Abstract
Metastatic lesions of the pancreas are uncommon. The incidence of pancreatic involvement with squamous cell carcinoma is the lowest among the different histological subtypes of lung cancer. We present the clinical data, pathological findings, and imaging features of two cases of pancreatic metastasis from squamous cell lung cancer with a literature review. A 75-year-old man and a 67-year-old man were both diagnosed with metastatic pancreatic tumors from squamous cell lung cancer during a multidisciplinary team discussion. Both patients were misdiagnosed as having a neuroendocrine tumor of the pancreas by imaging examination before surgery. Squamous cell lung cancer metastasis to the pancreas is extremely rare, and the atypical clinical and radiological presentations make it difficult to distinguish metastatic tumors from primary pancreatic tumors.
Keywords
Background
Metastatic lesions to the pancreas are uncommon and are estimated to account for up to 3% to 5% of all pancreatic lesions.1,2 When metastasis does occur, the most common primary tumor is renal cell carcinoma, followed by lung cancer, colorectal cancer, and sarcoma. 3 The most frequent sites of lung cancer metastases are the liver, bones, brain, and adrenal gland, whereas the pancreas is a rather uncommon site of metastatic lung cancer. Specifically, the majority of the pancreatic metastases arise from small-cell lung carcinoma, with an incidence of 10%, followed by adenocarcinoma (2.4%), large-cell carcinoma (1.9%), and finally, squamous cell carcinoma, with an incidence of only 1.1%. 4
The diagnosis of metastatic lesions in the pancreas is challenging because they are easily misdiagnosed as primary pancreatic tumors. We herein present two cases of metastatic squamous cell carcinoma involving the pancreas from a primary lung cancer and discuss the differential diagnosis of metastatic lesions of the pancreas, with a literature review.
Case presentation
Case 1
A 75-year-old man presented with a history of vomiting and abdominal distension for 5 months and obstructive jaundice for 2 weeks. He reported a history of left upper lobectomy for poorly-differentiated squamous cell lung cancer 1 year earlier. He received postoperative chemotherapy and immunotherapy (paclitaxel with lobaplatin and camrelizumab) and remained in good condition with no evidence of local or systemic recurrence. His carbohydrate antigen 19-9 concentration was 216.86 U/mL (normal range: 0–34 U/mL), carbohydrate antigen 125 concentration was 181.8 U/mL (normal range: 0–35 U/mL), and the heat shock protein 90α concentration was 111.92 ng/mL (normal range: 0–82.6 ng/mL). The total bilirubin concentration was 43.6 µmol/L (normal range: 0–23 µmol/L), direct bilirubin concentration was 39.9 µmol/L (normal range: 0–8 µmol/L), and indirect bilirubin concentration was 3.7 µmol/L (normal range: 0–20 µmol/L).
Abdominal computed tomography (CT) revealed an ill-defined solid tumor measuring 4 × 3.2 cm in the pancreatic head, resulting in enlargement of the pancreatic head, and dilation of the intrahepatic and extrahepatic biliary ducts and pancreatic duct. The tumor showed slight enhancement in the arterial phase and heterogeneous moderate enhancement in the portal venous phase (Figure 1). The pancreatic body and tail were edematous with peripancreatic inflammatory changes. The superior mesenteric vein was invaded by the tumor, and the portal vein was embedded within the tumor, leading to varicosity of the distal splenic vein and left gastric vein. Lymphadenopathy was also detected in the porta hepatis, portacaval space, hepatogastric ligament, para-aortic region, celiac trunk, mesentery, and behind the pancreatic head. On magnetic resonance imaging (MRI), the tumor showed hypointensity on T1-weighted images (T1WI) and slight hyperintensity on T2-weighted images (T2WI). The tumor showed slight enhancement during the arterial phase, heterogeneous moderate enhancement during the portal venous phase, and persistent heterogeneous enhancement during the delayed phase (Figure 2). Magnetic resonance cholangiopancreatography (MRCP) demonstrated dilation of both the biliary and pancreatic ducts. Lymphadenopathy was also detected using MRI. No evidence of other masses/metastatic disease was detected either clinically or by CT. For this reason, the pancreatic mass was considered to represent a primary pancreatic neoplasm, such as a neuroendocrine tumor.

Computed tomography (CT) images showing a tumor in the pancreatic head. The tumor shows slight enhancement in the arterial phase and heterogeneous moderate enhancement in the portal venous phase with contrast enhancement. Invasion to the superior mesenteric vein (white arrow, Figure 1e), pancreatitis (white arrow, Figure 1d), and lymphadenopathy are also seen: (a)-(e) were axial CT images with contrast enhancement and (f) was coronal CT image with contrast enhancement.

Magnetic resonance (MRI) images showing a tumor in the pancreatic head. The tumor shows hypointensity on T1-weighted images (T1WI) and slight hyperintensity on T2-weighted images (T2WI) and slight enhancement in the arterial phase, heterogeneous moderate enhancement in the portal venous phase, and persistent heterogeneous enhancement in the delayed phase with contrast-enhanced imaging: (a) was axial T1WI image, (b) was axial T2WI image, (c)-(f) were axial contrast-enhanced images and (g) was coronal T2WI image.
Considering all findings, an investigative laparotomy was performed, during which a tumor was found in the pancreatic head infiltrating surrounding structures. The tumor was not surgically resectable, and palliative operation was elected. Cholecystojejunostomy and gastrojejunostomy were performed to relieve the jaundice, and aspiration biopsy of the tumor was performed for cytopathological examination. The biopsy results identified dysplastic squamous epithelium, which was positive for immunostaining for P40, P63, CK19, and CK5/6 (Figure 3).
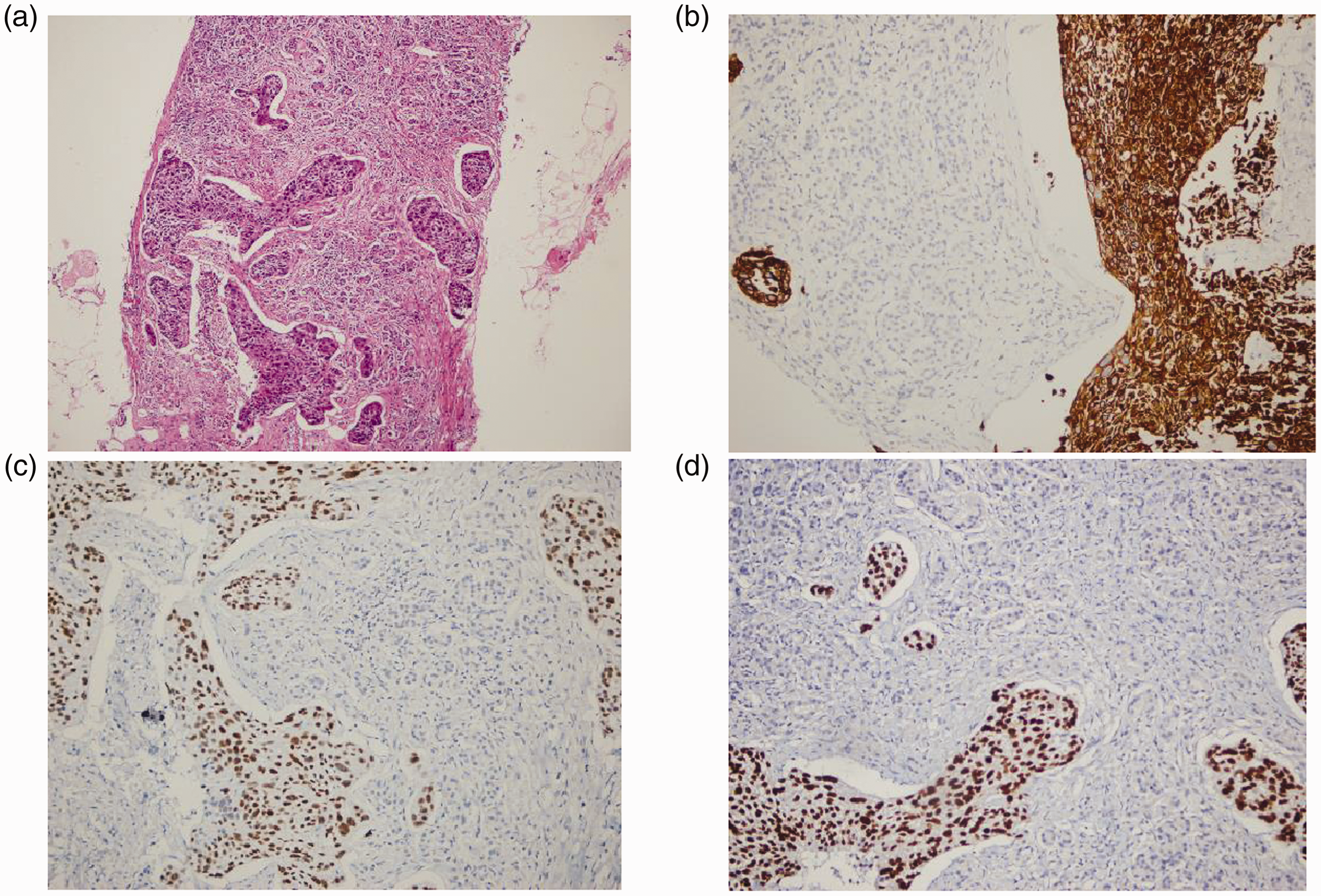
Histological images of the pancreatic tumor showing squamous cell carcinoma in the pancreas (hematoxylin and eosin staining (HE), ×10 magnification). The squamous component was positive with immunostaining for CK5/6, P40, and P63 (×10, ×10, and ×20 magnification, respectively): (a) was HE staining of the specimen, (b) was immunohistochemical staining with CK5/6, (c) was immunohistochemical staining with P40 and (d) was immunohistochemical staining with P63.
At a multidisciplinary team discussion, a diagnosis consistent with metastatic squamous cell carcinoma of the lung was agreed, and the patient was treated with chemotherapy and immunotherapy. To date, no other abnormalities have been found in the patient during 2 months of follow-up.
Case 2
A 67-year-old man presented with a 1-week history of vomiting and abdominal pain. He reported a history of left lower lobectomy for squamous cell lung cancer 3 years earlier. He also had diabetes mellitus for 8 years. The patient received postoperative chemotherapy and immunotherapy (paclitaxel with lobaplatin and camrelizumab) and remained in good condition for 3 years after surgery with no evidence of local or systemic recurrence; all tumor marker concentrations were normal. MRI at the current admission revealed a well-defined tumor measuring 4.6 × 4.1 cm in the pancreatic head (Figure 4). The tumor was hypointense on T1WI and slightly hyperintense on T2WI with slight enhancement in the arterial phase, heterogeneous moderate enhancement in the portal venous phase, and persistent heterogeneous enhancement in the delayed phase, without biliary obstruction or evidence of disease in the chest or elsewhere. No evidence of other masses/metastatic disease was detected either clinically or by CT. The tumor was initially diagnosed as a neuroendocrine tumor.
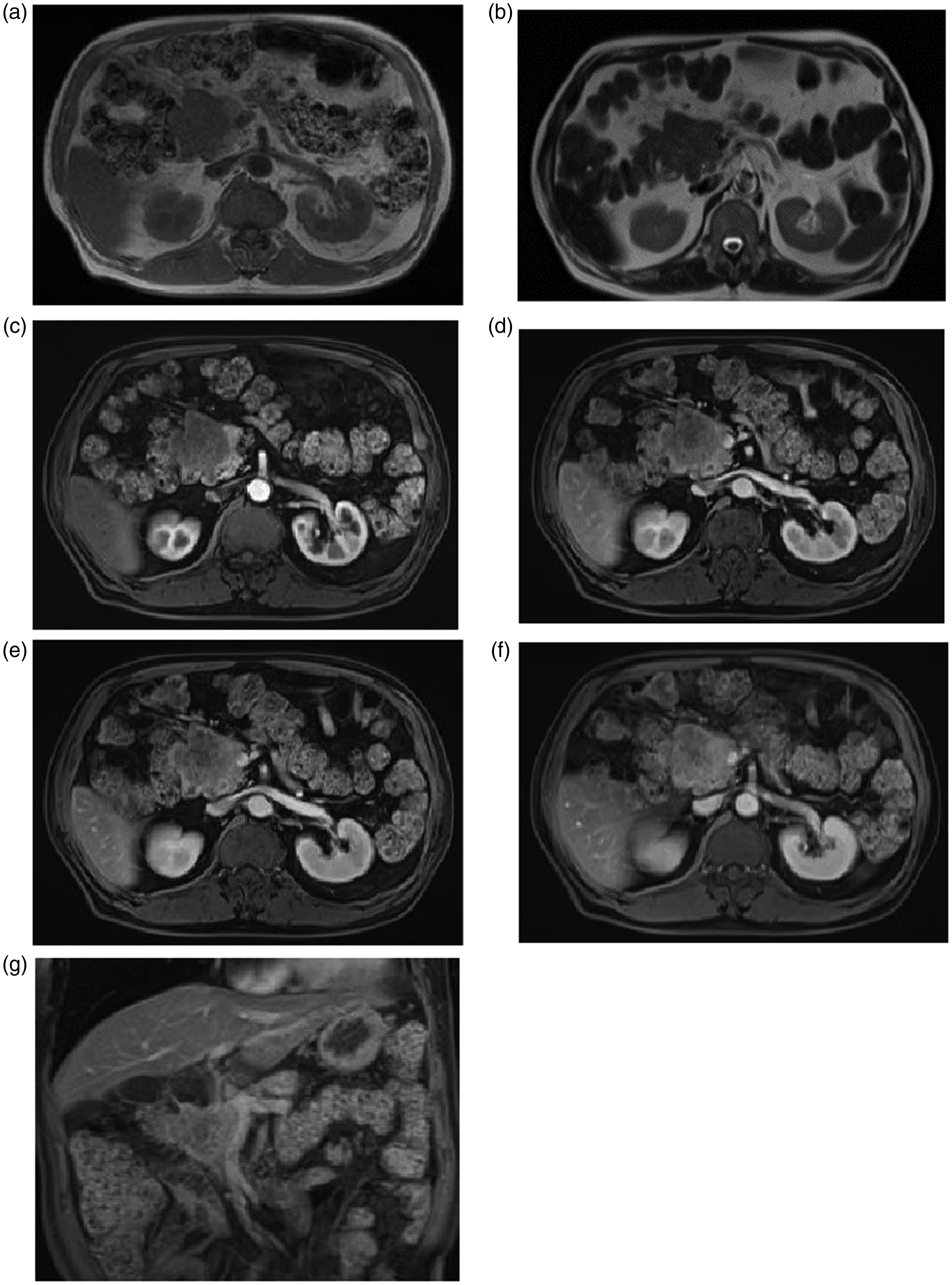
Magnetic resonance images showing a tumor in the pancreatic head. The tumor showed hypointensity on T1-weighted images (T1WI) and slight hyperintensity on T2-weighted images (T2WI), with slight enhancement in the arterial phase, heterogeneous moderate enhancement in the portal venous phase, and persistent heterogeneous enhancement in the delayed phase with contrast-enhanced imaging: (a) was axial T1WI image, (b) was axial T2WI image, (c)–(f) were axial contrast-enhanced images and (g) was coronal enhanced T1WI image.
Considering the patient’s good overall condition, surgical resection was proposed, and pancreaticoduodenectomy (Whipple procedure) was performed. During laparotomy, the tumor measuring 4 × 4 cm was seen in the pancreatic head and had invaded the common bile duct, duodenal wall, and mesentery of the transverse colon. Final histological examination of the completely resected specimen revealed squamous cell carcinoma with necrosis arising from the pancreas, and immunostaining showed positivity for P40, P63, CK19, and CK5/6 (Figure 5).

Histological images of the pancreatic tumor. The images show squamous cell carcinoma in the pancreas (hematoxylin and eosin staining (HE); ×10 magnification). The squamous component was positive with immunostaining for CK5/6, P40, and P63 (×200 magnification): (a) was HE staining of the specimen, (b) was immunohistochemical staining with CK5/6, (c) was immunohistochemical staining with P40 and (d) was immunohistochemical staining with P63.
At a multidisciplinary team discussion, a diagnosis consistent with metastatic squamous cell carcinoma of the lung was agreed, and the patient was treated with chemotherapy and immunotherapy. To date, no other abnormalities have been found in the patient during 6 months of follow-up.
Discussion
Pancreatic metastasis is rare in living patients, accounting for approximately 1% of patients with lung cancer with distal metastasis, 5 and is usually observed at post-mortem examination in appropriately 10% of patients with lung cancer. 6 Intra-abdominal metastasis from lung cancer is usually through the vascular route to the liver and adrenal gland, 7 whereas the pancreas is a rather uncommon site of metastatic lung cancer, with squamous cell carcinoma being the least frequent cytological type of lung cancer that metastasizes to the pancreas. 8
To our knowledge, only eight cases of metastatic lung squamous cell carcinoma in the pancreas have been reported in the English literature worldwide to date.4,9–15 We report the ninth and tenth cases of pancreatic metastasis secondary to squamous cell lung carcinoma (Table 1). In the 10 cases, the patients’ age ranged from 44 to 78 years, and there was an obvious predilection for men, with a female to male ratio of 1:4. The clinical presentations of metastatic tumors in the pancreas can be asymptomatic or nonspecific, namely epigastric pain (8/10), jaundice (3/10), vomiting (3/10), nausea (2/10), backache (1/10), loss of appetite (1/10), and weight loss (1/10).
Characteristics of the patients with pancreatic metastasis of squamous cell lung cancer.

M, male; F, female; ERCP, endoscopic retrograde cholangiopancreatography; N/A, not available.
Typically, metastatic carcinoma to the pancreas presents as part of widespread disease. Presentation as a single pancreatic mass in the absence of any visceral involvement is extremely rare.1,16 In the reported 10 cases, only four patients had metastasis in other locations, namely the brain, lung, liver, and lymph nodes.4,10,13,14 The time interval between the diagnosis of the primary tumor and metastasis to the pancreas varies widely. 17 In the reported 10 cases, pancreatic metastasis was synchronous, originating from squamous cell lung cancer, in 2 cases. The remaining 8 cases (80%) experienced metachronous pancreatic metastasis, and the time interval ranged from 6 months to 3 years.
CT is the most frequently chosen method for detecting pancreatic cancer because this imaging method can delineate the tumor as well as demonstrate the extent of vascular invasion and lymphadenopathy. In the 10 reported cases of secondary pancreatic tumor from squamous cell lung cancer, CT was used to demonstrate the tumor in 8 cases, MRI was used in 1 case, and endoscopic ultrasonography (EUS) was used in 1 case. All the tumors were single, with a maximum diameter of 2.5 cm to 4.6 cm. Nine of the 10 tumors were localized; 5 tumors were located at the pancreatic head, 2 at the pancreatic tail, 1 at the body and tail, 1 at the head and body, and 1 at the body. Abdominal lymphadenopathy was detected in two cases.
Only one case report described the MRI features of the tumor as hypointense on T1WI and hyperintense on T2WI 9 (Table 2). The tumor showed hypovascularity on enhanced images in the arterial, venous, and delayed phases, which made it difficult to differentiate from primary pancreatic ductal adenocarcinoma. 9 In our report, both cases had tumors at the pancreatic head. For case 1, both CT and MRI were performed. The tumor was isodense on precontrast CT images, slightly enhanced in the arterial phase, and heterogeneously moderately enhanced in the portal venous phase with contrast enhancement. In the MRI images, the tumor showed hypointensity on T1WI and slight hyperintensity on T2WI with slight enhancement in the arterial phase, heterogeneous moderate enhancement in the portal venous phase, and persistent heterogeneous enhancement in the delayed phase. No rim enhancement was demonstrated. Dilation of the intrahepatic and extrahepatic biliary ducts, pancreatitis, and abdominal lymphadenopathy were also detected. In case 2, only MRI was performed, and the tumor had similar imaging features to the first patient. The enhancement pattern of both patients demonstrated that the tumor was not hypovascular as with pancreatic adenocarcinoma and which differed from previous reports. Instead of peripheral rim enhancement, the tumors showed gradual enhancement, which reflected a certain degree of vascular perfusion similar to neuroendocrine tumors, which are relatively hypervascular with areas of necrosis or cystic degeneration and heterogeneous enhancement. Bile duct dilation and vascular invasion can also be observed in tumors with higher pathological grade.
Imaging characteristics of the pancreatic tumors.

N/A, not available; T1WI, T1-weighted imaging; T2WI, T2-weighted imaging; CT, computed tomography; MRI, magnetic resonance imaging; US, ultrasonography; EUS, endoscopic ultrasonography; PET, positron emission tomography.
Conclusion
From our two cases and previously reported cases of pancreatic metastasis from squamous cell lung cancer, these metastatic tumors are usually asymptomatic or the symptoms are nonspecific; tumors are usually metachronous and present as a solitary lesion without metastasis to other organs. These findings may easily result in confusion regarding whether the pancreatic tumor is primary or metastatic. With imaging, these tumors may be hypo- or hypervascular, making it difficult to distinguish them from pancreatic adenocarcinoma or neuroendocrine tumors. As in both of our cases, all tumors in previous reports were initially misdiagnosed as primary pancreatic tumors. Therefore, with a prior history of squamous cell carcinoma of the lung, a metachronous, isolated pancreatic tumor should be considered as pancreatic metastasis, with the differential diagnosis of pancreatic adenocarcinoma or neuroendocrine tumor depending on the tumor’s vascularity.
Footnotes
Ethics statement
This study is based on clinical cases and hence, ethical approval was not required. Written and verbal consent was obtained from the patients or the patients’ caregiver(s) for publication. This study complied with the CARE guidelines. Consent for treatment was obtained from the patients.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
