Abstract
The use of percutaneous ablation therapy to manage focal malignant liver lesions has been widely embraced and integrated into clinical practice. These minimally invasive procedures offer a successful alternative to surgery for patients limited by poor hepatic reserve or problematic anatomy. Radiofrequency is currently the most commonly performed ablation therapy technique; however, new research in the field of microwave technology suggests certain advantages over radio frequency. This article analyzes the differences between these two technologies and discusses the circumstances in which one method may be preferred over the other for hepatic malignancies.
Keywords
Primary and secondary hepatic malignancies are among the most common tumors seen throughout the world. 1 Most patients with either primary hepatocellular carcinoma (HCC) or hepatic metastasis are not candidates for surgery; this makes ablative therapy essential to their treatment plan. Hyperthermic ablations are cost-effective and repeatable, can be performed on an outpatient basis, are compatible with real-time imaging, and offer reduced morbidity and mortality. 2 The positive progression this field has seen leads many authors to believe that with continued development and research, ablative technology may surpass the preference for surgical resection in patients who are surgical candidates.
Tumor ablation refers to any direct administration of treatment to a lesion for destruction; it is done thermally and nonthermally. 3 The procedure is minimally invasive and comparable to a biopsy as the tissue in question is treated with the insertion of a needle into a lesion. Sonography is an ideal imaging modality to assist with an ablation because of its portability and real-time qualities. 4 The primary objective of ablation in the liver is to destroy a target volume of parenchyma associated with a focal malignancy with margins measuring at least 0.5 to 1.0 cm. 3 The specificity and accuracy of an ablation minimize iatrogenic damage to a cirrhotic liver; the amount of residual functioning liver is a strong predictive factor for long-term patient survival rates. The balance between complete eradication of tumor and sparing of healthy liver parenchyma requires discretion, but this is a key benefit ablation offers over surgery.
Chemical ablation is a nonthermal method typically done with acetic acid or ethanol; the cell’s cytoplasm is dehydrated, the proteins are denatured, and coagulation necrosis ensues. Thermal ablation delivers cytotoxic increases or decreases in temperature to the tumor through a radiofrequency (RF), microwave, laser, or cryotherapy technique; because of the lack of clinical prevalence, laser and cryotherapy will be discussed only briefly here. Cryotherapy is the oldest ablation technique; in practice since 1963, the tissue is cooled to lethal temperature levels with the use of cryoprobes and argon or nitrogen gas. 1 Cryoablation is not commonly performed on the liver; its typical use is for primary renal tumors and bone metastasis. 3 Although precise, laser ablation is not commonly used for interstitial lesions given the lack of penetration and small zone of ablation it offers due to scattering and absorption of light.
Review
Focal tissue heating in hyperthermic ablation is considered 50°C and above. Irreversible cellular necrosis occurs when a temperature of 46°C is maintained for 60 minutes. Cystolic and mitochondrial enzymes and nucleic acid–histone protein complexes are immediately compromised in the cell, and apoptosis is carried out over several days; this is referred to as “coagulation necrosis.” 3 Ideally, temperatures above 50°C are warranted, but some applications are limited to 100°C to avoid tissue vaporization (occurring at 110°C). These target temperatures are only a theoretical guideline as cellular necrosis is tissue specific with many variables. 5 Maintaining an appropriate temperature is crucial to the procedure; temperatures that are too high result in charring, and a limited radius of ablation is achieved, causing inadequately heated margins of the lesion. 2
Contrast-enhanced computed tomography (CT) is routinely used immediately following hyperthermic ablation to assess the therapeutic zone as compared with the amount of the viable residual tumor (Figure 1). 5 To determine the true borders of ablation to judge the resolution of a tumor, long-term diagnostic imaging follow-up is necessary. Sonography typically encounters poor visualization of ablated tissue; CT is the choice modality for follow-up. Regardless of which heat-based technique is used, combination therapy is strongly recommended for patients following ablation; chemotherapy or chemoembolization gives further assurance that there is complete eradication of malignant cells. Combination therapy is a standard suggestion for the resolution of any solid tumor regardless of whether it is confronted with surgery or ablation.
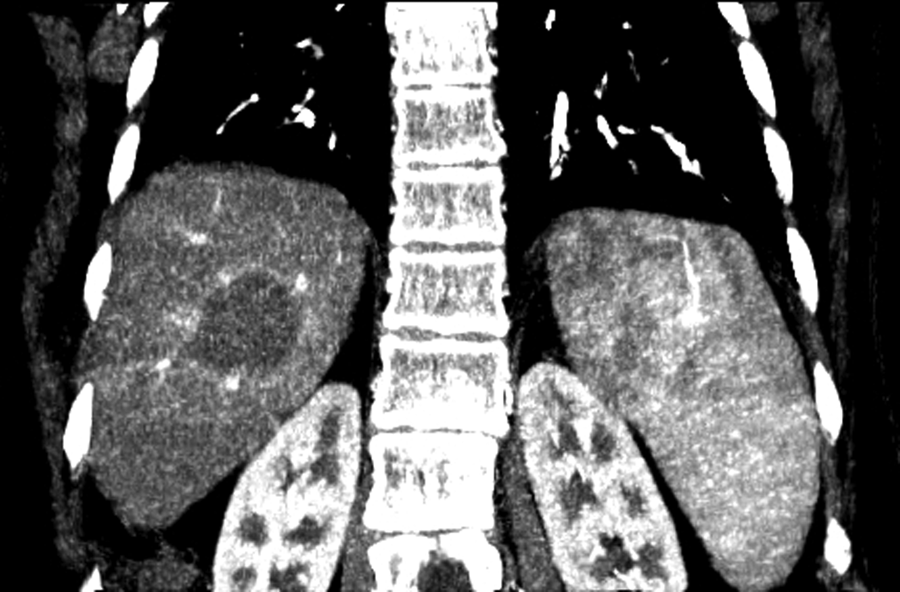
Abdominal computed tomography scan performed one day following ablation for hepatocellular carcinoma. Zone of ablation appears hypoechoic.
Radiofrequency
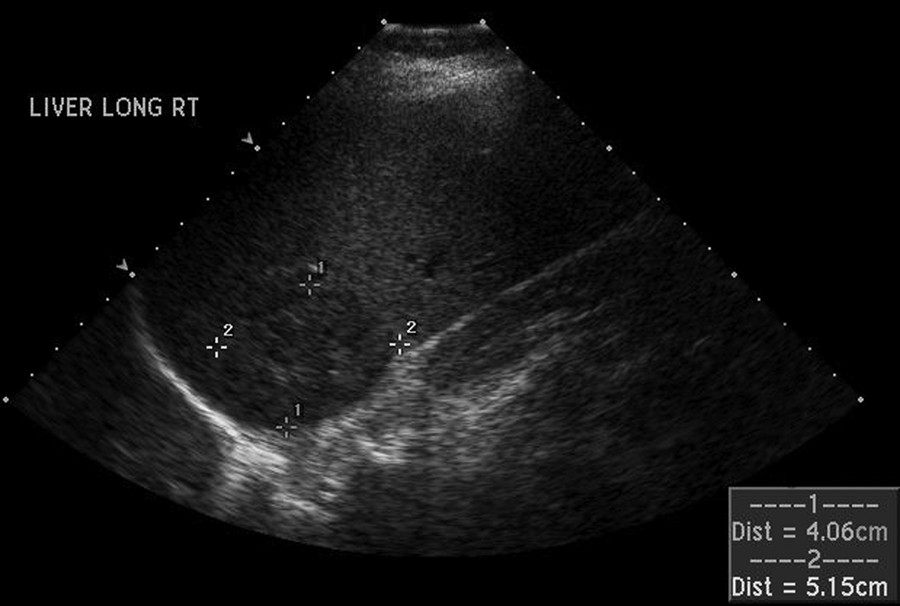
Since Food and Drug Administration (FDA) approval in 1997, the radiofrequency ablation (RFA) procedure has had tremendous success; it is the most widely used percutaneous ablative technique for nonsurgical cancer patients.3,4 With the use of this technology, lesions ranging from 2 to 5 cm in size can be fully ablated (Figure 2). 1 The treatment itself occupies roughly 8 to 20 minutes per ablation and can be done on an outpatient basis with conscious sedation; multiple ablations may be required. As many as four tumors can be treated per examination; any further metastatic recurrence has the potential for treatment as well. 4 Ideally, tumors that undergo ablation are fully encased within the liver, at least 1 cm deep to the capsule and at least 2 cm away from any vascularity. 1 A circumference of 5 to 10 mm of necrosis beyond the tumor margin is desired to ensure success. Lesions residing too near large hepatic vessels are at risk for incomplete ablation because the blood flow in the vessel cools the surrounding area. There is also increased risk noted for tumors located near the portal triad or biliary system; this may require the use of a different technique such as cryoablation. Another option that physicians use during an RF procedure is vascular occlusion of the nearby vessels; this reduces heat dispersion and accomplishes a larger zone of ablation. 6

Gray-scale ultrasonographic image of a focal hepatocellular carcinoma lesion located within the right lobe of the liver prior to radiofrequency ablation.
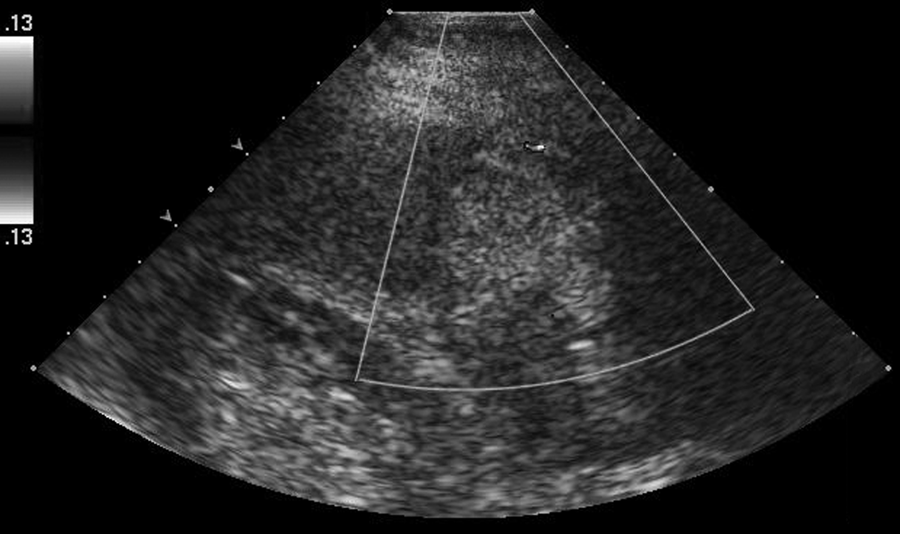
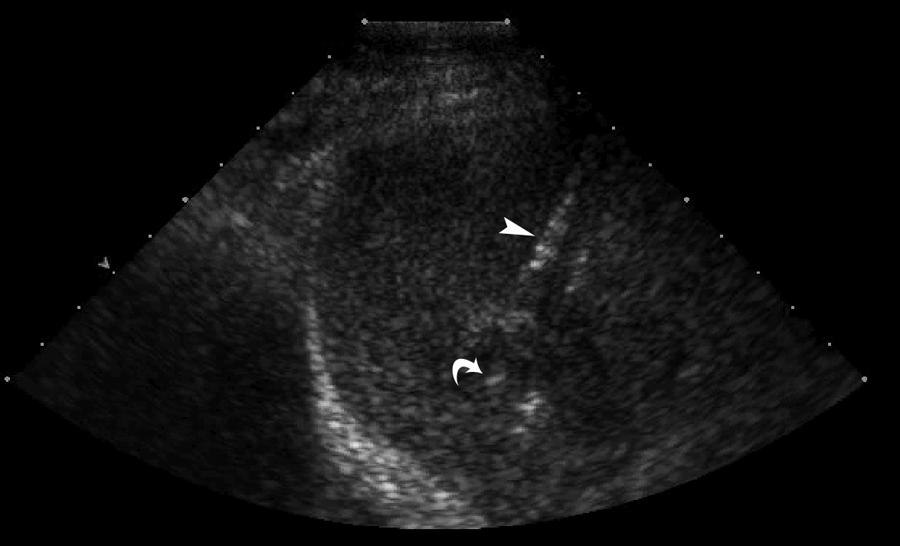
The RFA zone has a wide spectrum of appearances; this is largely attributed to the amount of time elapsed since treatment, the local environment, or the rare occurrence of a complication. 7 Ensuring correct insertion of the electrode needle tip into the lesion at the beginning of an ablation is when sonographic assistance plays its most significant role; a prominent needle tip and burn line are routinely observed (Figure 3,4). Sonographically, the zone of ablation is characterized intraoperatively by visualization of echogenic micro bubbles (Figure 5). 1 As the procedure progresses, sonographic visualization becomes limited by an “echogenic cloud” or “transient hyperechoic zone,” acoustic shadowing from microbubbles of water vapor, and other cellular components that are the result of active heating (Figure 6).7,8 The transient hyperechoic zone can be expected to persist for 30 to 90 minutes. 8 The ablation will typically be round or oval, but it can also be slender or irregular; large hepatic vessels may indent the area and cause deformities due to the heat sink affect. 7 CT is completed within one week to evaluate the margins of the lesion and determine if immediate retreatment is needed. 9 Over time, the volume of the RFA zone decreases and leaves a small scar-like lesion. 7 On rare occasions, a hepatic abscess, biloma, calcification, hepatic arterial pseudoaneurysm, or progression of the tumor can be visualized diagnostically within the ablated region or near the rim. Enlargement of the RFA zone is not common; if enlargement is seen, it is likely a complication has ensued. Color Doppler and pulsed-wave Doppler sonography are valuable tools immediately following ablation to detect hemorrhagic complications. 10 The color “line sign” may be visualized sonographically once the electrode is removed if the needle track remains patent; surveillance of the color line sign helps the team quickly address and prevent serious hemorrhagic complications. The use of electrode track coagulation during the ablation is recommended as it decreases the incidence of the color line sign.
Radiofrequency-cooled electrode needle.
Insertion of a radiofrequency electrode needle into a hepatocellular carcinoma lesion. Curved arrow demonstrates needle tip, arrowhead demonstrates needle shaft.
Appearance of echogenic microbubbles seen in the early stage of a radiofrequency ablation procedure.
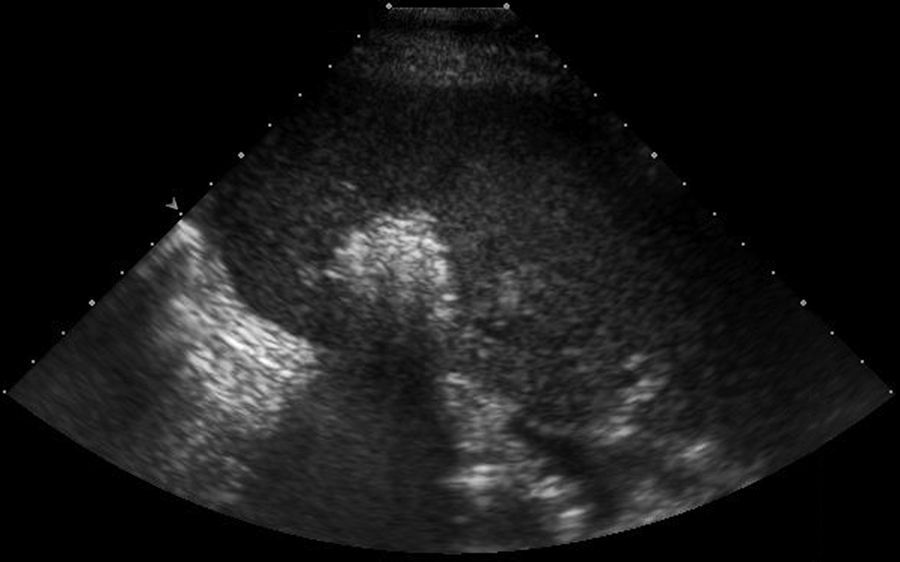
Appearance of the transient hyperechoic zone or echogenic cloud that is seen sonographically as an radiofrequency ablation procedure progresses.
The label radio that is used with regard to an ablative procedure does not describe the wave of energy emitted during the treatment but refers to the high-frequency electromagnetic current generated in the range of 200 to 1200 kHz. 9 The physical principles that drive this procedure are essentially derived by instigating a simple electrical circuit. 3 A generator sends an electrical current through electrodes with noninsulated ends that are placed into a tumor; the energy oscillates between the electrodes through ionic channels present in biologic tissue. The tissue’s ions follow the flow of the current and begin to agitate, and heat is then generated through friction; this process is referred to as the Joule effect.3,4 Higher current density results in more rapid tissue heating; the areas closest to the electrode experience the most effective heating, whereas the peripheral region is most affected by thermal conduction. 3 As intracellular dehydration and protein denaturation begins to occur, the tissue loses its ability to conduct electricity; this electrical impedance is measured in ohms and can be detected by the RF generator and used as a feedback signal.
Many different electrodes have been developed for RF ablations, and each generator is equipped with a specific algorithm depending on the electrodes available to it. 3 Electrode needles are sized at 14, 15, or 17 gauge with anywhere between 4 and 10 retractable curved prongs depending on the design. 1 Most systems function on the monopolar method, which uses an interstitial electrode and a dispersive electrode, or ground pad. The interstitial electrode is inserted into the tumor under sonographic guidance; the ground pad is placed over the patient’s legs to disperse energy over a large surface area as a means of closing the electrical circuit.3,4 It is important to monitor ground pads because skin burns can result from unequal placement or by using an inadequate amount. 3 Monopolar electrode systems may also incorporate a fluid, such as saline, which is circulated inside the active electrode to achieve internal cooling to increase the distribution of energy throughout the tumor (Figure 6). This allows a larger volume of ablation to be obtained because tissue impedance is reduced.
Bipolar electrodes are another type of needle used in RFAs. This mechanism necessitates accurate guidance as two interstitial electrodes are placed within the target area and an electric current oscillates between them. This gives the effect of faster, more focal heating as the current is directed only between the two electrodes; a diameter of destruction measuring as large as 3.5 cm has been produced with this method. 9 The benefit of this system is that there is no need for placement of ground pads on the patient. Although a slightly smaller zone of ablation is obtained with bipolar electrodes, this may be ideal for patients with a smaller focal malignancy because a greater amount of healthy parenchyma can be spared. A bipolar system can also function as multipolar; this entails placing pairs of electrodes and alternating between them.
A variety of coagulation algorithms exist for an RFA procedure. 11 The purpose of an algorithm is to regulate power output and control; this directly affects the volume of ablation that can be accomplished. Strategies of power delivery have been based on rapid tissue heating with the maximum amount of power applied, pulsing of electricity, or gradual increases in wattage. Recent research with an internally cooled electrode suggests that a pulsed algorithm with gradual “ramp up” of power levels increases the volume of coagulation and reduces procedural time. The gradual application of wattage avoids charring and allows the tissue to be heated more evenly and thoroughly than full power methods. Restarting the algorithm after the initial electrical impedance is reached will also result in larger volumes of liver coagulation.
Microwave
In a clinical setting, microwave electromagnetic energy operates at either 915 MHz or 2.45 GHz. 3 Microwave coagulation therapy is carried out much like RFA, one to three 14.5-gauge microwave antennas are inserted into the focal lesion; a coaxial cable extending from the antenna is connected to a generator, and an electromagnetic microwave is emitted through a noninsulated opening. 2 Grounding pads are not necessary for this type of ablation therapy. This procedure can be performed on an outpatient basis, percutaneously, laparoscopically, or during open surgery; it is well tolerated with a minimal rate of major complications.2,12 The time of ablation is approximately 10 minutes but varies with tumor size. 2 The patient selection is essentially the same as for RFA. 1 Ablation zone diameter for microwave radiation can be up to 5.5 cm. Sonographically, the zone of ablation will appear hyperechoic around the tip of the needle. Multiple ablation sessions may be necessary depending on the size of the malignancy.
The physics principle on which this technology is based differs from the Joule effect seen with RF. Microwave energy is made through dielectric hysteresis. 3 Certain molecules, such as water, have an intrinsic need to constantly realign with an electromagnetic current when it flows through the medium where the molecule resides. One end of a water molecule has a negative charge and the other positive; electromagnetic radiation is a wave that oscillates between positive and negative charges so this flow of energy disrupts the water molecule and the motion of the two becomes synchronized. 2 Kinetic energy is increased during the rotation of the dipole molecule, and heat is expended, creating an overall temperature elevation in the surrounding tissue and coagulation necrosis. 3 The frequency of the electromagnetic wave can be adjusted to determine the temperature increase and the zone of ablation. 2 An appreciated benefit of dipole rotation is that there is a minimal heat sink effect; the typical ablation zone distortion near large blood vessels is not seen. This clearly decreases some of the limitations of which lesions are able to undergo treatment.
Microwave energy passes through biologic substances readily; it is not limited by the same electrical impedances as seen with RF. 2 Microwaves pass through deficient conductors of electricity such as lung, bone, or charred (vaporized) tissue. The avoidance of electrical insulation takes away any restrictions put on the amount of power that can be applied to a tumor at one time; the power density of microwave is up to 2 cm peripheral to the antenna. This permission of energy freely allocates extreme intratumoral temperatures (greater than 150°C) to be maintained as needed. These high temperatures produce a larger ablation zone faster, making microwave heating more efficient than RFA. This improves overall efficacy of the procedure because thermal conduction to the surrounding tissue is increased. 3 Microwave also allows multiple antennae to be used at one time, adding to its efficiency; common antenna designs include monopole, dipole, triaxial, slotted, and choked.
Microwaves have a reputation of being more difficult to dispense. It is a drawback that coaxial cable is the medium for microwaves when referenced to the small wires that RF electrodes use. It is also a concern that coaxial cables are known to heat up when carrying large quantities of power; without a cooling jacket, this can injure the skin.
Analysis
Radiofrequency ablation has many advantages and is widely integrated among medical professionals, but there are limitations to address. Concerns mainly center on the recurrence rates associated with larger tumors treated and ability of current flow, be it heat sink or electrical impedance. 2 Microwave technology is still in its infancy and necessitates further development. In this way, it cannot compare to RF, but theoretically the physics of microwave make it well suited for ablation therapy. There are also potential advantages. RF only distributes millimeters of thermal conduction, constituting a smaller size of ablation than the volume microwave coagulation compensates. 3 With the larger zone of active heating during microwave ablation, greater homogeneity is established both within the target tissue and near blood vessels. 2 Microwave technology boasts efficiency, but RFAs are inexpensive and easily generated. 3
Early clinical trials conducted in the Department of Radiology at Kyoto University Graduate School of Medicine for ablation therapy between March 1999 and October 2000 compared microwave ablation with RF therapy, treating a total of 94 nodules in 72 patients; 36 received RF therapy, and 36 received microwave ablation. 13 The study concluded that the two methods gave relatively equivalent results, with the exception that microwave coagulation often required more treatment sessions than RF. This may explain why RFA has experienced more clinical use than microwave.
A more recent clinical study carried out between August 2003 and December 2004 focused solely on the adaptation of a cooled shaft antenna to increase the microwave ablation zone. This was a sequential study that used information from ex vivo and in vivo porcine livers followed up by clinical research on 90 patients with liver cancer, all of whom were nonsurgical candidates. 14 The findings suggested that incorporating a cooled-shaft microwave antenna substantially improves microwave delivery, therefore increasing the respective volume of coagulation. Microwave accomplishes better penetration in a cirrhotic liver than does RFA. Zones of ablation in the range of 4.8 to 8.0 cm were reported with multiple sessions, and up to 3 centimeters of tumor destruction were achieved with one session; however, it was recommended that more testing should be carried out on a larger patient population with hepatic masses measuring greater than 3 cm.
Another large study comparing cooled- and non-cooled-shaft microwave antennae done in 2007 reported that the cooled-shaft antenna reduced the risk of patient complications. 12 This 13-year study involved 1136 patients with a variety of liver tumors; 21 patients exposed to the noncooled shaft antenna experienced major complications, whereas only 9 of the cooled-shaft antenna patients showed serious complications. The study also concluded that fewer treatment sessions minimize the number of major complications.
Studies comparing survival rates of ablative technologies to surgical resection have also been recorded. Between January 1995 and December 2009, 478 cirrhotic patients with HCC were observed in one such study. 15 This study concluded that survival rates for resection are similar to those for ablations performed on HCC lesions measuring less than 5 cm and for treatment of up to three nodules measuring less than 3 cm; however, long-term survival is greatly improved by surgical resection in cases of solitary lesions larger than 5 cm or livers with multiple lesions above 3 cm.
Contrast-enhanced sonography is one way ablation therapy may expand and improve clinically in the future to guarantee desirable results. 16 In one clinical study performed by the Peking University Institutional Review Board, contrast-enhanced sonography was used to confirm patients with HCC as candidates ready to undergo RF ablation. The use of contrast-enhanced sonography determined fewer patients valid for ablative treatment than those within the control group who obtained the standard screening; the long-term results of those approved with contrast-enhanced sonography were better than those of patients in the control group. Contrast-enhanced sonography can not only determine the necessity of ablation therapy but also aid preemptively in guiding and planning for RFA. Contrast-enhanced sonography gives a specific outline of the border and location of a tumor, directly answering whether the tumor is too large or too closely related to a major vessel. This clarity can point the examiner in the right direction when deciding which type of ablative treatment is necessary. Contrast-enhanced sonography can highlight small HCC lesions not appreciated on computed tomography screening. For these reasons, contrast-enhanced sonography would be a useful addition to a protocol for ablative therapy; however, intravenous contrast agents in the United States are currently only approved for applications related to echocardiography. 17
Considering the variables and the myriad of options available to thermal ablative technology, there is no clear preference between microwave and radiofrequency technique for clinical use. 3 Each hyperthermic energy source is unique, must be matched to the particular application at hand, and is dependent on available tools, operator discretion, skill level, and preference. Patient characteristics as well as tumor size are key elements in determining appropriate ablative treatment options. This field is still encountering vast research and improvement, especially within the realm of equipment and devices. For this reason, it is crucial for those involved to remain updated on all further developments.
Footnotes
The author declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author received no financial support for the research and/or authorship of this article.
