Abstract
Open lung biopsy is the gold standard for diagnosing lung cancer, but it is an invasive approach. There have been several noninvasive methods developed to biopsy the lung. This literature review compares the use of endobronchial ultrasound (EBUS) and computed tomography with fluoroscopy (CTF) guidance in the biopsy of lung nodules. Computed tomography with fluoroscopy has a higher diagnostic yield than endobronchial ultrasound but has more postprocedural complications. Endobronchial ultrasound guided lung biopsy is effective when the lesions are located in the right middle lobe of the lung, and overall EBUS has a low rate of complications when compared to CTF.
Introduction
Lung cancer presents clinically late in its course, and therefore only has a median survival rate of 6 to 12 months following diagnosis. 1 Because of this low survival rate, quick and accurate diagnosis is crucial in treatment planning. The most important predictor of lesion resectability, treatability, and prognosis is accurate sampling technique, leading to accurate staging. 2 The gold standard to diagnose lung cancer is an open lung biopsy, a highly invasive and aggressive approach. 3 There are noninvasive methods available to biopsy the lung for malignancy determination and staging. Two of these are computed tomography with fluoroscopy (CTF) and endobronchial ultrasound (EBUS). While endobronchial biopsy, a valid biopsy technique, has been reported in the literature for over 20 years, endobronchial ultrasound guided biopsy was only sporadically reported in the literature in the 1990s and has been studied with increasing frequency only in the past 10 years. Computed tomography with fluoroscopy has been used to guide lung biopsies for over 20 years, and the more recent advances in multi-slice technology have refined the capabilities of this technique. The goal of this literature review is to compare EBUS and CTF guided lung biopsies and to determine when each is best used in the diagnosis of lung cancer.
Methods
An online search for articles related to lung biopsies guided by endobronchial ultrasound and computed tomography with fluoroscopy was conducted. Articles containing original research on these topics were considered for this review. Full-text analysis was performed on the selected articles. The results of each study were divided into categories and compared related to the size of the lesion biopsied, the location of the lesion biopsied, the overall diagnostic yield of the procedure, and the safety of the procedure.
Endobronchial Ultrasound Guidance
Procedure
Endobronchial ultrasound guided transbronchial biopsies (EBUS-TBB) are typically done with a flexible 20-MHz radial endocavitary transducer. The procedure is performed with the patient under conscious sedation or general anesthesia, and a local anesthetic is used to numb the area of interest. A flexible bronchoscope is inserted into the bronchus. The transducer is placed inside a guide sheath, which is then inserted into the working channel of the bronchoscope. It is advanced toward the pulmonary lesion or target of interest. Sonographically, alveolar tissue is white with a “snowstorm” appearance due to the presence of air. 4 Solid lesions will appear as dark, homogenous structures that contrast markedly with the normal lung tissue. Once a solid lesion is visualized, it is assumed that the transducer is located at the target. Once the target is located, the transducer may be removed from the guide sheath, but the guide sheath is left in place at the lesion in the event further imaging is necessary at some point later in the procedure. A bronchial brush, biopsy forceps, or both are inserted into the guide sheath, and the biopsy typically is performed blind, or without imaging guidance. An alternate approach includes leaving the EBUS transducer in place while the lesion is biopsied under real-time guidance, although this technique appears to be performed infrequently. 2
If the lesion(s) is not seen at the target area sonographically, then fluoroscopy can be used to assist in locating the lesion. In this case, the transducer is removed from the guide sheath and replaced by a curette, which is guided via fluoroscopy. 5 Once the target bronchus is identified, the curette is removed and the transducer is reinserted to allow the biopsy to continue as previously described. Another option is to continue with the biopsy blindly at the target area. 6
Diagnostic Yield of EBUS-TBB by Size
When comparing the diagnostic potential of EBUS with transbronchial biopsy (TBB), the diagnostic yield appears to increase as the size of the lesion increases (Table 1). Asahina et al. evaluated 30 lung nodules in 29 patients and found that EBUS-TBB provided diagnostic results in 44% of lesions (8 out of 18) that measured less than 20 mm and 92% of lesions (11 out of 12) that measured between 20 and 30 mm. 5 In a larger study of 150 patients, Kurimoto et al. detected diagnostic results in 73% of lesions (59 out of 81) that measured less than 20 mm, 77% of lesions (33 out of 43) that measured between 20 and 30 mm, and 92% of lesions (24 out of 26) that measured greater than 30 mm. 7 Eberhart et al. studied 39 patients with lung lesions by EBUS and found a higher diagnostic yield in smaller lesions than in the studies by Asahina and Kurimoto. 6 Endobronchial ultrasound with biopsy detected diagnostic results in 78% of lesions (7 out of 9) that measured less than 20 mm, 70% of lesions (16 out of 23) that measured between 20 and 30 mm, and 57% of lesions (4 out of 7) that measured over 30 mm.
Endobronchial Ultrasound Guided Transbronchial Biopsies (EBUS-TBB) Diagnostic Yield by Lesion Size
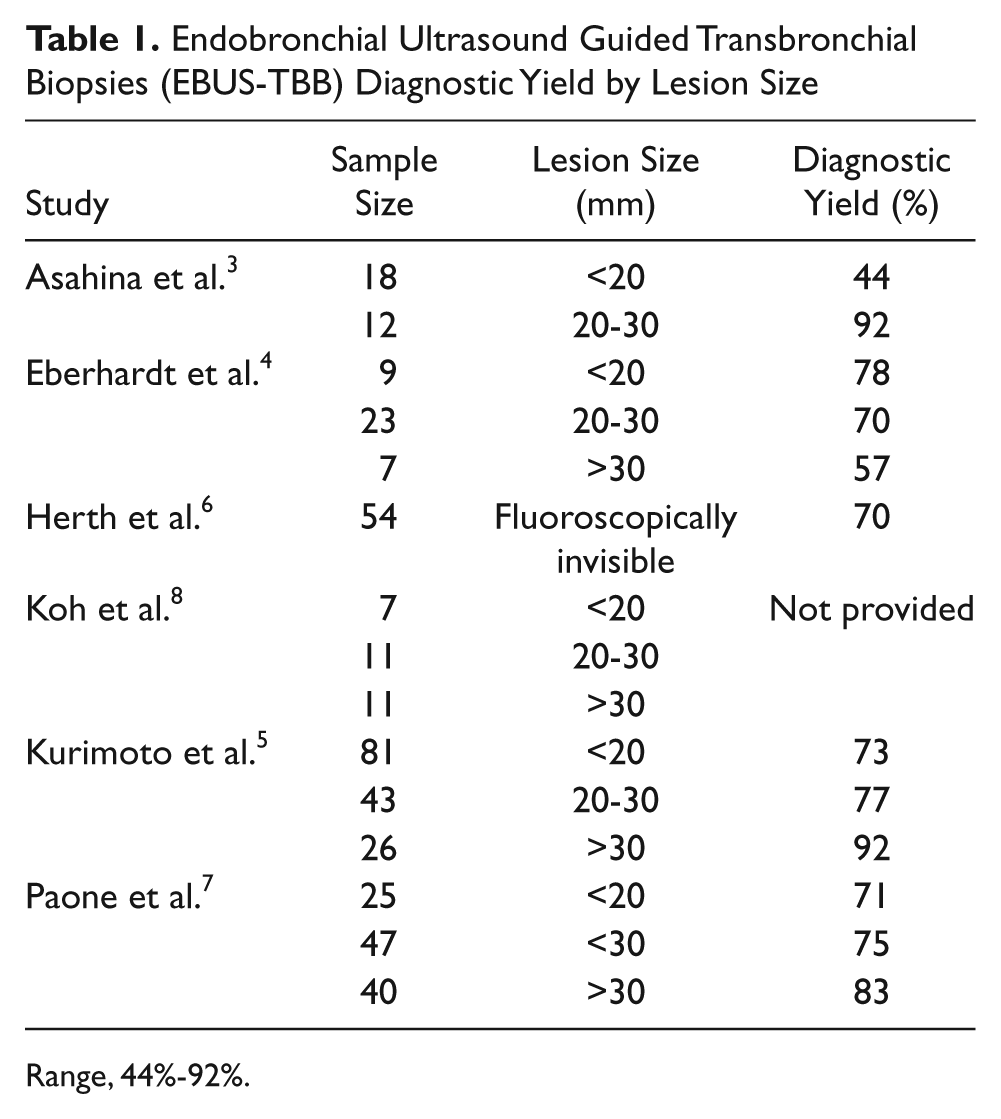
Range, 44%-92%.
Herth et al. performed EBUS-TBB on 138 patients whose lung nodules were not visible by fluoroscopy. 4 Their results showed that 70% of lesions (38 out of 54) could be successfully detected and biopsied with EBUS guidance. Paone et al. evaluated 206 patients, randomly assigning them to receive either EBUS or transbronchial biopsy alone. 8 Endobronchial ultrasound guided biopsy demonstrated a success rate of 71% for lesions measuring less than 20 mm, 75% for lesions measuring less than 30 mm, and 83% for lesions measuring larger than 30 mm. 8
Diagnostic Yield Using EBUS-TBB by Location
There is variance in the diagnostic yield of lesions depending on the lobe of the lung where the lesion is discovered. Of the EBUS research reviewed, the highest accuracy was noted when the biopsy occurred in the right middle lobe of the lung. Herth et al. had a 42% success rate (6 out of 14 lesions) in the right upper lobe, 100% success rate (8 out of 8 lesions) in the right middle lobe, 100% success rate (8 out of 8 lesions) in the right lower lobe, 50% success rate (5 out of 10 lesions) in the left upper lobe, and 42% success rate (11 out of 14 lesions) in the left lower lobe. 4 Kurimoto et al. showed success rates of 74% (34 out of 46 lesions) in the right upper lobe, 100% (14 out of 14 lesions) in the right middle lobe, 67% (20 out of 30 lesions) in the right lower lobe, 61% (22 out of 36 lesions) in the left upper lobe, and 79% (19 out of 24 lesions) in the left lower lobe. 7 Eberhart et al. showed 59% success (16 out of 27 lesions) in the upper lobes, 100% (3 out of 3 lesions) in the right middle lobe, and 89% (8 out of 9 lesions) in the lower lobes. 6 In contrast to these studies, Asahina et al. had a success rate of 55% (6 out of 11) of lesions located in the right upper lobe, 0% (0 out of 1) of lesions in the right middle lobe, 57% (4 out of 7) of lesions in the right lower lobe, 100% (7 out of 7) of lesions in the left upper lobe, and 50% (2 out of 4) of lesions in the left lower lobe. 5 However, no significant conclusions should be drawn from this report as only one lesion involved the right middle lobe.
Not all studies gave specific information about the diagnostic yield of their biopsies according to location of the targeted lesion. Paone et al. concluded that the success of the biopsies in their series was independent of the lesion location. 8 Although it appears the EBUS-TBB is most successful when the lesions are located in the right middle lobe of the lung, the actual number of lesions found in this location is less than those found in other lobes. These findings may be less significant due to the disparity in sample sizes among the different lobes of the lung.
Overall Diagnostic Yield and Safety of EBUS-TBB
The overall diagnostic yield for EBUS-TBB remained relatively consistent across the studies included in this literature review (Table 2). Although the reported EBUS biopsies did not always result in a diagnosis, a diagnosis was reached 63% to 77% of the time.4–9 Those series that were able to obtain yields in the 70th percentile may have been more successful due to the greater number of specimens obtained during those biopsies.7,8
Endobronchial Ultrasound Guided Transbronchial Biopsies (EBUS-TBB) Overall Diagnostic Yield

Range, 63%-77%
Complications reported across all the EBUS studies were minimal (Table 3). Few research studies reported major complications. Pneumothorax was reported from 0% to 5% of the time,4,6,9 and minimal to moderate bleeding was also reported on occasion.4,7 Endobronchial ultrasound with biopsy appears to have few complications, but the overall low complication rates may be due to the fact that EBUS-TBB is used less often than other biopsy methods and these patients may reflect a selection bias based on favorable anatomy.
Endobronchial Ultrasound Guided Transbronchial Biopsies (EBUS-TBB) Complications

Nine total complications of 389 patients or 2% overall complication rate.
Computed Tomography With Fluoroscopy Guidance
Procedure
Computed tomography with fluoroscopy guidance uses a computed tomography system equipped with software that allows it to act like a fluoroscope. The CT scanner is able to rotate quickly in one plane, creating a real-time CT image. Before the biopsy begins, a preliminary CT is done to localize the target lesion and optimize patient positioning. The patient position—supine, prone, or lateral decubitus—is chosen to allow the shortest and most direct route to the target lesion. After the lesion is identified, an introducer needle is advanced under real-time CTF imaging. The imaging is limited to intermittent views of the needle to observe its advancement. The needle may be held by surgical forceps to decrease the amount of radiation to the operator’s hand. After it is determined that the introducer needle is at the tip of the targeted lesion, the biopsy needle is then inserted through the introducer needle and the specimens are obtained.
A traditional CT guided biopsy would require that the operator leave the examination room to review images, but CTF allows the operator to view real-time needle placement while at the patient’s side. The real-time aspect of CTF lung biopsy is more efficient than conventional CT scanners in that it can reduce the examination time overall, but it does increase the radiation exposure to both the patient and the operator. 10 The operator is most at risk for scatter radiation from the patient, and the operator’s hands are at risk during the procedure because they may be directly exposed to radiation if they are in the CT beam during the biopsy procedure. 10 Although operator exposure data comparing conventional CT to CTF does not exist, Kim et al. found that the effective mean dose to the operator’s hands during a CTF lung biopsy procedure was 0.759 mSv as compared to 0.034 mSv for the conventional CT lung biopsy, a greater than 20-fold increase. This study also determined that patients received a higher radiation dose under CTF, 6.53 mSv for CTF compared to 2.72 mSV for conventional CT. 10 For the operator, the use of 6 mm slices and the addition of a needle holder may reduce radiation exposure.10,11 For the patient, application of the ALARA principle is the most appropriate method to minimize radiation exposure.
Directly after the biopsy, a CT scan is done to determine whether the patient has a pneumothorax or any other complications. Patients who demonstrate no pneumothorax may proceed with a chest radiograph several hours after the procedure to verify the absence of pneumothorax. Patients with a moderate or severe pneumothorax noted on the CT scan may be treated on the scanning table by aspiration or chest tube placement immediately after the biopsy. 12
Diagnostic Yield With CTF by Size
When assessing the diagnostic potential based on the size of a lesion using CTF, similar to EBUS, the diagnostic yield increases as the size of the lesion increases (Table 4). Hiraki et al. studied 1000 patients and yielded diagnostic results in 93% of lesions (140 out of 151) measuring less than 10 mm, 96% of lesions (414 out of 431) measuring 11 to 20 mm, 98% of lesions (208 out of 213) measuring 21 to 30 mm, and 93% of lesions (190 out of 205) measuring greater than 31 mm. 13 Yamagami et al. showed similar results in 110 patients with 88% of lesions (22 out of 25) measuring less than 10 mm, 96% of lesions (39 out of 40) measuring 10 to 20 mm, 100% of lesions (31 out of 31) measuring 20 to 30 mm, and 100% of lesions (8 out of 8) measuring greater than 30 mm. 12 Hirose et al. studied 50 patients and showed an accuracy rate of 89% of lesions (24 out of 27) measuring less than 20 mm and 91% of lesions (21 out of 23) measuring greater than 20 mm. 14 Some studies reviewed did not give diagnostic rates by size of the lesion but did give the median size, average size, or range of sizes overall.10,15,16 This finding may be attributed to the fact that most studies done using CTF have put more weight on overall diagnostic yield and complications than the lesion size.
Computed Tomography With Fluoroscopy (CTF) Guided Biopsy Diagnostic Yield by Lesion Size
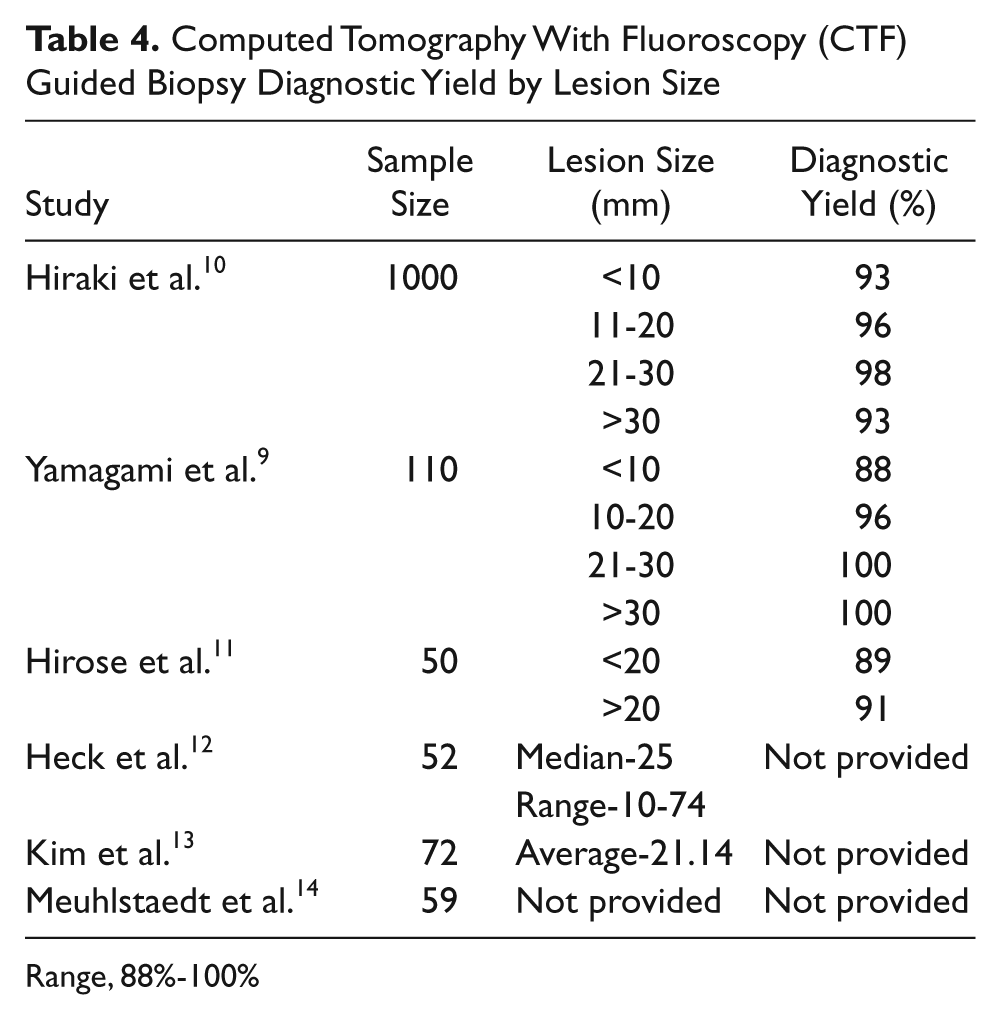
Range, 88%-100%
Diagnostic Yield by Location With CTF
As compared to diagnostic yield by size, not all CTF studies reviewed focused on the diagnostic yield by location of a lesion. Hiraki et al. was the only study to give diagnostic results both by lesion size and location of the lesion. 13 The study showed a diagnostic yield of 97% for lesions (583 out of 603) located in the upper and middle lobes and 93% for lesions (369 out of 397) located in the lower lobes. Studies by both Heck et al. and Kim et al. did not report diagnostic yield by lesion size, but did report diagnostic yield by lesion location.10,15 Heck et al., in 52 patients, reported that 33% of the lesions biopsied were in the upper lobes, 7% were in the right middle lobe, and 58% were in the lower lobes. 15 Kim et al. studied 72 patients and reported 44 lesions in the upper and middle lobes and 28 lesions in the lower lobes. 10
Since CTF guided biopsies are done from the external chest wall rather than endobronchially, the depth of the lesion of interest is an important aspect that can affect diagnostic yield. Hiraki et al. demonstrated a diagnostic yield of 95% for lesions (143 out of 151) with a 0 mm depth, 96% for lesions (177 out of 185) with a depth of 1 to 10 mm, 94% for lesions (208 out of 222) with a depth of 11 to 20 mm, 96% for lesions (189 out of 196) with a depth of 21 to 30 mm, and 96% for lesions (206 out of 215) with a depth greater than 31 mm. 13 Hirose et al. reported a diagnostic yield of 92% for lesions (36 out of 39) with a depth less than 60 mm and 82% for lesions (9 out of 11) with a depth greater than 60 mm. 14 While Hirose et al. found no significant difference in diagnostic yield based on the depths of the lesions, Hiraki et al. demonstrated a higher diagnostic value with superficial lesions at depths closer to the chest wall.13,14 The variations in results between the two studies can likely be attributed to the relatively small number of patients in the series of Hirose et al. compared to the much larger sample reported in the Hiraki et al. study.
Overall Diagnostic Yield and Safety With CTF
The overall diagnostic yield for biopsies guided by CTF remained relatively consistent over the studies included in this literature review (Table 5) with a diagnosis made 88% to 96% of the time.10,14–16 Several complications also were reported consistently across these studies (Table 6). Pneumothorax is the most common complication seen with CTF, with an incidence ranging from 2% to 42%.10,12–16 Heck et al. reported that number of pneumothorax cases, 15 out of 57 (26%) was not statistically different from the number of pneumothorax cases seen when biopsies were performed using conventional CT, 16 out of 42 (38%), but Kim et al. did report a statistically significant difference in the cases of pneumorthorax seen by CTF, 8 out of 72 (11.1%) versus 19 out of 72 (27%) by conventional CT. 10 In some instances, the biopsies were terminated due to a rapidly progressing pneumothorax. 13 Intrapulmonary bleeding was the next most commonly seen complication (Table 6).10,13–16 Hemoptysis,10,12,14 subcutaneous hematoma, 12 and plural effusion 16 were other complications that were noted, but these were seen infrequently.
Computed Tomography With Fluoroscopy (CTF) Guided Biopsy Overall Diagnostic Yield
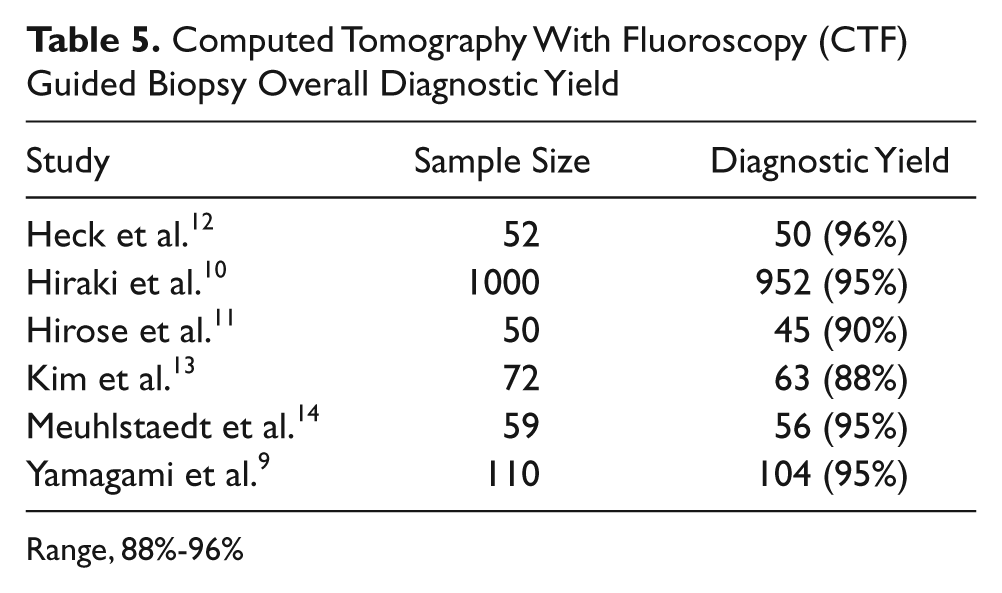
Range, 88%-96%
Computed Tomography With Fluoroscopy (CTF) Guided Biopsy Complications
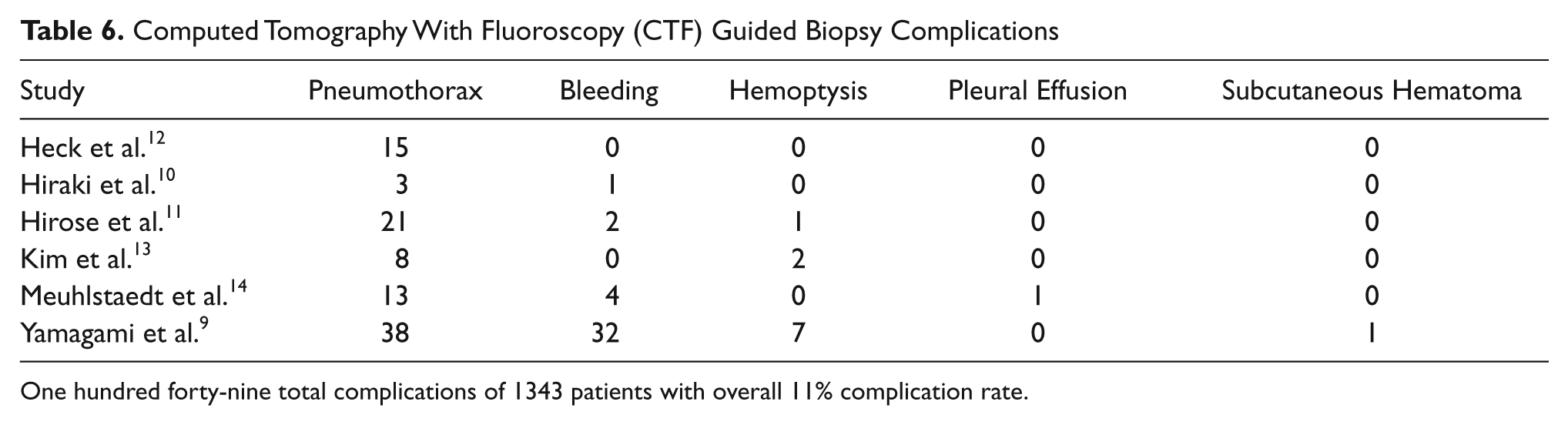
One hundred forty-nine total complications of 1343 patients with overall 11% complication rate.
Discussion
In lesions measuring less than 20 mm, CTF guided biopsies demonstrated a higher diagnostic yield in comparison with EBUS guided biopsies (Tables 1 and 4). For instance, CTF reported yields ranging from 88% to 96%, while EBUS reported yields ranging from 44% to 78%. This finding possibly indicates that the size of the lesion may not be a significant factor in the diagnostic success of CTF procedures, but it is likely an important consideration for EBUS. The diagnostic yield in lesions measuring greater than 20 mm were more comparable to each other, with CTF ranging from 91% to 100% and EBUS ranging from 57% to 92%. The success rate increased with increasing lesion size in both groups, though much more significantly for EBUS. It is worth noting that CTF can only biopsy lesions that are fluoroscopically visible, whereas EBUS has shown a diagnostic yield of 70% with fluoroscopically invisible lesions regardless of size. 4
Lesion location as well appears not to influence the diagnostic yield of CTF biopsies, but it does seem to be a more important factor for diagnostic yield by EBUS. A trend is observed in the reported literature that there is a better diagnostic yield in lesions that are located in the upper and middle lobes of the lung compared to lower lobes of the lung for both CTF and EBUS. When considering only the lower lobe, CTF shows an overall higher success rate. Reports also indicate that CTF guided procedures are more successful when the lesion is more superficial and closer to the chest wall. Since EBUS-TBB is done endobronchially, there is less of an issue with biopsies performed on lesions deeper in the chest. More studies that look specifically at lesion depth would be helpful to differentiate which modality has the higher diagnostic yield for superficial lesions versus deeper lesions.
Overall, the ability to make a diagnosis is higher with CTF guided biopsies than EBUS guided biopsies. Diagnostic yields ranged from 88% to 96% for CTF while EBUS showed a range of 63% to 77% (Tables 2 and 5). However, when selecting a biopsy method, all aspects of the modalities should be considered as well as the lesion characteristics, including size and location within the lung. While CTF is not the only imaging modality able to show real-time images during a biopsy, it is less common for EBUS-TBB to be done under real-time guidance. The advantages of real-time CTF guidance is at the cost of increased radiation exposure to the patient and operator. The patients’ radiation doses are further increased by the need of an immediate CT following the biopsy to determine if there are complications.
More complications were noted with biopsies using CTF guidance in comparison to EBUS guidance. There were complications 2% of the time (9 of 389 participants) with the EBUS studies reviewed (Table 3) compared to complications 11% of the time (149 of 1343 participants) noted in the CTF studies (Table 6). In a population-based risk study performed by Weiner et al., who looked at 15,865 patients who had conventional CT guided lung biopsy, pneumothorax appears to result about 15% of the time, and pneumothorax requiring a chest tube occurs about 6.6% of the time. 17 In the research reviewed, both CTF and EBUS fared better than conventional CT guided lung biopsies. Larger studies comparing EBUS to CTF for lung biopsy in a randomized fashion are warranted to determine if the favorable comparisons withstand additional research. Currently, the physician should consider lesion location, lesion size, lesion depth from the chest wall, and the potential for complications when deciding which biopsy method is best for the patient. In cases where radiation exposure is a concern, a physician can feel confident in the diagnostic yield of EBUS.
Conclusion
The overall diagnostic yield of CTF guided biopsies is higher than that of EBUS guided biopsies according to literature reviewed. The increased diagnostic accuracy, particularly for small lesions, and the ability to use real-time imaging during the biopsy are advantages of CTF biopsies. EBUS has advantages in that deeper lesions may offer less of a challenge to locate and it offers an option for lesions that are not visible by fluoroscope. Additional research is needed to isolate the best noninvasive option based on lesion location and size while minimizing the complications.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
