Abstract
Abdominal aortic aneurysm (AAA) is a common disease in today’s older population. When AAAs grow large, the risk for rupture becomes high, and the AAA should be treated. It can be treated in one of two ways: open or endovascular repair. With endovascular repair, an endograft is placed within the aorta using angiographic guidance to exclude the AAA. Intravascular sonography (IVUS) can be used as an adjunct to angiography for the endovascular repair. The endograft must then be routinely followed for complications such as endoleak. This case report identifies a symptomatic type III endoleak and describes how IVUS is used in conjunction with computed tomography and angiography to treat type III endoleaks.
Abdominal aortic aneurysm (AAA) is an enlargement of the abdominal aorta or a stretched and budging wall of the aorta. 1 Risk factors for developing AAA include smoking, high blood pressure, male gender, older age, family history, infection, inflammation, and trauma. 1 There are two types of AAA: fusiform and saccular. Clinical opinion holds that eccentric saccular aneurysms are at a greater risk for rupture than diffuse fusiform aneurysms. 2 If the AAA enlarges beyond 5 to 5.5 cm, the risk for rupture becomes high and the patient is considered for treatment. It may be treated by open surgery or endovascular surgery. 3
Endovascular AAA repair (EVAR) is a minimally invasive procedure where the AAA is excluded by an endograft. A surgeon or interventional radiologist uses angiography and percutaneous placement of catheters to gain access, usually through the common femoral arteries. Guidewires are advanced cephalad through the iliac arteries and aorta, proximal to the AAA. Catheters are introduced over the wires, and an aortogram is performed. Important arterial branches are identified, and sizing measurements are made. The endograft comes preloaded onto a catheter and confined within a sheath. This catheter is passed over the wire and placed in a position that, once deployed, will exclude the AAA from blood flow. Intravascular sonography (IVUS) is commonly used during EVARs for additional visualization and vessel sizing, specifically at the seal zones, where the endograft will “seal” to the native artery. IVUS uses a high-frequency catheter-tipped transducer that projects a 360-degree live image. IVUS is now being used for EVAR procedures to identify and mark essential arterial branches and for vessel sizing to correlate with computed tomography (CT) for endograft diameters and length.
Endografts are routinely followed by sonography for complications, and a common major complication is endoleak. Endoleaks occur when blood feeds into the native AAA sac, either secondary to incomplete graft attachment to the native vessel wall or through a patent branch artery such as the inferior mesenteric artery or lumbar arteries. With an endoleak present, there remains substantial risk for AAA growth and rupture.
Case Report
In 2005, an elderly man presented to an outside facility for acute onset of abdominal pain. He was found to be hypotensive, and a CT scan was performed. The CT showed a large saccular abdominal aneurysm with extension into the left common iliac artery. He was transferred to our facility, where he underwent an EVAR and the AAA was successfully occluded.
In 2011, he presented back to our emergency department for workup of new abdominal pain. Upon admission, an endograft surveillance duplex examination was ordered to rule out endoleak. A sonographic examination (Zonare Z-One sonography system; Zonare, Mountain View, California) was performed using a curvilinear (c6-2) transducer. The residual AAA sac was measured and was seen to have increased in size to 7.56 cm (Figure 1) from 4.57 cm, when the last surveillance duplex had been performed. In addition, blood flow was clearly visible outside of the endograft but within the residual AAA sac. Blood flow was confirmed with color flow (Figure 2) and spectral Doppler (Figure 3).
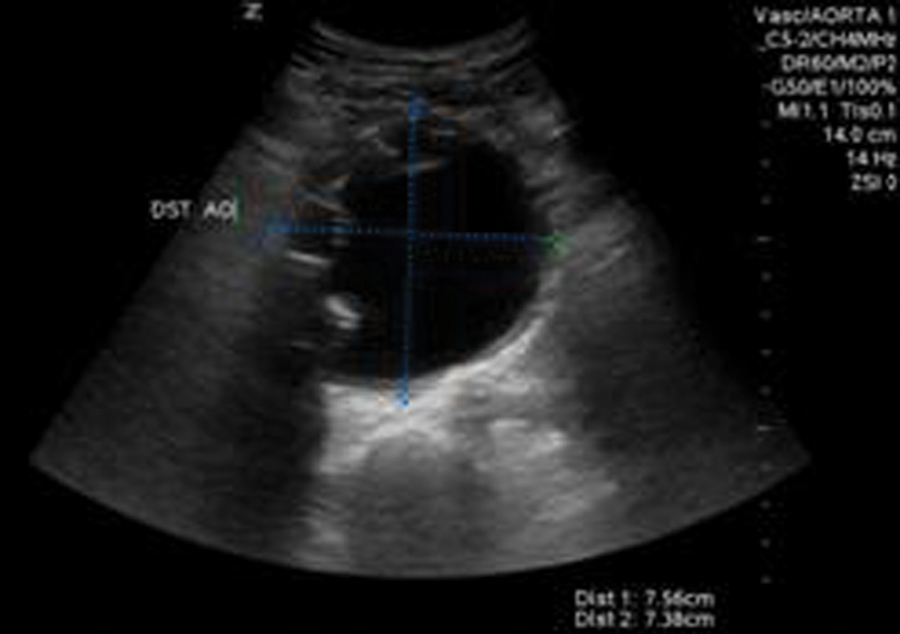
Duplex sonography image of the residual abdominal aortic aneurysm sac with endograft limbs visible on the right wall.
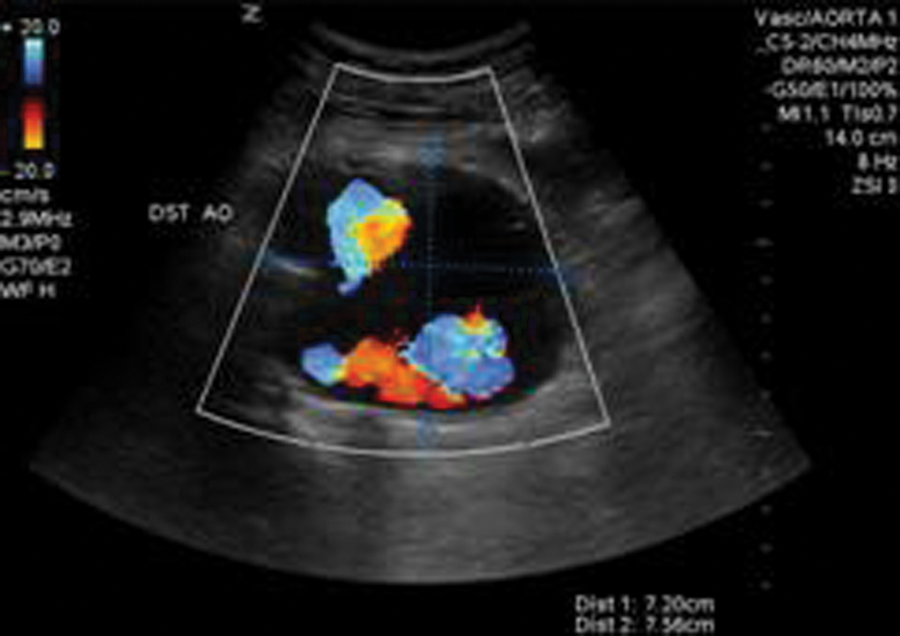
Duplex sonography with color flow demonstrating blood flow outside the endograft but within the residual sac.
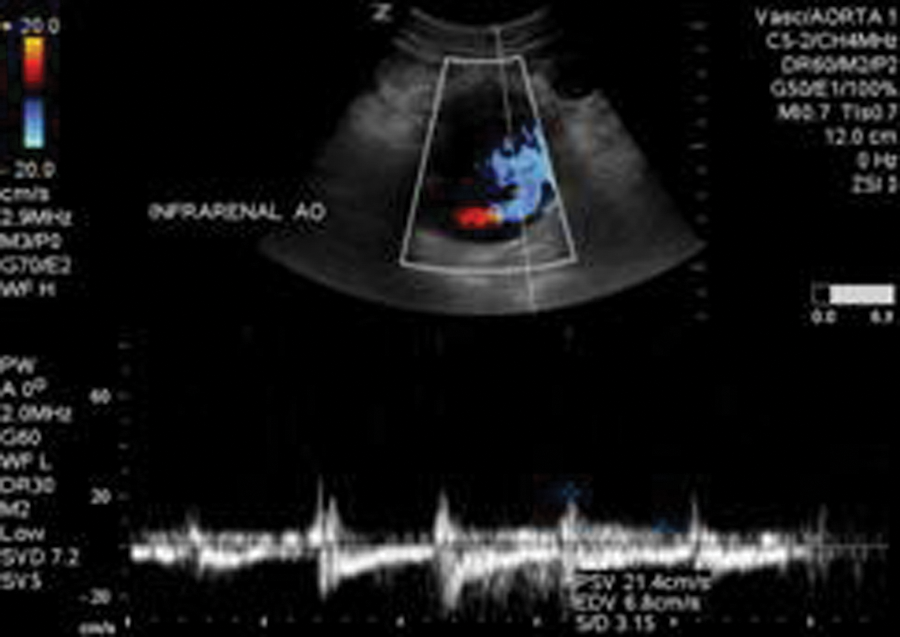
Duplex sonography with spectral Doppler demonstrating flow outside the endograft but within the residual sac.
A computed tomography angiogram (CTA) confirmed the residual sac size and endoleak. From the CTA, the surgeon was able to determine that the endoleak was a type III endoleak, with the left limb of the endograft having separated from the main body of the endograft. The graft separation was best seen on the CTA sagittal and coronal reconstructions (Figures 4–6). The sagittal images showed the proximal graft separation and distal graft separation. The coronal image demonstrated the residual sac expansion.
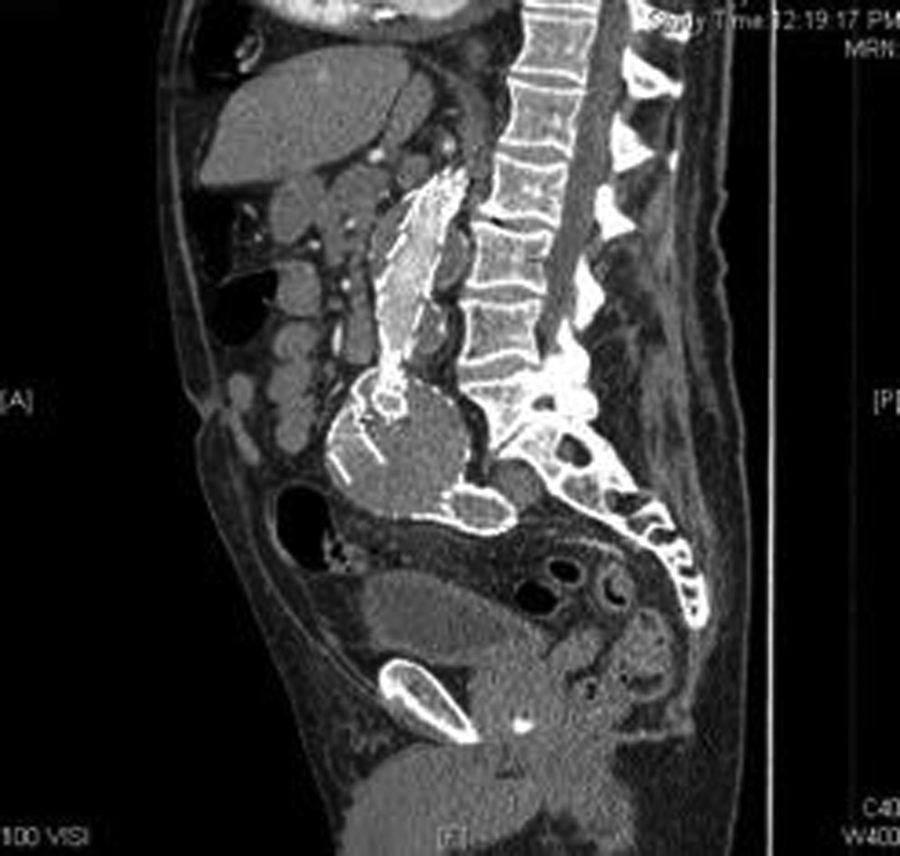
Computed tomography angiogram sagittal reconstruction showing the proximal graft separation within the residual sac.
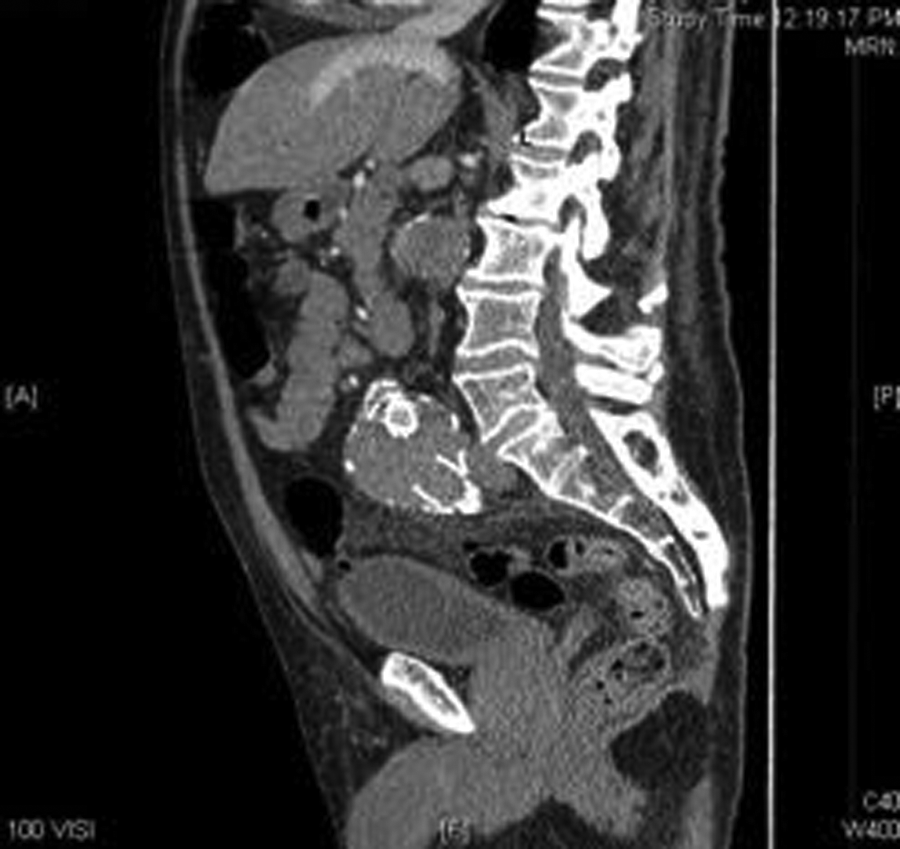
Computed tomography angiogram sagittal reconstruction showing the distal graft separation within the residual sac.
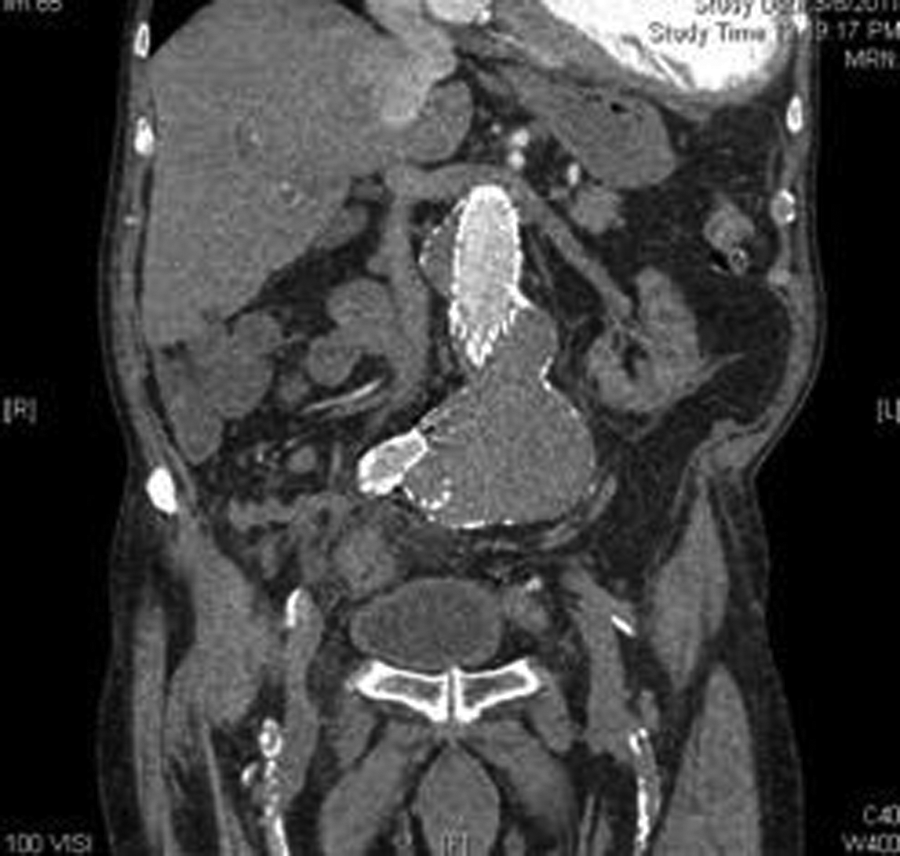
Computed tomography angiogram coronal reconstruction showing the residual abdominal aortic aneurysm sac and portions of the proximal endograft and right limb.
The patient was taken to surgery the following day to repair the type III endoleak with an endovascular approach. The plan was to deploy a graft that would connect the separated pieces and create a new left limb. Access was acquired under sonographic guidance through the left common femoral artery, and a guidewire was advanced cephalad through the left external iliac artery, past the hypogastric artery and into the common iliac artery, where the distal seal zone of the original endograft ended. The wire was then advanced through the distal left limb graft and eventually out of the graft at the separation through the residual AAA sac and cephalad to the proximal aorta. IVUS (Volcano Industries s5 imaging system; Volcano, San Diego, California) was brought in, and a sonographic catheter (Visions PV8.2F; Volcano) was advanced over the guidewire into the proximal aorta. The IVUS image was rotated for proper orientation with the left renal vein at the top of the screen (Figure 7). The surgeon then pulled the catheter back with the images saved as a video clip on the hard drive and later sent to the hospital’s Picture Archiving and Communication System (PACS). The composite loop was displayed as a longitudinal view called the in-line digital (ILD) image. The ILD displayed the abdominal aorta and left iliac artery, including the proximal graft separation and the distal graft separation (Figures 8 and 9).
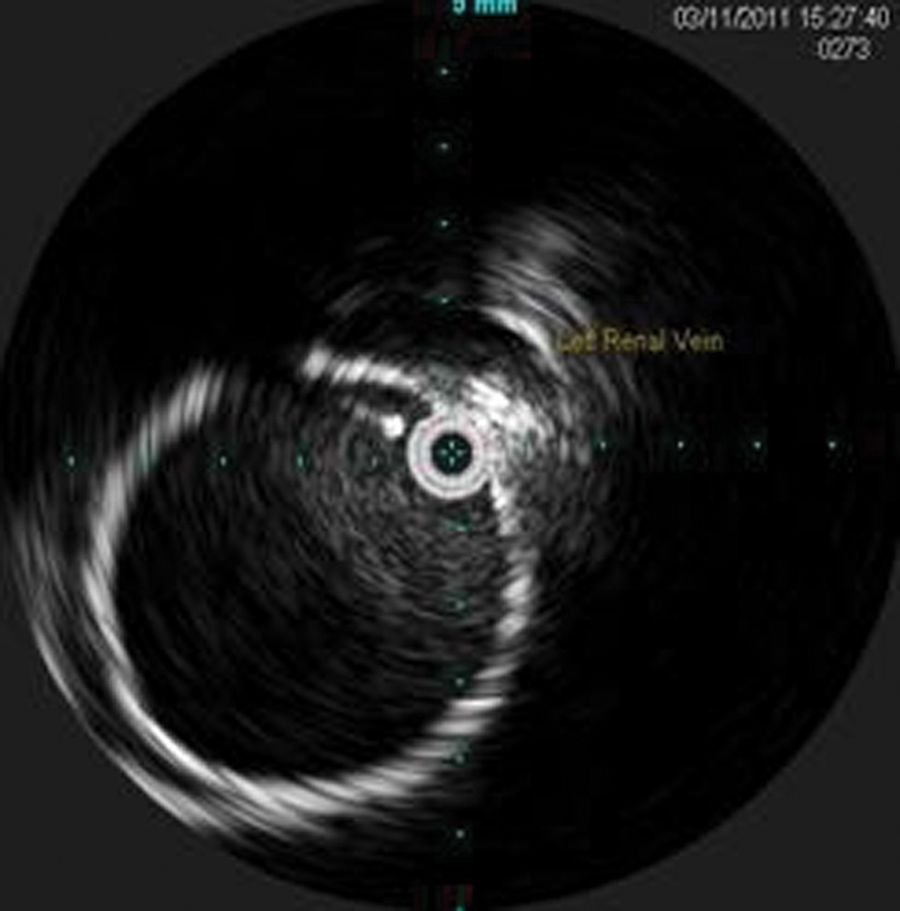
Intravascular sonography image of the proximal endograft with the left renal vein anterior.
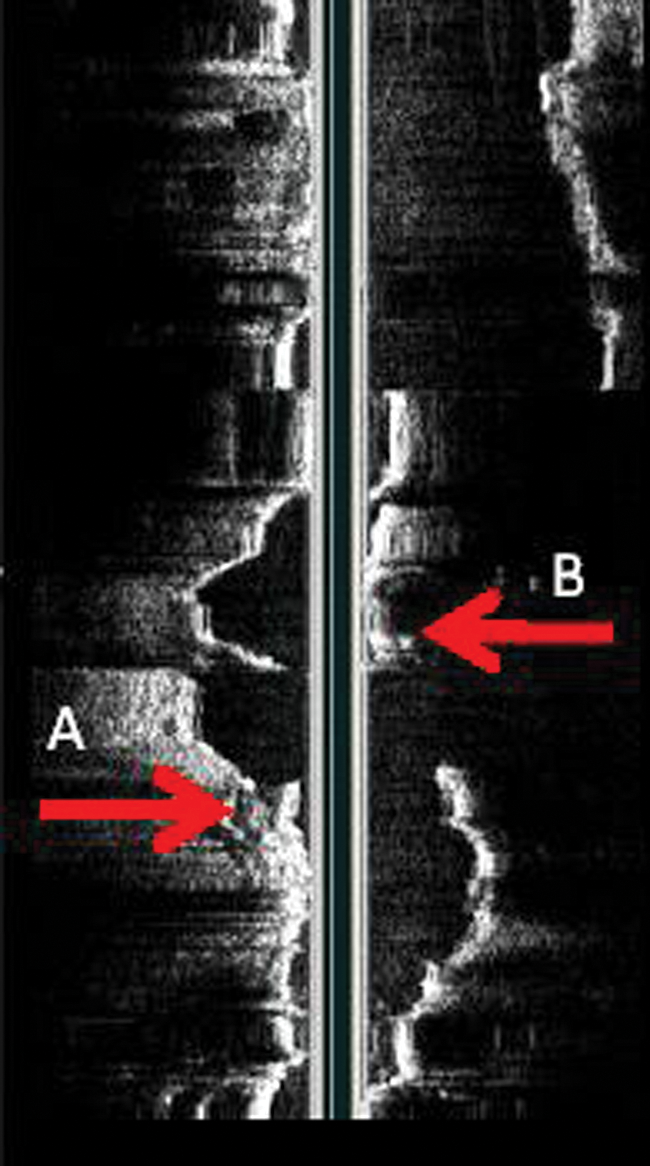
Originally obtained in-line digital image showing the proximal endograft and separated left limb. Arrow labeled B shows the proximal graft separation. Arrow labeled A shows the distal graft separation.
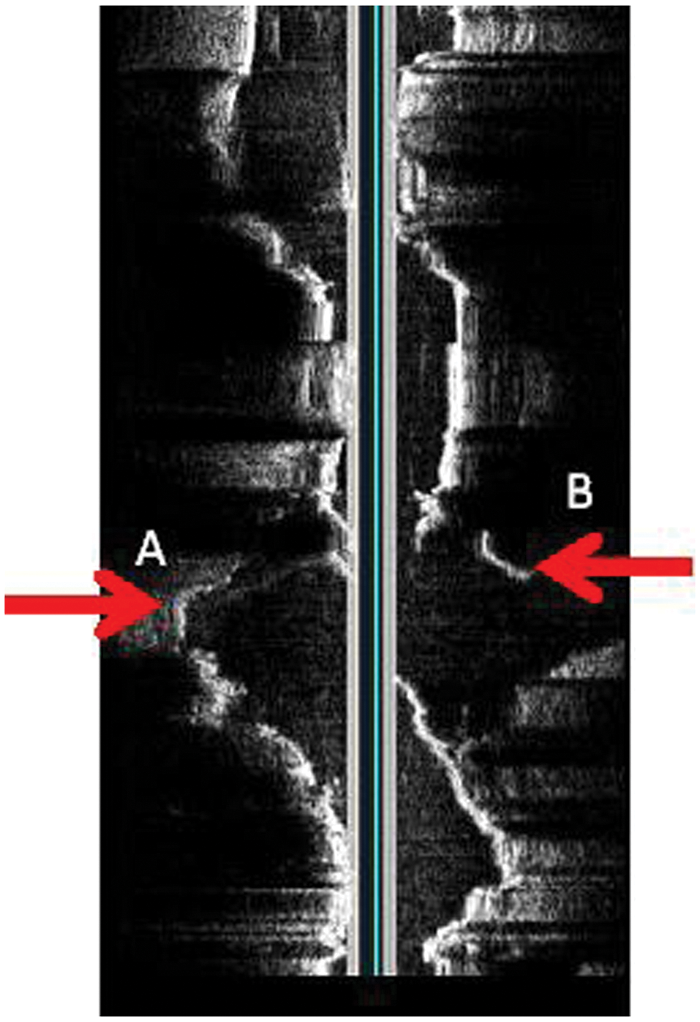
Rotated in-line digital image showing the separation in greater detail. Arrows A and B should line up and form a continuous graft.
During the pullback, the celiac artery, superior mesenteric artery, left renal vein (Figure 7), bilateral renal arteries, proximal seal zone of the endograft (Figure 10), bifurcation of the endograft (Figure 11), proximal left limb, graft separation, AAA sac (Figure 12), distal left limb, distal seal zone of the left endograft and left hypogastric artery (Figure 13), and left external iliac artery were identified.
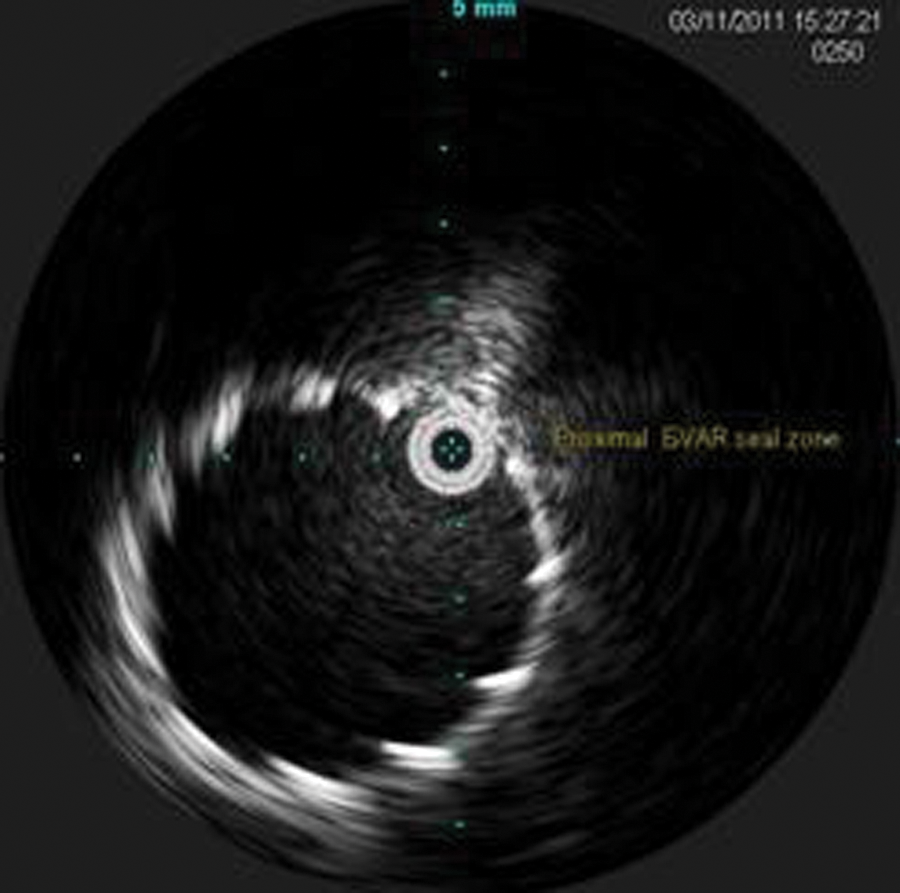
Intravascular sonography image of the proximal endograft.
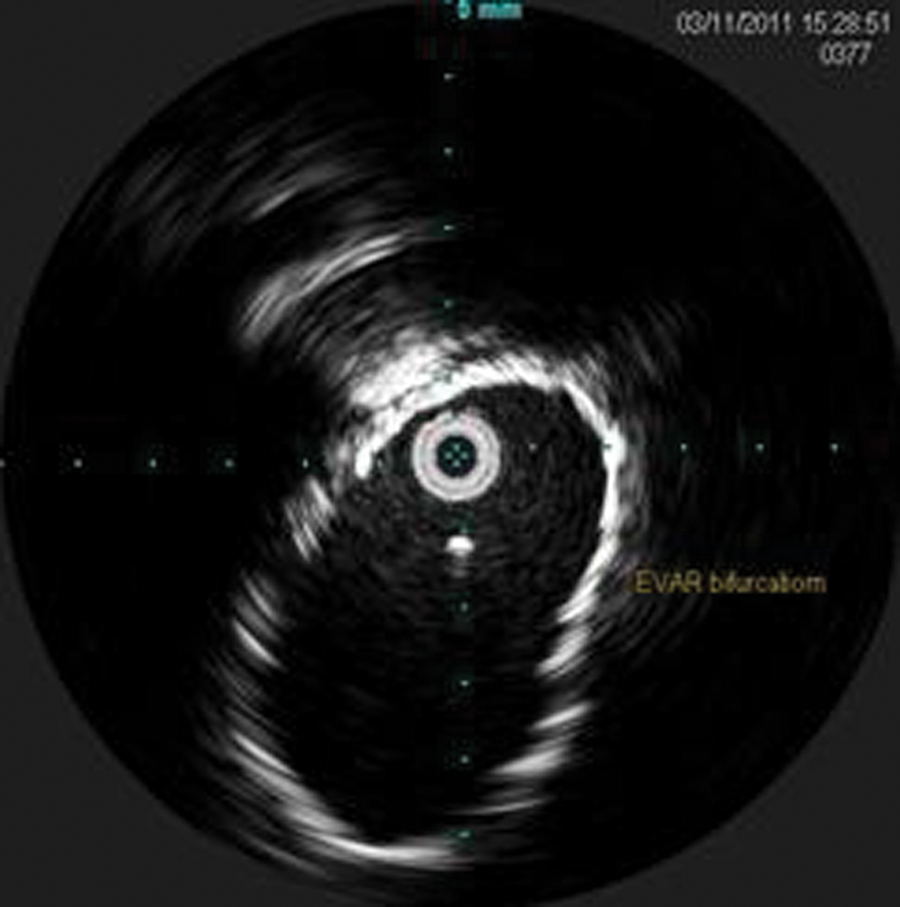
Intravascular sonography image of the endograft bifurcation.
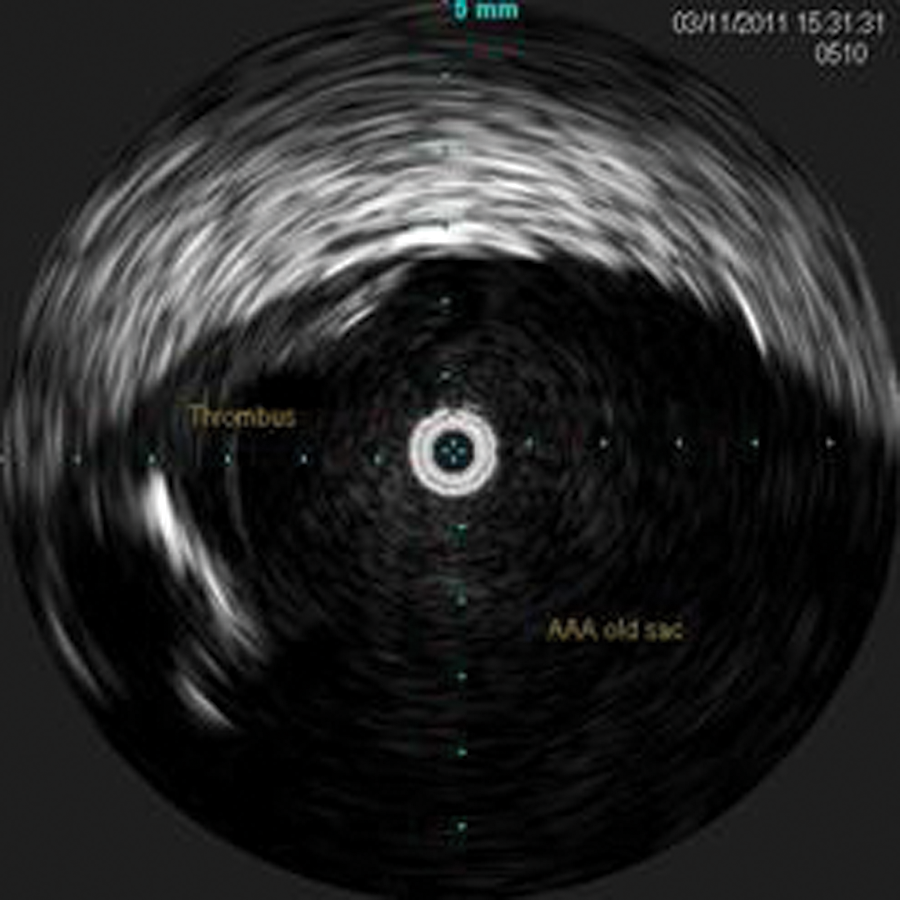
Intravascular sonography image of the residual abdominal aortic aneurysm sac with thrombus visible. This is the space between the two separated grafts where the active type III endoleak was identified.
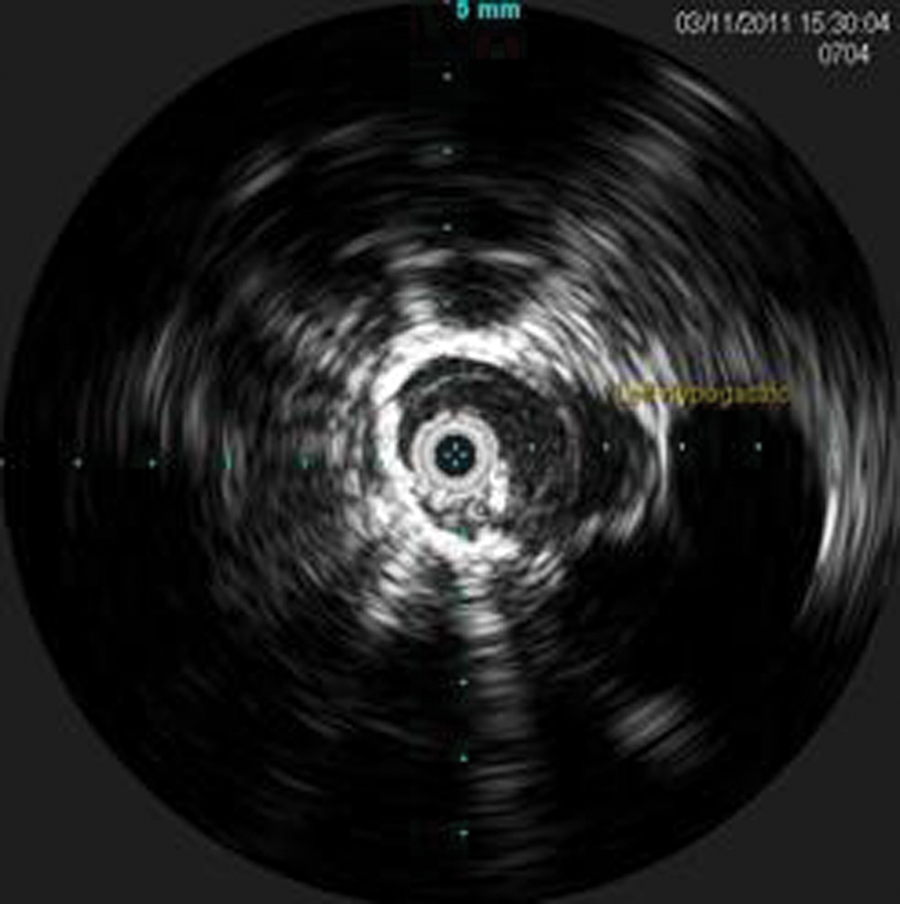
Intravascular sonography image of the distal left limb graft sealed to the left common iliac artery. Left hypogastric artery is seen to the right on the image.
The ILD was saved, and measurements of the separated grafts and the new seal zones were obtained. Measurements were taken for the new seal zones at the proximal left limb (new proximal seal zone—12.0 mm) (Figure 14), distal left limb, original distal seal zone (Figure 15), and distal left common iliac artery (CIA; new distal seal zone–11.3 mm) (Figure 16). These IVUS measurements were correlated with the CT measurements.
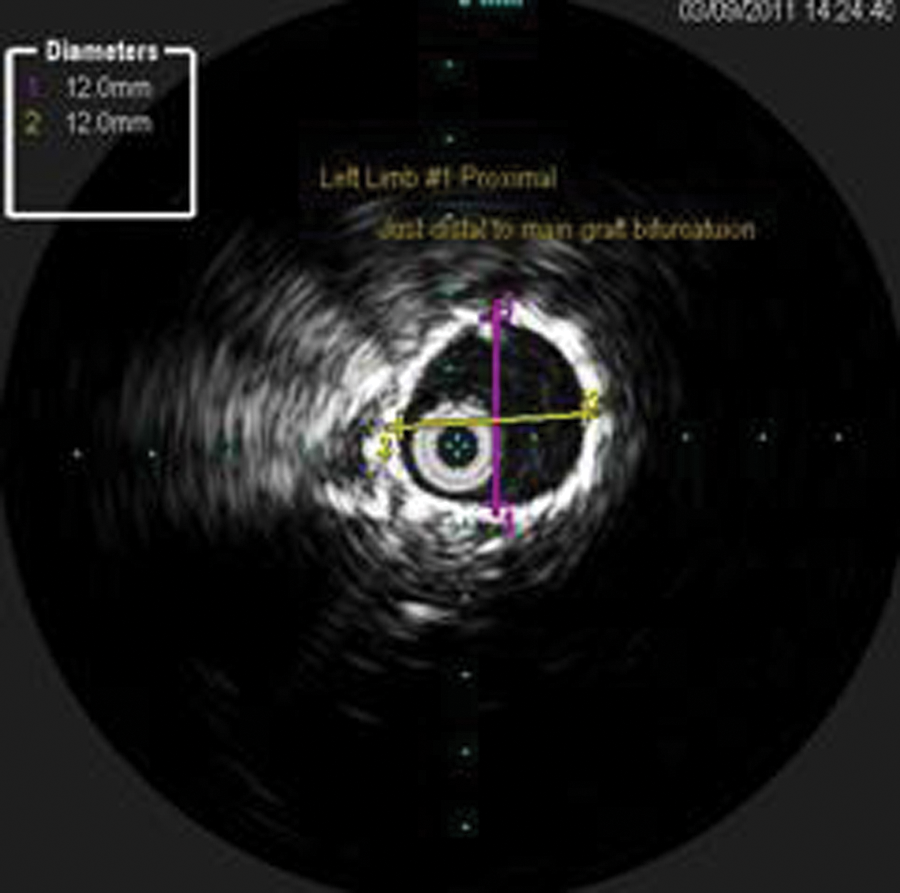
Intravascular sonography image with measurements of the original proximal left limb. This became the new seal zone.
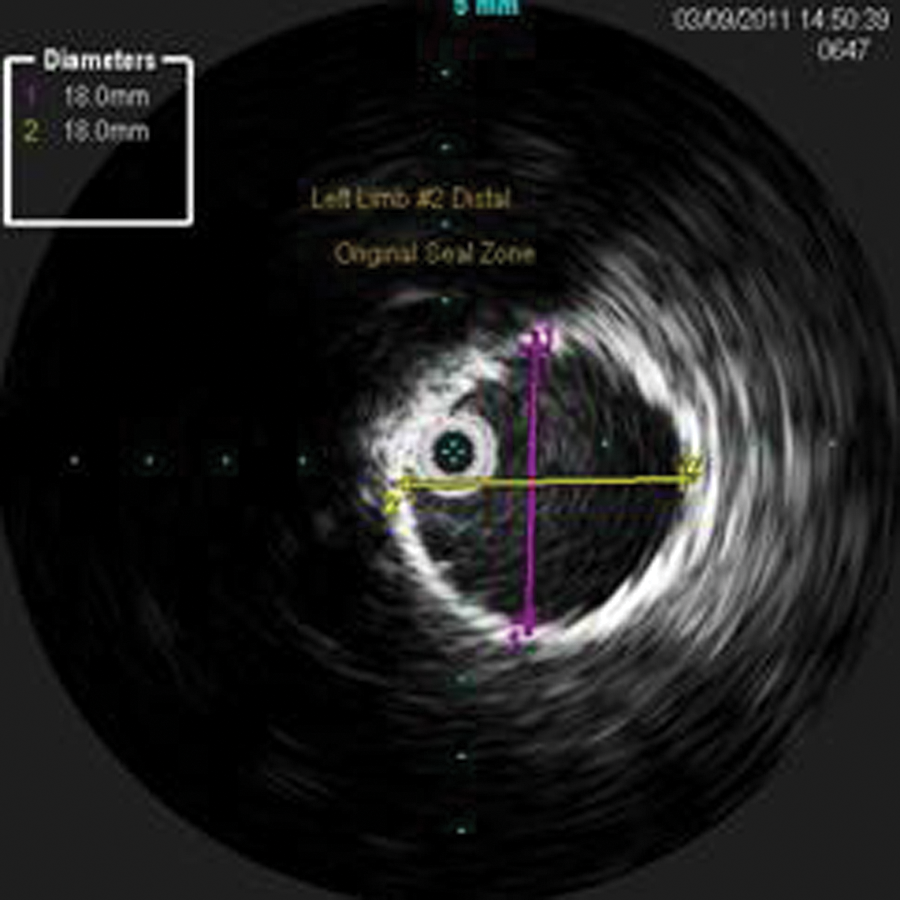
Intravascular sonography image with measurements of the original distal left limb and original seal zone.
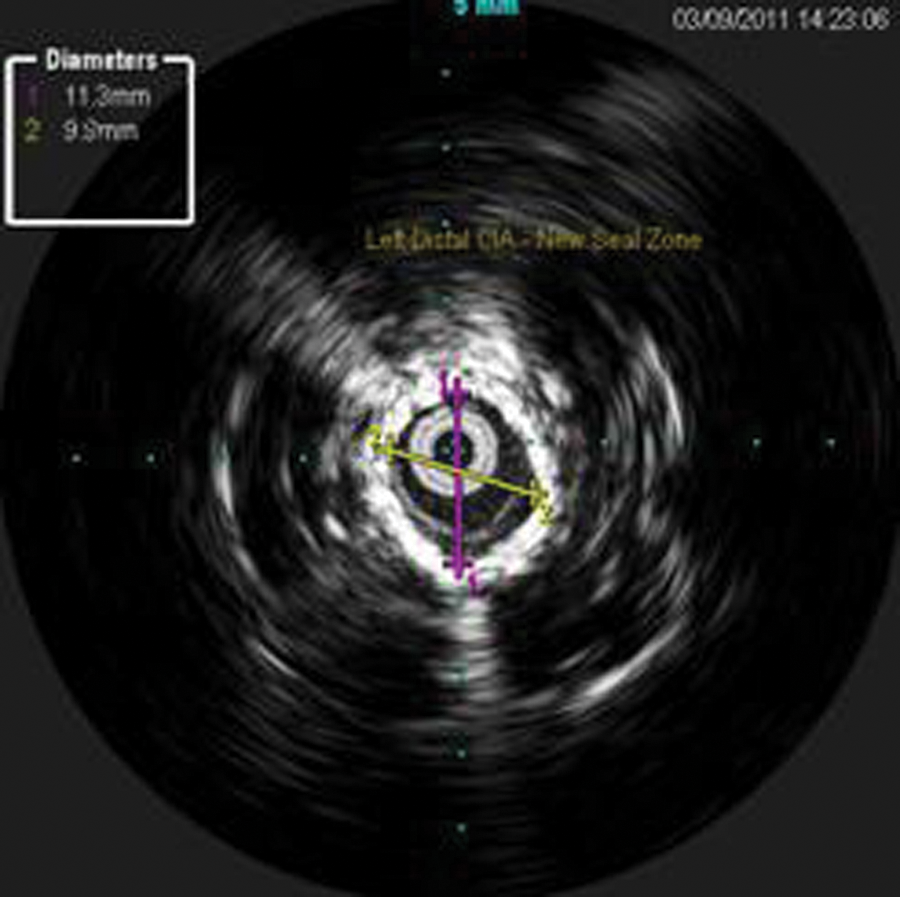
Intravascular sonography image with measurements of the left native distal common iliac artery, distal to the original endograft. This became the new distal seal zone.
CT measurements were obtained prior to the beginning of the case and were compared with the IVUS measurements. Based on the correlated CT and IVUS measurements, a 14 × 90-mm stent graft (Cook, Inc., Bloomington, Indiana) was selected. The graft was loaded over the wire and advanced to a position with a proximal landing zone in the proximal left limb and extending through the original left limb (the two separated stent grafts) and a distal landing zone past the original landing zone just proximal to the left hypogastric artery. A completion angiogram was performed that showed complete exclusion of the type III endoleak, and no new endoleaks were visualized after the deployment of the new left limb. A follow-up sonogram, performed the following day, showed no signs of endoleak (Figure 17). The patient was discharged with no complications.
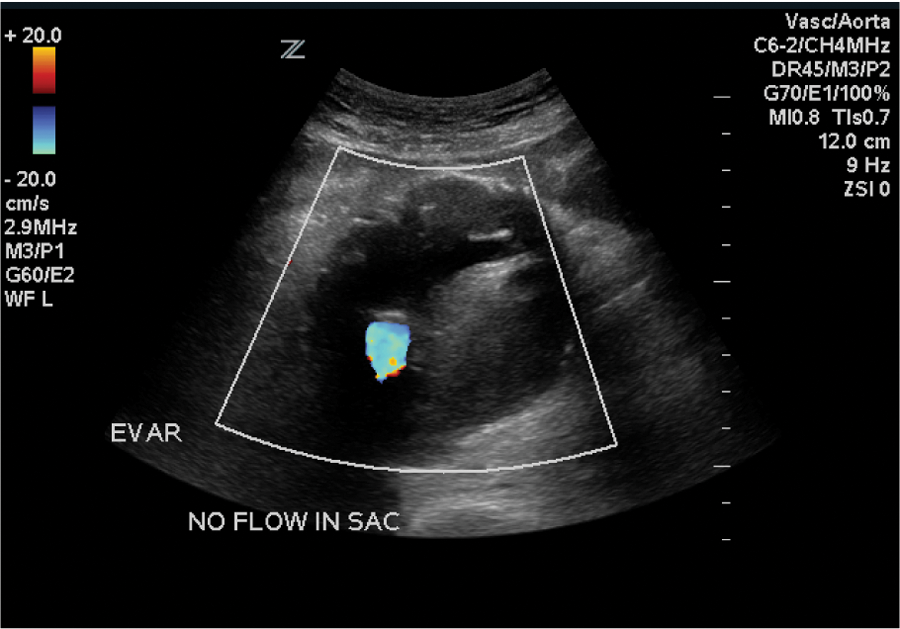
Duplex sonography of the repaired endograft showing blood flow only within the endograft and fresh thrombus in the residual sac.
Discussion
Abdominal aortic aneurysm is a common disease in today’s population. It is the 10th leading cause of death in men aged 65 to 74 years in the United States. 1 AAA occurs when the walls of the abdominal aorta weaken and over time the aorta expands in size. Patients may present with a pulsatile abdominal mass or dull back pain; however, most AAAs are asymptomatic and are usually found incidentally on a screening sonography examination or other unrelated imaging study. 3 An aneurysm is defined as a permanent dilation of an artery, with an increase in diameter greater than 1.5 times its normal diameter. 3 For most patients, this means that an AAA may be defined as an aorta that measures equal to or greater than 3.0 cm. 4 Although the gold standard for AAA diagnosis is CT, 5 sonography has also proved to be useful in the diagnosis and surveillance of AAA. Current recommendations are that AAAs 3 to 4 cm should be assessed with sonography every 12 months, and those 4 to 4.5 cm should be followed every 6 months. AAAs greater than 4.5 cm should be considered for referral to a vascular specialist. 3 Once the AAA is deemed high risk for rupture, it is considered for repair. Based on the current evidence, 5.5 cm appears to be the best threshold for repair. 2 Any AAA that grows faster than a rate of 0.6 to 0.8 cm annually is also considered for repair. 3 Much debate still remains about whether open repair or EVAR is the treatment of choice. Before 1991, open repair was the only widely available treatment for AAA. Since its introduction, EVAR has evolved as an option for patients who are not candidates for open repair. In high-risk patients with life-threatening AAA, EVAR may the only treatment option. 6 EVAR has significantly reduced healing time, hospital stay, and overall cost of care for the treatment of AAA.7,8 It has also resulted in significantly better perioperative outcomes, such as less blood loss, less operating time, and a lower rate of mechanical ventilation. 8 With its growing popularity, EVAR has introduced a new need for surveillance as new potential complications have arisen, including endoleak, graft migration, and endograft occlusion. 9 As a result of these complications, EVAR is associated with a significantly higher rate of reintervention than open repair. 10
During an EVAR procedure, many interventionalists elect to use IVUS as an adjunct to angiography for additional visualization, identify important aortic branches, and obtain measurements for graft sizing. 11 The residual aneurysm sac is left in the body and typically will thrombose over time. Ideally, it should contract around the endograft as the body naturally reabsorbs the thrombotic material. White et al 12 state, “Endoleak is a condition associated with endoluminal vascular grafts, defined by the persistence of blood flow outside the lumen of the endoluminal graft but within an aneurysm sac or adjacent vascular segment being treated by the graft. Endoleak is due to incomplete sealing or exclusion of the aneurysm sac or vessel segment, as evidenced by imaging studies, such as contrast-enhanced CT scanning, ultrasonography, or angiography.” There are four types of endoleak (Table 1). Endoleaks are important to identify as they can lead to aneurysm growth and rupture. 13
Types of Endoleak 17

A type III endoleak is a separation of the graft. This type of endoleak is especially critical to diagnose and treat because it allows unobstructed aortic blood flow under systemic arterial pressure directly into the residual AAA sac as if there were no graft ever placed, increasing the risk for rupture. Type III endoleaks are considered medical emergencies and must be treated immediately. One treatment option is a secondary endovascular approach to close an endoleak by placing an additional endoluminal graft or covered stent. 12 Intravascular sonography is especially useful during this procedure as it gives a detailed image of the artery and graft from the inside. The identification of target sites with IVUS is easy and reliable. 14 It is possible to identify important aortic branches, graft beginning and ending zones, and previous graft sizes using IVUS. Measurements first obtained from CT can then be correlated with IVUS, increasing the confidence in selecting the proper-sized graft. The use of IVUS for measurements to select the accurate graft size and for visualization during graft deployment also helps minimize the chance of developing an endoleak in the postoperative period.14,15 It is especially valuable when operating on patients with chronic renal failure, as the amount of contrast is reduced by using IVUS for branch location and graft apposition. 16 It is particularly valuable in the treatment of type III endoleaks as it is able to identify the actual graft separation as well as provide accurate measurements at multiple locations.
Conclusion
This case report presents one of the less common types of endoleaks that can arise following an EVAR procedure. The risk for endoleak mandates the need for surveillance by either CT or duplex sonography. In this case, both CT and duplex sonography were able to identify a type III endoleak that needed immediate repair. This report describes the initial EVAR procedure as well as a secondary endovascular procedure to exclude the endoleak. It also demonstrates how intravascular sonography can be used in conjunction with CT measurements for case management: to help choose the appropriate size graft for the repair, to delineate important anatomy, and how it can complement angiography during the procedure.
Footnotes
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author received no financial support for the research, authorship, and/or publication of this article.
