Abstract
A literature review has been conducted to demonstrate the role of duplex sonography in evaluating renal artery stenosis (RAS) as a screening method for patients prior to percutaneous transluminal renal artery stent (PTRAS) placement. The review also evaluates the role of ultrasound surveillance following PTRAS and establishes the overall influence of ultrasound on defining stent efficacy for managing hemodynamically significant renovascular disease. Recent studies have narrowed criteria for intervention through duplex sonographic assessment of RAS to better predict patient benefit and response to stent placement. Studies using sonographic parameters evaluating patients for renal artery stenosis, criteria-dependent stent placement, and postintervention follow-up have shown improved patient response, helping ensure effective and appropriate care in RAS management.
Keywords
Renal artery stenosis (RAS) is most commonly caused by atherosclerotic disease of the aorta and renal artery resulting in obstruction of flow, typically occurring at the origin and in the proximal one-third of the renal artery. 1 Luminal narrowing of the artery results in reduced perfusion of the kidney and possible loss of renal parenchyma secondary to chronic ischemia. Hemodynamically significant obstruction is characterized by the clinical presentation of renovascular hypertension (HTN), renal dysfunction, flash pulmonary edema, and congestive heart failure (CHF). 2 From the time of initial diagnosis, more than half of RAS patients demonstrate progressive stenotic changes within five years, resulting in renal atrophy, renal failure, and interventional hemodialysis. 2 Factors that increase the risk for RAS development include increased age, male sex, HTN, diabetes, coronary artery disease, smoking, aortoiliac disease, and peripheral arterial disease (PAD).1,2
Clinical Findings and Medical Management
Abnormal clinical laboratory values consistent with renal failure secondary to RAS include increased serum creatinine (CRTN), decreased urinary clearance of CRTN, and declining glomerular filtration rate (GFR).2,3 Clinical presentation of medication-resistant HTN, pulmonary edema, or unexplained renal insufficiency indicated by laboratory values must be corroborated by imaging to confirm RAS and to determine the appropriate plan of care.
Traditional treatment for HTN and associated cardiovascular complications includes medications such as diuretics, angiotensin-converting enzyme (ACE) inhibitors, beta-blockers, and calcium channel blockers. This management of renovascular HTN may be effective; however, adverse reactions to medication, an ineffective medication type, atherosclerotic disease progression, or noncompliance can all hinder patient response. In addition, decreasing blood pressure via antihypertensive medication can compromise renal function in the presence of significantly stenotic renal arteries, resulting in ischemic renal atrophy and facilitating failure. 3 Intervention will be contingent upon the degree of stenosis and progression of renovascular disease. Medical management likely will still be prescribed to preserve renal function, however, even following angioplasty and stent placement.
Intervention
Surgical revascularization through renal artery bypass or endarterectomy has historically been the primary alternative to medical management in indicated patients. 3 Open surgery is now used much less frequently because of its increased morbidity compared with the percutaneous techniques available. Surgical intervention is now typically reserved for patients with contraindications to stenting or to those having renal artery revascularization in conjunction with an aortic repair. 4
Angioplasty, with or without stenting, comprises a significantly less invasive method of revascularization of stenotic renal vessels. It is performed by percutaneously inserting an expandable balloon-tipped catheter in the femoral artery at the groin and using fluoroscopy to guide the balloon into the site of stenosis within the renal artery. The balloon is inflated with several atmospheres of pressure to fracture the atherosclerotic lesion and stretch the native arterial walls, dilating the narrowed vessel lumen. This method poses less risk to patients than traditional open surgical revascularization, but balloon angioplasty alone has been shown to have a significant rate of restenosis. 5 Much of this is due to the limited ability to achieve adequate dilation within the ostium of the renal artery (the renal segment within 5 mm of origin at the aorta), which accounts for 75% of RAS. 2
Percutaneous transluminal renal artery stenting (PTRAS) combines the approach of balloon angioplasty with stent placement to overcome the problems of elastic recoil of the lesion, dissection, and restenosis.3,6 Percutaneous intervention has become the procedure of choice, preferred for the advantages of decreased cost and decreased risk of complications, with comparable potential to preserve renal function.
Imaging Methods
Clinical indicators including HTN and/or renal dysfunction can exist independent of renal artery disease or be a manifestation of RAS. Ultimately, the percentage of lumen reduction within the renal artery dictates the treatment for RAS patients; to determine the likelihood of benefit from PTRAS, patients must fall under selective criteria defining a hemodynamically significant stenosis. Patients with increased likelihood of a favorable response are identified through preprocedural imaging, the accuracy of which is critical in administering the most effective treatment to salvage renal and cardiovascular function. Noninvasive methods used to screen patients for RAS have been a source of controversy regarding their accuracy and limitations in detecting stenotic vessels when compared with angiography.
Angiography
Catheter-based digital subtraction angiography (DSA) has been the gold standard in detecting RAS for its sensitivity and specificity in defining the degree of lumen reduction but carries a risk of procedure-related complications and damage to renal function. 7 Imaging of the renal arteries with angiography is achieved by advancing a catheter from the groin through the iliac and aortic vessels to the renal artery at the aortic origin. Contrast material is administered through the catheter into the vessel and exposed to fluoroscopic x-rays highlighting vessel anatomy (Figure 1). Benefits of catheter-based angiography include high resolution and accuracy of RAS detection. However, the increased cost, exposure to radiation, risk of contrast nephrotoxicity, and possible puncture site complications are disadvantages that make this invasive technique somewhat better suited to confirming RAS following noninvasive screening if a test of acceptable accuracy is available. As an alternative to DSA, studies have been conducted to compare the value of noninvasive assessment of RAS, which historically had equivocal results secondary to strong operator dependence. However, advancements in technology have encouraged continued exploration and refinement of these techniques, which would be preferred based on cost, availability, decreased risks of morbidity, and lack of any radiation exposure.
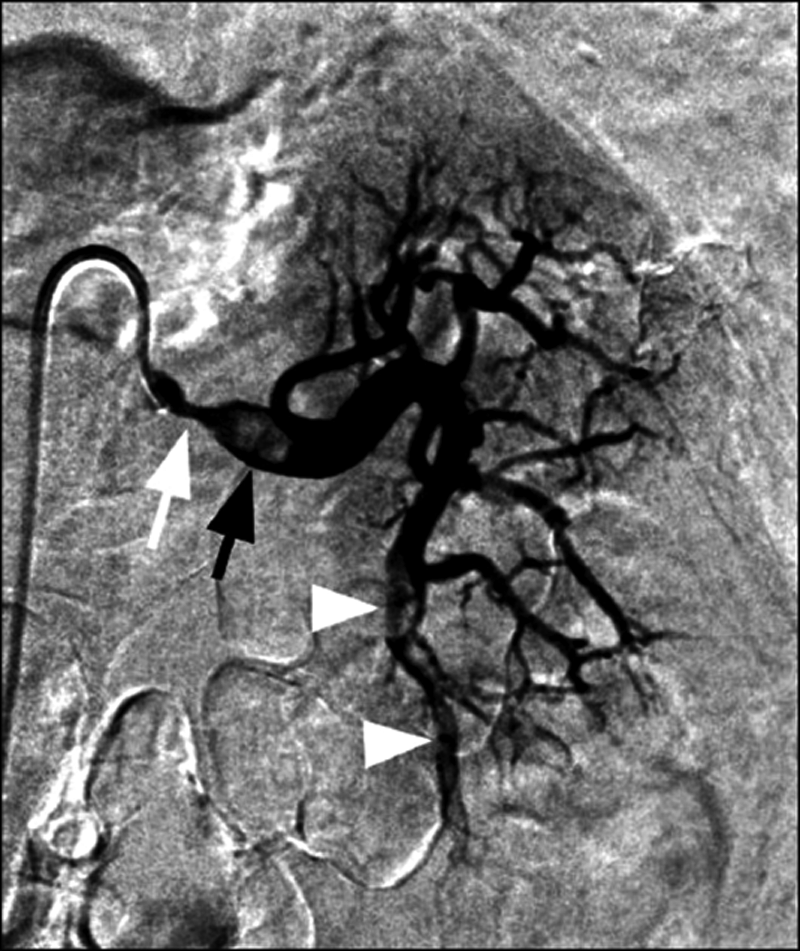
Catheter-based renal arteriogram demonstrating significant stenosis of the renal artery. Source: Arabi et al. 7 © 2011 Arabi et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. No copyright release required.
Computed Tomographic Angiography
Computed tomographic angiography (CTA) generates multiple cross-sectional images of anatomy through transmission and absorption of x-rays. These are received as digital signals by the detector and translated into 2D and 3D images. The combination of intravenously administered contrast increases resolution of targeted vessels, allowing abnormal vascular anatomy to be more easily visualized. This modality has demonstrated notable accuracy in determining the degree of renal artery stenosis; in a published report comparing CTA detection of RAS with angiography, 8 sensitivity measured in the range of 91% to 94% and specificity between 93% and 97%; however, positive predictive values of CTA were reported to be only 75% by Rountas et al, 9 well short of optimal compared with angiography (Figure 2). Additional limitations of CTA include exposing the patient to a substantial amount of radiation, the use of contrast, the risk of allergic reaction, limitations of body habitus, and artifacts related to motion or previous surgical clips and stents.10,11
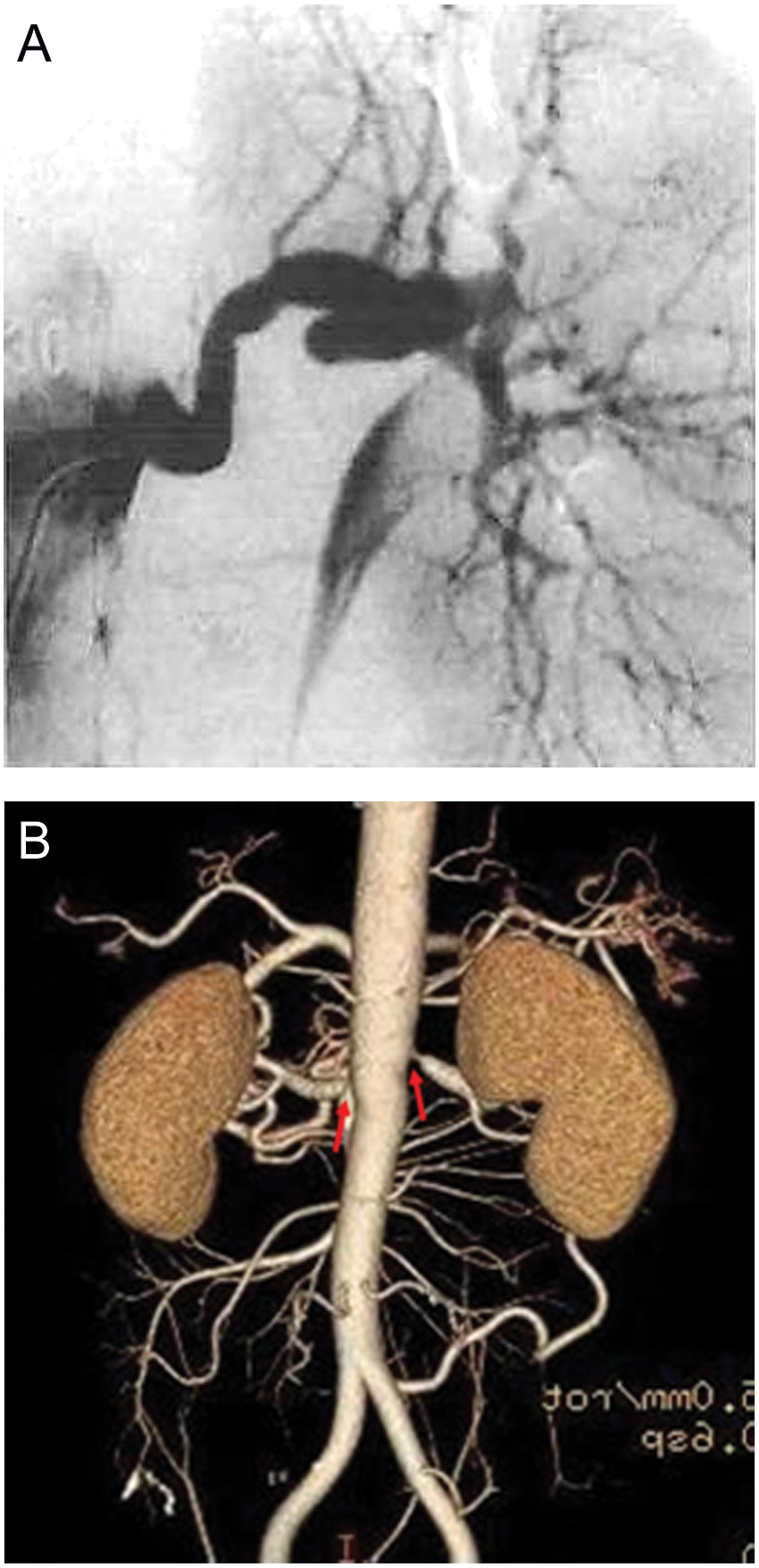
(a) Arteriogram showing no significant stenosis within the left renal artery. (b) Corresponding computed tomographic angiogram showing a stenotic area within the left renal artery. 10
Magnetic Resonance Angiography
Magnetic resonance angiography (MRA) uses nuclear magnetic resonance detection of radiofrequency signals emitted by hydrogen atoms within the body that are excited by a strong magnetic field. These signals are digitally processed to render 2D and 3D images of target organs (Figure 3). MRA uses gadolinium-based contrast to increase visualization of the vasculature and is capable of assessing indirect indications of RAS through volume-rendering methods that enhance visualization of turbulent flow within stenotic vessels. The benefits of MRA include lack of radiation and iodinated contrast, as well as superior resolution of renal parenchyma compared with CTA. Limitations include body habitus restrictions, claustrophobia, contraindications in patients with renal insufficiency or metal implants, and artifacts from motion and previous surgery and/or metal clips.
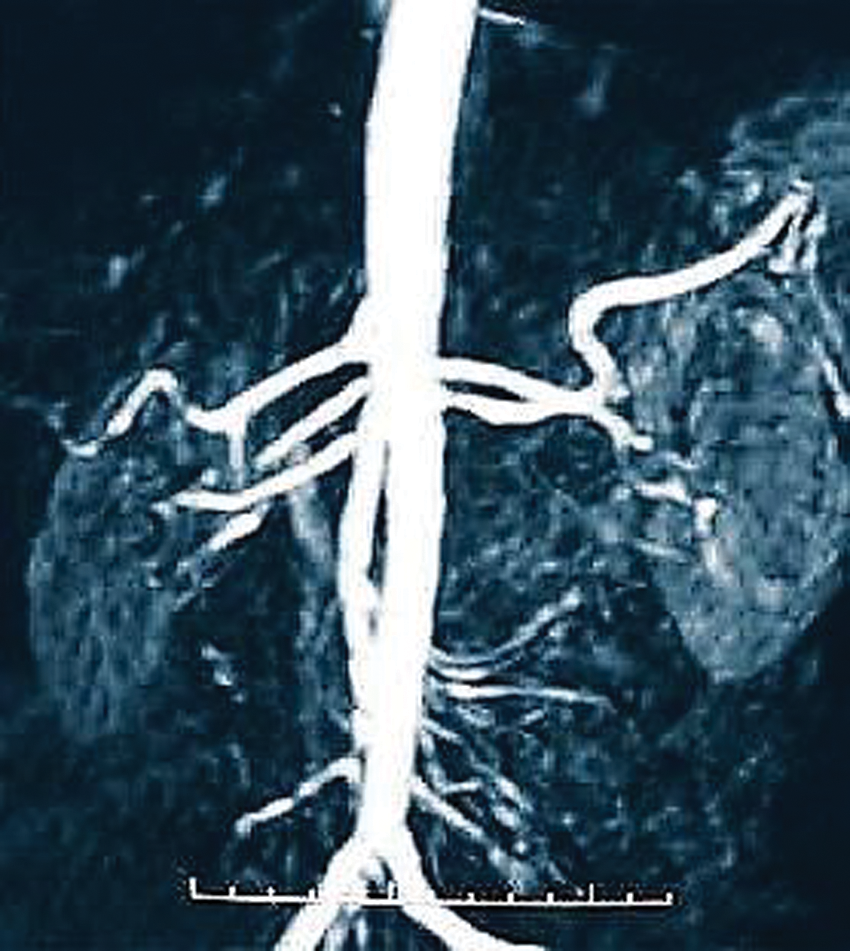
Magnetic resonance angiography of renal artery stenosis at the left renal artery ostium. Reprinted by permission from Macmillan Publishers Ltd: Natural Clinical Practice: Cardiology 2009. 11
Overall sensitivity and specificity of MRA in detecting RAS have been reported to be 100% and 96%, respectively. 1 In contrast to these reports, a study by Gilfeather et al 12 assessed the value of MRA in hemodynamically significant stenoses of equal to or greater than 70% luminal reduction; 54 patients and 107 kidneys were evaluated by gadolinium-enhanced MRA and compared with conventional angiography. The MRA results showed a less than 10% difference in the average degree of stenosis obtained compared with angiography results in 65% of the vessels evaluated; however, overestimation of stenosis occurred in 21% of patients and underestimation of luminal reduction in 14% of patients. 13
The variability of imaging accuracy compared with angiography has been the core of controversy regarding the pursuit of noninvasive or minimally invasive imaging in accurately defining the degree of stenosis and planning the management and care for RAS patients. Published studies using noninvasive imaging as a modality to screen for RAS in patients prior to angioplasty and stent placement without angiographic confirmation have shown an increased potential to include patients with more moderate RAS, lessening the likelihood of benefit and affecting the reported efficacy of stents placed for RAS intervention.
Duplex Sonography
In evaluating renal artery blood flow, duplex sonography uses gray-scale and color Doppler imaging to visualize the artery and the direction of blood flow (Figure 4). Turbulent flow may also be identified as an area where small-scale color variation occurs, indicating a chaotic pattern of flow caused by sudden narrowing and subsequent expansion of the lumen. Spectral Doppler evaluation is used to determine the morphology of the blood flow waveform and the velocity of blood flow. Increased peak systolic velocities, the presence or absence of an early systolic peak, and parenchymal resistive indices are aspects of the spectral waveform that can be used to indicate directly or indirectly the presence of RAS (Figures 5–7). To fully evaluate a suspected RAS, velocities and spectral waveforms should be obtained at the proximal, mid, and distal segments of the renal artery. A peak systolic velocity in the abdominal aorta just proximal to the origin of the renal artery is also sampled to compare to proximal renal artery velocities. The renal aortic ratio (RAR) measures the greatest peak systolic velocity (PSV) within the renal artery divided by the aortic PSV; a ratio ≥3.5 and/or a PSV greater than 180 cm/s is consistent with an angiographic confirmed stenosis of ≥60%.14,15 An elevated end-diastolic velocity (EDV) ≥150 cm/s has been suggested to be consistent with a renal artery stenosis of ≥80%. 8 Indirect measures of RAS, based on velocity data from the segmental arteries at the hilum of the kidney, have included an acceleration time (the time interval from onset of systole to reaching peak systolic flow) of greater than 0.07 seconds and an absolute acceleration in early systole of less than 300 cm/s2 (Figures 6 and 7).16–18
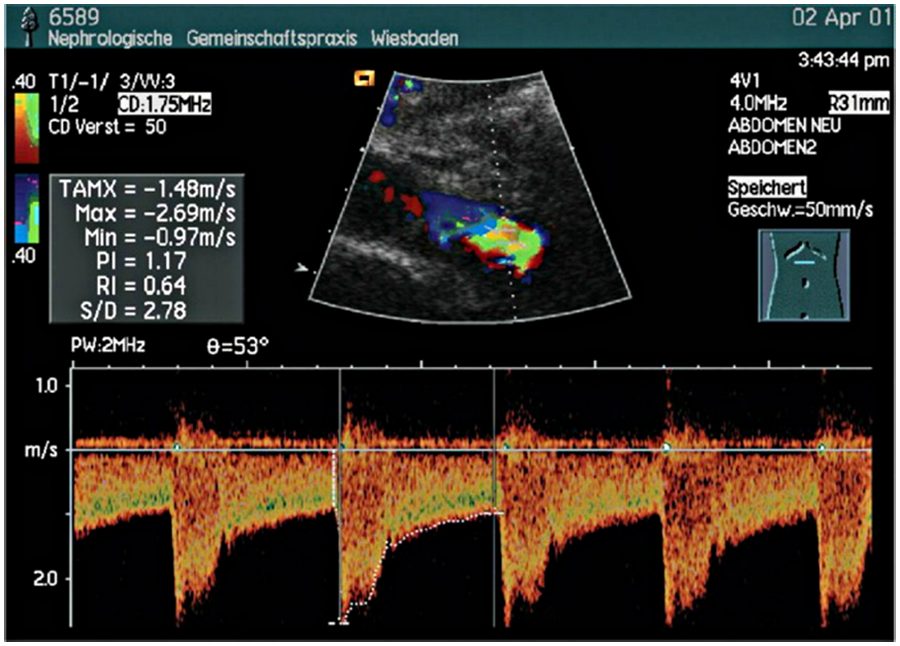
Duplex sonogram showing high blood flow velocities and turbulence near the ostium of the left renal artery scanned with the patient in a supine position. Peak systolic velocity of 269 cm/s indicates renal artery stenosis. 14
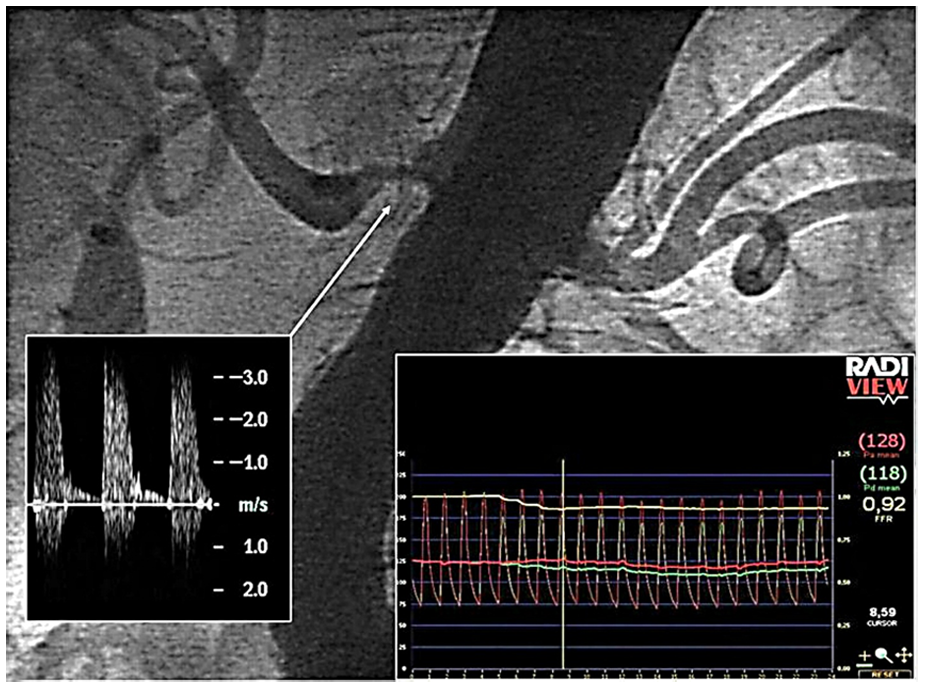
Comparison of angiography, sonographic measurements, and transstenotic pressure gradient in a right-sided renal artery stenosis. Angiography demonstrates a >50% stenosis. The left insert shows Doppler signals at the level of the stenosis (300 cm/s). 15
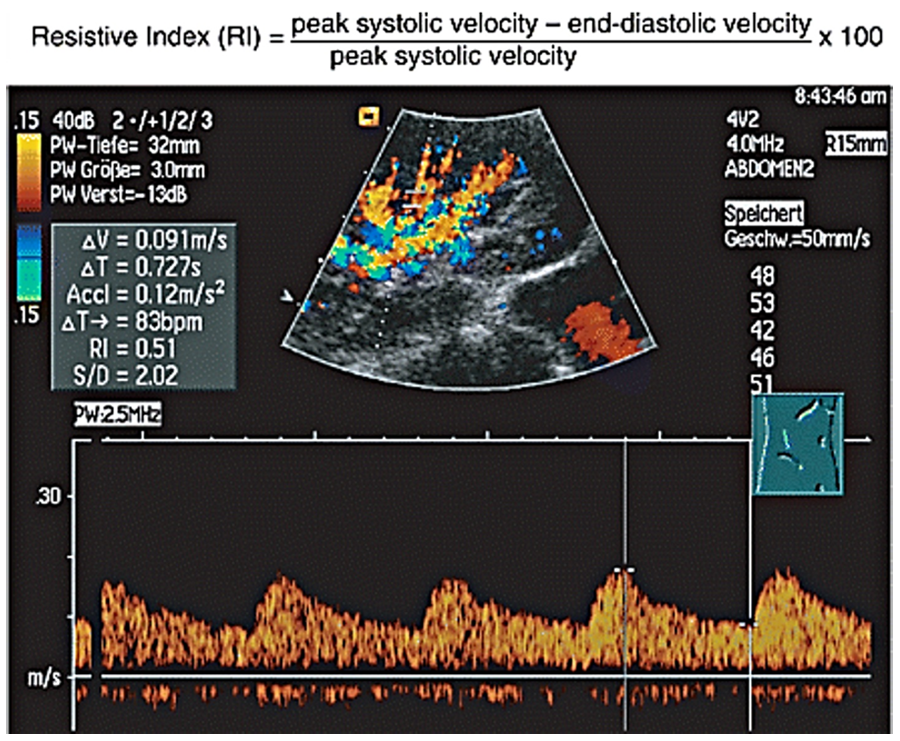
The “tardus-parvus” flow pattern reveals low peak systolic and high end-diastolic velocity resulting in low resistive index values from 0.42 to 0.53. 14
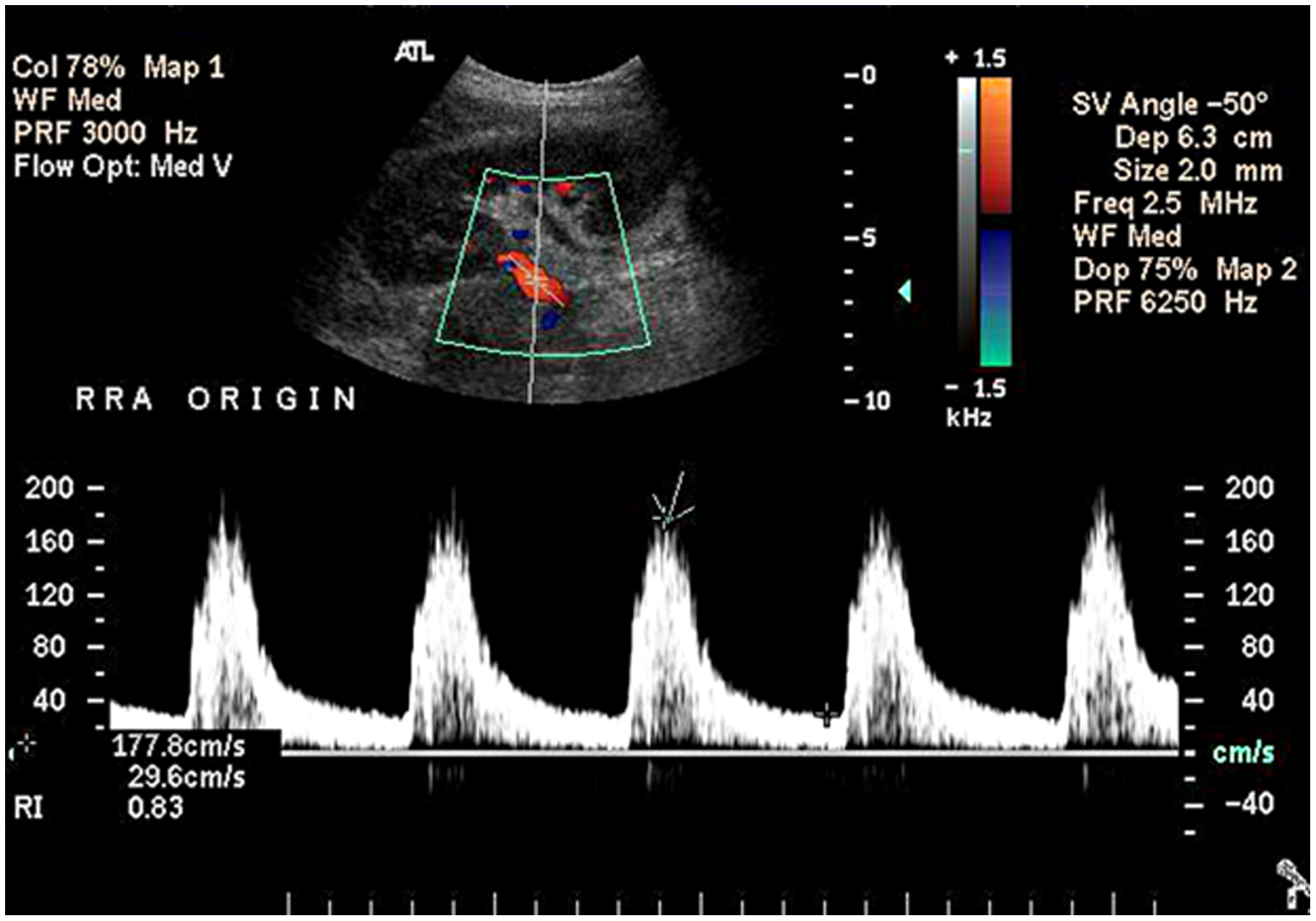
Duplex sonography of right renal artery origin showing increased peak systolic velocity indicative of borderline renal artery stenosis. 16
Duplex sonography has traditionally been regarded as having equivocal value in detecting significant RAS due to the demonstrable variation in accuracy as a highly operator-dependent modality. Recently published studies performed comparing sonographic accuracy of RAS detection to angiography have shown sensitivities ranging between 93% and 98%, a specificity of approximately 98%, positive predictive values between 98% and 99%, negative predictive values between 94% and 97%, and overall accuracy between 96% and 98%.3,19,20 Such accuracy results from suitable adaptation of techniques and criteria for RAS assessment and, when achieved, allows patients the alternative of a widely available, noninvasive imaging modality that is low in cost and poses no radiation exposure or risk of adverse contrast reactions that might impair renal function. Limitations of duplex sonography have been studied by Hedayati et al, 20 who determined the clinical and technical factors that significantly influence the ability to obtain a complete duplex-sonographic examination of renal arteries in RAS assessment. A regression analysis based on 250 patient examinations demonstrated the factors that most strongly predicted an incomplete or nondiagnostic examination were the technologist’s years of experience, recent abdominal surgery, and the technology available on the equipment used for the study, affecting 35% of attempted examinations.
Published Data
Early published studies evaluating the efficacy of PTRAS in managing renovascular HTN and renal dysfunction in RAS exhibited widely variable patient selection criteria, methods, postintervention follow-up techniques, and the end points used to define success. As a result, many of these early analyses of stent efficacy versus medical management were largely inconclusive because of the liberal inclusion of patients and underdeveloped indicators for intervention.2,21,22 One result of these trials was the need to more closely define RAS prior to intervention, and most later trials used 60% or 70% diameter reduction as the threshold for hemodynamic significance; research has demonstrated a greater likelihood of positive outcome on RAS-induced HTN and renal dysfunction at this level of stenosis or greater. A further confounding factor was the limited accuracy of nonstandardized noninvasive imaging techniques and the parameters used to define the significance of stenosis. Much of the early published literature suggested little or no benefit of stent placement in patients’ outcomes compared with medication management for RAS-induced HTN and preservation of renal function.
A study involving 140 RAS patients, STAR (Stent Placement in Patients with Atherosclerotic Renal Artery Stenosis and Impaired Renal Function), aimed to determine the value of PTRAS over medical management. 23 The published results favored medical management over percutaneous angioplasty and stenting based on lack of improved renal function and complication rates. Review of the methods showed multiple deficiencies in study design, including the liberal inclusion criteria of 50% or greater stenosis as opposed to the recommended 70% to establish hemodynamically significant RAS; more than 30% of patients enrolled had less than 70% stenosis in the renal artery.
ASTRAL (Angioplasty and Stenting for Renal Artery Lesions) was one of the largest early published trials, enrolling 806 RAS patients to determine the effect of PTRAS on renovascular HTN and function. 24 Published reports showed no evidence of benefit to RAS patients; however, the trial enrolled patients with uncertainty of benefit based on clinical presentation without angiographic confirmation of the degree of renal artery stenosis. Twenty-five percent of enrolled patients had no previous renal dysfunction, 40% had less than 70% stenosis, and only 28% had RAS of at least 70% luminal reduction. A third study, DRASTIC (Dutch Renal Artery Stenosis Intervention Cooperative), also contributed to the inconsistent reports on stent efficacy, allowing inclusion of more than 30% of patients without hemodynamically significant stenoses in defining the benefit of stent placement for RAS. 25
The published data involved evaluation of RAS with loosely related physiological markers as indicators and anatomic assessment with noninvasive methods that had a documented potential of over- and underestimating vessel stenoses. The inclusion criteria greatly affected recommendations for intervention, placing stents in patients unlikely to benefit. The prevalence of inaccurate data used in evaluating the degree of RAS significantly affected the plan of care, application, recognized value, and ability to measure the progression and postprocedural efficacy of stents as interventional methods for RAS control.
The scarcity of trials and inconsistencies in reports resulted in a sharp decline in 2001 of the use and application of renal artery stents following publication of early trials. 4 Limited sample sizes, biased selection in retrospective analyses, failure to define standards of inclusion criteria, and using poor indicators and alignment of outcome influenced American College of Radiology (ACR) guidelines of stent placement indication and reimbursement. The Centers for Medicaid & Medicare Services (CMS) issued a policy for surgical intervention of RAS, restricting reimbursement of PTRAS for “patients in whom there is an inadequate response to a thorough medical management of symptoms for whom surgery is the likely alternative. Percutaneous transluminal renal artery angioplasty for this group is an alternative to surgery, not simply an addition to medical management.” 4 The implication of restricted reimbursement for renal intervention demanded evidence-based medicine through an increase in randomized trials that supported improved outcomes of stent placement for RAS patients.
A limitation of many of the earlier trials was a lack of inclusion of any hemodynamic screening followed by angiographic confirmation of significant RAS in patients as an indicator of a potential positive response to stent placement. Inclusion criteria for more recent studies have required noninvasive vascular interrogation in conjunction with physiological factors to ensure patient benefit. A trial on PTRAS efficacy on RAS-induced HTN and renal dysfunction using narrowed selection criteria required angiographically confirmed stenoses between 70% and 99% prior to stent placement. Of the 64 enrolled patients, an overall benefit rate of 87.5% for HTN response and 86.4% with improved renal function after 12 months was reported, countering earlier published reports of failed efficacy and bolstering the incorporation of sensitive imaging modalities in patient selection. 26 Comparative reports on angiographic evaluation of RAS and duplex sonography have demonstrated favorable correlation in defining hemodynamically significant stenosis compared with other methods (Figure 8).3,15,18
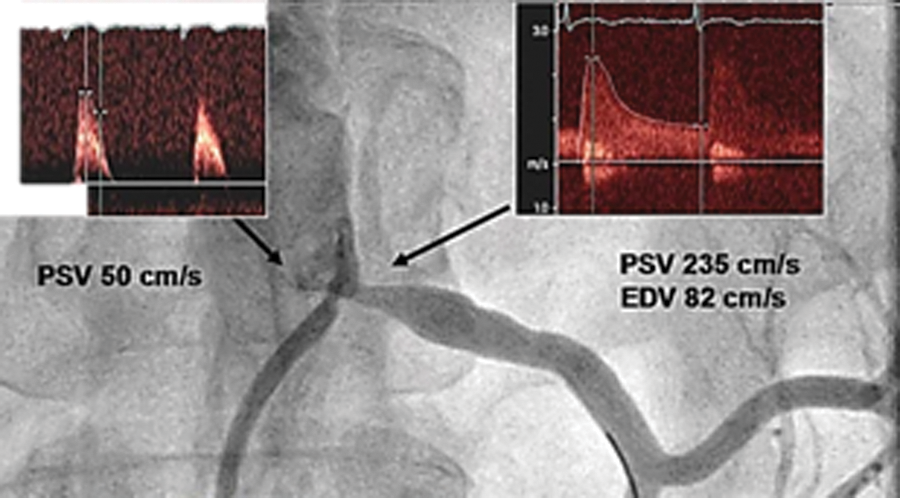
Angiography compared with sonographic measurements and transstenotic pressure gradient measurements in a left renal artery stenosis. Inserts in the upper part of the figure depict peak systolic velocity (PSV) in the aorta (left) and at the level of the stenosis. 15
The Role of Sonography in RAS
The inclusion of duplex sonography pre- and postprocedurally by Sharafuddin et al 5 demonstrated improved selection of patients appropriate for PTRAS placement in a study of 18 patients with 22 renal arteries confirmed by angiography as ≥70%. The enhanced role of sonography in follow-up examinations also demonstrated a positive correlation with stent efficacy and outcome in RAS patients. Quantitative analyses of waveforms in follow-up were compared with preprocedural waveforms, showing normalization of previously abnormal Doppler data in hemodynamically successful stent placements (Figure 9).
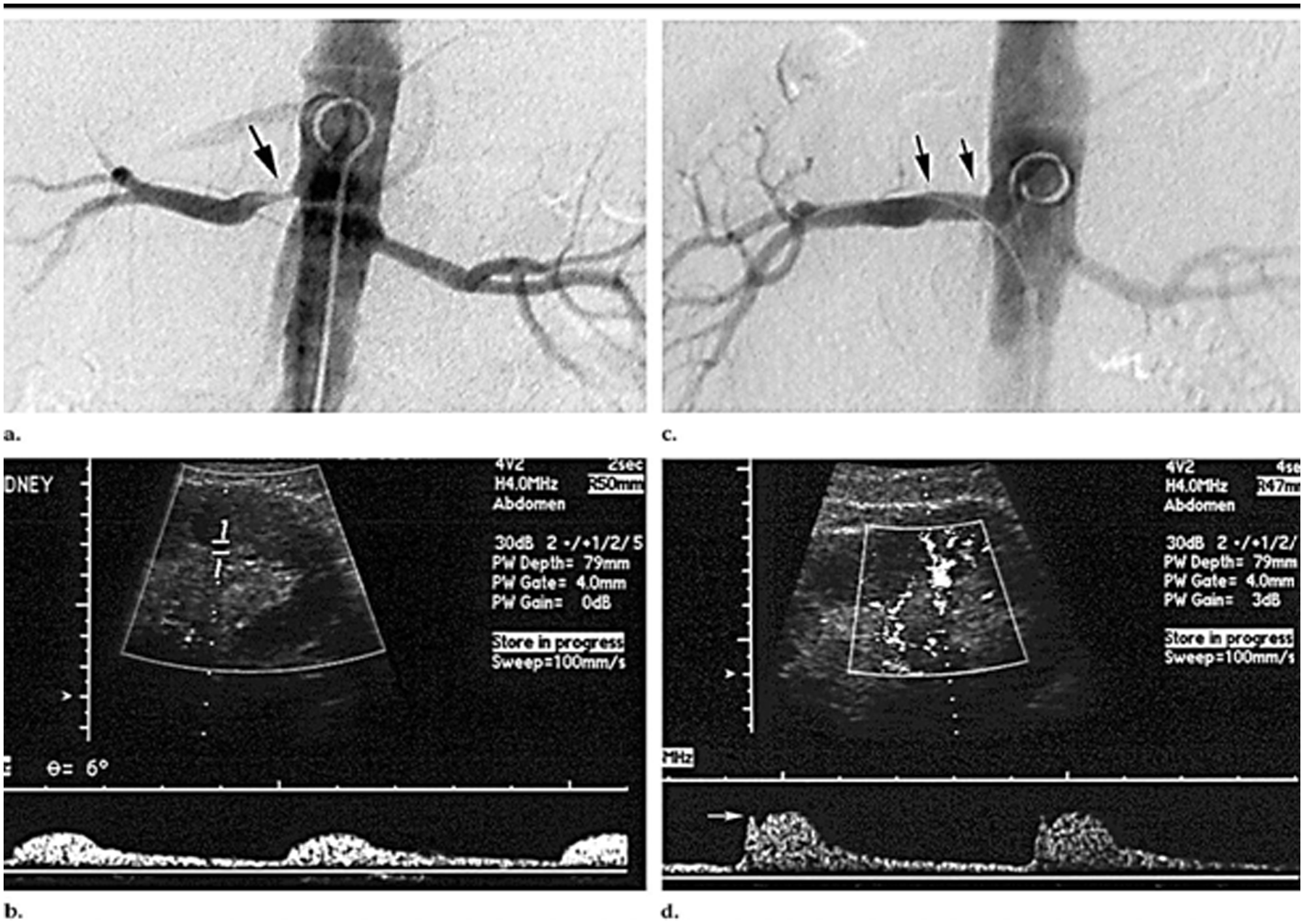
(a) Angiogram of right renal artery stenosis (arrow). (b) Corresponding longitudinal duplex sonogram. Doppler waveform from a lower-pole segmental artery demonstrates a tardus-parvus pattern. (c) Angiogram following percutaneous transluminal renal artery stent (PTRAS) demonstrates widely patent right renal artery ostium and lumen (arrows). (d) Corresponding longitudinal duplex sonogram. Doppler waveform from a lower-pole segmental artery, obtained after PTRAS, demonstrates marked improvement in waveform morphology. 5
Watson et al 27 used duplex sonography to obtain peak systolic velocities throughout renal artery stents in 41 patients postprocedurally and found an increased incidence of elevated velocities characteristic of some degree of residual stenosis, suggesting that outcomes were possibly less related to the efficacy of stents in RAS but in some cases to a partial technical failure of the procedure despite the appearance of anatomic success. ODORI was a multicenter prospective study, carried out between 2005 and 2007, that evaluated stent efficacy on renovascular HTN and function in 251 patients. The study used duplex sonography preprocedurally to confirm a renal artery PSV of ≥180 cm/s and/or a RAR of ≥3.5. The interventional end point was <30% residual stenosis with end-procedural measured residual stenosis averaging only 2.5%; using the same sonographic parameters in follow-up examinations, postprocedural stenoses were noted in only 6.6% of patients at one year. 28
In a study by Gill et al, 29 sonography was used to determine hemodynamically significant RAS in 100 patients (126 vessels), which was compared to angiography prior to stent placement. Stabilized or improved renal function was seen after a mean follow-up of 25 months in 72.5% of 65 patients treated for renal impairment; 79% of 48 patients treated for medication-resistant hypertension showed improvement in blood pressure control, and 4.2% were cured of their HTN, a testament to the essential need for accuracy in screening patients prior to PTRAS. Riehl et al 30 were able to demonstrate angiographically confirmed ≥70% RAS by measuring the resistance index and the right-left variation of resistive indices (RIs) between normal and stenosed renal arteries. In a similar study, Schwerk et al 31 equated a right-left difference in RI of >5% with a hemodynamically significant >60% RAS with good sensitivity and specificity. A large ongoing RAS study on the cardiovascular outcomes following stent placement, CORAL (Cardiovascular Outcomes in Renal Atherosclerotic Lesions), includes 1080 patients screened by duplex sonography with angiographic confirmation and uses renal length as part of the inclusion criteria. 2 A smaller study by Watson et al 27 on 33 patients and 61 vessels combined creatinine levels and pre- and postprocedural renal length to determine stent efficacy in treating RAS. A right-left renal pole-to-pole length discrepancy of ≥1.5 cm has also been suggested as an indicator of unilateral RAS and can be used to assess postprocedural renal atrophy caused by stent failure. 8
The RADAR Trial is an ongoing prospective study of 300 patients using duplex sonography as an integral component of assessing hemodynamically significant RAS. 17 RAS patients receive a baseline sonographic assessment of RAS severity confirmed by angiography to establish accuracy as well as a postoperative assessment at 6 and 12 months using the criteria for hemodynamically significant stenosis. The study criteria for stenosis are a right-left intrarenal resistive index ((PSV-EDV)/PSV) difference that is equal to or greater than 0.05, indicating a unilateral hemodynamically significant stenosis to be confirmed by angiography for lumen reduction of greater than or equal to 70%. For bilateral renal artery stenoses, the criterion is an increased acceleration time distal to the stenotic sites of greater than or equal to 0.07 seconds.
Conclusion
The increasing number of studies implementing duplex sonography in RAS assessment indicates the increased value and credibility that this modality has gained over the years. The accuracy demonstrated by sonographic evaluation using standardized protocols creates a valuable imaging modality for vessel assessment. The direct and indirect evaluation of renal disease with sonography provides a more complete overview of renal pathology in RAS and is relevant as well in postoperative stent response determination. The use of state-of-the-art sonography in the noninvasive evaluation of RAS more accurately identifies the affected patient population and has improved patient selection for angioplasty and stenting to those who have the greatest potential to benefit. The improved identification of hemodynamically as well as anatomically significant RAS will likely result in better definition of appropriate patients for the application of intervention in renovascular disease and in improved stent efficacy in reported results. The implementation of duplex sonography in RAS provides the patient a safe, inexpensive, effective evaluation of a disease that is difficult to detect, manage, and treat and provides ready access to appropriate care that measurably improves patients’ quality of life.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
