CI, 0.49-0.63) to a maximum of 0.59 m/s (95% CI, 0.51-0.69) over time. Among the obese patients, the mean PSV was more variable, ranging from a minimum of 0.59 (95% CI, 0.48-0.70) to a maximum of 0.67 (95% CI, 0.56-0.78). A repeated measures ANOVA indicated that the PSV changed significantly over time between the baseline (0 minutes) and 3 minutes (F = 3.62, P = .032, η2 = .096). There were, however, no significant differences (F = 1.65, P = .208) between the mean PSV of the cases (0.57; 95% CI, 0.51-0.66) and the controls (0.64; 95% CI, 0.56-0.72). The effects of BMI were also not significant (F = 1.76, P = .84).
Temporal variation (post-cuff inflation) in mean peak systolic velocity ± 95% confidence interval with respect to body mass index among cases and controls.
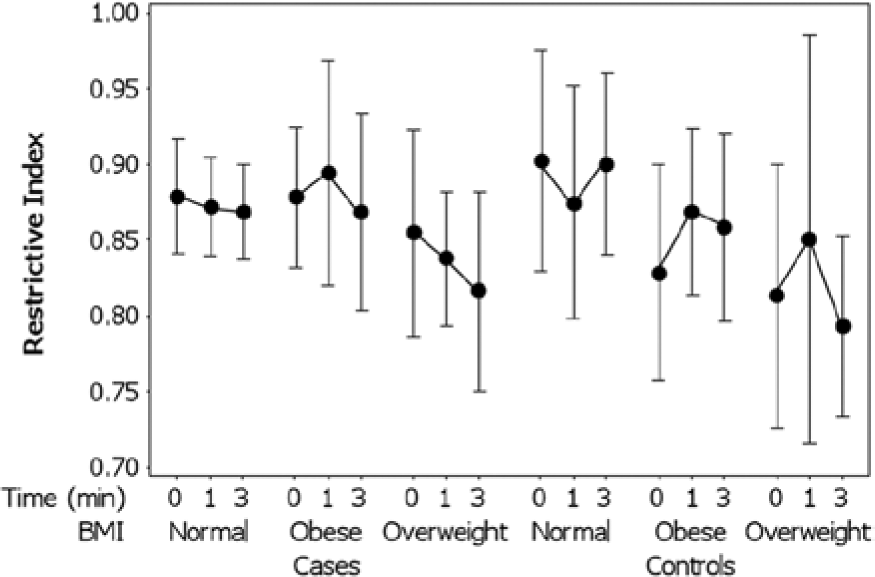
No significant correlations were found between the RI, brachial artery diameters, or BMI. The temporal variation in the RI ± 95% CI from 0 to 3 minutes post-cuff inflation with respect to BMI among the controls and cases is illustrated in Figure 4. Among the normal weight patients, the mean RI remained relatively constant over time, ranging from a minimum of 0.87 (95% CI, 0.84-0.90) to a maximum of 0.88 (95% CI, 0.85-0.91). In contrast, among the obese patients, the RI was more variable over time, ranging from a minimum of 0.85 (95% CI, 0.81-0.89) to a maximum of 0.89 (95% CI, 0.86-0.91). A repeated measures ANOVA indicated that the RI varied significantly with respect to time between the baseline (0 minutes) and 3 minutes (F = 4.64, P = .038, η2 = .120). Although the mean RI did not vary significantly (F = 0.085, P = .772) between the cases (0.87; 95% CI, 0.85-0.89) versus the controls (0.82; 95% CI, 0.81-0.88), there was a significant difference in the RI between the three BMI groups (F = 4.39, P = .020, η2 = .206).
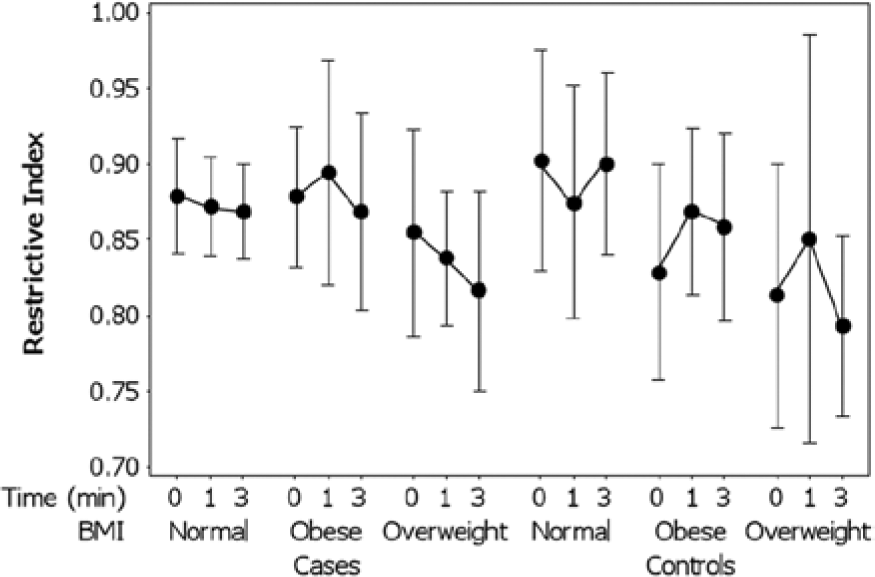
Temporal variation (post-cuff inflation) in mean resistive index ± 95% confidence interval with respect to body mass index among cases and controls.
Discussion
This prospective case-control study is the first to evaluate potential endothelial dysfunction among compensated hepatitis C patients by means of flow-mediated dilation, peak systolic velocity, and resistive index in the brachial artery. After carefully controlling for confounding demographic factors (age and sex), cardiovascular risk factors (including BMI), and the effects of other chronic liver or renal diseases, there was a lack of association between hepatitis C and the endothelial dysfunction of the brachial artery.
Although there is growing evidence that infectious diseases may contribute to the pathogenesis of carotid and coronary atherosclerosis,19–22 this association is still incompletely understood. It has been speculated that because viruses can incite arterial injury, mutagenesis, or altered lipid metabolism, they may contribute to the pathogenesis of atherosclerosis. The infection burden for atherosclerosis development and progression is mainly sustained by infectious agents that possess particular tropism for cells of the vascular wall. For example, Chlamydia pneumonia is capable of infecting vascular endothelium and smooth muscle cells, whereas cytomegalovirus and herpes virus foster the recruitment of monocyte/macrophages and T cells for atherosclerotic lesions. 21 The evidence to suggest that HCV infection is also involved in the pathogenesis of atherosclerosis is, however, not well established.
Our results do not support previous suggestions that posit an association between hepatitis C and atherosclerosis.1–3 From our data, we concluded that HCV positivity in compensated patients, in the absence of cardiovascular risk factors and controlling for BMI, does not appear to be independently associated with endothelial dysfunction, a measure of subclinical atherosclerosis. The results of our study are consistent with others showing no statistically significant association between HCV infection and atherosclerosis.4,5 One possible explanation for the lack of association in our study is that, although HCV infection may stimulate the inflammatory cascade in compensated patients, the resulting inflammation from cytokine release may not be of sufficient magnitude to cause significant endothelial dysfunction. Thus, in the early stages of hepatitis C infectivity (i.e., in patients with a low viral load, or in patients with shorter duration of the disease), it might not be possible to detect endothelial damage by flow-mediated dilatation. Another possible explanation is that, although the infectivity burden of hepatitis C is known to induce plaque formation and endothelial dysfunction in larger vessels, such as the carotid arteries, 22 the infection is not associated with endothelial dysfunction in medium-size brachial vessels. This is not altogether likely, however, as the brachial artery is approximately the same size as the major coronary arteries, an arterial bed that is also prone to plaque formation. It is also possible that, because chronic hepatitis C infection is associated with lower serum lipid levels, this may counteract the pro-inflammatory effect, and indirectly the pro-atherogenic effects, of HCV infection.
This study was subject to a number of limitations, so the absence of conclusive statistical evidence implies that judgment must be suspended. 23 The results of this study were limited by (a) the relatively small sample size resulting from the strict inclusion criteria, chosen to eliminate as many risk factors as possible that could potentially lead to endothelial dysfunction but to restrict the power to conduct significance testing, 24 and (b) the lack of control of other factors that may confound the interpretation of measures of endothelial dysfunction, including the ethnicity and psychological condition of the patients, which might have affected vasomotor tone.13–15 Further prospective studies, with larger samples, are needed in order to draw more definitive conclusions concerning the possible role of hepatitis C infection in the pathogenesis of atherosclerosis.
Footnotes
Acknowledgements
The authors have no industrial links, affiliations, or financial disclosures.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
