Abstract
Visualization of a first trimester pregnancy, whether normal or abnormal, is best done using sonography. Heterotopic pregnancies, the combination of an intrauterine and an extrauterine pregnancy, provide a difficult diagnosis as the intrauterine pregnancy gives a sense of false assurance. This case study presents a spontaneous heterotopic pregnancy with one viable intrauterine pregnancy and one ruptured ectopic pregnancy and discusses the role that sonography plays in the detection and management of this problem.
Heterotopic pregnancy is a rare clinical condition accounting for 1 in 30,000 natural pregnancies. 1 It occurs when two or more fertilized ova implant simultaneously; one pregnancy implants in the uterine cavity and the other implants outside of the uterus, most commonly occurring in the fallopian tubes and rarely in the cervix or ovary. 2 For this reason, heterotopic pregnancies are just as a dangerous as an ectopic pregnancy. The extrauterine or ectopic pregnancy can lead to life-threatening hemorrhagic shock if not diagnosed early, and it requires immediate surgery or treatment. 1 However, in the case of heterotopic pregnancy additional considerations in the treatment of the ectopic are often necessary, given the parents’ preference to continue only the intrauterine pregnancy.
Transabdominal sonography and transvaginal sonography are imaging modalities ideally suited to evaluate heterotopic pregnancies. The case presented below illustrates a patient with an intrauterine and an extrauterine pregnancy with sonography as the main diagnostic imaging modality used in the detection and diagnosis of the pathology. Transabdominal sonography was the primary imaging method used to obtain and evaluate more global views of the adnexa, fundal, and abdominal regions, while transvaginal sonography provided a more detailed visualization of the intrauterine pregnancy, ovaries, and adnexa.
Case Report
A nulliparous woman in her early thirties, approximately nine weeks pregnant, presented to the emergency room with complaints of abdominal pain and severe cramping. She was previously hospitalized a week earlier with similar symptoms and was discharged after sonography verified a viable intrauterine pregnancy. She appeared pale and diaphoretic and was hypotensive with a blood pressure of 84 mmHg systolic and 51 mmHg diastolic. During her initial evaluations it was noted that her blood pressure continued to drop. She was unable to walk and complained of severe pain radiating to her shoulders. Laboratory blood work was ordered that showed an increased white blood cell count of 17.60 K/mm3 (normal values are 4.0-10.00 K/mm3). The blood count also showed extremely low levels of red blood cells, hemoglobin, and hematocrit. Her serum quantitative beta human chorionic gonadotropin (B-hCG) level was 99,137.0 mIU/mL (normal values in nonpregnant females are <5 mIU/mL). Based on the patient’s symptoms and laboratory results, a transabdominal pelvic sonogram was ordered.
Sonography was done using an iU22 (Philips Ultrasound, Bothel, Washington) with a curved array C5-2 probe (2- to 5-MHz bandwidth) or a transvaginal curved array C8-4v probe (4- to 8-MHz bandwidth). A transabdominal sonogram was performed first and demonstrated a viable intrauterine gestation with a heart rate of 165 beats per minute (Figures 1 and 2). As shown in Figures 3 and 4, a complex left adnexal lesion was visualized in the left lower quadrant. The lesion was noted to be anterior with free fluid adjacent to and continuous with the splenic gutter. A measurement of the mass showed it to be approximately 12.7 cm long, 10.6 cm wide, and 4.6 cm in the anterior-posterior (AP) dimension. (Because of the excessive amount of free fluid and bowel around the mass, it was difficult to obtain precise measurements, which is why the calipers were placed where there was no peristalsis seen.) The mass was felt to be highly suspicious for a thrombus. Free fluid was also seen in both the abdomen and the pelvis, with fluid demonstrated in the perihepatic and perisplenic spaces (Figures 5 and 6). A transvaginal sonogram was then performed and confirmed the findings of a viable intrauterine pregnancy with a crown-rump length of 2.3 cm, which correlated with a gestational age of 9 weeks 0 days (Figure 7). A small subchorionic hemorrhage was noted. The right ovary measured 2.9 cm long, 2.7 cm wide, and 3.3 cm AP, with a hemorrhagic or corpus luteum cyst measuring 2.0 cm (Figures 8-10). Transvaginally, another ill-defined mass was noted in the left adnexa posterior and adjacent to the uterus measuring 7.25 cm long, 4.57 cm wide, and 3.94 cm AP, which was highly suspicious for an ectopic pregnancy (Figures 11 and 12). Because of the location and size of the transabdominal mass and given the limited field of view of the transvaginal probe, it was not possible to show the two separate masses in the same image. The sonographic results were called to the emergency department physician by the reading radiologist as soon as the examination was completed.
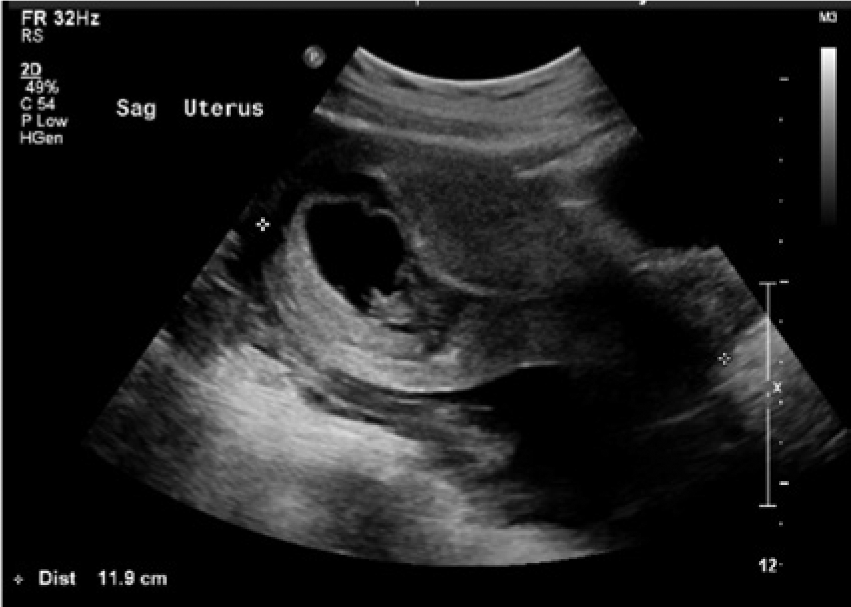
Transabdominal sonogram demonstrating the intrauterine pregnancy in the sagittal plane.
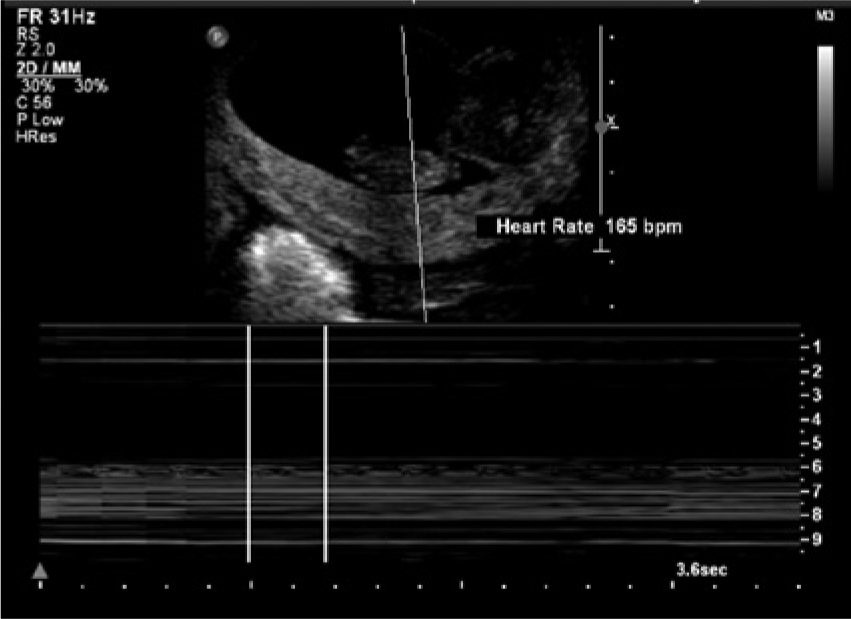
M-mode recording showing a fetal heart rate of the intrauterine gestation of 165 beats per minute.
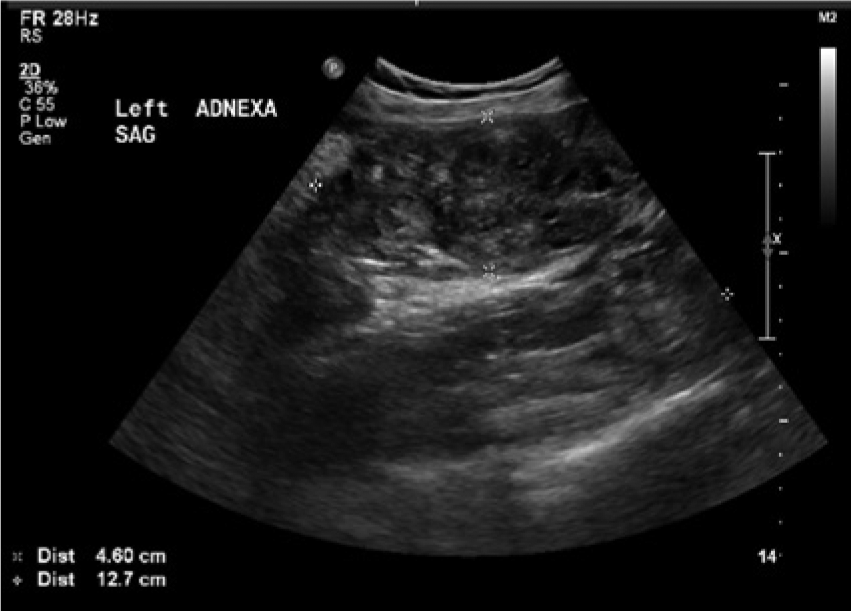
Transabdominal sonogram showing the complex left adnexal mass in the sagittal plane.
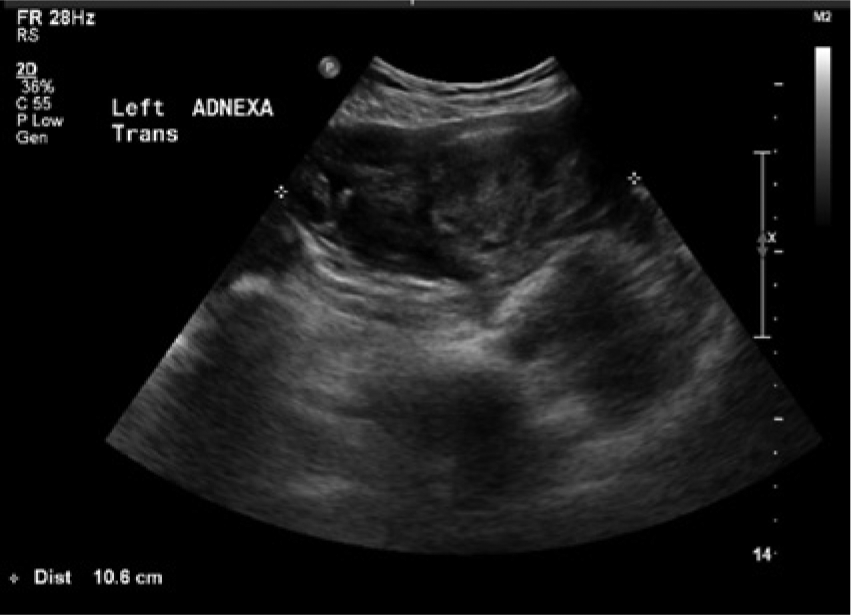
Transabdominal sonogram showing the same complex left adnexal mass in the transverse plane.
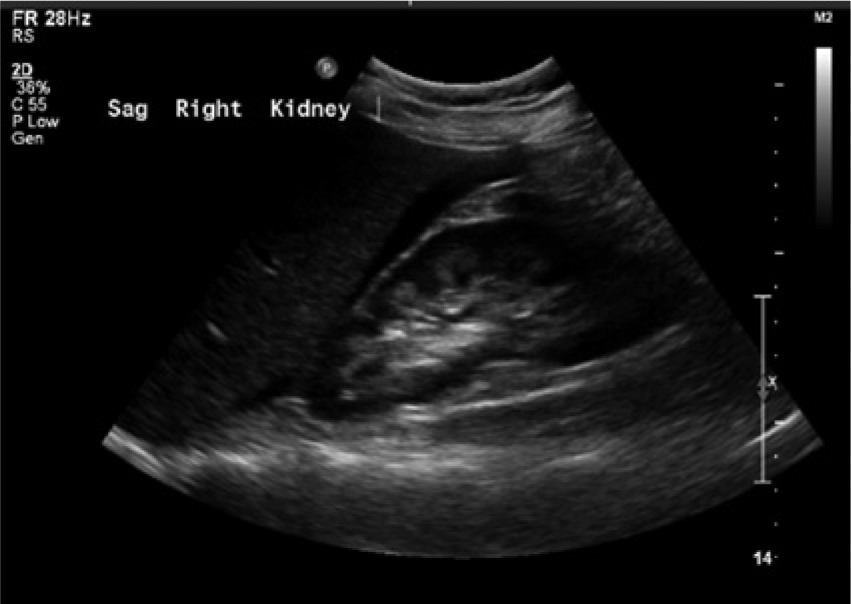
Transabdominal sonogram showing free fluid in Morison’s pouch around the right kidney.
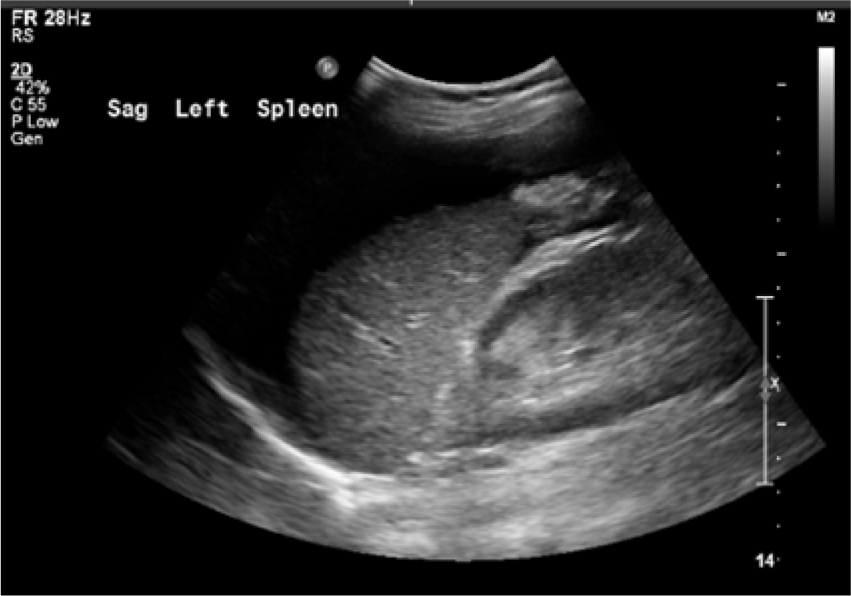
Transabdominal sonogram showing free fluid in the perisplenic region.
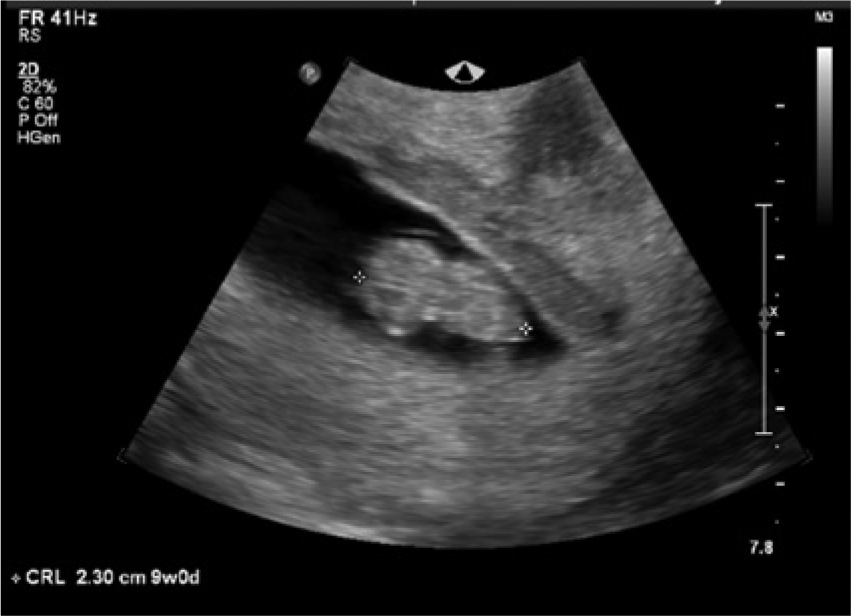
Transvaginal sonogram showing an intrauterine pregnancy with a crown-rump length measurement of 2.3 cm.
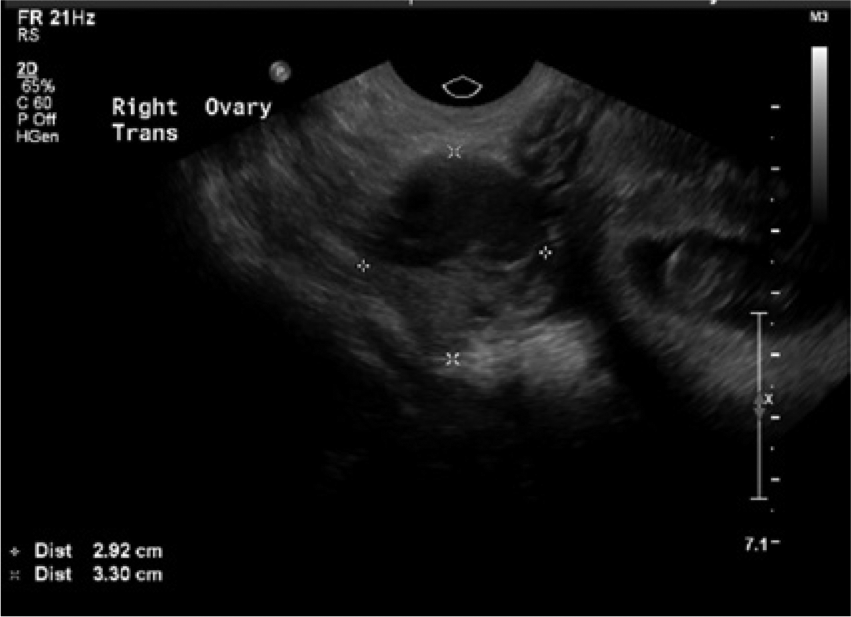
Transvaginal sonogram showing the right ovary in the transverse plane with cursors denoting the measurement.
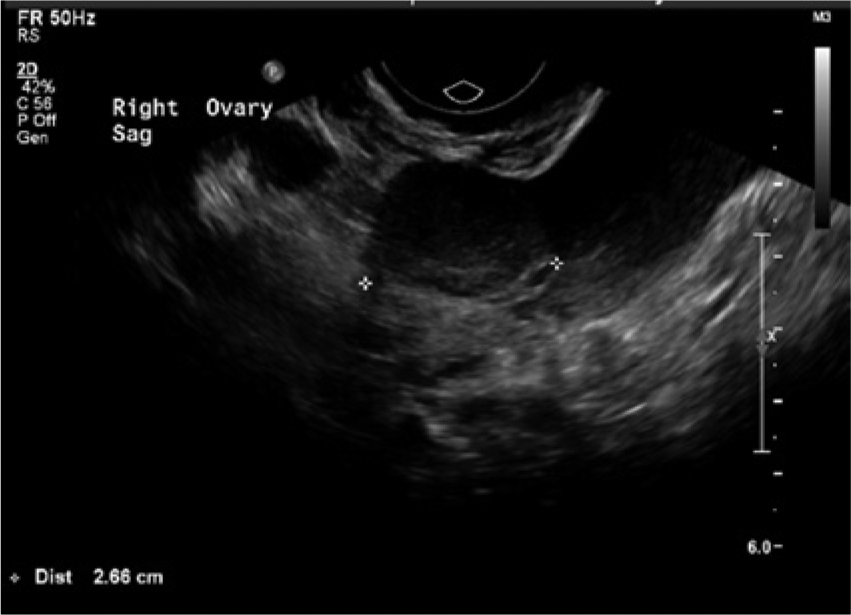
Transvaginal sonogram showing the right ovary in the sagittal plane with cursors denoting the measurement.
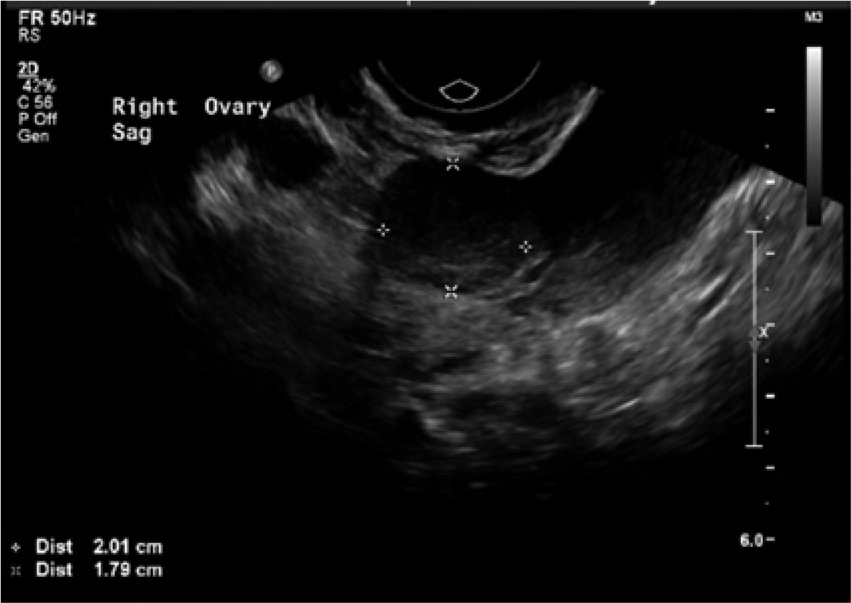
Transvaginal sonogram showing the 2.0-cm hemorrhagic corpus luteum cyst on the right ovary.
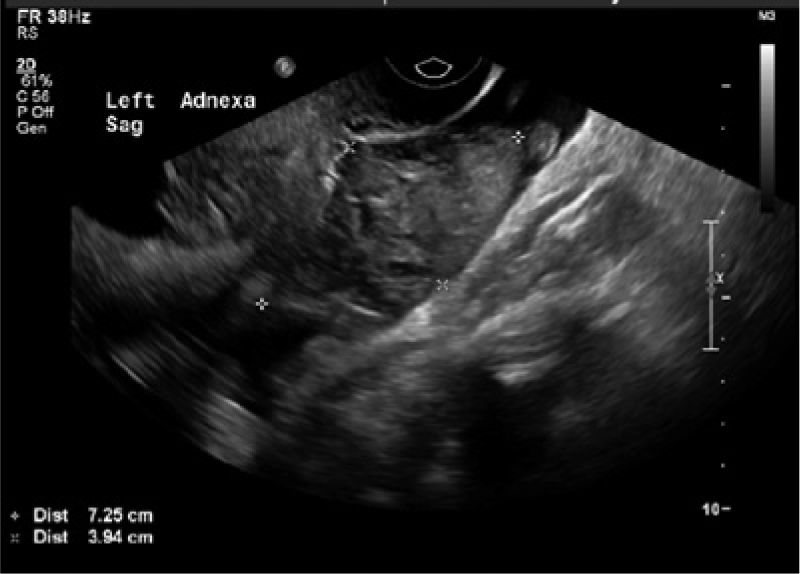
Transvaginal sonogram showing the left adnexal mass in the sagittal plane with measurements; this was determined to be a ruptured ectopic pregnancy.
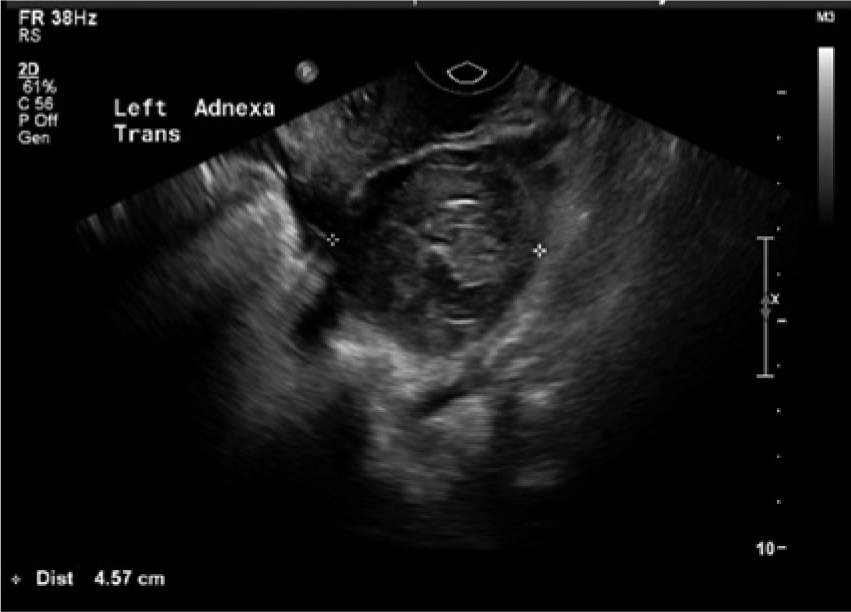
Transvaginal sonogram showing the transverse image of the same left adnexal mass later determined to be a ruptured ectopic pregnancy.
Immediately after the sonographic examination, the patient was taken to the operating room with a probable left ruptured ectopic with hemoperitoneum. The uterus was enlarged, consistent with an intrauterine pregnancy. Intraoperative findings showed the right fallopian tube and ovary to be normal; however, the left fallopian tube had a tubal mass consistent with an ectopic. The tubal segment was excised using the LigaSure apparatus with good hemostasis. The pelvis and abdomen were vigorously irrigated numerous times, with a total of approximately 2000 mL of both blood and thrombus removed.
The left fallopian tube, measuring 5.5 cm in length and varying from 1.0 cm to 4.5 cm in diameter, was submitted for laboratory evaluation. Sectioning revealed tan-red and tan-brown hemorrhagic soft tissue within the lumen. Specimens were obtained and sent for pathological analysis, which confirmed chorionic villi suggestive of a heterotopic pregnancy with a ruptured left adnexal gestation. The patient made an unremarkable recovery from surgery, and the intrauterine live gestation continued uninterrupted.
Discussion
Sonographic imaging was vital in the rapid detection, evaluation, and ultimate management of this heterotopic pregnancy. Both transabdominal imaging and transvaginal imaging were necessary to detect both adnexal masses. Transabdominally, the complex mass that was detected in the pelvic area was felt to be consistent with a thrombus secondary to intraperitoneal bleeding. Free fluid was also found in the perihepatic and perisplenic spaces, classic sonographic indicators of a ruptured ectopic. Transvaginal sonography allowed a more detailed evaluation of specific regions, enabling the detection of a mass in the left adnexa, representative of a ruptured ectopic. Reports in the literature indicate that transvaginal sonography has a 41% to 84% detection rate of a heterotopic pregnancy with the mean gestational age at time of diagnosis of 8 weeks. 2
Heterotopic pregnancy in natural conception is extremely rare. Historically, the prevalence is estimated at 1 in 30,000; however, more recent studies have demonstrated that with pregnancies involving ovulation induction and in vitro fertilization, the incidence has increased to 1 in 100.1,3 Other investigators estimate the risk of heterotopic pregnancy at 1 in 119, and this number rises to 1 in 45 with embryo transfer. 4 With embryo transfer, the excessive number of embryos causes multiple pregnancies, ectopic pregnancies, and heterotopic pregnancies. 5 Women with the highest risk of an ectopic pregnancy are those with a history of an ectopic, prior tubal surgery, or a history of pelvic inflammatory disease (PID). 2 Investigators have theorized that heterotopic pregnancies occur because the fertilized ovum fails to enter the uterine cavity due to tubal adhesions. 1 This leads to the implantation of the ovum in the fallopian tube. 1 However, the heterotopic pregnancy in this case study occurred in a nulliparous woman with no history of assisted reproductive technology, PID, tubal surgeries, or previous ectopic pregnancies.
Diagnosing heterotopic pregnancies early can be very challenging, and a missed diagnosis will often lead to a ruptured ectopic. Abdominal pain is the most common symptom associated with a heterotopic pregnancy, along with an adnexal mass, peritoneal irritation, and an enlarged uterus.4,6 Unlike ectopic pregnancies, heterotopic pregnancies lack vaginal bleeding due to the intact intrauterine pregnancy. 4 However, the presenting signs and symptoms of a heterotopic pregnancy are nonspecific and can be commonly confused for other normal or abnormal findings during pregnancy. 6 In the case above, the presence of the right ovarian hemorrhagic corpus luteum cyst could be mistaken for an extrauterine pregnancy. Serial B-hCG levels have not been shown to assist in the diagnosis of heterotopic pregnancies because both gestations produce hCG.
Of note, only approximately 10% of heterotopic pregnancies are identified preoperatively, and approximately one-third of intrauterine gestations in a heterotopic pregnancy spontaneously abort.7,8 Heterotopic pregnancies nonetheless are life-threatening and require a timely diagnosis and treatment. The most common treatment option for the management of heterotopic pregnancies is surgical removal of the ectopic gestation by salpingectomy or salpingostomy. 2 Laparotomy, or the more commonly preferred laparoscopy, is widely used. Laparotomy is the preferable surgical modality in cases with severe intra-abdominal bleeding or for patients in hemorrhagic shock. 2 It has been estimated that positive outcomes are seen in roughly 66% of heterotopic pregnancies. 1
The goal of a sonogram in cases of pregnancy is to determine the presence and characteristics of an intrauterine pregnancy. However, this does not eliminate the possibility of an ectopic gestation. It is important to keep the possibility of an ectopic pregnancy in mind when a patient presents with a current intrauterine pregnancy and abdominal pain. Sonography is a useful tool in identifying a heterotopic pregnancy, but the lack of investigation of this possibility has led to the diagnosis being missed in up to 50% of cases.9,10 Sonographers must have a thorough understanding of normal and abnormal sonographic features, implantation sites, and different scanning techniques in conjunction with the patient’s medical and surgical history in order to correlate the patient’s clinical presentation with sonographic images.
Conclusion
Although heterotopic pregnancy is a rare clinical presentation in normal conception, it should be considered in viable intrauterine pregnancies with clinical and sonographic presentations of abdominal pain, adnexal mass, and free fluid in the abdominal cavity. Early intervention and detection allow appropriate early management and the preservation of the current intrauterine pregnancy and future fertility.
Footnotes
Acknowledgements
The author would like to thank JoAnn Pingleton, RDMS, and the University of Missouri Diagnostic Medical Ultrasound staff for their assistance, leadership, and guidance.
Declaration of Conflicting Interests
The author declares no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
